Abstract
Background/objectives
Although relatively less muscle mass has been associated with greater diabetes prevalence, whether there is an association between muscle mass and diabetes prevalence independent of body fat distribution is unknown. The objective was to determine whether less skeletal muscle mass is associated with greater diabetes prevalence in young men and women independent of body fat distribution.
Subjects/methods
One thousand seven hundred and sixty-four adults, aged 20–49 years old, from the United States National Health and Nutrition Examination Survey (2005–2006). Body composition, including appendicular lean mass (ALM), was measured by dual-energy x-ray absorptiometry. Diabetes was defined as fasting blood glucose ≥7 mmol/l, 2-h blood glucose ≥11.1 mmol/l on 75 g OGTT, HbA1c ≥ 48 mmol/mol (6.5%), use of diabetes medications, or self-reported diagnosis of diabetes.
Results
The odds of diabetes were 1.31 times higher in men [OR 1.31 (1.18–1.45), p = 0.0001], and 1.24 times higher in women [OR 1.24 (1.05–1.46), p = 0.01], per percent decrease in ALM/weight after controlling for age, race, height, smoking, and education. After additionally controlling for android/gynoid fat, the odds of diabetes were 1.20 times higher per percent decrease in ALM/weight in men [OR 1.20 (1.04–1.37), p = 0.01]; an inverse association was also observed in women, albeit was not statistically significant [OR 1.08 (0.90–1.30), p = 0.42].
Conclusions
Less muscle mass was associated with greater diabetes prevalence independent of body fat distribution in young men. The association was not statistically significant in women after controlling for android and gynoid adiposity. Low muscle mass could be a causal factor in the development of type 2 diabetes or a correlated marker of higher metabolic risk.
Similar content being viewed by others

Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Although obesity is a strong risk factor for type 2 diabetes, there is significant variation in the prevalence of type 2 diabetes among adults of comparable body mass index (BMI), demonstrating the importance of risk factors other than overall body weight [1, 2]. This variable risk is partially attributable to differential adipose depots, with trunk and upper body (i.e., android) fat being more pro-inflammatory and having a more adverse cardiometabolic effect than gluteofemoral (i.e., gynoid) fat [3]. However, adipose depots alone do not fully explain differential type 2 diabetes risk. Considering that skeletal muscle is the most insulin-sensitive tissue and responsible for 70–90% of post-prandial glucose disposal [4], lower skeletal muscle mass relative to adiposity may be an important marker of type 2 diabetes risk.
Studies using data from the United States National Health and Nutrition Examination Survey (NHANES) have demonstrated that lower appendicular lean mass (ALM)/BMI is associated with greater insulin resistance in older adults [5], lower skeletal muscle index [the ratio of skeletal muscle mass (estimated by bioelectrical impedance) to total body weight] is associated with both insulin resistance and diabetes in adults younger and older than 60 years [6], and percent lean mass is negatively associated with HbA1c in both men and women without diabetes, and in men <40 years old with diabetes [7]. However, none of these studies controlled for fat deposition patterns that vary by sex and are also associated with type 2 diabetes risk, and only one of the studies performed sex-stratified analyses [7], which are critical given known sex differences in muscle and type 2 diabetes risk. In addition, data in older adults cannot be extrapolated to younger adults (i.e., under 50 years) because younger adults have not experienced age-related declines in muscle mass and generally develop type 2 diabetes at a higher BMI than older adults [8]. Finally, whether there are race-specific differences in the association between skeletal muscle mass and diabetes prevalence in young American adults is unknown. Understanding whether there are sex and race differences in the association between muscle mass and type 2 diabetes may yield important insights into known sex and race disparities in type 2 diabetes.
We hypothesized that less skeletal muscle mass is associated with greater diabetes prevalence in both young men and women in the United States, independent of body fat distribution. In a secondary analysis, we sought to determine whether less skeletal muscle is associated with greater diabetes prevalence independent of body fat distribution in Hispanics vs non-Hispanic Blacks vs non-Hispanic Whites.
Materials/subjects and methods
We analyzed cross-sectional data from the 2005–2006 NHANES, as that was the most recent year with body composition data. The survey uses a multistage, complex, stratified probability sampling design that oversamples minorities and is representative of non-institutionalized adults in the United States, providing excellent external generalizability. The survey has been conducted and managed by the Centers for Disease Control and Prevention since 1971, and its contents and procedure manuals are available online at http://www.cdc.gov/nchs/nhanes/htm. This study was exempt from local Institutional Review Board review due to the de-identified nature of the data analyzed.
We restricted our sample to individuals aged 20–49 years old. We excluded subjects who were pregnant, nursing, or status-post bilateral oophorectomy given changes in body weight, body composition, endocrine hormones, and/or type 2 diabetes risk in these groups. We also excluded subjects who were prescribed testosterone, growth hormone, or glucocorticoids given the known effects of these endocrine hormone deficiencies and/or their replacement on body composition and/or type 2 diabetes risk. Individuals whose height was >192.5 cm and/or whose weight was >136.4 kg were excluded due to limitations of the DXA table. Of 10 348 participants in NHANES 2005–2006, 1764 eligible participants were included in this analysis.
Body composition variables
All body composition measures were assessed using a DXA QDR-4500 Hologic scanner (Bedford, MA). Android and gynoid regions were defined by the Hologic APEX software used in the scan analysis. The android region is the area around the waist between the mid-point of the lumbar spine and the top of the pelvis; the gynoid area lies between the head of the femur and mid-thigh. Appendicular lean mass (ALM) was defined as the sum of the muscle mass of both legs and arms.
Covariates
The following data were self-reported using questionnaires and included in analyses because they have been independently associated with type 2 diabetes risk: age, sex, race/ethnicity [9, 10], education [11], smoking status [12], and physical inactivity [13]. Race/ethnicity was categorized as Hispanic (combining Mexican American and other Hispanic), non-Hispanic White, non-Hispanic Black, and other (including multi-racial). For race-stratified analyses, “other” was excluded. Education was categorized as (1) less than 12th grade, (2) high school graduate or General Education Diploma (GED) equivalent, or (3) higher. For race-stratified analyses, education was collapsed into two categories of (1) less than 12th grade or (2) high school graduate, GED equivalent, or higher. Smoking status was categorized as never smoker (smoked <100 cigarettes in life), former smoker (do not now smoke cigarettes), or current smoker (smoke cigarettes every day or some days). Individuals were defined as physically inactive if they reported no vigorous or moderate activity of at least 10 min over the past 30 days that caused light to heavy sweating or slight to large increases in breathing according to the standard NHANES physical activity questionnaire. This definition of physical inactivity was chosen because prospective studies in both women and men have demonstrated that any level of self-reported physical activity is associated with a lower risk of type 2 diabetes compared to no physical activity [14, 15]. In a secondary analysis, physical activity was coded according to whether the individual met the American Heart Association’s (AHA) recommendations for physical activity in adults (150 min of moderate-to-vigorous aerobic activity per week, or 75 min of vigorous aerobic activity per week) [16]. An electronic digital scale, calibrated in kilograms, was used to assess weight, and a stadiometer was used to measure height after a deep inhalation. BMI was calculated as weight in kilograms divided by height in meters squared. Height was included as a covariate in analyses given the linear relationship between height and muscle mass [17].
Diabetes variables
All techniques in NHANES followed the guidelines put forth by the American Diabetes Association. A fasting glucose blood test was performed in the morning after a 9-hour fast; subsequently, a 75-g oral glucose tolerance test (OGTT) was performed. Exclusion criteria for oral glucose tolerance testing included hemophilia or chemotherapy safety exclusions, fasting <9 h, taking insulin or oral medications for diabetes, refusing phlebotomy, and not drinking the entire Trutol™ solution within the allotted time. Plasma glucose was measured using a hexokinase method (Roche/Hitachi 911), and samples were processed, stored, and shipped to Fairview Medical Center Laboratory at the University of Minnesota for analysis. Glycosylated hemoglobin (HbA1c) was measured using HPLC (Tosoh Medics, Inc., San Francisco, CA). HbA1c samples were processed, stored, and shipped to the Diabetes Laboratory at the University of Minnesota for analysis.
Diabetes was defined by the presence of one or more of the following conditions: (1) HbA1c ≥ 48 mmol/mol (6.5%); (2) fasting glucose ≥7 mmol/l (126 mg/dl); (3) a 2-h glucose on an OGTT of ≥11.1 mmol/liter (200 mg/dl); (4) self-reported diagnosis of diabetes; or (4) self-reported use of diabetes medications (oral hypoglycemic agents and/or insulin) as previously defined [18]. Because 73 subjects were missing HbA1c, fasting glucose, and 2-h glucose on OGTT, diabetes was defined as self-reported diagnosis or self-reported use of diabetes medications in a secondary analysis.
Statistical analysis
All data were downloaded, merged according to NHANES guidelines, and analyzed incorporating sampling weights, primary sampling units, and strata as supplied by NHANES. Continuous variables are represented as mean ± SD and categorical variables as count (percent).
To determine sex-stratified differences in clinical characteristics between those with and without diabetes, we performed Wilcoxon tests to compare continuous variables, Chi-squared tests to compare categorical variables, and Fisher’s exact tests to compare categorical variables if count <10 in either cell.
To determine the association between percent ALM/weight (primary predictor) and diabetes prevalence (primary outcome), we performed a series of multivariate logistic regression models: (1) a sex-stratified model (primary hypothesis), (2) a sex-combined model that included an interaction term for percent ALM/weight with sex, and (3) a sex- and race-stratified model. We adjusted for demographic covariates (i.e., age, sex, race, smoking, and education) known to be associated with type 2 diabetes risk, height, and physical inactivity, as well as the ratio of android/gynoid fat because it varies by sex and because android fat is detrimental towards, whereas gynoid fat is protective against, type 2 diabetes. A power calculation for the primary endpoints was as follows: in a sample of 1764 participants, assuming alpha = 0.05, power = 0.8, and a diabetes prevalence of 5%, we could detect an odds ratio of 1.15 between ALM/weight below the median vs ALM/weight above the median. Data are presented as an odds ratio with 95% confidence interval and associated p value for each model.
We used SAS (version 9.2 or 9.3; SAS Institute, Cary, NC) for all analyses and applied procedures to account for NHANES 2005–2006 sampling probabilities and complex sampling design in all models. Multiple imputations was applied to address potential bias resulting from nonrandom missing DXA data [19]. Five complete data files that contained both the non-missing and imputed values (generated using sequential multivariate imputation) were created. A two-sided p value ≤0.05 was considered a statistically significant test of the hypothesis that less skeletal muscle mass is associated with greater diabetes prevalence independent of body fat distribution in both young men and young women in the United States.
Results
Clinical characteristics
The clinical characteristics of 958 men and 806 women in our study sample, by diabetes status, are listed in Table 1. Diabetes was present in 5.2% of men and 5.1% of women, consistent with the young age of this population. Individuals with diabetes were older, had higher BMI, lower ALM/weight, higher android/gynoid fat, and were more physically inactive than those without diabetes. Using AHA guidelines, 77% of women with diabetes (n = 27), 63% of women without diabetes (n = 293), 63% of men with diabetes (n = 26), and 55% of men without diabetes (n = 345) did not meet recommended physical activity guidelines. In men, there was a larger proportion of Hispanic adults, and a smaller proportion of non-Hispanic White adults, with diabetes than without diabetes. In women, there was a larger proportion of the less than 12th grade education category, and a smaller proportion of the more than 12th grade education category, among those with diabetes compared to those without diabetes. Correlations between ALM/weight and anthropometric or DXA-derived adiposity measures are provided in Table 2.
Association between skeletal muscle mass and diabetes prevalence
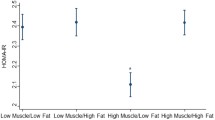
The odds of diabetes were 1.31 times higher in men [OR 1.31 (1.18–1.45), p = 0.0001], and 1.24 times higher in women [OR 1.24 (1.05–1.46), p = 0.01], per percent decrease in ALM/weight after controlling for age, race, height, smoking, and education (Fig. 1). After additionally controlling for android/gynoid fat, the odds of diabetes were 1.20 times higher per percent decrease in ALM/weight in men [OR 1.20 (1.04–1.37), p = 0.01]; an inverse association was also observed in women, albeit was not statistically significant [OR 1.08 (0.90–1.30), p = 0.42] (Fig. 1). After additionally controlling for physical inactivity, the odds of diabetes were 1.18 times higher per percent decrease in ALM/weight in men [OR 1.18 (1.02–1.37), p = 0.02]; an inverse association was also observed in women, albeit was not statistically significant [OR 1.07 (0.87–1.31), p = 0.54] (Fig. 1). If physical activity was instead categorized according to AHA-recommended physical activity guidelines, the results were comparable. In men, the odds of diabetes were 1.19 times higher per percent decrease in ALM/weight in men [OR 1.19 (1.01–1.39), p = 0.04]. In women, an inverse association was also observed, albeit was not statistically significant [OR 1.13 (0.99–1.31), p = 0.095).
After controlling for android/gynoid (A/G) fat, the odds of diabetes were 1.20 times higher for each percent decrease in ALM/weight in men an inverse association was also observed in women albeit was not statistically significant. After additionally controlling for physical inactivity (PA), the odds of diabetes were 1.18 times higher for each percent decrease in ALM/weight in men an inverse association was also observed in women albeit was not statistically significant.
In a sex-combined interaction model that included age, race, height, smoking, and education, the odds of diabetes were 1.26 times higher per percent decrease in ALM/weight [OR 1.26 (1.14–1.39), p = 0.0001] (Fig. 1). After additionally controlling for android/gynoid fat, the odds of diabetes were 1.12 times higher per percent decrease in ALM/weight [OR 1.12 (1.01–1.25), p = 0.04]. After additionally controlling for physical inactivity, an inverse association was also observed albeit was not statistically significant [OR 1.11 (0.98–1.25), p = 0.10]. If physical activity was instead categorized according to AHA recommended physical activity guidelines, the results were similar. The odds of diabetes were 1.14 times higher per percent decrease in ALM/weight [OR 1.14 (1.05–1.24), p = 0.003]. There was no statistically significant sex interaction in any of these models.
When the diabetes definition was changed to diabetes by self-report only so as not to introduce bias for or against subjects with missing biochemical data, effect estimates were similar, although not statistically significant, likely owing to the smaller number of diabetes cases.
Racial differences in the association between skeletal muscle mass and diabetes prevalence
The odds of diabetes trended higher per percent decrease in ALM/weight in non-Hispanic White men after controlling for age, height, smoking, education, and android/gynoid fat [OR 1.30 (0.97–1.73), p = 0.08]. In non-Hispanic Black men, Hispanic men, non-Hispanic White women, non-Hispanic Black women, and Hispanic women, the association was not significant.
Discussion
We demonstrated that less skeletal muscle mass is associated with higher odds of prevalent diabetes independent of body fat distribution in young men. For example, a 91 kg man with 28.2 kg of muscle (31% ALM/weight) had a 1.20-times higher odds of diabetes than a 91 kg man with 29.1 kg of muscle (32% ALM/weight). We add to the literature by demonstrating that this association is independent of body fat distribution, which is known to be associated with type 2 diabetes risk. However, the association was not statistically significant among women after controlling for android and gynoid adiposity. Whether low muscle mass is a causal factor in the development of type 2 diabetes, or whether it is simply a marker of higher metabolic risk, requires further investigation.
Our data are in agreement with previous sex-stratified cross-sectional studies, which reported that relatively lower muscle mass is associated with insulin resistance [20] and diabetes [7] in men, but not women. We add to the literature by demonstrating that this association between muscle mass and diabetes prevalence remains significant in men after controlling for android and gynoid adiposity. Although there is no consensus about the best biomarker to capture the association between fat distribution and type 2 diabetes risk, and we demonstrated a widespread correlation between adiposity measures, we chose the android/gynoid fat ratio for our models because it makes a distinction between android fat, which confers increased risk for type 2 diabetes, and gynoid fat, which may be protective against type 2 diabetes [17, 21, 22]. In contrast, other adiposity measures do not distinguish between different fat depots with differential risks for diabetes and whose distribution varies by sex. In contrast to the results we report in men, we found that the association between skeletal muscle mass and diabetes prevalence was not statistically significant in women after controlling for android and gynoid adiposity. We may have had more power to detect an association in men because men have more muscle mass and higher type 2 diabetes prevalence than BMI- and age-matched women [23]. Alternatively, since women have less muscle mass, but more gynoid fat, than men, perhaps muscle is a less important, while gynoid fat is a more important, depot in women to protect against diabetes risk compared to men. In addition, the exploratory race-stratified analysis was limited by small numbers, making it difficult to draw conclusions from the data. Further studies are warranted to determine whether relationships between muscle mass and diabetes are influenced by sex and race.
One strength of this cross-sectional study is that it uses a more comprehensive definition of diabetes that includes both self-reported and biochemical data, whereas previous studies investigating the association between muscle mass and prevalent diabetes did not include HbA1c and/or OGTT data [6, 7]. As expected, this resulted in a higher prevalence of diabetes than reported by the 2005–2006 National Health Interview Survey (2.4–2.7% among U.S. adults aged 18–44 years), which only defined diabetes by self-report, and thus did not include adults unaware of their diabetes diagnosis [24]. One limitation of this study is that correlation in a cross-sectional study does not prove causation. Since ALM/weight is correlated with multiple adiposity measures, the association between ALM/weight and type 2 diabetes could be subject to residual confounding by adiposity even after controlling for android/gynoid fat. Previous prospective studies in middle-aged and older adults have produced discordant results with respect to the association between muscle mass and incident diabetes [25,26,27], and other studies have demonstrated that type 2 diabetes is a risk factor for muscle loss with aging [28]. Since muscle and fat mass are correlated both cross-sectionally and longitudinally, interventions targeting muscle mass in the setting of randomized, placebo-controlled trials are needed in order to determine whether low muscle mass is a type 2 diabetes risk factor independent of adiposity. In addition, since we restricted our study to adults aged 20–49 years old, the results of this study may not be generalizable to older adults, who lose muscle mass with aging and have a higher prevalence of diabetes.
In summary, we demonstrate that less skeletal muscle mass independent of android and gynoid adiposity is associated with higher diabetes prevalence in young men. The association was not statistically significant in women after controlling for body fat distribution. Further study is needed to determine whether low muscle mass itself is a causal factor in the development of type 2 diabetes, or whether it is simply a marker of higher metabolic risk.
Data availability
All data generated and analyzed during this study are available in the following repository: National Health and Nutrition Examination Survey, https://www.cdc.gov/nchs/nhanes/index.htm.
References
Stefan N, Haring HU, Hu FB, Schulze MB. Metabolically healthy obesity: epidemiology, mechanisms, and clinical implications. Lancet Diabetes Endocrinol. 2013;1:152–62.
Abstracts of the 8th International Conference on Cachexia, Sarcopenia and Muscle Wasting, Paris, France, 4–6 December 2015. J Cachexia Sarcopenia Muscle. 2015;6:398–509.
Karpe F, Pinnick KE. Biology of upper-body and lower-body adipose tissue–link to whole-body phenotypes. Nat Rev Endocrinol. 2015;11:90–100.
Baron AD, Brechtel G, Wallace P, Edelman SV. Rates and tissue sites of non-insulin- and insulin-mediated glucose uptake in humans. Am J Physiol. 1988;255:E769–74.
Batsis JA, Mackenzie TA, Jones JD, Lopez-Jimenez F, Bartels SJ. Sarcopenia, sarcopenic obesity and inflammation: results from the 1999-2004 National Health and Nutrition Examination Survey. Clin Nutr. 2016;35:1472–83.
Srikanthan P, Hevener AL, Karlamangla AS. Sarcopenia exacerbates obesity-associated insulin resistance and dysglycemia: findings from the National Health and Nutrition Examination Survey III. PLoS ONE. 2010;5:e10805.
Bower JK, Meadows RJ, Foster MC, Foraker RE, Shoben AB. The association of percent body fat and lean mass with HbA1c in US adults. J Endocr Soc. 2017;1:600–8.
Hillier TA, Pedula KL. Characteristics of an adult population with newly diagnosed type 2 diabetes: the relation of obesity and age of onset. Diabetes Care. 2001;24:1522–7.
Cowie CC, Rust KF, Ford ES, Eberhardt MS, Byrd-Holt DD, Li C, et al. Full accounting of diabetes and pre-diabetes in the U.S. population in 1988-1994 and 2005-2006. Diabetes Care. 2009;32:287–94.
Shai I, Jiang R, Manson JE, Stampfer MJ, Willett WC, Colditz GA, et al. Ethnicity, obesity, and risk of type 2 diabetes in women: a 20-year follow-up study. Diabetes Care. 2006;29:1585–90.
Maty SC, Everson-Rose SA, Haan MN, Raghunathan TE, Kaplan GA. Education, income, occupation, and the 34-year incidence (1965-99) of Type 2 diabetes in the Alameda County Study. Int J Epidemiol. 2005;34:1274–81.
Maddatu J, Anderson-Baucum E, Evans-Molina C. Smoking and the risk of type 2 diabetes. Transl Res. 2017;184:101–7.
O’Donovan G, Hillsdon M, Ukoumunne OC, Stamatakis E, Hamer M. Objectively measured physical activity, cardiorespiratory fitness and cardiometabolic risk factors in the Health Survey for England. Prev Med. 2013;57:201–5.
Manson JE, Rimm EB, Stampfer MJ, Colditz GA, Willett WC, Krolewski AS, et al. Physical activity and incidence of non-insulin-dependent diabetes mellitus in women. Lancet. 1991;338:774–8.
Wannamethee SG, Shaper AG, Alberti KG. Physical activity, metabolic factors, and the incidence of coronary heart disease and type 2 diabetes. Arch Intern Med. 2000;160:2108–16.
Arnett DK, Blumenthal RS, Albert MA, Buroker AB, Goldberger ZD, Hahn EJ, et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;140:e596–646.
Janssen I, Heymsfield SB, Wang ZM, Ross R. Skeletal muscle mass and distribution in 468 men and women aged 18-88 yr. J Appl Physiol. 2000;89:81–8.
Srikanthan P, Karlamangla AS. Relative muscle mass is inversely associated with insulin resistance and prediabetes. Findings from the third National Health and Nutrition Examination Survey. J Clin Endocrinol Metab. 2011;96:2898–903.
Centers for Disease Control and Prevention (CDC) NCHS & US Department of Health and Human Services. NHANES: technical documentation for the 1999-2004 dual energy x-ray absorptiometry (DXA) multiple imputation data files. 2008. https://wwwn.cdc.gov/Nchs/data/nhanes/dxa/dxa_techdoc.pdf. Access on 22 Oct 2021.
Schorr M, Dichtel LE, Gerweck AV, Valera RD, Torriani M, Miller KK, et al. Sex differences in body composition and association with cardiometabolic risk. Biol Sex Differ. 2018;9:28.
Azuma K, Heilbronn LK, Albu JB, Smith SR, Ravussin E, Kelley DE, et al. Adipose tissue distribution in relation to insulin resistance in type 2 diabetes mellitus. Am J Physiol Endocrinol Metab. 2007;293:E435–42.
Snijder MB, Visser M, Dekker JM, Goodpaster BH, Harris TB, Kritchevsky SB, et al. Low subcutaneous thigh fat is a risk factor for unfavourable glucose and lipid levels, independently of high abdominal fat. The Health ABC Study. Diabetologia. 2005;48:301–8.
Logue J, Walker JJ, Colhoun HM, Leese GP, Lindsay RS, McKnight JA, et al. Do men develop type 2 diabetes at lower body mass indices than women? Diabetologia. 2011;54:3003–6.
Center for Disease Control. Diabetes Atlas. https://gis.cdc.gov/grasp/diabetes/DiabetesAtlas.html#. 2005–2006. Access on 10 Mar 2022.
Li JJ, Wittert GA, Vincent A, Atlantis E, Shi Z, Appleton SL, et al. Muscle grip strength predicts incident type 2 diabetes: population-based cohort study. Metab Clin Exp. 2016;65:883–92.
Larsen BA, Wassel CL, Kritchevsky SB, Strotmeyer ES, Criqui MH, Kanaya AM, et al. Association of muscle mass, area, and strength with incident diabetes in older adults: The Health ABC Study. J Clin Endocrinol Metab. 2016;101:1847–55.
Kalyani RR, Metter EJ, Xue QL, Egan JM, Chia CW, Studenski S, et al. The relationship of lean body mass with aging to the development of diabetes. J Endocr Soc. 2020;4:bvaa043.
Park SW, Goodpaster BH, Strotmeyer ES, Kuller LH, Broudeau R, Kammerer C, et al. Accelerated loss of skeletal muscle strength in older adults with type 2 diabetes: the health, aging, and body composition study. Diabetes Care. 2007;30:1507–12.
Funding
MSH is supported by K23DK115903. KKM is supported by K24HL092902. AL is supported by Grant 2020096 from the Doris Duke Charitable Foundation. JBM is supported by UM1DK078616 and R01HL151855.
Author information
Authors and Affiliations
Contributions
MSH, AL, JBM, and KKM contributed to the conception, design, and acquisition of data. All authors made substantial contributions to the analysis and interpretation of data, drafting of the article and revising it critically for important intellectual content, and gave approval of the version to be published. MSH is the guarantor of this work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Haines, M.S., Leong, A., Porneala, B.C. et al. Association between muscle mass and diabetes prevalence independent of body fat distribution in adults under 50 years old. Nutr. Diabetes 12, 29 (2022). https://doi.org/10.1038/s41387-022-00204-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41387-022-00204-4
- Springer Nature Limited
This article is cited by
-
A multifaceted and inclusive methodology for the detection of sarcopenia in patients undergoing bariatric surgery: an in-depth analysis of current evidence
Reviews in Endocrine and Metabolic Disorders (2024)
-
Association between depression and endometriosis using data from NHANES 2005–2006
Scientific Reports (2023)
-
Epidemiological, mechanistic, and practical bases for assessment of cardiorespiratory fitness and muscle status in adults in healthcare settings
European Journal of Applied Physiology (2023)
-
The Effects of Sodium–Glucose Cotransporter 2 Inhibitors on Body Composition in Type 2 Diabetes Mellitus: A Narrative Review
Diabetes Therapy (2023)