Abstract
While pharmacological, behavioral and psychosocial treatments are available for substance use disorders (SUDs), they are not always effective or well-tolerated. Neuromodulation (NM) methods, including repetitive transcranial magnetic stimulation (rTMS), transcranial direct current stimulation (tDCS) and deep brain stimulation (DBS) may address SUDs by targeting addiction neurocircuitry. We evaluated the efficacy of NM to improve behavioral outcomes in SUDs. A systematic literature search was performed on MEDLINE, PsychINFO, and PubMed databases and a list of search terms for four key concepts (SUD, rTMS, tDCS, DBS) was applied. Ninety-four studies were identified that examined the effects of rTMS, tDCS, and DBS on substance use outcomes (e.g., craving, consumption, and relapse) amongst individuals with SUDs including alcohol, tobacco, cannabis, stimulants, and opioids. Meta-analyses were performed for alcohol and tobacco studies using rTMS and tDCS. We found that rTMS reduced substance use and craving, as indicated by medium to large effect sizes (Hedge’s g > 0.5). Results were most encouraging when multiple stimulation sessions were applied, and the left dorsolateral prefrontal cortex (DLPFC) was targeted. tDCS also produced medium effect sizes for drug use and craving, though they were highly variable and less robust than rTMS; right anodal DLPFC stimulation appeared to be most efficacious. DBS studies were typically small, uncontrolled studies, but showed promise in reducing misuse of multiple substances. NM may be promising for the treatment of SUDs. Future studies should determine underlying neural mechanisms of NM, and further evaluate extended treatment durations, accelerated administration protocols and long-term outcomes with biochemical verification of substance use.
Similar content being viewed by others
Introduction
Substance use disorders (SUDs) account for 500,000 deaths annually in the U.S alone [1, 2]. Moreover, SUDs frequently co-occur with psychiatric disorders, including schizophrenia and mood disorders [3,4,5]. Although there are validated pharmacologic and psychotherapeutic treatments available for SUDs, relapse rates are high [6, 7]. Thus, development of neuroscience-informed therapeutics for SUDs is critical. Neuromodulation (NM) may offer such opportunities [8, 9].
Reinforcing effects of substances are primarily mediated by mesocorticolimbic systems, which include midbrain dopamine (DA) projections to prefrontal cortex (PFC) and ventral striatum [nucleus accumbens (NAc)] [10, 11]. Substance misuse is associated with mesolimbic hypodopaminergia [12], and dysfunction of dorsolateral prefrontal cortex (DLPFC) and dorsal anterior cingulate cortex (dACC), which are involved in decision-making and self-control. Moreover, the ventral PFC, including the orbitofrontal cortex (OFC) and ventral anterior cingulate cortex (vACC), is involved in limbic arousal and emotional processing [13]. Hence, dysfunction in these systems has been associated with SUDs [14]. Furthermore, left DLPFC mediates reward-based motivation, while right DLPFC is involved in withdrawal-related behaviors and inhibition [15]. Thus, use of NM to stimulate right DLPFC may strengthen executive functions by inhibiting the left DLPFC to counterbalance hemispheric imbalance, which may contribute to reduction of substance consumption and craving [16, 17]. Invasive and/or non-invasive NM may be promising brain-based approaches since they modulate SUD-related mesolimbocortical circuitry [8, 9, 18]. Such interventions include repetitive transcranial magnetic stimulation (rTMS), transcranial direct current stimulation (tDCS), and deep brain stimulation (DBS).
Repetitive transcranial magnetic stimulation (rTMS)
rTMS is a non-invasive NM technique that has shown utility for neurological and psychiatric disorders [19]. Application of alternating magnetic fields to the scalp through a copper wire induces temporary electrical currents and modulates cortical excitability in localized brain tissue [20] (Fig. 1a). Numerous studies have demonstrated enduring functional and structural neuroplastic changes in target regions [21, 22], and increased DA release in the mesolimbic system [23,24,25,26].
Stimulation parameters vary significantly with respect to stimulus intensity, frequency and total number of pulses, which can produce differential effects [27]. Typically, low frequency (LF; ≤1 Hz) stimulation produces local inhibitory effects while high frequency (HF; ≥5 Hz) stimulation produces local excitatory effects on motor cortex [28, 29]. rTMS primarily alters motor cortical excitability and inhibition, with indirect effects on craving or motivation. Frequency-dependent rTMS effects on regional brain activity may have implications for clinical therapeutics in neuropsychiatric disorders [30, 31]. Coil type can also modulate effects; while traditional TMS employs a figure-8 coil design and can only reach depths of 0.7 cm, deep TMS, wherein a three-dimensional H-coil helmet design is used, can stimulate a deeper and broader brain area, reaching a depth of 3.2 cm [32].
Two robust rTMS adaptations have emerged wherein bursts of magnetic pulses, referred to as theta burst stimulation (TBS), are applied. In intermittent theta burst stimulation (iTBS), a two second train of TBS bursts is repeated every ten seconds, inducing long-term potentiation and cortical excitability [33, 34]. Contrastingly, continuous theta-burst stimulation (cTBS) applies trains of uninterrupted TBS bursts and induces long-term depression and inhibitory effects [34].
rTMS appears safe when administered according to recommended guidelines [35]. There is little risk beyond local discomfort at the site of stimulation and other minor side effects (e.g. mild headache, dizziness) [36]. Importantly, a deep-TMS system was recently cleared by the Food and Drug Administration (FDA) for smoking cessation [37]. However, long-term effects of repeated rTMS sessions are unknown [38].
Transcranial direct current stimulation (tDCS)
Using two or more electrodes (i.e., anodal, cathodal), tDCS delivers a low intensity current (0.5–2.0 milliamps [mA]) to a targeted brain region for several minutes (Fig. 1b). This allows for polarity-dependent modulation of the neuronal resting membrane potential and cortical excitability. Cathodal current decreases while anodal current increases cortical excitability [39, 40]. Similar to rTMS, tDCS protocols can vary with respect to numerous parameters such as current strength, electrode size and placement, stimulation duration and frequency [41].
tDCS is an accessible, low-cost stimulation method that is well-tolerated, though minor side effects such as scalp irritation are reported [42]. Similar to rTMS, tDCS has been used to effectively treat neuropsychiatric conditions such as Parkinson’s disease, chronic pain, and major depression [43]. Although underlying mechanisms for tDCS are not fully understood, induction of neurochemical changes in targeted brain tissue is being investigated for SUD treatment.
Deep brain stimulation (DBS)
DBS is an invasive NM technique used to treat Alzheimer’s disease, Parkinson’s disease, and obsessive compulsive disorder [44]. It involves a neurosurgical procedure wherein implanted electrodes deliver electrical pulses directly to targeted brain regions, which modulates neural circuitry and subsequently alters neuroplasticity (Fig. 1c). While rTMS and tDCS use lower frequencies to induce excitation or inhibition of neurons, DBS blocks neural transmission with high-frequency stimulation [45]. Implanted electrodes are connected to an implantable pulse generator placed under the skin of the chest wall, allowing for continuous stimulation at a pre-set frequency [46]. Thus, stimulation parameters can be modulated as a patient’s condition changes.
Unlike other surgical interventions, DBS does not damage brain tissue [47], but given its invasive nature, is associated with infection, seizures or stroke. DBS is well-tolerated once the patient has recovered from the primary surgical procedure [48]. Focal stimulation of deep brain regions involved in addiction neurocircuitry (e.g. NAc) may facilitate SUD treatment.
We conducted a systematic review and meta-analysis to determine the efficacy of NM for improving addiction outcomes (e.g., drug craving, consumption, and relapse). As significant progress has been made in this area, a systematic review and meta-analysis building on previous narrative reviews [8, 9] with quantification of NM effects in SUDs is warranted.
Methods
Search strategy
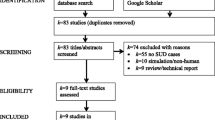
A comprehensive literature search by two authors (D.M. and A.P., trained on Covidence) was conducted using Medline, PubMed and PsycINFO databases, in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [49] (Supplementary Fig. 1), through October, 2023. Articles published after 2000 in peer-reviewed journals were considered. A list of keywords and search terms for four key concepts (SUD, rTMS, tDCS, DBS) was applied (See Supplementary Table 1 for Search Strategy). Reference lists of relevant reviews were also screened for applicable articles. The review was registered at PROSPERO (CRD42023475165).
Eligibility criteria
Using PICOS [50], studies were included if they satisfied the following criteria – Population (P): Studies recruiting participants (18+ years of age) diagnosed with SUD/dependence of alcohol, tobacco, cocaine, methamphetamine, opioids, or cannabis, according to standardized criteria (e.g., DSM-IV or DSM-5); Intervention (I): Intervention employing either rTMS, tDCS, or DBS; Comparison (C): Studies including either sham stimulation, a control group receiving no intervention or an active control arm were included. DBS studies were exempted considering the ethical constraints on the use of control groups with invasive brain surgery/stimulation; Outcomes (O): Studies investigating substance-related outcomes (consumption, craving, cue-induced craving, abstinence, relapse) as primary or secondary outcomes of interest using a validated measurement tool (e.g. Obsessive Compulsive Drinking Scale [OCDS]); Study Design (S): Studies employing either a parallel (between-subject) or cross-over (within-subject) randomized controlled trial (RCT). For DBS, case series (N ≥ 2) were permitted.
Studies were excluded if: (1) recruited participants without a SUD and/or a standardized criteria for diagnosis (e.g., “heavy drinkers”); (2) lacked a well-defined control group (rTMS and tDCS studies); (3) literature review, meta-analysis, dissertation, abstract, conference presentation or case report.
Study selection
Two authors (D.M. and A.P.) independently screened titles and abstracts obtained on Covidence to determine eligibility for full-text review, and subsequently reviewed the full text of the screened studies. Disagreements were resolved by consensus, and review with the senior author (T.P.G.).
Data extraction and risk of bias
For included studies, two authors (D.M. and A.P.) extracted author information, sample size, study design, stimulation parameters, primary substance use outcomes (craving and consumption), and any secondary outcomes. Effect sizes (Hedge’s g) of substance use and other outcomes were calculated for each study using post-treatment data of active and control (sham and/or no treatment) groups, respectively (see Tables 1–4). Due to the heterogeneity in follow-up periods across studies, treatment effects were determined using end-of-treatment data, unless otherwise stated. For DBS studies with no control conditions, within-subject (pre-post treatment) effect sizes were calculated.
The Cochrane Risk-of-Bias Tool (RoB-2) [51] assessed quality of included RCTs. Studies with a high risk of bias were subsequently excluded if at least four domains were considered of moderate risk, or if two or more domains were flagged as high risk. The Risk Of Bias In Non-randomized Studies of Interventions (ROBINS-I) [52] tool assessed risk of bias in non-randomized studies (DBS Studies); all extracted DBS studies were included in this review.
Meta-analysis
To quantify NM effects, we performed meta-analyses on rTMS and tDCS studies investigating alcohol and tobacco use disorders. Acute versus repeated stimulation were independently evaluated. Meta-analyses were conducted when three or more studies evaluated a synonymous outcome (craving, cue-induced craving, and/or consumption).
We utilized standardized mean difference (SMD; Hedge’s g) with 95% confidence intervals (CI’s) in each selected meta-analysis to calculate the effect size of NM-related changes in alcohol and tobacco craving, cue-induced craving, and/or consumption (p ≤ 0.05, two-tailed). Random-effects models pooled individual SMDs, and used data from studies that reported end-of-treatment substance use data from active and control treatment arms. Negative values indicated that active stimulation produced greater reductions in craving, cue-induced craving, and/or consumption compared to sham treatment. The I2 statistic estimated between-trial heterogeneity; I2 of ≤40% was considered low heterogeneity, 40–60% moderate heterogeneity, and >60% high heterogeneity [53]. Meta-analyses were performed using R version 4.3.1 [54] with package metafor [55].
Results
We identified a total of 94 studies that met our inclusion criteria, with a total of 4306 participants.
Repetitive transcranial magnetic stimulation (rTMS)
Fifty-one studies investigating rTMS as treatment for SUDs were identified, with 2406 participants receiving either active or control treatment (sham stimulation or no treatment; Table 1).
Alcohol
Sixteen studies [56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71] investigated the effects of rTMS for alcohol use disorder (AUD). Eleven studies used multiple active sessions (10–20 sessions) with HF stimulation (10–20 Hz) targeting right, left, or bilateral dorsolateral prefrontal cortex (DLPFC), medial prefrontal cortex (mPFC) or insula [61,62,63,64,65,66,67,68,69,70,71]. Findings were mixed, with seven studies [61, 63,64,65, 68, 71] demonstrating significant post-TMS reductions in alcohol craving and/or consumption compared to sham stimulation. Notably, 3/7 positive studies applied deep TMS using various H-coils as opposed to the traditional Figure-8 coil, suggesting that this technology may be particularly efficacious in treating AUD. Two studies employed the H-1 coil to target the mPFC and bilateral DLPFC, respectively, whilst one opted for the H-7 coil to target both the mPFC and anterior cingulate cortex (ACC) concurrently. One study [71] applied a 10 session cTBS stimulation protocol to the mPFC, with significant reductions in alcohol craving.
Of these eleven studies, ten were combined in a meta-analysis to determine the effects of repeated rTMS stimulation on alcohol craving (n = 447). Active rTMS significantly reduced craving scores in AUD compared to sham (SMD = −1.25, 95% CI: −2.34 to −0.15, p = 0.02, I2 = 95.8%; Fig. 2B). Similarly, meta-analysis of five repeated rTMS trials (n = 184) demonstrated that multiple rTMS sessions produced greater reductions in alcohol consumption than sham (SMD = −1.39, 95% CI: −2.37 to −0.41, p < 0.01, I2 = 86.2%; Fig. 2C).
Five studies [56,57,58,59,60] evaluated the effects of a single active (10–20 Hz) stimulation session and found no significant improvements in alcohol craving or consumption post-TMS when compared to sham. Four studies [56,57,58, 60] targeted the right DLPFC, while one [59] targeted the left frontal pole with cTBS. Accordingly, meta-analysis of craving outcomes in these five trials (n = 149) revealed that acute active versus sham rTMS did not significantly decrease craving (SMD = −0.16, 95% CI: −0.42 to 0.09, p = 0.21, I2 = 0%; Fig. 2A).
Tobacco
Sixteen studies [37, 72,73,74,75,76,77,78,79,80,81,82,83,84,85,86] examined efficacy of rTMS for tobacco use disorder (TUD). All studies demonstrated reductions in tobacco craving/cue-induced craving and/or cigarette consumption following active versus sham rTMS, with the exception of Li et al. [72] and Kozak et al. [73]. While Li et al. applied a single 10 Hz stimulation session targeting the left DLPFC, Kozak et al. [73] tested multiple HF sessions (20 Hz) targeting the bilateral DLPFC in individuals with comorbid schizophrenia (SCZ). However, Moeller et al. [85] applied deep-TMS to the PFC and insula using the H-4 coil in nicotine-dependent SCZ patients and found that active stimulation increased the latency to smoke, suggesting reduced motivation. Similarly, Ibrahim and colleagues [86] applied multiple sessions of active versus sham deep TMS to insular cortex in smokers receiving concurrent varenicline treatment, and found significant rTMS-related effects in smoking abstinence at Week 12.
Dinur-Klein et al. [84] and Zangen et al. [37] also applied deep-TMS to the lateral PFC and insula using the H-ADD and H-4 coils respectively, and found significant reductions in tobacco consumption and craving [37, 84]. Importantly, Dinur-Klein et al. [84] applied both 1 Hz (LF) and 10 Hz (HF) stimulation to the lateral prefrontal cortex (PFC) and insula, finding that cigarette consumption decreased significantly only in the 10 Hz condition. These studies were amongst the largest studies of NM for SUDs, with sample sizes of 115 and 262 respectively. The study by Zangen et al. [37] is the only multisite clinical trial in the addiction NM field, and led to FDA clearance of the H-4 coil for smoking cessation.
Notably, while Trojak et al. [81] reported positive results, findings were not maintained at follow-up (12 weeks), signifying a lack of durability in long-term outcomes, though this was the only study to apply LF stimulation (1 Hz) exclusively.
Additionally, two studies [77, 84] investigated cue-induced provocation prior to stimulation, and found that presentation of smoking cues reduced cigarette consumption and cue-induced craving, respectively.
Meta-analyses were performed on acute and repeated rTMS for TUD. Of four single-session rTMS studies, three reported cue-induced craving (n = 40) and were subsequently evaluated, indicating no significant effect of a single active versus sham stimulation session (SMD = −0.95, 95% CI: −2.30 to 0.41, p = 0.17, I2 = 87.4%; Fig. 3A). Of twelve multi-session studies, six reported tobacco consumption (n = 342) and eight reported subjective craving (n = 593). While repeated rTMS significantly reduced cigarette use (SMD = −1.65, 95% CI: −3.00 to −0.30, p = 0.01, I2 = 95.1%; Fig. 3C), there was no significant effect of active versus sham stimulation on craving (SMD = −0.86, 95% CI: −1.80 to 0.08, p = 0.07, I2 = 94.8%; Fig. 3B).
Cannabis
Only two RCTs [87, 88] examined the use of rTMS for cannabis use disorder (CUD). Sahlem et al. [87] used a randomized, sham-controlled, crossover design to investigate therapeutic effects of a single 10 Hz stimulation session applied to left DLPFC, finding no significant differences in cannabis craving compared to sham. Kozak-Bidzinski et al. [88] applied 20 sessions of 20 Hz rTMS to bilateral DLPFC using a parallel groups design in participants with CUD and schizophrenia. Non-significant reductions in cannabis consumption were noted post-TMS versus sham (60 versus 5%), and trends towards reductions in urine toxicology (carboxy-tetrahydrocannabinol) and craving were observed.
Cocaine
Six studies [59, 89,90,91,92,93] investigated rTMS for cocaine use disorder. Two studies demonstrated a significant decrease in cocaine craving following multiple sessions of 15 Hz rTMS to the left DLPFC. Martinez et al. [91] applied both 1 Hz and 10 Hz stimulation to mPFC and ACC using the H-7 coil, finding no significant effect on cocaine craving, though a reduction in cocaine self-administration was present in the 10 Hz condition versus 1 Hz rTMS and sham. Conversely, Bolloni et al. [89] found no significant effects of deep TMS on cocaine consumption when targeting the PFC with H-1 coil, though there was a trend for decreased consumption between baseline and 6-months post-TMS in the active group. Hanlon et al. [59] applied a single stimulation session, finding no treatment-related effects on craving following cTBS to the left frontal pole.
Methamphetamine
Eight studies [94,95,96,97,98,99,100,101] investigated the use of rTMS for methamphetamine (MA) use disorder. Seven studies [95,96,97,98,99,100,101] exhibited significant improvements in MA unconditioned and cue-induced craving and/or consumption following multiple active rTMS sessions (5–20) targeting the left DLPFC or left PFC (1–10 Hz), compared to sham treatment. Interestingly, Li et al. [94] found that a single 1 Hz stimulation session applied to the left DLPFC increased cue-induced MA craving compared to sham. Notably, three studies [98,99,100] adopted iTBS and/or cTBS stimulation parameters and reported positive results consistent with standard rTMS.
Opioids
Four studies [102,103,104,105] evaluated outcomes in opioid use disorder (OUD) patients following multiple HF rTMS sessions (5–40) targeting the left DLPFC. Three studies [103,104,105] reported significant improvements in opioid craving and/or cue-induced craving, with the exception of Tsai et al. [102] who evaluated treatment effects in participants receiving concurrent methadone maintenance therapy. Although there was no significant impact on opioid craving or consumption, an improvement in depressive symptoms was present post-treatment. Li et al. [105] also observed improvements in depressive symptoms, in conjunction with reduced opioid craving, though their participants received concurrent occupational therapy. Liu et al. [104] applied both 1 Hz and 10 Hz stimulation to the left DLPFC, finding that both conditions produced similar reductions in cue-induced opioid craving compared to no treatment.
Transcranial direct current stimulation (tDCS)
Thirty-six studies investigating tDCS as treatment for SUDs, with 1582 participants receiving either active or control treatment (sham stimulation or no treatment; Table 2).
Alcohol
Fourteen studies [106,107,108,109,110,111,112,113,114,115,116,117,118,119] examined the effects of tDCS for AUD. Nine [106, 108,109,110, 113, 114, 116, 117, 119] demonstrated positive effects on alcohol craving and/or consumption following right or left anodal tDCS to DLPFC. While single stimulation sessions of right anodal and left anodal tDCS to the DLPFC demonstrated comparable effects, multi-session studies showed that right anodal DLPFC stimulation was consistently effective [113, 114, 119] but left anodal DLPFC stimulation was inconsistent [108, 110,111,112]. Variations of stimulation intensity (1–2 mA) and duration (10–30 min) were explored, though these differences did not produce consistent outcomes.
While nine studies reported positive effects on alcohol use outcomes following active tDCS, meta-analyses of craving and consumption outcomes in single- and multi-session studies did not reveal significant SMDs for active versus sham stimulation. Analysis of subjective craving from four single-session trials (n = 187) were non-significant (SMD = −0.60, 95% CI: −1.22 to 0.01, p = 0.06, I2 = 69.0%; Fig. 4A), as were sub-group analyses of craving (n = 777, SMD = −0.14, 95% CI: −0.57 to 0.28, p = 0.51, I2 = 80.6%; Fig. 4B) and consumption (n = 242, SMD = −0.08, 95% CI: −0.39 to 0.23, p = 0.62, I2 = 0%; Fig. 4C) from eight multi-session trials.
Tobacco
Eleven studies [120,121,122,123,124,125,126,127,128,129,130] were conducted on tDCS in TUD. All studies applied 2.0 mA stimulation for 15–30 min, except for Falcone et al. [123] and Meng et al. [121] both of whom applied 1.0 mA stimulation for 20 min. Seven studies, including Falcone et al. and Meng et al. reported positive effects on tobacco craving and/or cigarette consumption [121, 123,124,125,126,127, 129], with right anodal DLPFC stimulation being most effective, particularly with multi-session protocols [125,126,127,128,129]. Notably, Ghorbani-Behnam et al. [129] compared extended tDCS treatment (20 sessions over 12 weeks) with a shorter treatment duration (20 sessions over 4 weeks), with 8 weeks of bupropion and sham stimulation. Results showed that longer durations of tDCS resulted in the highest abstinence rate at 6 months post-treatment (25.7%).
While seven studies reported independent improvements in tobacco-related outcomes, meta-analysis did not reflect similar effects. From four single-session studies, sub-group analyses of craving (n = 72, SMD = −0.27, 95% CI: −0.60 to 0.06, p = 0.11, I2 = 0%; Fig. 5A) and consumption (n = 79, SMD = −0.79, 95% CI: −2.07 to 0.49, p = 0.22, I2 = 84.7%; Fig. 5B) did not produce significant effects with active versus sham stimulation. Similarly, in four multi-session trials, subgroup analyses of craving (n = 101, SMD = −0.50, 95% CI: −1.24 to 0.24, p = 0.19, I2 = 70.5%; Fig. 5C) and consumption (n = 86, SMD = −0.47, 95% CI: −1.49 to 0.56, p = 0.37, I2 = 79.2%; Fig. 5D) were non-significant.
Cocaine
Three studies [131,132,133] examined tDCS on cocaine craving using right anodal DLPFC stimulation, reporting conflicting results. While Batista et al. [131]. observed a reduction in cocaine craving after 5 sessions of 2 mA/20 min tDCS, Verveer et al. [132]. and Gaudreault et al. [133]. found no significant effects on craving following 10 active 2 mA/13 min or 15 active 2 mA/20 min sessions, respectively.
Methamphetamine
Five studies [134,135,136,137,138] investigated the effects of tDCS on MA use disorder, all of which reported a significant reduction in MA unconditioned or cue-induced craving compared to sham following right anodal DLPFC tDCS. Four studies [134,135,136,137] applied 2.0 mA stimulation for 20 min, whereas Xu et al. [138] combined 1.5 mA tDCS with computerized cognitive addiction therapy (CCAT). While both studies by Shahbabaie et al. [134, 135] examined effects of a single stimulation session, the remaining three studies [136,137,138] opted for a multi-session protocol (5–20 sessions). Notably, 4/5 [134,135,136,137] of these studies evaluated males only, while the remaining study examined only female participants [138].
Opioids
Three studies [139,140,141] were conducted on tDCS treatment efficacy for OUD. Two studies [139, 140] applied ten sessions of 2.0 mA tDCS to the DLPFC for 20 min. Taremian et al. [140] evaluated opioid craving and depressive symptoms in participants receiving methadone, and compared right anodal DLPFC stimulation with sham. Active tDCS significantly reduced opioid craving and depressive symptoms, compared to sham, and methadone alone. Eskandari et al. [139] compared left anodal DLPFC stimulation with right anodal DLPFC stimulation and sham, observing a significant reduction in craving in all groups; active groups exhibited greater effects. Wang et al. [141] applied a single stimulation session targeting the fronto-parietal-temporal area at 1.5 mA for 20 min. Despite these differences, a significant decline in heroin craving was observed, which persisted with the presentation of opioid-related cues.
Deep brain stimulation (DBS)
Seven studies investigated DBS as SUD treatment, with 48 participants receiving active or sham stimulation (Table 3).
Alcohol
Four studies [142,143,144,145] investigated effects of DBS on AUD by targeting the NAc. All studies observed significant decreases in alcohol consumption and/or craving post-treatment. Notably, Bach et al. [145] (N = 12) was the first to compare active and sham DBS and found significant improvements in substance use and craving following 6-months of active stimulation.
Tobacco
One study examined the use of DBS on TUD by targeting the NAc. Kuhn et al. [146] found that 3/10 TUD participants in their study quit smoking post-treatment, while the remaining seven participants showed a significant decline in tobacco craving and cigarette consumption.
Opioids
Two studies [147, 148] examined effects of DBS treatment in heroin-dependent participants and reported significant reductions in opioid craving and an increase in opioid abstinence. While Kuhn et al. [147] targeted the NAc exclusively, Chen et al. [148] applied simultaneous stimulation to anterior limb of the internal capsule (ALIC) and NAc.
Discussion
We systematically reviewed the cumulative literature on the efficacy of NM (rTMS, tDCS, DBS) for SUD treatment (Table 4). Findings were inconsistent across each stimulation methodology, and varied significantly with respect to SUD. This may be attributed to variations in treatment parameters, symptom severity across SUD participants, use of adjunctive treatment interventions and population heterogeneity, including the presence of comorbid psychiatric disorders, age, sex, and treatment history.
Nonetheless, findings from rTMS and tDCS studies demonstrated several commonalities. For rTMS, positive outcomes when treating tobacco, stimulant and opioid use disorders were observed, as indicated by post-treatment reductions in subjective and cue-induced substance craving and/or consumption when compared to sham treatment. Accordingly, effect sizes were clinically relevant (Hedge’s g > 0.5) but highly variable, consistent with heterogeneity of the published literature [9]. Furthermore, meta-analyses found that multi-session active versus sham rTMS was particularly effective in reducing tobacco consumption, but effects on tobacco craving were non-significant. Interestingly, effects of rTMS on AUD were less consistent, with 7/16 studies demonstrating significant improvements. Subsequent meta-analyses found that multi-session rTMS produced significantly greater reductions in alcohol craving and consumption. tDCS studies were promising in the treatment of tobacco, alcohol, stimulant, and opioid use disorders, as suggested by medium effect sizes (Table 2). However, meta-analyses of tDCS trials for AUD and TUD found that both single- and multi-session stimulation were not superior to sham stimulation in reducing craving or consumption, suggesting that rTMS may be superior to tDCS for these SUDs.
DBS produced reductions in craving, consumption and/or abstinence in alcohol, tobacco, and opioid use disorders. Available data is limited to case-series making it difficult to calculate effect sizes (Table 3), with the exception of one randomized sham-controlled study in AUD [145]. Sample sizes in DBS studies were low (ranging 2–12, averaging 6.9 ± 3.1 participants), suggesting the need for larger samples and randomized controlled trials.
Treatment parameters
Variability in treatment efficacy across NM studies may be attributed to differences in stimulation parameters (e.g., stimulation target, frequency, intensity, treatment duration and sample size/demographics). For both rTMS and tDCS studies, multi-session protocols are more effective than single-sessions protocols, as indicated by larger effect sizes and the number of positive outcome studies (see Tables 1–4). This is consistent with previous reports in the addictions neuromodulation literature [149]. However, total number of sessions needed to produce long-lasting effects is unclear and requires further investigation. For rTMS, the most commonly used paradigm across substances was 10–20 sessions once daily. In contrast, studies investigating TMS in depression suggest ≥30 sessions are needed for treatment durability [150]. While studies demonstrated persistent effects, including post-TMS reductions in 3-month alcohol [71] and cigarette consumption [82] after only 10 sessions of rTMS, durability of these effects remains uncertain as there is lack of long-term follow-up and biochemical verification beyond 1-month. Amiaz et al. [77] found that reductions in cigarette consumption after 10 sessions of rTMS were not maintained at 6-months. Similarly, number of tDCS sessions needed remains unclear due to lack of long-term follow-up. tDCS protocols were also considerably shorter, with all but two studies [129, 138] applying ≤10 sessions overall. Interestingly, Ghorbani Behnam et al. [129] applied 20 total sessions and found that when these sessions were distributed over a longer period of time (12 versus 4 weeks), tobacco abstinence was considerably higher at 6-month follow-up. Accordingly, session frequency may also play an important role. Moreover, potential effects of an accelerated stimulation paradigm (e.g. more than one session daily) should also be further investigated. Studies in depression have found that accelerated protocols are safe and well-tolerated, and perform comparably to standard once-daily rTMS [151,152,153]. Martinotti et al. [93] conducted the only randomized sham-controlled addictions study to adopt such an accelerated stimulation approach, but reported unfavourable cocaine use outcomes following twice daily stimulation. Nonetheless, Steele and colleagues [154] have found that three iTBS sessions/day for 10 days was tolerable and reduced cocaine consumption.
The need for maintenance sessions following initial stimulation treatment should be further evaluated to increase durability [155]. Two studies incorporated weekly reminder sessions following 15 daily HF deep-TMS sessions, and found that reductions in alcohol consumption [68] and tobacco craving [37] persisted 3-months post-treatment. However, Amiaz et al. [77] found that improvements in tobacco use outcomes following 10 HF rTMS sessions and 8 maintenance sessions did not persist at 6-months; this may reflect the effects of the coil (Figure-8 vs. H-coil) or the number of initial sessions (10 versus 15).
Four rTMS studies [83, 84, 91, 104] compared the effects of LF (1 Hz) and HF (10 Hz) stimulation and found that 10 Hz rTMS significantly reduced substance craving and/or consumption, suggesting that HF rTMS stimulation parameters have greater therapeutic potential in comparison to LF stimulation. Accordingly, most rTMS studies used HF stimulation (e.g., ≥5 Hz) regardless of SUD. For tDCS studies, the effects of stimulation intensity (1 mA vs. 2 mA) were less clear. However, tDCS outcomes were more promising when stimulation sessions were of longer duration (>15 min).
Cue-exposure prior to rTMS may activate craving-related neurocircuitry, and subsequent stimulation could then disrupt drug-related memory consolidation [156]. Accordingly, Dinur-Klein et al. [84] incorporated smoking cue exposure prior to HF deep TMS and found that it reduced cigarette consumption. Amiaz et al. [77] evaluated differential effects of both neutral and smoking cues prior to HF rTMS, finding that smoking cues reduced cue-induced tobacco craving. This expands on previous findings in both PTSD [157] and OCD [158], wherein provocation using brief cue exposure prior to treatment alleviated symptoms compared to no cue provocation. Future studies should determine whether cue exposure should be utilized in all rTMS and tDCS protocols.
There were inconsistencies for rTMS in AUD treatment, with positive outcomes reported in 44% of studies. Nonetheless, deep TMS was effective when compared to rTMS using a Figure-8 coil, suggesting that the H-coil may be advantageous when treating AUD due to targeting of deep brain structures (e.g., insula, nucleus accumbens). Subsequent meta-analyses did find positive effects of multi-session rTMS on alcohol craving and consumption. However, given that there are several evidence-based treatments available for AUD [159], we suggest that neuromodulation treatment development should be focused on SUDs with a lack of evidence-based biological treatments, such as cannabis and stimulants.
Target brain region
Substance use outcomes with NM are influenced by targeted brain region, as well as the subsequent bilateral or unilateral stimulation of regions of interest. Most rTMS studies for SUDs have targeted the DLPFC (38/50 studies). rTMS targeting the left DLPFC produced predominantly positive effects and clinically relevant effect sizes when treating tobacco, stimulant and opioid use disorders, while those stimulating the right or bilateral DLPFC were less effective (Table 1). In contrast, studies in AUD were not responsive to left DLPFC rTMS, though right and bilateral DLPFC stimulation was effective when multiple sessions were conducted. Alternative regions were less commonly studied. Notably, the mPFC/frontal pole (with or without concurrent stimulation of ACC) emerged as a novel therapeutic target, particularly with a deep TMS protocol with H-coil technology, as indicated by studies with alcohol [63, 68] and cocaine [91]. Targeting bilateral PFC and insular cortex with deep TMS may also be effective in alcohol and tobacco treatment [37, 66, 84, 86].
Both DLPFC and mPFC have emerged as leading rTMS targets; much remains unknown about the mechanism by which rTMS induces its therapeutic effects in SUDs. An understanding of rTMS-induced alterations in SUD-related brain circuitry is limited as very few studies have incorporated neuroimaging. Furthermore, there is much uncertainty surrounding optimal target locations, both for specific SUDs and individual patients, as there have been no direct head-to-head comparisons of different active rTMS targets. Consequently, it is possible that alternate targets may be required for distinct SUDs. Interestingly, there is evidence that the Default Mode Network may be a SCZ-specific network of tobacco dependence [160]. It is critical that rTMS clinical trials include brain-based measures (e.g., MRI, EEG) in order to elucidate mechanisms of action and identify optimal treatment targets.
With respect to tDCS, right anodal DLPFC stimulation appears to be most efficacious across all substances. However, right anodal DLPFC studies had considerably more stimulation sessions (≥5 sessions) than those applying left anodal DLPFC (≤5 sessions) stimulation. Thus, observed differences may be related to treatment duration, and future studies should explore longer durations of left anodal DLPFC tDCS.
Importantly, stimulation sites for rTMS and tDCS are conventionally identified using the 10–20 EEG system or by measuring distances from predefined external landmarks. While this one-size-fits-all approach produces approximate targeting of specified regions, it does not consider inter-individual differences in brain morphology and network architecture. Neuronavigation-guided NM with magnetic resonance imaging (MRI) may achieve greater precision with personalized targets. rTMS studies in depression have demonstrated the benefits of such an approach and found that clinical outcomes were significantly improved when patients were stimulated closer to fMRI-personalized targets [161]. Selected rTMS studies integrated MRI-neuronavigation [56,57,58, 60, 75, 81, 90], though the number of studies was insufficient to distinguish its effectiveness in comparison to non-personalized targeting. No tDCS studies were present. Consequently, future randomized control trials are warranted to assess the clinical potential of neuronavigation-guided personalized rTMS and tDCS. Most DBS studies targeted the NAc, and were consistently positive.
Alternate neuromodulation modalities
Other NM methods that are less frequently used and excluded from this review include Electroconvulsive Therapy (ECT) [162], Magnetic Seizure Therapy (MST) and Transcranial Alternating Current Stimulation (tACS) [163]. Studies examining their effects on SUDs are limited. We also excluded invasive ACC stimulation; ACC implants have shown positive effects, particularly for AUD, although adverse events have been reported [164].
Psychiatric comorbidities
Only a few studies have tested neuromodulation interventions in populations with comorbid psychiatric disorders. Notably, 3/4 of rTMS studies that examined TUD participants with co-occurring SCZ observed significant reductions in tobacco craving and consumption [78, 80, 85] (Table 1). Prevalence of tobacco use in SCZ is 60–80% and contributes to a 25-year decreased life expectancy in SCZ [165], emphasizing the therapeutic potential of rTMS for this comorbidity. Moreover, SCZ patients have high rates of cannabis misuse [166]. Kozak-Bidzinski et al. [88] studied rTMS in outpatients with SCZ and CUD (N = 19). Although the difference in cannabis use was not statistically significant, larger reductions (~60%) were observed in the active (n = 9) versus sham (n = 10) group, highlighting its treatment potential. Ultimately, these NM methods show promise in treating co-occurring SUD and psychiatric disorders, warranting further research in clinical trials with larger sample sizes.
Strengths and limitations
This comprehensive systematic review and meta-analysis contributes substantially to the literature on NM for SUDs for the following reasons: (1) We calculated effect sizes for each study across all three stimulation modalities, and where applicable, conducted a meta-analysis of the published data, to compare and contrast these treatment outcomes. This is the first comprehensive systematic review of the addiction NM literature to include meta-analytic comparisons; (2) We evaluated the treatment efficacy of each stimulation technique, with respect to each SUD and the stimulation parameters applied, to identify their differential effects across substances; (3) We included several new studies that have been published since the reviews by Salling and Martinez [8] and Coles and colleagues [9].
However, there were some limitations. First, there was significant variability in the number of studies for each SUD and NM methodology. Many of these studies were also preliminary (sample size <40 participants). Second, studies were not balanced for sex, with an emphasis on males. Thus, sex-related differences in treatment outcomes are unclear. Third, there was variability in outcomes evaluated (e.g., craving vs. consumption) and in methods used to measure them (e.g., biochemical verification versus self-report). Fourth, as substance use was the primary outcome of interest, associated outcomes such as psychiatric symptoms and cognition were secondary and not always reported. Finally, treatment effects were quantitively assessed using end-of-treatment data due to heterogeneity in follow-up periods. Thus, enduring effects of NM interventions cannot be adequately determined.
Conclusions and future directions
There is considerable promise for the use of NM therapies in SUDs. Nonetheless, further research is required to determine clinical safety and efficacy. Future studies should focus on optimizing stimulation parameters and regimens for these NM methods, with emphasis on stimulation duration, number of treatment sessions needed to produce enduring effects, accelerated treatment paradigms, stimulation frequency and intensity and targeted brain region. Assessment of enduring effects of NM treatment using biochemical verification at extended time-points and the need for maintenance sessions following treatment cessation to optimize clinical outcomes should be emphasized. Neuroimaging data (fMRI) should be acquired prior to, during, and following treatment to elucidate the underlying neural mechanisms mediating treatment effects. Moreover, MRI-neuronavigation may address potential discordance between coil/electrode placement and region of interest, potentially improving treatment efficacy.
Finally, greater emphasis on co-occurring psychiatric disorders is needed. rTMS may be a promising intervention for patients with SCZ and concurrent SUDs, warranting larger randomized sham-controlled trials. Finally, the potential of adjunctive psychotherapeutic and/or pharmacological intervention should be determined, which may improve substance use outcomes [81]. While some studies have implemented concurrent pharmacological interventions [78], few have parsed the clinical impact of each therapy for augmentation of NM outcomes.
References
Substance Abuse and Mental Health Services Administration. Key substance use and mental health indicators in the United States: results from the 2021 National Survey on Drug Use and Health. 2022. https://www.samhsa.gov/data/sites/default/files/reports/rpt39443/2021NSDUHFFRRev010323.pdf.
United Nations Office on Drugs and Crime. World Drug Report 2020. 2020. https://www.unodc.org/unodc/data-and-analysis/world-drug-report-2022.html.
Swendsen JD, Merikangas KR. The comorbidity of depression and substance use disorders. Clin Psychol Rev. 2000;20:173–89.
Starzer MSK, Nordentoft M, Hjorthøj C. Rates and predictors of conversion to schizophrenia or bipolar disorder following substance-induced psychosis. Am J Psychiatry. 2018;175:343–50.
Kandel DB, Johnson JG, Bird HR, Weissman MM, Goodman SH, Lahey BB, et al. Psychiatric comorbidity among adolescents with substance use disorders: findings from the MECA study. J Am Acad Child Adolesc Psychiatry. 1999;38:693–9.
National Institute on Drug Abuse. Drugs, Brains and Behaviour: The Science of Addiction. 2007. http://www.drugabuse.gov/scienceofaddiction/brain.html.
Kabisa E, Biracyaza E, Habagusenga Jd’Amour, Umubyeyi A. Determinants and prevalence of relapse among patients with substance use disorders: case of Icyizere Psychotherapeutic Centre. Subst Abus Treat Prev Policy. 2021;16:13.
Salling M, Martinez D. Brain stimulation in addiction. Neuropsychopharmacology. 2016;41:2798–809.
Coles AS, Kozak K, George TP. A review of brain stimulation methods to treat substance use disorders. Am J Addict. 2018;27:71–91.
Volkow ND, Fowler JS, Wang GJ, Baler R, Telang F. Imaging dopamine’s role in drug abuse and addiction. Neuropharmacology. 2009;56:3–8.
Koob GF, Volkow ND. Neurobiology of addiction: a neurocircuitry analysis. Lancet Psychiatry. 2016;3:760–73.
Nutt DJ, Lingford-Hughes A, Erritzoe D, Stokes PRA. The dopamine theory of addiction: 40 years of highs and lows. Nat Rev Neurosci. 2015;16:305–12.
Goldstein RZ, Volkow ND. Dysfunction of the prefrontal cortex in addiction: neuroimaging findings and clinical implications. Nat Rev Neurosci. 2011;12:652–69.
McClure SM, Bickel WK. A dual-systems perspective on addiction: contributions from neuroimaging and cognitive training. Ann N. Y Acad Sci. 2014;1327:62–78.
Balconi M, Finocchiaro R, Canavesio Y. Reward-system effect (BAS rating), left hemispheric ‘unbalance’ (alpha band oscillations) and decisional impairments in drug addiction. Addict Behav. 2014;39:1026–32.
Balconi M, Finocchiaro R. Decisional impairments in cocaine addiction, reward bias, and cortical oscillation “unbalance”. Neuropsychiatr Dis Treat. 2015;11:777–86.
Maiti R, Mishra BR, Hota D. Effect of high-frequency transcranial magnetic stimulation on craving in substance use disorder: a meta-analysis. J Neuropsychiatry Clin Neurosci. 2017;29:160–71.
Steele VR. A circuit-based approach to treating substance use disorders with noninvasive brain stimulation. Biol Psychiatry. 2021;89:944–6.
McNamara B, Ray JL, Arthurs OJ, Boniface S. Transcranial magnetic stimulation for depression and other psychiatric disorders. Psychol Med. 2001;31:1141–46.
Lefaucheur J-P, Aleman A, Baeken C, Benninger DH, Brunelin J, Di Lazzaro V, et al. Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS): An update (2014–2018). Clin Neurophysiol. 2020;131:474–528.
Tang AD, Bennett W, Bindoff AD, Bolland S, Collins J, Langley RC, et al. Subthreshold repetitive transcranial magnetic stimulation drives structural synaptic plasticity in the young and aged motor cortex. Brain Stimul Basic Transl Clin Res Neuromodulation. 2021;14:1498–507.
Jin Y, Bai X, Jiang B, Guo Z, Mu Q. Repetitive transcranial magnetic stimulation induces quantified functional and structural changes in subcortical stroke: a combined arterial spin labeling perfusion and diffusion tensor imaging study. Front Hum Neurosci. 2022;16:829688.
Keck ME, Welt T, Müller MB, Erhardt A, Ohl F, Toschi N, et al. Repetitive transcranial magnetic stimulation increases the release of dopamine in the mesolimbic and mesostriatal system. Neuropharmacology. 2002;43:101–9.
Strafella AP, Paus T, Fraraccio M, Dagher A. Striatal dopamine release induced by repetitive transcranial magnetic stimulation of the human motor cortex. Brain. 2003;126:2609–15.
Strafella AP, Paus T, Barrett J, Dagher A. Repetitive transcranial magnetic stimulation of the human prefrontal cortex induces dopamine release in the caudate nucleus. J Neurosci. 2001;21:RC157.
Cho SS, Strafella AP. rTMS of the left dorsolateral prefrontal cortex modulates dopamine release in the ipsilateral anterior cingulate cortex and orbitofrontal cortex. PLOS ONE. 2009;4:e6725.
de Jesus D, Favalli G, Hoppenbrouwers S, Barr M, Chen R, Fitzgerald P, et al. Determining optimal rTMS parameters through changes in cortical inhibition. Clin Neurophysiol. 2014;125:755–62.
Speer AM, Kimbrell TA, Wassermann EM, D. Repella J, Willis MW, Herscovitch P, et al. Opposite effects of high and low frequency rTMS on regional brain activity in depressed patients. Biol Psychiatry. 2000;48:1133–41.
Fitzgerald PB, Fountain S, Daskalakis ZJ. A comprehensive review of the effects of rTMS on motor cortical excitability and inhibition. Clin Neurophysiol. 2006;117:2584–96.
Martin-Rodriguez JF, Ruiz-Veguilla M, Alvarez de Toledo P, Aizpurua-Olaizola O, Zarandona I, Canal-Rivero M, et al. Impaired motor cortical plasticity associated with cannabis use disorder in young adults. Addict Biol. 2021;26:e12912.
Nardone R, Bergmann J, Christova M, Lochner P, Tezzon F, Golaszewski S, et al. Non-invasive brain stimulation in the functional evaluation of alcohol effects and in the treatment of alcohol craving: a review. Neurosci Res. 2012;74:169–76.
Levkovitz Y, Isserles M, Padberg F, Lisanby SH, Bystritsky A, Xia G, et al. Efficacy and safety of deep transcranial magnetic stimulation for major depression: a prospective multicenter randomized controlled trial. World Psychiatry. 2015;14:64–73.
Di Lazzaro V, Pilato F, Dileone M, Profice P, Oliviero A, Mazzone P, et al. The physiological basis of the effects of intermittent theta burst stimulation of the human motor cortex. J Physiol. 2008;586:3871–9.
Huang Y, Edwards M, Rounis E, Bhatia K, Rothwell J. Theta burst stimulation of the human motor cortex. NEURON. 2005;45:201–6.
Rossi S, Hallett M, Rossini PM, Pascual-Leone A. Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin Neurophysiol. 2009;120:2008–39.
Slotema CW, Blom JD, Hoek HW, Sommer IEC. Should we expand the toolbox of psychiatric treatment methods to include Repetitive Transcranial Magnetic Stimulation (rTMS)? A meta-analysis of the efficacy of rTMS in psychiatric disorders. J Clin Psychiatry. 2010;71:873–84.
Zangen A, Moshe H, Martinez D, Barnea-Ygael N, Vapnik T, Bystritsky A, et al. Repetitive transcranial magnetic stimulation for smoking cessation: a pivotal multicenter double-blind randomized controlled trial. World Psychiatry J World Psychiatr Assoc WPA. 2021;20:397–404.
Steele VR, Maxwell AM. Treating cocaine and opioid use disorder with transcranial magnetic stimulation: a path forward. Pharm Biochem Behav. 2021;209:173240.
Priori A, Berardelli A, Rona S, Accornero N, Manfredi M. Polarization of the human motor cortex through the scalp. NeuroReport. 1998;9:2257.
Nitsche MA, Paulus W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J Physiol. 2000;527:633–9.
Dedoncker J, Brunoni AR, Baeken C, Vanderhasselt M-A. A systematic review and meta-analysis of the effects of transcranial direct current stimulation (tDCS) over the dorsolateral prefrontal cortex in healthy and neuropsychiatric samples: influence of stimulation parameters. Brain Stimul. 2016;9:501–17.
Kessler SK, Turkeltaub PE, Benson JG, Hamilton RH. Differences in the experience of active and sham transcranial direct current stimulation. Brain Stimul. 2012;5:155–62.
Camacho-Conde JA, del Rosario Gonzalez-Bermudez M, Carretero-Rey M, Khan ZU. Therapeutic potential of brain stimulation techniques in the treatment of mental, psychiatric, and cognitive disorders. CNS Neurosci Ther. 2023;29:8–23.
Perlmutter JS, Mink JW. Deep brain stimulation. Annu Rev Neurosci. 2006;29:229–57.
Dostrovsky JO, Lozano AM. Mechanisms of deep brain stimulation. Mov Disord. 2002;17:S63–S68.
Herrington TM, Cheng JJ, Eskandar EN. Mechanisms of deep brain stimulation. J Neurophysiol. 2016;115:19–38.
Haberler C, Alesch F, Mazal PR, Pilz P, Jellinger K, Pinter MM, et al. No tissue damage by chronic deep brain stimulation in Parkinson’s disease. Ann Neurol. 2000;48:372–6.
Hariz MI. Complications of deep brain stimulation surgery. Mov Disord. 2002;17:S162–S166.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Int J Surg. 2021;88:105906.
Schardt C, Adams MB, Owens T, Keitz S, Fontelo P. Utilization of the PICO framework to improve searching PubMed for clinical questions. BMC Med Inf Decis Mak. 2007;7:16.
Corbett MS, Higgins JPT, Woolacott NF. Assessing baseline imbalance in randomised trials: implications for the Cochrane risk of bias tool. Res Synth Methods. 2014;5:79–85.
Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919
Fletcher J. What is heterogeneity and is it important? BMJ. 2007;334:94–96.
R Core Team. R Foundation for Statistical Computing, Vienna, Austria, 2023.
Viechtbauer W. Conducting meta-analyses in R with the metafor package. J Stat Softw. 2010;36:1–48.
Herremans S, Baeken C, Vanderbruggen N, Vanderhasselt M, Zeeuws D, Santermans L, et al. No influence of one right-sided prefrontal HF-rTMS session on alcohol craving in recently detoxified alcohol-dependent patients: results of a naturalistic study. Drug Alcohol Depend. 2012;120:209–13.
Herremans S, Vanderhasselt M, De Raedt R, Baeken C. Reduced intra-individual reaction time variability during a Go-NoGo task in detoxified alcohol-dependent patients after one right-sided dorsolateral prefrontal HF-rTMS session. Alcohol Alcohol. 2013;48:552–7.
Herremans S, Van Schuerbeek P, De Raedt R, Matthys F, Buyl R, De Mey J, et al. The Impact of Accelerated Right Prefrontal High-Frequency Repetitive Transcranial Magnetic Stimulation (rTMS) on Cue-Reactivity: An fMRI Study on Craving in Recently Detoxified Alcohol-Dependent Patients. PLOS ONE. 2015;10:e0136182.
Hanlon CA, Dowdle LT, Correia B, Mithoefer O, Kearney-Ramos T, Lench D, et al. Left frontal pole theta burst stimulation decreases orbitofrontal and insula activity in cocaine users and alcohol users. Drug Alcohol Depend. 2017;178:310–7.
Jansen JM, van den Heuvel OA, van der Werf YD, de Wit SJ, Veltman DJ, van den Brink W, et al. The Effect of High-Frequency Repetitive Transcranial Magnetic Stimulation on Emotion Processing, Reappraisal, and Craving in Alcohol Use Disorder Patients and Healthy Controls: A Functional Magnetic Resonance Imaging Study. Front Psychiatry. 2019;10:272.
Mishra B, Nizamie S, Das B, Praharaj S. Efficacy of repetitive transcranial magnetic stimulation in alcohol dependence: a sham-controlled study. Addiction. 2010;105:49–55.
Hoppner J, Broese T, Wendler L, Berger C, Thome J. Repetitive transcranial magnetic stimulation (rTMS) for treatment of alcohol dependence. World J Biol Psychiatry. 2011;12:57–62.
Ceccanti M, Inghilleri M, Attilia M, Raccah R, Fiore M, Zangen A, et al. Deep TMS on alcoholics: effects on cortisolemia and dopamine pathway modulation. A pilot study. Can J Physiol Pharm. 2015;93:283–90.
Girardi P, Rapinesi C, Chiarotti F, Kotzalidis G, Piacentino D, Serata D, et al. Add-on deep transcranial magnetic stimulation (dTMS) in patients with dysthymic disorder comorbid with alcohol use disorder: a comparison with standard treatment. World J Biol Psychiatry. 2015;16:66–73.
Addolorato G, Antonelli M, Cocciolillo F, Vassallo G, Tarli C, Sestito L, et al. Deep transcranial magnetic stimulation of the dorsolateral prefrontal cortex in alcohol use disorder patients: effects on dopamine transporter availability and alcohol intake. Eur Neuropsychopharmacol. 2017;27:450–61.
Perini I, Kämpe R, Arlestig T, Karlsson H, Löfberg A, Pietrzak M, et al. Repetitive transcranial magnetic stimulation targeting the insular cortex for reduction of heavy drinking in treatment-seeking alcohol-dependent subjects: a randomized controlled trial. Neuropsychopharmacol Publ Am Coll Neuropsychopharmacol. 2020;45:842–50.
Raikwar S, Divinakumar KJ, Prakash J, Khan SA, GuruPrakash KV, Batham S. A sham-controlled trial of repetitive transcranial magnetic stimulation over left dorsolateral prefrontal cortex and its effects on craving in patients with alcohol dependence. Ind Psychiatry J. 2020;29:245–50.
Harel M, Perini I, Kämpe R, Alyagon U, Shalev H, Besser I, et al. Repetitive transcranial magnetic stimulation in alcohol dependence: a randomized, double-blind, sham-controlled proof-of-concept trial targeting the medial prefrontal and anterior cingulate cortices. Biol Psychiatry. 2022;91:1061–9.
Zhang T, Song B, Li Y, Duan R, Gong Z, Jing L, et al. Neurofilament light chain as a biomarker for monitoring the efficacy of transcranial magnetic stimulation on alcohol use disorder. Front Behav Neurosci. 2022;16:831901.
Hoven M, Schluter RS, Schellekens AF, van Holst RJ, Goudriaan AE. Effects of 10 add-on HF-rTMS treatment sessions on alcohol use and craving among detoxified inpatients with alcohol use disorder: a randomized sham-controlled clinical trial. Addiction. 2023;118:71–85.
McCalley DM, Kaur N, Wolf JP, Contreras IE, Book SW, Smith JP, et al. Medial prefrontal cortex theta burst stimulation improves treatment outcomes in alcohol use disorder: a double-blind, sham-controlled neuroimaging study. Biol Psychiatry Glob Open Sci. 2023;3:301–10.
Li X, Du L, Sahlem G, Badran B, Henderson S, George M. Repetitive transcranial magnetic stimulation (rTMS) of the dorsolateral prefrontal cortex reduces resting-state insula activity and modulates functional connectivity of the orbitofrontal cortex in cigarette smokers. Drug Alcohol Depend. 2017;174:98–105.
Kozak K, Sharif-Razi M, Morozova M, Gaudette EV, Barr MS, Daskalakis ZJ, et al. Effects of short-term, high-frequency repetitive transcranial magnetic stimulation to bilateral dorsolateral prefrontal cortex on smoking behavior and cognition in patients with schizophrenia and non-psychiatric controls. Schizophr Res. 2018;197:441–3.
Li X, Hartwell K, Owens M, LeMatty T, Borckardt J, Hanlon C, et al. Repetitive transcranial magnetic stimulation of the dorsolateral prefrontal cortex reduces nicotine cue craving. Biol Psychiatry. 2013;73:714–20.
Pripfl J, Tomova L, Riecansky I, Lamm C. Transcranial magnetic stimulation of the left dorsolateral prefrontal cortex decreases cue-induced nicotine craving and EEG delta power. Brain Stimul. 2014;7:226–33.
Eichhammer P, Johann M, Kharraz A, Binder H, Pittrow D, Wodarz N, et al. High-frequency repetitive transcranial magnetic stimulation decreases cigarette smoking. J Clin Psychiatry. 2003;64:951–3.
Amiaz R, Levy D, Vainiger D, Grunhaus L, Zangen A. Repeated high-frequency transcranial magnetic stimulation over the dorsolateral prefrontal cortex reduces cigarette craving and consumption. Addiction. 2009;104:653–60.
Wing V, Bacher I, Wu B, Daskalakis Z, George T. High frequency repetitive transcranial magnetic stimulation reduces tobacco craving in schizophrenia. Schizophr Res. 2012;139:264–6.
Dieler A, Dresler T, Joachim K, Deckert J, Herrmann M, Fallgatter A. Can intermittent theta burst stimulation as add-on to psychotherapy improve nicotine abstinence? Results from a pilot study. Eur Addict Res. 2014;20:248–53.
Prikryl R, Ustohal L, Kucerova H, Kasparek T, Jarkovsky J, Hublova V, et al. Repetitive transcranial magnetic stimulation reduces cigarette consumption in schizophrenia patients. Prog Neuropsychopharmacol Biol Psychiatry. 2014;49:30–5.
Trojak B, Meille V, Achab S, Lalanne L, Poquet H, Ponavoy E, et al. Transcranial magnetic stimulation combined with nicotine replacement therapy for smoking cessation: a randomized controlled trial. Brain Stimul. 2015;8:1168–74.
Abdelrahman AA, Noaman M, Fawzy M, Moheb A, Karim AA, Khedr EM. A double-blind randomized clinical trial of high frequency rTMS over the DLPFC on nicotine dependence, anxiety and depression. Sci Rep. 2021;11:1640.
Rose J, McClernon F, Froeliger B, Behm F, Preud’homme X, Krystal A. Repetitive transcranial magnetic stimulation of the superior frontal gyrus modulates craving for cigarettes. Biol Psychiatry. 2011;70:794–9.
Dinur-Klein L, Dannon P, Hadar A, Rosenberg O, Roth Y, Kotler M, et al. Smoking cessation induced by deep repetitive transcranial magnetic stimulation of the prefrontal and insular cortices: a prospective, randomized controlled trial. Biol Psychiatry. 2014;76:742–9.
Moeller SJ, Gil R, Weinstein JJ, Baumvoll T, Wengler K, Fallon N, et al. Deep rTMS of the insula and prefrontal cortex in smokers with schizophrenia: proof-of-concept study. Schizophrenia. 2022;8:1–9.
Ibrahim C, Tang VM, Blumberger DM, Malik S, Tyndale RF, Trevizol AP, et al. Efficacy of insula deep repetitive transcranial magnetic stimulation combined with varenicline for smoking cessation: a randomized, double-blind, sham controlled trial. Brain Stimul. 2023;16:1501–9.
Sahlem GL, Baker NL, George MS, Malcolm RJ, McRae-Clark AL. Repetitive transcranial magnetic stimulation (rTMS) administration to heavy cannabis users. Am J Drug Alcohol Abus. 2018;44:47–55.
Bidzinski KK, Lowe DJE, Sanches M, Sorkhou M, Boileau I, Kiang M, et al. Investigating repetitive transcranial magnetic stimulation on cannabis use and cognition in people with schizophrenia. NPJ Schizophr. 2022;8:2.
Bolloni C, Panella R, Pedetti M, Frascella A, Gambelunghe C, Piccoli T, et al. Bilateral transcranial magnetic stimulation of the prefrontal cortex reduces cocaine intake: a Pilot study. Front Psychiatry. 2016;7:133.
Terraneo A, Leggio L, Saladini M, Ermani M, Bonci A, Gallimberti L. Transcranial magnetic stimulation of dorsolateral prefrontal cortex reduces cocaine use: a pilot study. Eur Neuropsychopharmacol. 2016;26:37–44.
Martinez D, Urban N, Grassetti A, Chang D, Hu M-C, Zangen A, et al. Transcranial magnetic stimulation of medial prefrontal and cingulate cortices reduces cocaine self-administration: a pilot study. Front Psychiatry. 2018;9:80.
Lolli F, Salimova M, Scarpino M, Lanzo G, Cossu C, Bastianelli M, et al. A randomised, double-blind, sham-controlled study of left prefrontal cortex 15 Hz repetitive transcranial magnetic stimulation in cocaine consumption and craving. PloS One. 2021;16:e0259860.
Martinotti G, Pettorruso M, Montemitro C, Spagnolo PA, Acuti Martellucci C, Di Carlo F, et al. Repetitive transcranial magnetic stimulation in treatment-seeking subjects with cocaine use disorder: a randomized, double-blind, sham-controlled trial. Prog Neuropsychopharmacol Biol Psychiatry. 2022;116:110513.
Li X, Malcolm R, Huebner K, Hanlon C, Taylor J, Brady K, et al. Low frequency repetitive transcranial magnetic stimulation of the left dorsolateral prefrontal cortex transiently increases cue-induced craving for methamphetamine: a preliminary study. Drug Alcohol Depend. 2013;133:641–6.
Su H, Zhong N, Gan H, Wang J, Han H, Chen T, et al. High frequency repetitive transcranial magnetic stimulation of the left dorsolateral prefrontal cortex for methamphetamine use disorders: a randomised clinical trial. Drug Alcohol Depend. 2017;175:84–91.
Liang Y, Wang L, Yuan T-F. Targeting withdrawal symptoms in men addicted to methamphetamine with transcranial magnetic stimulation. Jama Psychiatry. 2018;75:1199–201.
Liu T, Li Y, Shen Y, Liu X, Yuan T-F. Gender does not matter: add-on repetitive transcranial magnetic stimulation treatment for female methamphetamine dependents. Prog Neuropsychopharmacol Biol Psychiatry. 2019;92:70–5.
Chen T, Su H, Li R, Jiang H, Li X, Wu Q, et al. The exploration of optimized protocol for repetitive transcranial magnetic stimulation in the treatment of methamphetamine use disorder: a randomized sham-controlled study. EBioMedicine. 2020;60:103027.
Su H, Chen T, Jiang H, Zhong N, Du J, Xiao K, et al. Intermittent theta burst transcranial magnetic stimulation for methamphetamine addiction: a randomized clinical trial. Eur Neuropsychopharmacol J Eur Coll Neuropsychopharmacol. 2020;31:158–61.
Su H, Liu Y, Yin D, Chen T, Li X, Zhong N, et al. Neuroplastic changes in resting-state functional connectivity after rTMS intervention for methamphetamine craving. Neuropharmacology. 2020;175:108177.
Yuan J, Liu W, Liang Q, Cao X, Lucas MV, Yuan T-F. Effect of low-frequency repetitive transcranial magnetic stimulation on impulse inhibition in abstinent patients with methamphetamine addiction. Jama Netw Open. 2020;3:e200910.
Tsai T-Y, Wang T-Y, Liu YC, Lee P-W, Chang WH, Lu T-H, et al. Add-on repetitive transcranial magnetic stimulation in patients with opioid use disorder undergoing methadone maintenance therapy. Am J Drug Alcohol Abus. 2021;47:330–43.
Shen Y, Cao X, Tan T, Shan C, Wang Y, Pan J, et al. 10-Hz repetitive transcranial magnetic stimulation of the left dorsolateral prefrontal cortex reduces heroin cue craving in long-term addicts. Biol Psychiatry. 2016;80:e13–14.
Liu X, Zhao X, Liu T, Liu Q, Tang L, Zhang H, et al. The effects of repetitive transcranial magnetic stimulation on cue-induced craving in male patients with heroin use disorder. EBioMedicine. 2020;56:102809.
Li X, Song G, Yu J, Ai S, Ji Q, Peng Y, et al. Effectiveness and safety of repetitive transcranial magnetic stimulation for the treatment of morphine dependence. Med (Baltim). 2021;100:e25208.
Boggio P, Sultani N, Fecteau S, Merabet L, Mecca T, Pascual-Leone A, et al. Prefrontal cortex modulation using transcranial DC stimulation reduces alcohol craving: a double-blind, sham-controlled study. Drug Alcohol Depend. 2008;92:55–60.
Claus ED, Klimaj SD, Chavez R, Martinez AD, Clark VP. A randomized trial of combined tDCS over right inferior frontal cortex and cognitive bias modification: null effects on drinking and alcohol approach bias. Alcohol Clin Exp Res. 2019;43:1591–9.
da Silva M, Conti C, Klauss J, Alves L, Cavalcante H, Fregni F, et al. Behavioral effects of transcranial Direct Current Stimulation (tDCS) induced dorsolateral prefrontal cortex plasticity in alcohol dependence. J Physiol-PARIS. 2013;107:493–502.
den Uyl T, Gladwin T, Wiers R. Transcranial direct current stimulation, implicit alcohol associations and craving. Biol Psychol. 2015;105:37–42.
den Uyl TE, Gladwin TE, Wiers RW. Electrophysiological and behavioral effects of combined transcranial direct current stimulation and alcohol approach bias retraining in hazardous drinkers. Alcohol Clin Exp Res. 2016;40:2124–33.
den Uyl T, Gladwin T, Rinck M, Lindenmeyer J, Wiers R. A clinical trial with combined transcranial direct current stimulation and alcohol approach bias retraining. Addict Biol. 2017;22:1632–40.
den Uyl TE, Gladwin TE, Lindenmeyer J, Wiers RW. A clinical trial with combined transcranial direct current stimulation and attentional bias modification in alcohol-dependent patients. Alcohol Clin Exp Res. 2018;42:1961–9.
Klauss J, Pinheiro L, Merlo B, Santos G, Fregni F, Nitsche M, et al. A randomized controlled trial of targeted prefrontal cortex modulation with tDCS in patients with alcohol dependence. Int J Neuropsychopharmacol. 2014;17:1793–803.
Klauss J, Anders QS, Felippe LV, Nitsche MA, Nakamura-Palacios EM. Multiple sessions of transcranial direct current stimulation (tDCS) reduced craving and relapses for alcohol use: a randomized placebo-controlled trial in alcohol use disorder. Front Pharmacol. 2018;9:716.
Nakamura-Palacios E, Benevides M, Zago-Gomes M, de Oliveira R, de Vasconcellos V, de Castro L, et al. Auditory event-related potentials (P3) and cognitive changes induced by frontal direct current stimulation in alcoholics according to Lesch alcoholism typology. Int J Neuropsychopharmacol. 2012;15:601–16.
Vanderhasselt M-A, Allaert J, De Raedt R, Baeken C, Krebs RM, Herremans S. Bifrontal tDCS applied to the dorsolateral prefrontal cortex in heavy drinkers: influence on reward-triggered approach bias and alcohol consumption. Brain Cogn. 2020;138:105512.
Wietschorke K, Lippold J, Jacob C, Polak T, Herrmann M. Transcranial direct current stimulation of the prefrontal cortex reduces cue-reactivity in alcohol-dependent patients. J Neural Transm. 2016;123:1173–8.
Witkiewitz K, Stein ER, Votaw VR, Wilson AD, Roos CR, Gallegos SJ, et al. Mindfulness-based relapse prevention and transcranial direct current stimulation to reduce heavy drinking: a double-blind sham-controlled randomized trial. Alcohol Clin Exp Res. 2019;43:1296–307.
Dubuson M, Kornreich C, Vanderhasselt M-A, Baeken C, Wyckmans F, Dousset C, et al. Transcranial direct current stimulation combined with alcohol cue inhibitory control training reduces the risk of early alcohol relapse: a randomized placebo-controlled clinical trial. Brain Stimul Basic Transl Clin Res Neuromodulation. 2021;14:1531–43.
Xu J, Fregni F, Brody AL, Rahman AS. Transcranial direct current stimulation reduces negative affect but not cigarette craving in overnight abstinent smokers. Front Psychiatry. 2013;4:112.
Meng Z, Liu C, Yu C, Ma Y. Transcranial direct current stimulation of the frontal-parietal-temporal area attenuates smoking behavior. J Psychiatr Res. 2014;54:19–25.
Kroczek A, Haussinger F, Rohe T, Schneider S, Plewnia C, Batra A, et al. Effects of transcranial direct current stimulation on craving, heart-rate variability and prefrontal hemodynamics during smoking cue exposure. Drug Alcohol Depend. 2016;168:123–7.
Falcone M, Bernardo L, Ashare R, Hamilton R, Faseyitan O, McKee S, et al. Transcranial direct current brain stimulation increases ability to resist smoking. Brain Stimul. 2016;9:191–6.
Yang L-Z, Shi B, Li H, Zhang W, Liu Y, Wang H, et al. Electrical stimulation reduces smokers’ craving by modulating the coupling between dorsal lateral prefrontal cortex and parahippocampal gyrus. Soc Cogn Affect Neurosci. 2017;12:1296–302.
Fregni F, Liguori P, Fecteau S, Nitsche M, Pascual-Leone A, Boggio P. Cortical stimulation of the prefrontal cortex with transcranial direct current stimulation reduces cue-provoked smoking craving: a randomized, sham-controlled study. J Clin Psychiatry. 2008;69:32–40.
Boggio P, Liguori P, Sultani N, Rezende L, Fecteau S, Fregni F. Cumulative priming effects of cortical stimulation on smoking cue-induced craving. Neurosci Lett. 2009;463:82–6.
Fecteau S, Agosta S, Hone-Blanchet A, Fregni F, Boggio P, Ciraulo D, et al. Modulation of smoking and decision-making behaviors with transcranial direct current stimulation in tobacco smokers: a preliminary study. Drug Alcohol Depend. 2014;140:78–84.
Smith R, Boules S, Mattiuz S, Youssef M, Tobe R, Sershen H, et al. Effects of transcranial direct current stimulation (tDCS) on cognition, symptoms, and smoking in schizophrenia: a randomized controlled study. Schizophr Res. 2015;168:260–6.
Ghorbani Behnam S, Mousavi SA, Emamian MH. The effects of transcranial direct current stimulation compared to standard bupropion for the treatment of tobacco dependence: a randomized sham-controlled trial. Eur Psychiatry J Assoc Eur Psychiatr. 2019;60:41–8.
Müller T, Shevchenko Y, Gerhardt S, Kiefer F, Vollstädt-Klein S. The influence of perceived stress and self-control on efficacy of repeated transcranial direct current stimulation in non-treatment-seeking smokers. Drug Alcohol Depend. 2021;226:108861.
Batista E, Klauss J, Fregni F, Nitsche M, Nakamura-Palacios E. A randomized placebo-controlled trial of targeted prefrontal cortex modulation with bilateral tDCS in patients with crack-cocaine dependence. Int J Neuropsychopharmacol. 2015;18:pii:pyv066.
Verveer I, van der Veen FM, Shahbabaie A, Remmerswaal D, Franken IHA. Multi-session electrical neuromodulation effects on craving, relapse and cognitive functions in cocaine use disorder: a randomized, sham-controlled tDCS study. Drug Alcohol Depend. 2020;217:108429.
Gaudreault P-O, Sharma A, Datta A, Nakamura-Palacios EM, King S, Malaker P, et al. A double-blind sham-controlled phase 1 clinical trial of tDCS of the dorsolateral prefrontal cortex in cocaine inpatients: craving, sleepiness, and contemplation to change. Eur J Neurosci. 2021;53:3212–30.
Shahbabaie A, Golesorkhi M, Zamanian B, Ebrahimpoor M, Keshvari F, Nejati V, et al. State dependent effect of transcranial direct current stimulation (tDCS) on methamphetamine craving. Int J Neuropsychopharmacol. 2014;17:1591–8.
Shahbabaie A, Ebrahimpoor M, Hariri A, Nitsche MA, Hatami J, Fatemizadeh E, et al. Transcranial DC stimulation modifies functional connectivity of large-scale brain networks in abstinent methamphetamine users. Brain Behav. 2018;8:e00922.
Rohani Anaraki M, Dolatshahi B, Nosratabadi M, Nouri Yalghouzaghaji M, Rezaei Mashhadi S. Repeated transcranial direct current stimulation (tDCS) on methamphetamine craving: a randomized, sham-controlled study. Iran Rehabil J. 2019;17:385–94.
Alizadehgoradel J, Nejati V, Sadeghi Movahed F, Imani S, Taherifard M, Mosayebi-Samani M, et al. Repeated stimulation of the dorsolateral-prefrontal cortex improves executive dysfunctions and craving in drug addiction: a randomized, double-blind, parallel-group study. Brain Stimul. 2020;13:582–93.
Xu X, Ding X, Chen L, Chen T, Su H, Li X, et al. The transcranial direct current stimulation over prefrontal cortex combined with the cognitive training reduced the cue-induced craving in female individuals with methamphetamine use disorder: a randomized controlled trial. J Psychiatr Res. 2021;134:102–10.
Eskandari Z, Mostafavi H, Hosseini M, Mousavi SE, Ramazani S, Dadashi M. A sham-controlled clinical trial to examine the effect of bilateral tDCS on craving, TNF-α and IL-6 expression levels, and impulsivity of males with opioid use disorder. J Addict Dis. 2021;39:347–56.
Taremian F, Nazari S, Moradveisi L, Moloodi R. Transcranial direct current stimulation on opium craving, depression, and anxiety: a preliminary study. J Ect. 2019;35:201.
Wang Y, Shen Y, Cao X, Shan C, Pan J, He H, et al. Transcranial direct current stimulation of the frontal-parietal-temporal area attenuates cue-induced craving for heroin. J Psychiatr Res. 2016;79:1–3.
Voges J, Muller U, Bogerts B, Munte T, Heinze H. Deep brain stimulation surgery for alcohol addiction. World Neurosurg. 2013;80:S28.e21–S28.e31.
Muller U, Sturm V, Voges J, Heinze H, Galazky I, Buntjen L, et al. Nucleus accumbens deep brain stimulation for alcohol addiction - safety and clinical long-term results of a pilot trial. Pharmacopsychiatry. 2016;49:170–3.
Davidson B, Giacobbe P, George TP, Nestor SM, Rabin JS, Goubran M, et al. Deep brain stimulation of the nucleus accumbens in the treatment of severe alcohol use disorder: a phase I pilot trial. Mol Psychiatry. 2022;27:3992–4000.
Bach P, Luderer M, Müller UJ, Jakobs M, Baldermann JC, Voges J, et al. Deep brain stimulation of the nucleus accumbens in treatment-resistant alcohol use disorder: a double-blind randomized controlled multi-center trial. Transl Psychiatry. 2023;13:11.
Kuhn J, Bauer R, Pohl S, Lenartz D, Huff W, Kim E, et al. Observations on unaided smoking cessation after deep brain stimulation of the nucleus accumbens. Eur Addict Res. 2009;15:196–201.
Kuhn J, Moller M, Treppmann J, Bartsch C, Lenartz D, Gruendler T, et al. Deep brain stimulation of the nucleus accumbens and its usefulness in severe opioid addiction. Mol Psychiatry. 2014;19:145–6.
Chen L, Li N, Ge S, Lozano AM, Lee DJ, Yang C, et al. Long-term results after deep brain stimulation of nucleus accumbens and the anterior limb of the internal capsule for preventing heroin relapse: an open-label pilot study. Brain Stimul. 2019;12:175–83.
Song S, Zilverstand A, Gui W, Li H-J, Zhou X. Effects of single-session versus multi-session non-invasive brain stimulation on craving and consumption in individuals with drug addiction, eating disorders or obesity: a meta-analysis. Brain Stimul. 2019;12:606–18.
McClintock SM, Reti IM, Carpenter LL, McDonald WM, Dubin M, Taylor SF, et al. Consensus recommendations for the clinical application of repetitive transcranial magnetic stimulation (rTMS) in the treatment of depression. J Clin Psychiatry. 2018;79:16cs10905.
Fitzgerald PB, Hoy KE, Elliot D, Susan McQueen RN, Wambeek LE, Daskalakis ZJ. Accelerated repetitive transcranial magnetic stimulation in the treatment of depression. Neuropsychopharmacology. 2018;43:1565–72.
Chen L, Hudaib A-R, Hoy KE, Fitzgerald PB. Efficacy, efficiency and safety of high-frequency repetitive transcranial magnetic stimulation applied more than once a day in depression: a systematic review. J Affect Disord. 2020;277:986–96.
Cole EJ, Stimpson KH, Bentzley BS, Gulser M, Cherian K, Tischler C, et al. Stanford accelerated intelligent neuromodulation therapy for treatment-resistant depression. Am J Psychiatry. 2020;177:716–26.
Steele VR, Maxwell AM, Ross TJ, Stein EA, Salmeron BJ. Accelerated intermittent theta-burst stimulation as a treatment for cocaine use disorder: a proof-of-concept study. Front Neurosci. 2019;13:1147.
Rachid F. Maintenance repetitive transcranial magnetic stimulation (rTMS) for relapse prevention in with depression: a review. Psychiatry Res. 2018;262:363–72.
Barr MS, George TP. Deep repetitive transcranial magnetic stimulation for smoking cessation: is going deeper better? Biol Psychiatry. 2014;76:678–80.
Isserles M, Shalev AY, Roth Y, Peri T, Kutz I, Zlotnick E, et al. Effectiveness of deep transcranial magnetic stimulation combined with a brief exposure procedure in post-traumatic stress disorder – a pilot study. Brain Stimul. 2013;6:377–83.
Carmi L, Tendler A, Bystritsky A, Hollander E, Blumberger DM, Daskalakis J, et al. Efficacy and safety of deep transcranial magnetic stimulation for obsessive-compulsive disorder: a prospective multicenter randomized double-blind placebo-controlled trial. Am J Psychiatry. 2019;176:931–8.
Berglund M, Thelander S, Salaspuro M, Franck J, Andréasson S, Öjehagen A. Treatment of alcohol abuse: an evidence-based review. Alcohol Clin Exp Res. 2003;27:1645–56.
Ward HB, Beermann A, Nawaz U, Halko MA, Janes AC, Moran LV, et al. Evidence for schizophrenia-specific pathophysiology of nicotine dependence. Front Psychiatry. 2022;13:804055.
Cash RFH, Cocchi L, Lv J, Fitzgerald PB, Zalesky A. Functional magnetic resonance imaging–guided personalization of transcranial magnetic stimulation treatment for depression. Jama Psychiatry. 2021;78:337–9.
Ahmadi J, Ekramzadeh S, Pridmore S. Remission of methamphetamine-induced withdrawal delirium and craving after electroconvulsive therapy. Iran J Psychiatry Behav Sci. 2015;9:e1793.
Daughters SB, Yi JY, Phillips RD, Carelli RM, Fröhlich F. Alpha-tACS effect on inhibitory control and feasibility of administration in community outpatient substance use treatment. Drug Alcohol Depend. 2020;213:108132.
Leong SL, Glue P, Manning P, Vanneste S, Lim LJ, Mohan A, et al. Anterior cingulate cortex implants for alcohol addiction: a feasibility study. Neurotherapeutics. 2020;17:1287–99.
de Leon J, Diaz FJ. A meta-analysis of worldwide studies demonstrates an association between schizophrenia and tobacco smoking behaviors. Schizophr Res. 2005;76:135–157.
Hasin D, Walsh C. Cannabis use, cannabis use disorder, and comorbid psychiatric illness: a narrative review. J Clin Med. 2021;10:15.
Funding
This project was supported by a National Institute on Drug Abuse (NIDA) grant (R21-DA-043949) to Dr. Tony George.
Author information
Authors and Affiliations
Contributions
TG conceptualized the presented review. DM and AP conducted the literature searches, quality assessments, and extraction of data. DM conducted the meta-analysis and created the Forest Plots, with help and guidance from MSan and MSor DM and AP analyzed and interpreted the results, and designed the tables. DM designed the figures. DM and AP wrote the first draft of the manuscript. HW, VT, VS, CH, and TG oversaw its revision and encouraged DM and AP to explore specific topics for the review. All authors contributed to and have approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
TPG is a co-principal editor at Neuropsychopharmacology. CAH is employed by BrainsWay and has a financial interest in the company. The remaining authors have nothing to disclose.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mehta, D.D., Praecht, A., Ward, H.B. et al. A systematic review and meta-analysis of neuromodulation therapies for substance use disorders. Neuropsychopharmacol. 49, 649–680 (2024). https://doi.org/10.1038/s41386-023-01776-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-023-01776-0
- Springer Nature Switzerland AG
This article is cited by
-
Discriminative functional connectivity signature of cocaine use disorder links to rTMS treatment response
Nature Mental Health (2024)
-
What can neuroimaging of neuromodulation reveal about the basis of circuit therapies for psychiatry?
Neuropsychopharmacology (2024)
-
Pump the brakes: identifying neurobiological targets to enhance inhibitory control in drug addiction
Neuropsychopharmacology (2024)
-
Understanding and Treating Dual Diagnosis: Double the Trouble
Current Addiction Reports (2024)
-
The Brain Salience Network at the Intersection of Pain and Substance use Disorders: Insights from Functional Neuroimaging Research
Current Addiction Reports (2024)