Abstract
Background
Effective positive-pressure ventilation is a critical factor in newborn resuscitation. Neonatal endotracheal intubation (ETT) needs considerable training and experience, which poses a human factor challenge. Laryngeal mask airway (LMA) ventilation can be a secure and viable alternative during the initial stages of newborn resuscitation. However, there is limited evidence for its use during chest compression (CC).
Methods
Seventeen lambs were randomized into LMA or ETT ventilation post cord occlusion induced cardiac arrest. After 5 min of cardiac arrest, resuscitation was initiated as per NRP recommendations. Ventilation, oxygenation, systemic and pulmonary hemodynamic parameters were recorded till the return of spontaneous circulation (ROSC) or 20 min.
Results
Baseline characteristics were similar between the groups. The incidence of ROSC was 75% (6/8) in the LMA group and 56% (5/9) in the ETT group (p = 0.74). The median (IQR) time to achieve ROSC was 6.85 min (6 min–9.1 min) in the LMA group and 7.50 min (5.33 min–18 min) in the ETT group (p = 0.65).
Conclusion
LMA ventilation during CC is feasible and non-inferior to ETT in this model.
Impact
-
Laryngeal mask airway (LMA) ventilation with chest compression is feasible and non-inferior to endotracheal tube ventilation in this experimental near-term lamb model of asphyxial cardiac arrest.
-
First translational study to evaluate the use of LMA as an airway device with chest compression. Evidence primer for clinical studies to evaluate and confirm the feasibility and efficacy of LMA ventilation with chest compression are necessary before randomized clinical trials in neonates.
-
LMA use in neonatal cardiopulmonary resusciation (CPR) could have the potential to optimize advanced resuscitation, especially in resource-limited healthcare settings.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
The effective delivery of positive-pressure ventilation (PPV) is the most important factor in the successful resuscitation of a depressed newborn.1 If the infant’s heart rate remains <60 bpm after at least 30 s of PPV that inflates the lungs, as evidenced by chest movement with ventilation, chest compressions (CC) are indicated. Neonatal resuscitation program (NRP) strongly recommends endotracheal tube (ETT) insertion if CC are necessary during resuscitation because ETT will maximize the efficacy of each positive-pressure breath and allow the compressor to give compressions from the head of the bed.2
ETT insertion in a newborn requires considerable training and experience.3 Even resuscitators with advanced training sometimes require prolonged and/or multiple attempts to successfully intubate the neonatal trachea.4 Study conducted using the National Emergency Airway Registry for Neonates (NEAR4NEOS), an international multi-site prospective database, found that first attempt success rates in neonatal intubations are <50% with considerable risk of procedure-related adverse events.5 Availability of healthcare personnel with expertise in advanced resuscitation, including newborn ETT insertion, is a practical challenge, especially in limited-resource (human and technical) settings. With the change in practice of resuscitating infants born through meconium-stained amniotic fluid in 2015, where routine endotracheal intubation and tracheal suctioning are no longer needed, intubation skills have become even harder to acquire and maintain.6 PPV with laryngeal mask airway (LMA) is a reasonable alternative.2,7,8
NRP recommends LMA as a rescue airway device when a resuscitator “cannot ventilate and cannot intubate.”1 LMA insertion in the newborn can be easily taught and successfully performed on the first attempt to establish a clinically patent airway within 10 s and restore oxygenation in 30 s.9 Studies have shown that providers can achieve proficiency in LMA insertion after a 15-min educational training on manikins alone (mean time to successful insertion of an LMA was 5 s).10 Laryngeal mask ventilation may be a more effective technique for healthcare personnel with limited expertise in endotracheal intubation in newborns.11
Prior randomized controlled trials had shown that LMA is more effective than face mask in delivering PPV in newborns >34 weeks/birth weight >1500 g.11,12 LMA use had better odds of successful resuscitation, 5-minute Apgar scores, and decreased the need for NICU admissions.12 Trials comparing LMA to ETT found ventilation of a depressed newborn with LMA to be equally effective.13,14 Oxygenation, ventilation, and Apgar scores were not different with either airway device during neonatal resuscitation.14 A meta-analysis found similar evidence for comparable efficacy between LMA and ETT in the initial stages of neonatal resuscitation.15 In addition, animal studies have shown that the chest compressions do not affect the seal created by the LMA.16
Current evidence suggests that LMA ventilation can be a secure and viable alternative to manage the airway of a depressed newborn during the initial stages of resuscitation; however, there is limited evidence for its use during chest compressions.15,17,18 We hypothesized that PPV through LMA during chest compression would have a similar time to return of spontaneous circulation (ROSC) compared to ETT. We defined ROSC as heart rate (HR) > 60 beats per minute (bpm) with systolic blood pressure (BP) > 30 mmHg.19
Primary outcome
The primary outcome of our study was the time taken to achieve the return of spontaneous circulation (ROSC).
Secondary outcome
The secondary outcome measures are:
-
1.
Incidence of ROSC.
-
2.
Number of epinephrine doses required during the resuscitation.
-
3.
Adequacy of ventilation and oxygenation measured by tidal volume (TV), mean airway pressures (MAP), partial pressure of carbon dioxide (paCO2), and partial pressure of oxygen (paO2).
-
4.
Systemic and pulmonary hemodynamic parameters measured as average left carotid blood flow(Qca), mean blood pressure (BP), and average left pulmonary blood flow(Qp).
-
5.
Procedure-related complications specifically abdominal distension, measured as abdominal girth at the level of umbilicus before and after resuscitation.
Methods
Ethics statement
The study protocol was approved by the Institutional Animal Care and Use Committee (IACUC, protocol # PED10085N) at the University at Buffalo. All experiments were performed according to animal ethical guidelines, in compliance with National Research Council guidelines for the care and use of laboratory animals.20
Study design
Randomized controlled trial (non-inferiority trial)
Study population
A transitioning asphyxiated cardiac arrest lamb model is an established model to study the gas exchange, systemic and pulmonary hemodynamics during neonatal resuscitation.19
Animal preparation
Time-dated near-term (139–141-day gestation; term being 145 days) pregnant ewes (2 different breeds – Polypay and combination of Suffolk, Dorset, Katahdin) were procured from Maple Twig Farms (Meyersville, PA) and May Family Farms (Buffalo Mills, PA). Following an overnight fast, the ewe was medicated with intravenous diazepam and ketamine. The ewe was intubated with a 10.0-mm-cuffed endotracheal tube (ETT), and general anesthesia was provided by 2–5% inhaled isoflurane. A cesarean section was performed. Fetal lambs were partially exteriorized and intubated with a cuffed endotracheal tube to remove lung liquid by gravity and occluded to prevent gas exchange. Flow transducers (Transonic, Ithaca, NY, USA) were placed around the left carotid artery and the left pulmonary artery. Catheters were placed in the right carotid artery and right jugular vein for pressure monitoring and blood gas analysis.21 Pulse oximetry probe and electrocardiogram (ECG) leads were placed to monitor preductal oxygen saturation and heart rate, respectively.
Randomization
We used opaque envelopes to randomize the lambs into either experimental or control groups before C-section. Resuscitators could not be blinded due to the nature of the intervention being studied (ETT vs LMA).
Experimental protocol
Asphyxia-induced cardiac arrest was achieved by umbilical cord occlusion. Asystole was defined as the absence of heart rate by ECG and absence of carotid flows. Post asystole lambs were left in asystole for an additional 5 min before resuscitation to ensure complete cardiac arrest. During this period, we removed the ETT, moved the lamb to the infant warmer, and inserted ETT or LMA SupremeTM (Laryngeal Mask Airway Co. Ltd, Jersey, UK) depending on the group to which the lamb was randomized, (A) LMA ventilation group (B) ETT ventilation group (controls). We used LMA SupremeTM because of the advantage of an esophageal drain tube. We used LMA Supreme size 1 for lambs <5 kgs and size 1.5 for lambs ≥5 kgs. We used continuous suction set at 80 cm H2O through a 6 Fr suction catheter inserted through the esophageal drain tube at the start of resuscitation. We initiated PPV as per NRP recommendations precisely at 5 min after asystole in both the groups. Figure 1 shows the proper placement of a LMA in a lamb model. We continuously measured end-tidal carbon dioxide, peak inspiratory pressure, positive end-expiratory pressure, and the tidal volume using a respiratory profile monitor (Respironics NM3, Philips). Arterial blood gases were drawn at 4 min of asystole, every minute during resuscitation, at ROSC, and every 5 min for 30 min following ROSC. Arterial blood flows and pressures were continuously recorded using a computer with BIOPAC systems (Goleta, California, USA). Resuscitation was continued until ROSC (defined as HR > 60 with a systolic BP > 30 mmHg) was attained or 20 min elapsed, whichever occurred first.
Sample size calculation
The duration of CPR associated with successful ROSC using an endotracheal tube as the airway device reported from similar experimental protocols in lambs was reviewed. The median (IQR) time to ROSC reported in a couple of recently published similar animal protocols from our lab was 8 min (6.3–10) and 6.1 min (2.9–7.1)22,23. Based on the review of published work from our lab, we calculated the standard deviation of the time to ROSC(outcome) in the control group (comparator) as 3 min.
We calculated the sample size based on the non-inferiority margin of 5%, power of 90%, and standard deviation of the outcome of 3 min. If there is truly no difference between the standard and experimental treatment, then 14 subjects (7 in each group) are required to be 90% sure that the lower limit of a one-sided 95% confidence interval (or equivalently a 90% two-sided confidence interval) will be above the non-inferiority limit of −5.
Data analysis
Continuous variables are reported as mean and standard deviation (SD) or median and interquartile range (IQR) and analyzed by Student t-test, Mann–Whitney U test, or repeated-measures ANOVA variance as appropriate. Categorical variables are reported as percentages and analyzed using the χ2 test with Yates correction. XLSTAT 2021.1 (Addinsoft, NY, USA) was used for statistical analysis. We defined statistical significance as p < 0.05 and analyzed all the lambs that were randomized.
Results
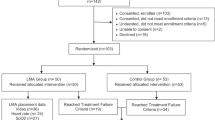
Seventeen lambs (8 in LMA and 9 in the ETT group) were randomized, asphyxiated, and resuscitated. Baseline characteristics were similar between the two groups as shown in Table 1.
Incidence and time to ROSC
The incidence of ROSC was 75% (6/8) in the LMA group and 56% (5/9) in the ETT group (p = 0.74). The median (interquartile range) time to achieve ROSC was 6 min 51 s (6 min 0 s–9 min 6 s) in the LMA group and 7 min 30 s (5 min 20 s–18 min 0 s) in the ETT group (p = 0.65) (Table 2).
Epinephrine during resuscitation
Lambs that achieved ROSC in the LMA group required one dose of epinephrine. In the ETT group, one lamb achieved ROSC without epinephrine, two lambs required four doses of epinephrine and two lambs achieved ROSC with one dose of epinephrine. The percentage of lambs that required more than one dose of epinephrine to achieve ROSC was 0% (0/6) in the LMA group and 40% (2/5) in the ETT group (p = 0.35) (Table 2).
Ventilation parameters
There was no difference between the mean (SD) peak inspiratory pressure used during CC in the LMA group (33 ± 5 cm H2O) and in the ETT group (33 ± 7 cm H2O) (p = 0.78). Mean airway pressure used during CC in the LMA group 15(5) cm H2O and ETT group 16(5) cm H2O was also similar (p = 0.84). The mean (SD) positive end-expiratory pressure used during chest compressions in the LMA group was 7(4) cm H2O and 6(3) cm H2O in the ETT group (p = 0.84). The mean (SD) expired tidal volume per kilogram body weight delivered during chest compressions in the LMA group was 9.9(3.6) ml/kg and 10.7(2.6) ml/kg in the ETT group (p = 0.59). We analyzed the mean airway pressure and expired tidal volume per kilogram body weight across various time points during the resuscitation and post-resuscitation for the lambs, which achieved ROSC and were not different (p = 0.28 and p = 0.63, respectively) (Fig. 2).
Bar graph comparing the a mean airway pressure in centimeter water and b expired tidal volume in milliliter/kilogram bodyweight in the lambs that achieved ROSC along the y axis and various time points of resuscitation along the x axis namely PPV, PPV + CC, Post ROSC. The dark bars represent LMA group, and the light bars represent the ETT group. There was no statistical difference in the mean airway pressures used and the tidal volumes delivered between the two groups.
Arterial blood gas
The mean arterial pH, paCO2, and paO2 during chest compression were not statistically different between the LMA group and the ETT group as shown in Table 2. However, the mean lactate during chest compression was significantly lower in the LMA group (p = 0.003). The statistical significance persisted in the subgroup analysis of the lambs that achieved ROSC (p = 0.03). The arterial paCO2 and paO2 in mmHg across various time points during the resuscitation and post-resuscitation for the lambs, which achieved ROSC were not different (p = 0.47 and p = 0.59, respectively) (Fig. 3).
Bar graph comparing the mean arterial a paCO2 levels and b paO2 in milliliter mercury in the lambs that achieved ROSC along the y axis and various time points of resuscitation along the x axis namely baseline, asystole, PPV, PPV + CC, Post ROSC. The dark bars represent LMA group, and the light bars represent the ETT group. There was no statistical difference in the mean arterial paCO2 and paO2 between the groups (p = 0.47 and p = 0.59, respectively).
Hemodynamic parameters
The mean Qca, mean BP, and mean Qp during chest compression were not statistically different between the LMA group and the ETT group as shown in Table 2. We analyzed the mean Qca, mean BP, and mean Qp across various time points during the resuscitation and post-resuscitation for the lambs, which achieved ROSC and they were not different (p = 0.75, p = 0.66, and p = 0.58, respectively) (Fig. 4).
Bar graph comparing the a mean carotid artery flow in milliliter per kilogram per minute, b mean arterial blood pressure in millimeter mercury, c mean pulmonary artery flow in milliliter per kilogram per minute in the lambs that achieved ROSC along the y axis and various time points of resuscitation along the x axis. The dark bars represent the LMA group, and the light bars represent the ETT group. There was no difference in the mean carotid artery flow, mean arterial blood pressure, mean pulmonary artery flow between the groups (p = 0.75, 0.66, 0.58, respectively).
Abdominal distension
The median (IQR) increase in the abdominal girth in centimeters measured before and after resuscitation in the lambs randomized to the LMA group was 3.8 (3.1–4.1) vs. 0.5 (0–1) in the lambs randomized to the ETT group (p = 0.008). The median increase in the abdominal girth during resuscitation was 11.5% from baseline in the LMA group.
Discussion
A knowledge gap persists in identifying the most effective airway device and interface during neonatal resuscitation.8 Novel techniques that require lower skills to perform, are easy to acquire, and have longer retention can help in the effective delivery of advanced CPR steps like CC and medications in low-resource settings. LMA is an alternative airway device that healthcare personnel can safely use even without extensive training in advanced resuscitation.24 In this randomized controlled trial in a near-term lamb model of perinatal asphyxial cardiac arrest we find that LMA ventilation during CC is feasible and non-inferior to ETT in resuscitation. Compared to ETT, LMA ventilation during chest compressions required similar mean airway pressures to achieve adequate tidal volumes. LMA ventilation resulted in similar ventilatory parameters as measured by arterial paCO2 and PaO2. Pulmonary and systemic hemodynamic measures were not different with either of the airway devices during chest compression. LMA ventilation during chest compression resulted in significantly lower blood lactate levels without any difference in other blood gas parameters.
World Health Organization’s “Every Newborn Action Plan” to end preventable deaths in low and middle-income countries envisions reducing the neonatal mortality rate to less than 10 per 1000 live birth by 2035.25 Although the implementation of basic resuscitation programs like Helping Baby Breathe with a focus on “The Golden Minute” (first minute after birth) has contributed to substantial progress,26 there is a need and utility for developing capabilities of mid-level resuscitation in low-resource settings.27 Such capabilities might include securing a LMA as an alternative to endotracheal intubation before CC and intraosseous access as an alternative to umbilical venous access for medications. That would enable healthcare personnel with limited skills to deliver advanced neonatal resuscitation in appropriate settings.28 In the developed countries, LMA use is more prevalent in adult resuscitative medicine, especially in out-of-hospital cardiac arrest(OHCA). A randomized trial that compared the effect of the initial airway strategy found that the Laryngeal tube arm had better 72 h-survival compared to the ETT arm in OHCA in adults.29
Newborns requiring CC during resuscitation at delivery are rare. It is estimated to occur in 0.08% of all term and near-term deliveries.30 The rarity of the problem makes it difficult to conduct a clinical study to evaluate the efficacy and safety of LMA ventilation during chest compression. Ethical concerns also surround conducting such resuscitation trials before gathering strong evidence for the proposed intervention. Animal experiments can bridge this knowledge gap. The sheep larynx is an appropriate experimental model for various surgical and functional investigations in human laryngology because of the similarities in the dimensions.31 A study that aimed to test the applicability of LMA in several animal species, including dogs (n = 33), cats (n = 9), swine (n = 9), rabbits (n = 5), sheep (n = 7), and roe deer (n = 1) and to compare its effectiveness in securing sealed airway respiration as an alternative to endotracheal tubes found that LMA designed for humans can be used for airway management in veterinary medicine. The rate of successful insertion of the LMA in the first attempt was highest in sheep in that study.32 In our study, we found that the insertion of LMA in the lambs was easy. We maintained the LMA in a relatively stable position (Fig. 2) during the entire duration of resuscitation and post resuscitative mechanical ventilation in lambs that achieved ROSC. In order to minimize variability in the insertion times of the airway devices33 and the efficacy of CC, we had the same trained personnel performing the assigned roles in all the experiments.
The LMA provides PPV with low pressures in the respiratory passage and reduced air losses compared to an uncuffed ETT with the advantage of lower airway resistance.34 A pediatric study done during the operative room anesthesia had shown a significant difference between ventilation with LMA compared to ETT in the peak inspiratory pressures (PIP) (10.4 ± 1.19 vs. 13.2 ± 1.25 cm H2O) required to achieve similar tidal volumes (TV) (140.8 ± 13.85 ml vs. 139.67 ± 28.95 ml). PIP with LMA was lesser compared to PIP with ETT. Pulmonary dynamic compliance (TV/PIP-PEEP) with LMA (13.62 ± 3.03 l/cm H2O) was significantly higher than that with ETT (10.84 ± 2.98) in children.35 However, in our study, we did not find a difference in the PIP during LMA ventilation. Our results could be due to the ventilation of the fluid-filled lung in our experimental model.
Theoretical concerns exist regarding the supraglottic airway devices (SGD) increasing the pressure in the retropharyngeal space that could be transmitted to the carotid arteries and reduce carotid blood flow. SGD was associated with decreased carotid blood flow compared to ETT during cardiopulmonary resuscitation in experimental porcine models.36,37 However, a study that evaluated carotid artery compression in patients with SGD did not demonstrate radiographically evident mechanical compression of the carotid artery.38 Hemodynamic data from our study supports this observation. We did not find any significant difference in the carotid blood flow during or after resuscitation. The difference in these results could be related to the difference in the induction of experimental cardiac arrest (ventricular fibrillation vs. umbilical cord occlusion)
The main limitation of our study is the inherent concern of species differences. Because of the lack of evidence from clinical studies to understand the effect of time difference to achieve ROSC in neonates,39 we based our sample size calculation on data from translational studies. In addition, we cannot extrapolate the results of our study to preterm gestation with surfactant deficient lungs. Our preliminary studies in lambs with the classic LMA showed us that abdominal distension with prolonged PPV could be a procedure-related adverse effect. We aimed to minimize this effect by using a second-generation LMA with an esophageal drain tube. Despite this experimental set up we found that LMA ventilation lead to abdominal distension of 11.5% increase from baseline. There are significant species differences in the GI anatomy between humans and lambs. The stomach constitutes 19% of humans’ total digestive tract volume compared to sheep, about 67%. We believe that the species difference in the GI anatomy contributed partly to our study’s findings related to abdominal distension. Nevertheless, we did not observe any significant differences in the tidal volumes obtained using LMA compared to ETT. We used cuffed ETT in our experimental model, contrary to the standard clinical practice of using uncuffed ETT in newborn infants.
To the best of our knowledge, this is the first translational study to evaluate the use of LMA as an airway device during chest compressions in a term animal model of perinatal asphyxia as an alternative to ETT. The strength of our study is the evidence primer available for undertaking clinical studies to evaluate the feasibility and efficacy of LMA use during chest compressions in newborn infants. Such studies would have the potential to optimize neonatal resuscitation in resource-limited healthcare settings.
A randomized trial conducted in Uganda comparing the use of cuffless LMA and face mask in neonates requiring positive-pressure ventilation by midwives found no difference in the early neonatal death or the NICU admission rates for moderate or severe hypoxic-ischemic encephalopathy. One of the secondary outcomes studied in this trial was the need for advanced resuscitation (endotracheal intubation, chest compression, and medications) initiated if a physician was available. The need for advanced resuscitation was ~7% in both groups. The use of LMA as the airway device during advanced resuscitation must be studied systematically in similar resource-limited settings to determine if the use of LMA could bring a meaningful improvement in the short-term survival and reduction in morbidity.
In addition to the importance of early effective airway access, the cost-effectiveness of LMA use is an essential factor to be considered apart from the safety and efficacy before considering the widespread use of LMA during neonatal resuscitation, especially in low-resource settings. The supreme LMA used in our study was a disposable device. However, there are other types of second-generation LMAs that can be autoclaved and reused. An educational interventional study conducted in a developing country found that the study participants (physician and midwives) considered LMA a sustainable, cost-effective method to resuscitate newborn infants.40 Another study that evaluated the cost analysis of LMA use in anesthesia practice found that the cost-efficiency of LMA will depend on the reuse rates.41
Conclusion
Laryngeal mask airway ventilation during chest compression is feasible and non-inferior to endotracheal tube ventilation in this experimental near-term lamb model of asphyxial cardiac arrest induced by umbilical cord occlusion. Further clinical studies confirming the feasibility and efficacy of LMA ventilation during chest compression in newborn infants are necessary before undertaking randomized clinical trials.
Change history
18 May 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41390-022-02106-8
References
Kattwinkel, J. et al. Part 15: Neonatal resuscitation: 2010 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation 122, S909–S919 (2010).
Weiner, G. M., Zaichkin, J. & Kattwinkel, J., American Academy of Pediatrics & American Heart Association. Textbook of Neonatal Resuscitation (2016).
Falck, A. J., Escobedo, M. B., Baillargeon, J. G., Villard, L. G. & Gunkel, J. H. Proficiency of pediatric residents in performing neonatal endotracheal intubation. Pediatrics 112, 1242–1247 (2003).
Carbine, D. N., Finer, N. N., Knodel, E. & Rich, W. Video recording as a means of evaluating neonatal resuscitation performance. Pediatrics 106, 654–658 (2000).
Foglia, E. E. et al. Neonatal intubation practice and outcomes: An International Registry Study. Pediatrics 143, e20180902 (2019).
Myers, P. & Gupta, A. G. Impact of the Revised Nrp Meconium Aspiration Guidelines on Term Infant Outcomes. Hosp Pediatr 10, 295–299 (2020)
Zanardo, V. et al. Delivery room resuscitation of near-term infants: role of the laryngeal mask airway. Resuscitation 81, 327–330 (2010).
Aziz, K. et al. Part 5: Neonatal resuscitation: 2020 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation 142, S524–S550 (2020).
Paterson, S. J., Byrne, P. J., Molesky, M. G., Seal, R. F. & Finucane, B. T. Neonatal resuscitation using the laryngeal mask airway. Anesthesiology 80, 1248–1253 (1994).
Gandini, D. & Brimacombe, J. Manikin training for neonatal resuscitation with the laryngeal mask airway. Paediatr. Anaesth. 14, 493–494 (2004).
Pejovic, N. J. et al. Neonatal resuscitation using a laryngeal mask airway: a randomised trial in Uganda. Arch. Dis. Child 103, 255–260 (2018).
Trevisanuto, D. et al. Supreme laryngeal mask airway versus face mask during neonatal resuscitation: a randomized controlled trial. J. Pediatr. 167, 286–291.e281 (2015).
Yang, C. et al. Randomized, controlled trial comparing laryngeal mask versus endotracheal intubation during neonatal resuscitation—a secondary publication. BMC Pediatr. 16, 17 (2016).
el Shimi, M., S. Abusaif, I. & Khafagy, S. Efficacy of laryngeal mask airway in neonatal resuscitation. The Egyptian. J. Hosp. Med. 70, 1767–1772 (2018).
Qureshi, M. J. & Kumar, M. Laryngeal mask airway versus bag-mask ventilation or endotracheal intubation for neonatal resuscitation. Cochrane Database Syst. Rev. 3, CD003314 (2018).
Carlson, J. N., Suffoletto, B. P., Salcido, D. D., Logue, E. S. & Menegazzi, J. J. Chest compressions do not disrupt the seal created by the laryngeal mask airway during positive pressure ventilation: a preliminary porcine study. CJEM 16, 378–382 (2014).
Bansal, S. C., Caoci, S., Dempsey, E., Trevisanuto, D. & Roehr, C. C. The laryngeal mask airway and its use in neonatal resuscitation: a critical review of where we are in 2017/2018. Neonatology 113, 152–161 (2018).
Shea, J. E., Scrivens, A., Edwards, G. & Roehr, C. C. Safe emergency neonatal airway management: current challenges and potential approaches. Arch. Dis. Child. Fetal Neonatal Ed. https://doi.org/10.1136/archdischild-2020-319398 (2021).
Rawat, M. et al. Oxygenation and hemodynamics during chest compressions in a lamb model of perinatal asphyxia induced cardiac arrest. Children 6, 52 (2019).
Council, N. R. Guide for the Care and Use of Laboratory Animals 8th edn (The National Academies Press, 2011).
Vali, P. et al. Hemodynamics and gas exchange during chest compressions in neonatal resuscitation. PLoS ONE 12, e0176478 (2017).
Vali, P. et al. Continuous chest compressions with asynchronous ventilations increase carotid blood flow in the perinatal asphyxiated lamb model. Pediatr. Res. https://doi.org/10.1038/s41390-020-01306-4 (2021).
Sankaran, D. et al. Randomised trial of epinephrine dose and flush volume in term newborn lambs. Arch. Dis. Child. Fetal Neonatal Ed. 106, 578–583 (2021).
Pejovic, N. J. et al. A randomized trial of laryngeal mask airway in neonatal resuscitation. N. Engl. J. Med. 383, 2138–2147 (2020).
WHO. Every Newborn: An Action Plan to End Preventable Deaths (World Health Organization, Geneva, 2014).
Versantvoort, J. M. D. et al. Helping babies breathe and its effects on intrapartum-related stillbirths and neonatal mortality in low-resource settings: a systematic review. Arch. Dis. Child. 105, 127 (2020).
Umphrey, L. et al. When helping babies breathe is not enough: designing a novel, mid-level neonatal resuscitation algorithm for médecins sans frontières field teams working in low-resource hospital settings. Neonatology 114, 112–123 (2018).
Niermeyer, S., Robertson, N. J. & Ersdal, H. L. Beyond basic resuscitation: what are the next steps to improve the outcomes of resuscitation at birth when resources are limited? Semin. Fetal Neonatal Med. 23, 361–368 (2018).
Wang, H. E. et al. Effect of a strategy of initial laryngeal tube insertion vs endotracheal intubation on 72-hour survival in adults with out-of-hospital cardiac arrest: a randomized clinical trial. JAMAM 320, 769–778 (2018).
Wyckoff, M. H., Perlman, J. M. & Laptook, A. R. Use of volume expansion during delivery room resuscitation in near-term and term infants. Pediatrics 115, 950–955 (2005).
Zrunek, M., Happak, W., Hermann, M. & Streinzer, W. Comparative anatomy of human and sheep laryngeal skeleton. Acta Otolaryngol. 105, 155–162 (1988).
Vidricková, P. & Boldižár, M. Usage of laryngeal mask airway devices in veterinary medicine. Macedonian Vet. Rev. 43, 131–139 (2020).
Mani, S. & Rawat, M. Proficiency of laryngeal mask airway insertion skill in NRP certified providers. Am. J. Perinatol. https://doi.org/10.1055/s-0040-1721379 (2020).
Tartari, S., Poole, D., Bocchi, A., Sgarbi, A. & Alvisi, R. Laryngeal mask vs tracheal intubation during mechanical ventilation in pediatric anesthesia. Miner. Anestesiol. 66, 33–37 (2000).
Mahdavi, A., Razavi, S. S., Malekianzadeh, B. & Sadeghi, A. Comparison of the peak inspiratory pressure and lung dynamic compliance between a classic laryngeal mask airway and an endotracheal tube in children under mechanical ventilation. Tanaffos 16, 289–294 (2017).
Segal, N. et al. Impairment of carotid artery blood flow by supraglottic airway use in a swine model of cardiac arrest. Resuscitation 83, 1025–1030 (2012).
Kim, T. H. et al. Effect of endotracheal intubation and supraglottic airway device placement during cardiopulmonary resuscitation on carotid blood flow over resuscitation time: an experimental porcine cardiac arrest study. Resuscitation 139, 269–274 (2019).
White, J. M., Braude, D. A., Lorenzo, G. & Hart, B. L. Radiographic evaluation of carotid artery compression in patients with extraglottic airway devices in place. Acad. Emerg. Med. 22, 636–638 (2015).
Foglia, E. E. et al. Duration of resuscitation at birth, mortality, and neurodevelopment: a systematic review. Pediatrics 146, e20201449 (2020).
Zanardo, V. et al. Laryngeal mask airway for neonatal resuscitation in a developing country: evaluation of an educational intervention. neonatal LMA: an educational intervention in DRC. BMC Health Serv. Res. 10, 254 (2010).
Macario, A., Chang, P. C., Stempel, D. B. & Brock-Utne, J. G. A cost analysis of the laryngeal mask airway for elective surgery in adult outpatients. Anesthesiology 83, 250–257 (1995).
Acknowledgements
The research in this manuscript was supported by the following grants: S.M. was supported by the American Academy of Pediatrics Neonatal Resuscitation Program (AAP NRP) young investigator grant 2019. P.C. was supported by the National Institutes of Health (NIH)/National Heart Lung and Blood Institute (NHLBI) K12 HL138052, National Institute of Child Health and Human Development (NICHD) R03HD096510, (NICHD) R01HD104909, AAP NRP Research grant 2020 & ZOLL Foundation grant 2020. M.R. was supported by NIH/NICHD R03 HD10406 & AAP NRP Research Grant. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH, AAP NRP, and the ZOLL Foundation.
Author information
Authors and Affiliations
Contributions
S.M. and M.R. conceptualization, methodology, data acquisition and analysis, interpretation, and writing—original draft, critically reviewing and editing. S.G., J.H., M.B., J.N., and P.C. data acquisition, extraction, critical reviewing, and editing of the manuscript. All authors approved the final version for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: Table 2 has been corrected.
Rights and permissions
About this article
Cite this article
Mani, S., Gugino, S., Helman, J. et al. Laryngeal mask ventilation with chest compression during neonatal resuscitation: randomized, non-inferiority trial in lambs. Pediatr Res 92, 671–677 (2022). https://doi.org/10.1038/s41390-021-01820-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-021-01820-z
- Springer Nature America, Inc.
This article is cited by
-
Laryngeal mask airway: an alternate option for all phases of neonatal resuscitation
Pediatric Research (2022)