Abstract
Postpartum adversity is among the strongest predictors for the emergence of postpartum depression (PPD) in humans and a translational risk factor employed in rodent models. Parental care is disturbed under conditions of environmental adversity, including low resource environments, and in PPD. Nonetheless, the neural changes associated with these adversity-induced maladaptive behavioral states remain poorly understood. Postpartum scarcity-adversity can be modeled in rats by providing the dam with limited bedding and nesting (LBN) materials, which mimics the effects of a stressful low resource environment in potentiating maltreatment/neglect in humans. Indeed, LBN exposure from postpartum days (PD) 2–9 increased adverse maternal behaviors, impaired pup retrieval, and increased passive stress coping responses. Since mesolimbic dopamine (DA) activity is an important mechanism for motivated maternal behavior and is implicated in PPD, we assessed the impact of postpartum scarcity-adversity on in vivo electrophysiological properties of ventral tegmental area (VTA) DA neurons at two timepoints. We found reduced numbers of active VTA DA neurons in LBN dams at PD 9-10 but not PD-21, suggesting a transient impact on VTA population activity in LBN dams. Finally, we assessed the impact of early life scarcity-adversity on VTA DA function by conducting VTA recordings in adult female offspring and found a long-lasting attenuation in DA activity. These findings highlight a link between adversity-induced deficits in DA function and disrupted maternal behavior, suggesting the VTA/mesolimbic DA system as a potential mechanism by which postpartum scarcity-adversity drives aberrant maternal behavior, and early postnatal programming of adult VTA function in the offspring.
Similar content being viewed by others
Introduction
Exposure to chronic stress or adversity shortly after giving birth is one of the strongest predictors for the emergence of postpartum depression (PPD) in humans [1,2,3] and a translational risk factor widely employed in relevant rodent models [4,5,6,7]. Postpartum stressors (chronic social stress, learned helplessness) impair maternal care, disrupt mother-infant interactions, and increase negative affect (i.e., anxiety and depressive-like behaviors) in rat dams [8,9,10,11]. This is consistent with clinical literature indicating that postpartum adversity compromises the quality of mother-infant interactions and that depressed mothers have difficulty caring for and are less responsive to their infant’s needs [12,13,14,15]. Thus, postpartum adversity drives aberrant maternal behaviors and disrupted mother-infant interactions, which are hallmark features of PPD.
Parental care is disturbed under conditions of adversity and/or chronic stress, which can take a variety of forms in both humans and rodents. In humans, low socioeconomic status (SES) or poverty is associated with higher levels of physiological and psychological stress in the mother-infant dyad due to unpredictable circumstances, restricted resources in their environment [16,17,18] as well as disrupted mother–child interactions, which negatively influence growth and development in the offspring [19,20,21]. Indeed, growing up in a low SES environment is linked to substantially worse health outcomes in the offspring, including impaired emotional development and higher rates of mood disorders [16, 18, 22,23,24,25,26]. Low SES can be modeled in rodents by depriving the rat dam of adequate nesting materials during the early postpartum period, which results in an impoverished nesting environment, thereby representing a form of scarcity-adversity for the dam and pups [27, 28]. Since bedding type and volume are important components of the dam’s nesting environment, limited bedding and nesting (LBN) constitutes a continuous stressor and results in higher corticosterone (CORT) levels as well as unpredictable, adverse maternal caregiving of pups (i.e., roughhousing, maltreatment) [28,29,30,31]. Most studies have employed LBN within the context of early life adversity and focused on long-term effects in the male offspring, which exhibit increased depression-related behaviors such as: passive coping, social withdrawal, blunted reward-related responses (i.e., anhedonia) and compromised cortical and limbic function and connectivity [32,33,34,35,36,37,38]. However, the offspring effects of early life scarcity-adversity are thought to be mediated by disrupted caregiving behaviors in the dam/mother [19, 27, 39, 40], suggesting maladaptive maternal behaviors as the behavioral mechanism by which postpartum scarcity-adversity programs offspring development. Yet, there is no data on the neural changes induced by LBN exposure in the dam that coexist with aberrant maternal behavior. This underscores the need to understand neural processes by which scarcity-adversity influences maternal brain function to drive maladaptive behavioral states.
Maternal behaviors including pup contact and nipple attachment activate the mesolimbic dopamine (DA) system and increase DA release, suggesting high reward value of these pup-dam interactions [41,42,43,44]. This is consistent with the VTA’s role as part of the maternal caregiving neuronal network, which is highly sensitive to interactions occurring between dams and their offspring [44,45,46,47]. Importantly, lesions of the ventral tegmental area (VTA) disrupt maternal behavior in postpartum rats [48]. Moreover, structural and functional deficits within the mesolimbic DA system have also been reported in PPD, and these are linked to impaired maternal care and responses to infants among women [7, 14]. Thus, compromised activity of VTA DA neurons induced by postpartum adversity could not only contribute to postpartum negative affect (depressive-like behavior) but may also interfere with reward-related processes necessary for maternal motivation and responsiveness.
We assessed the effects of postpartum environmental adversity (i.e., scarcity-adversity via LBN) on mother-infant interactions, motivated maternal behavior, stress coping and VTA DA neuron activity in the rat dam. We hypothesized that LBN would have a negative impact on these parameters. To evaluate intergenerational effects of postpartum scarcity-adversity on VTA activity in females, a subset of female pups was retained until adulthood and underwent electrophysiological recordings. We focused on females to evaluate the intergenerational impact of scarcity-adversity on the female DA system by studying early postpartum adversity effects (dams) and early life adversity effects (F1- pups) and because most LBN studies have emphasized long-term effects in male offspring, although LBN dams more often maltreat female pups [49]. Consistent with prior studies showing early life LBN rearing results in long-term changes in offspring reward- and depression-related behaviors suggesting mesolimbic DA system dysfunction (i.e., anhedonia) [32, 37, 38, 50], we hypothesized that early life LBN rearing would program an enduring attenuation of VTA DA activity in females.
Materials and methods
Animals
Primiparous timed-pregnant Sprague-Dawley rat dams were obtained from Envigo (Indianapolis, IN) on gestational day 13, housed individually and maintained in a quiet, temperature-controlled room on a 12-h light/dark cycle (7 PM lights off/7 AM lights on) with unlimited access to food and water. Parturition was verified daily; the day of birth was designated as postpartum day (PD) 0. Dams were randomly assigned to experimental conditions on PD 2 and remained in their condition until VTA recording (PD 9–10). A separate cohort of dams were used for behavioral testing, and a subset of female offspring were weaned at PD 23 and used for electrophysiological recordings during adulthood (+PD 90; Fig. 1). All experiments were carried out according to NIH guidelines and were approved by the University of Pittsburgh Institutional Animal Care and Use Committee.
A Dams gave birth (PD 0) and were assigned to control (CON) or postpartum scarcity-adversity (LBN) conditions from PD 2–9. Home cage observations were conducted 3× per day, daily from PD 2–6 during both light/dark cycle periods. Dams underwent electrophysiological recordings of the VTA during the last days of LBN (PD 9–10) using an acute, terminal procedure. A separate cohort of dams underwent LBN from PD 2–9 and VTA recordings at PD 20–21 to test for duration of effects. B A separate cohort of dams were housed in a CON or LBN cage from PD 2–9 and were tested for pup retrieval at PD7 and in the FST at PD 9. LBN was ended on the afternoon of PD 9 and dams were kept in normal bedding conditions until weaning of pups. A subset of female pups was kept for assessing long-term effects of early postnatal scarcity-adversity in the adult offspring. C Female pups were born and reared with a CON or LBN mother from PD 2–9 and weaned at PD 21. Rats were kept in pairs and undisturbed (except for weekly animal care) until they reached adulthood, at which point they were used for VTA DA neuron recordings. PD postpartum day, CON control, LBN limited bedding and nesting, VTA ventral tegmental area, FST forced swim test, DA dopamine.
LBN and maternal behavior observations
On PD 2, litters were culled to 10 pups with equal sex distribution (5 male, 5 female) and dams were randomly assigned to LBN (scarcity-adversity: 500 mL, 0.5′′ layer) or control bedding cages (2500 mL, ~4′′ layer), where they remained until the end of PD9. Naturalistic maternal behavior observations were conducted within the home cage and recorded three times a day (daily, PD 2–6) in 30-min sessions (morning/afternoon/night), as described previously [29, 36, 38]. The rationale for selecting these days was based on a prior study in LBN dams which identified the most marked behavioral effects within this range (PD2-5) [29]. We focused on adverse caregiving behaviors, which refer to aggressive behavior of the dam towards the pup, including behaviors that place pups at increased risk of threat or deprivation such as: rough/improper transport (carrying pup by limbs), dragging pups, stepping on pups, shoving pups away, trampling the nest, as these are consistently disrupted by adversity [27, 51, 52]. Each 30-min observation period was divided into 5-min segments during which behaviors were scored as occurring or not. Percentages of behaviors were obtained by dividing the number of segments in which a particular behavior was scored over the total number of observation segments on all days. Observation sessions conducted during light cycle (morning/afternoon) were grouped together and scored separately from those occurring during the dark cycle (night).
Behavioral testing
Testing occurred between 11:00 AM and 4:00 PM. Behavioral tests were videotaped and scored by a blind experimenter.
Pup retrieval
Cages were moved into the testing room for a 30-min acclimation period. The dam was placed in a testing apparatus (56 × 43 × 44 cm, made of opaque black Plexiglas) for a 5-min habituation period that also served as a short separation from pups. After 5 min, each of the 4 pups were placed in a Petri dish (1 per dish) containing a handful of home cage bedding in each corner of the apparatus (1 dish × corner). Latency to retrieve at least 75% of the pups was scored over a 10-min test period. This criterion was selected given that some dams in both groups retrieved 3 pups to the same corner (already containing a pup).
Forced swim test (FST)
Rats underwent the FST using a standard procedure consisting of 15-min swimming session on day 1 and a 5-min test on the following day in a cylinder filled with water (25 ± 1 °C) up to 38 cm, as described previously [53,54,55].
In vivo electrophysiology of VTA DA neurons
Electrophysiological recordings were conducted in rat dams at PD 9–10 or PD 20–21 (no behavioral testing exposure) and in a subset of adult female offspring as described previously [53,54,55]. Briefly, rats were anesthetized with chloral hydrate (400 mg/kg, i.p.; Sigma), mounted on a stereotaxic frame (Kopf, Tujunga, CA) and glass electrodes were lowered through 6–9 vertical tracks in a predetermined pattern within the VTA [AP: 5.4–5.7 mm; ML: 0.6–1.0 mm from bregma, and dorsoventral (DV): 6.5–9.0 mm from dura]. Only animals with a minimum of 6 tracks within 0.4 mm of target coordinates were included. All DA neurons recorded were found between 5.0 and 6.1 mm AP from bregma, and primarily from rostral VTA [56, 57], which is consistent with our prior work regarding stress effects on VTA population activity [53, 55, 58, 59]. Spontaneously active DA neurons were identified based on well-established criteria [60]. See Supplementary Methods for details.
Statistical analysis
Behavioral and electrophysiological data with a normal distribution were analyzed using unpaired t-tests; data sets deviating from the normal distribution were analyzed using Mann–Whitney U tests. Electrophysiological data of DA neurons was collected with Powerlab Lab Chart (AD Instruments) to identify spike time courses and exported to Neuroexplorer (NEX Technologies, NexTech Systems) software to calculate firing rate and burst firing. Statistics were calculated using GraphPad Prism 9.0 and differences were considered significant at p < 0.05. Statistical outliers were identified using QuickCalcs Grubbs test (GraphPad) and excluded from analysis.
Results
Postpartum scarcity-adversity, as modeled by LBN from PD 2–9, increases adverse maternal behavior
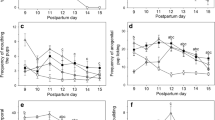
LBN dams exhibited increases in negative maternal behaviors directed at pups and nest-building compared to control dams (n = 7/group) across the light/dark cycle (Fig. 2). LBN dams exhibited increased percentages of time stepping on pups (U = 1.5, p = 0.0017), dragging pups (t12 = 4.268, p = 0.0011), transporting pups (U = 0, p = 0.0006), and shoving pups away (t12 = 3.859, p = 0.0023) during the daytime observation sessions (Fig. 1A–D). LBN dams also spent greater percentages of time nest-building (t12 = 3.077, p = 0.0096) and with pups scattered throughout the cage (U = 2.5, p = 0.0029), (Fig. 1E, F). LBN dams exhibited greater percentages of time stepping on pups (U = 0; p = 0.0006), dragging pups (t12 = 3.742, p = 0.0028), transporting pups (t12 = 4.596, p = 0.0006), pushing/shoving pups away (t12 = 2.274, p = 0.0422), nest-building (U = 0.5, p = 0.0012) and with a scattered litter (t12 = 2.307, p = 0.0397) during nighttime observation sessions (Fig. 1G–L). These data are summarized in Supplementary Table 1.
During the light cycle observation sessions, LBN dams exhibited increased percentages of A stepping (p = 0.0017), B dragging (p = 0.0011), C transporting pups (p = 0.0006), and D shoving pups away (p = 0.0023) (n = 7/group). LBN dams also spent more time nest-building (p = 0.0096) and with scattered litter (p = 0.0029). LBN dams also spent greater percentages of time G stepping pups (p = 0.0006), H dragging pups (p = 0.0028), I transporting pups (p = 0.0006), J pushing or shoving pups away (p = 0.04), K nest-building (p = 0.0012) and L with a scattered litter (p = 0.04) during nighttime observation sessions (n = 7/group). Error bars represent mean ± SEM. Gray bars represent control dams (CON), pink bars represent dams that underwent scarcity-adversity (LBN). *p < 0.05, **p < 0.01, ***p < 0.001.
LBN dams exhibit impaired pup retrieval and passive FST coping
A separate cohort of dams (CON: n = 7, LBN: n = 9) was used to determine the effects of postpartum scarcity-adversity on pup retrieval (PD 7) and the FST (PD 9) (Fig. 1C). Compared to controls, LBN dams exhibited longer latencies to retrieve pups (Mann–Whitney: U = 8, p = 0.0107) (Fig. 3A) and exhibited increased FST immobility compared to CON dams (t-test: t14 = 2.684, p = 0.0178) (Fig. 3B).
A Compared to controls, LBN dams exhibited longer latencies to retrieve pups (Mann–Whitney: U = 8, p = 0.01) at PD 7 (n = 7–9/group). B When tested in the FST at PD 9, these same dams (i.e., LBN) also exhibited increased immobility duration compared to CON (t-test: t14 = 2.684, p = 0.02; n = 7–9/group), suggesting an enhanced passive coping response induced by postpartum adversity. Error bars represent mean ± SEM. *p < 0.05.
LBN dams exhibit attenuated VTA population activity on PD 9–10 but not PD 20–21
Electrophysiological recordings were conducted on PD 9-10 (last LBN day) (Fig. 4A, B) in the same cohort of CON/LBN dams monitored previously for maternal behavior (CON: n = 5, 47 neurons; LBN: n = 5, 17 neurons; Fig. 1A). LBN dams exhibited attenuated VTA population activity, as indexed by lower numbers of spontaneously active DA neurons compared to CON dams (t8 = 5.419, p = 0.0006; Fig. 4C). No differences were detected for average firing rate (t62 = 0.1844, p = 0.8543; Fig. 4D) or percentage of spikes fired in bursts (U = 388, p = 0.8683; Fig. 4E).
A VTA DA neuron sampling: population activity was assessed by passing the electrode trough the VTA in a predetermined grid pattern (3 × 3) of 9 tracks separated by 0.2 mm. B A representative dopaminergic neuron waveform. C At PD 9–10, LBN dams exhibited attenuated VTA population activity, as indexed by a reduction in the number of spontaneously active DA neurons found, compared to CON dams (t-test: p = 0.0006, n = 5/group). D No effect of LBN on DA neuron firing rate (p = 0.85) at PD 9–10. E No effect of LBN was found for percentage of spikes firing in burst (p = 0.87) at PD-9–10. F When assessed at PD 20–21, no differences in VTA population were found between CON and LBN dams for VTA population activity (p = 0.54; n = 5/group). G At PD 20–21, LBN and CON dams exhibited comparable DA neuron firing rate (p = 0.34). H No difference between LBN and CON dams in percentage of spikes firing in burst (p = 0.97) at PD 20–21. Error bars represent mean ± SEM. ***p < 0.001.
To determine whether there are distinct short- and long-term effects of postpartum adversity on maternal VTA function, a separate cohort of dams underwent CON/LBN from PD 2–9 and recordings on PD 20–21, ~11–12 days post-LBN exposure (CON: n = 5, 25 neurons; LBN: n = 5: 25 neurons; Fig. 4A, B). No differences were found between CON and LBN dams for VTA population activity (t8 = 0.6439, p = 0.5377; Fig. 4F), basal firing rate (t48 = 0.9620, p = 0.3409; Fig. 4G) or percentage of spikes fired in bursts (U = 310, p = 0.9693; Fig. 4H), suggesting that LBN-induced attenuation of VTA population activity is transient and likely confined to the duration of scarcity-adversity exposure. A summary of electrophysiological results in CON and LBN dams at both timepoints are summarized in Supplementary Table 2.
Adult female offspring of LBN dams exhibit long-lasting attenuation in VTA DA activity
Electrophysiological recordings were conducted in a subset of adult female offspring (CON: n = 8, 55 neurons; LBN: n = 8: 36 neurons; Fig. 5). Adult female rats reared with an LBN dam from PD 2–9 exhibited 75% fewer active VTA DA cells (blunted VTA population activity) compared to rats reared with CON dams (Mann–Whitney: U = 4.5, p = 0.0022; Fig. 5A), 26% increased basal firing rate (U = 486, p = 0.0091; Fig. 5B) but no change in percentage of spikes firing in burst (U = 775, p = 0.0785; Fig. 5C). These data are summarized in Supplementary Table 3.
A Adult female rats that were reared under early life scarcity-adversity conditions (i.e., LBN dam PD 2–9) had a reduced number of active VA DA neurons compared to adult female rats reared with CON dams (Mann–Whitney: U = 4.5, p = 0.0022; n = 8/group). B Adult female rats reared with LBN dams exhibited increased basal DA neuron firing rate (U = 486, p = 0.0091). C No difference between CON and LBN offspring in percentage of spikes firing in burst (p = 0.08). Error bars represent mean ± SEM. **p < 0.01.
Discussion
In humans, low SES environments constitute a form of scarcity-adversity that places parents at greater risk for disrupted caregiving, including increased negative parenting behaviors [21, 61]. We show that scarcity-adversity can be modeled in rodents by depriving the dam of adequate nesting materials during the early postpartum (i.e., LBN PD 2–9), which increased negative maternal behaviors directed at pups and impaired maternal responses, thereby mimicking the effects of adverse environments (low SES) in potentiating negative parenting behaviors such as maltreatment and neglect in humans [27, 61, 62].
Postpartum scarcity-adversity increases negative maternal behaviors and impairs goal-directed and stress-related responses in the dam
Compared to CON dams, LBN dams exhibited increased adverse maternal behaviors that are disruptive to pups: stepping, dragging, shoving away, and improper transport. Similar results were obtained during the dark cycle, suggesting that the LBN-induced expression of negative pup-directed maternal behaviors is present throughout the circadian cycle. These findings are consistent with prior reports indicating that dams exposed to LBN during PD 8–12 spent higher percentages of time stepping on pups, transporting, and dragging of pups, which coexisted with an increase in nest-building by the dam and a fragmented, unpredictable nest environment for pups (scattered litter) [27, 36, 51, 63, 64]. Thus, the effects of LBN exposure during the first 2 weeks postpartum on maternal behavior parallel the clinical literature showing that children living in low SES or poverty conditions are more likely to experience increased exposure to negative parenting behaviors such as maltreatment and/or neglect [61, 62].
We also examined the impact of postpartum scarcity-adversity on goal-directed maternal responses and coping responses to an acute uncontrollable stressor. When tested for pup retrieval in a novel environment, LBN dams exhibited longer latencies compared to CON dams. Similarly, dams or adult female offspring (F1) exposed to chronic social stress from PD 2-16 also take longer to gather/retrieve their pups on PD 9 [10, 65], suggesting that postpartum stressors converge in impairing pup retrieval behavior. LBN dams also exhibited greater FST immobility at PD 9, suggesting a passive coping response which has previously been interpreted as higher depressive-like behavior [66,67,68]. Early postpartum dams exposed to gestational restraint stress also spend more time immobile in the FST [69, 70], suggesting that psychosocial stressors during pregnancy/postpartum converge in augmenting passive FST coping responses. Furthermore, increased FST immobility is commonly observed after chronic stress exposure and in preclinical models useful for the study of depression and postpartum mood disorders ([5, 6, 55, 71,72,73,74]). Collectively, these data show that postpartum scarcity-adversity impairs maternal goal-directed pup responses and produces divergent (i.e., passive) stress coping strategies. This is in agreement with prior reports indicating both deficits in maternal behavior and passive FST coping as common behavioral endpoints in a variety of preclinical rodent models relevant for the study of PPD, including non-stress based (e.g., genetic, hormone) models [72, 75,76,77,78]. Thus, our findings recapitulate the link between disrupted maternal behaviors and increased depression-related phenotypes in rodent models of PPD.
Postpartum scarcity-adversity induces a transient hypodopaminergic state in the rat dam
This study extends the current knowledge regarding adversity-induced changes in the maternal brain by demonstrating that early postpartum scarcity-adversity (LBN PD 2–9) compromises in vivo maternal mesolimbic DA function in the dam and induces distinct short- versus long-term effects. This was characterized by an hypodopaminergic state in LBN dams compared to CON dams at PD 9–10 but not PD 20–21, as indexed by a reduction in the number of active DA neurons within the VTA (i.e., blunted population activity = lower numbers of cells per track). This finding is in accordance with our prior studies on stress-induced plasticity of VTA DA neurons showing that adult male and female rats exposed to learned helplessness or chronic mild stress exhibit a selective attenuation in VTA population activity indicative of DA system downregulation, particularly in the medial reward-related population [53, 58, 73, 79,80,81,82]. Moreover, reduced VTA population activity in LBN dams is also consistent with our recent study showing a hypodopaminergic state in a novel rodent model useful for the study of PPD based on social adversity (permanent pup removal at PD1) [55]. Thus, postpartum scarcity-adversity mimics the effects of adult stress paradigms on VTA DA neuron function previously reported by our group and is consistent with stress-induced VTA plasticity in preclinical rodent models of depression [83, 84]. This is significant because the LBN paradigm is used by >15 groups as a model of early life stress, but this model’s effectiveness is dependent on adversity-induced changes in maternal caregiving behaviors, which negatively impact offspring development [28]. Within this context, our study is the first to show that postpartum scarcity-adversity (LBN) compromises maternal mesolimbic DA function by decreasing the number of active VTA DA neurons, thereby resulting in attenuation of DA neuron response to stimuli that we propose drives negative affect, anhedonia (e.g., depression-like states) and aberrant maternal behavior in postpartum females [44, 85]. Importantly, our findings of increased negative maternal behaviors towards pups, impaired pup retrieval, and VTA DA deficit in LBN dams are consistent with prior studies reporting disruptions in rat maternal behavior, including deficits in pup retrieval, following temporary pharmacological VTA inactivation [86, 87], 6-OHDA VTA lesions [88] or systemic DA signaling blockade via DA receptor antagonism [88,89,90,91]. Taken together, these data highlight the importance of mesolimbic DA system integrity, particularly within the VTA, for the adequate expression of maternal behaviors and suggest compromised VTA function (i.e., VTA hypoactivity leading to mesolimbic DA downregulation) as a mechanism by which LBN disrupts mother-infant interactions and drives maladaptive behavioral states in the dam.
Our prior work has shown normative, time-dependent effects of parturition on immobility behavior and VTA DA neuron activity, in which early postpartum dams (PD 1–3) exhibit increased immobility and blunted VTA population activity compared to virgins [54, 74]. Thus, early postpartum rodents exhibit a transient, normative negative affect state shortly after giving birth, which is analogous to the postpartum blues commonly reported in human mothers [92,93,94]. Within the context of this work, early postpartum LBN exposure (PD 2–9) appears to prolong depression-related neurobehavioral phenotypes (e.g., FST immobility, hypodopaminergic state), as suggested by our finding of higher immobility duration and reduced VTA DA activity during PD 9–10 in LBN dams. In accordance, postpartum adversity is a risk factor that can interact with postpartum blues and lead to PPD in human mothers [3, 15], suggesting translational relevance of early postpartum LBN exposure and usefulness for modeling constructs relevant to the study of PPD.
Postpartum scarcity-adversity induces an enduring hypodopaminergic state in adult female offspring
Exposure to early life adversity results in later-life changes in reward-related behaviors, which likely reflect long-term alterations in mesolimbic DA function [50]. For example, adolescent and adult rats exposed to early life LBN exhibit decreases in sucrose consumption and palatable food preference, reduced social play, and attenuated social motivation [32, 35, 37, 38, 95]. Furthermore, adolescent rats exposed to early life LBN display blunted social motivation that is linked to reduced Fos activation within mesolimbic DA targets, including the nucleus accumbens, in response to typically rewarding (social) stimuli [37]. Thus, early life LBN exposure induces persistent anhedonia in the offspring, as indexed by decreases in reward-related behaviors throughout the lifetime.
Although previous studies using LBN have identified long-term reward-related deficits in LBN offspring, little is known regarding the impact of early life LBN exposure on adult mesolimbic DA function. Therefore, we assessed long-term effects of LBN exposure on VTA function in the adult female offspring. We focused on females to evaluate the intergenerational impact of scarcity-adversity on the female DA system by comparing early postpartum adversity effects (dams) and early life adversity effects (F1- pups) and because female LBN dams more often maltreat female pups [49]. Early life scarcity-adversity induced an enduring attenuation in VTA population activity and increased firing rate, although the increase in firing rate was small in magnitude compared to the decreased responsivity engendered by a 75% decrease in population activity. The effect on population activity in adult female offspring was comparable to LBN dams at PD 9–10, which also exhibited VTA DA neuron hypoactivity, but divergent from those observed after the cessation of LBN exposure (PD 20–21) in which there was no difference in VTA activity between CON and LBN dams. This finding is significant because it highlights: (i) the intergenerational impact of scarcity-adversity in VTA function of female rats (dam + pup), and (ii) that the impact of scarcity-adversity on female VTA function is dependent on the developmental stage at which it occurs: postpartum LBN exposure induces a transient hypodopaminergic state observed at PD 9–10 but not PD 20–21 in the rat dam, whereas early life LBN exposure (via rearing with an LBN dam) induces a long-lasting hypodopaminergic state (>PD90) in the adult offspring. In accordance, a recent study conducted by our group in female rats showed no long-term impact of adolescent stress exposure on adult VTA population activity [96]. Collectively, these data suggest that early postnatal life (PD 2–9) may constitute a sensitive period for scarcity-adversity induced programming of an adult hypodopaminergic state in female rats.
In sum, postpartum scarcity-adversity induced aberrant (i.e., adverse) maternal behaviors in the dam, impaired pup retrieval responses and increased passive FST coping. These results overlap with those observed in rodent models relevant to PPD, in which deficits in maternal care and FST immobility are common behavioral outputs. These behavioral changes were accompanied by blunted VTA DA neuron activity at PD 9–10 but not PD 20–21, indicating temporary DA downregulation (i.e., hypodopaminergic state) in LBN dams that recovers after LBN cessation. In contrast, long-lasting VTA effects were observed in adult female offspring reared by LBN dams, suggesting early life programming of adult VTA function by scarcity-adversity and an intergenerational impact of scarcity-adversity on mesolimbic DA function. These findings highlight a link between adversity-induced deficits in DA function and disrupted maternal behavior, suggesting the VTA/mesolimbic system as a potential mechanism by which postpartum scarcity-adversity drives disrupted maternal behavior and therapeutic target for ameliorating impairments in maternal care induced by stress exposure and/or PPD.
References
Bloch M, Daly RC, Rubinow DR. Endocrine factors in the etiology of postpartum depression. Compr Psychiatry. 2003;44:234–46.
Payne JL, Maguire J. Pathophysiological mechanisms implicated in postpartum depression. Front Neuroendocrinol. 2019;52:165–80.
Yim IS, Tanner Stapleton LR, Guardino CM, Hahn-Holbrook J, Dunkel Schetter C. Biological and psychosocial predictors of postpartum depression: systematic review and call for integration. Annu Rev Clin Psychol. 2015;11:99–137.
Brummelte S, Galea LA. Depression during pregnancy and postpartum: contribution of stress and ovarian hormones. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34:766–76.
Li M, Chou SY. Modeling postpartum depression in rats: theoretic and methodological issues. Dongwuxue Yanjiu. 2016;37:229–36.
Perani CV, Slattery DA. Using animal models to study post-partum psychiatric disorders. Br J Pharm. 2014;171:4539–55.
Post C, Leuner B. The maternal reward system in postpartum depression. Arch Women’s Ment Health. 2019;22:417–29.
Brummelte S, Galea LA. Postpartum depression: etiology, treatment and consequences for maternal care. Horm Behav. 2016;77:153–66.
Kurata A, Morinobu S, Fuchikami M, Yamamoto S, Yamawaki S. Maternal postpartum learned helplessness (LH) affects maternal care by dams and responses to the LH test in adolescent offspring. Horm Behav. 2009;56:112–20.
Murgatroyd CA, Taliefar M, Bradburn S, Carini LM, Babb JA, Nephew BC. Social stress during lactation, depressed maternal care, and neuropeptidergic gene expression. Behav Pharm. 2015;26:642–53.
Nephew BC, Bridges RS. Effects of chronic social stress during lactation on maternal behavior and growth in rats. Stress. 2011;14:677–84.
Field T. Postpartum depression effects on early interactions, parenting, and safety practices: a review. Infant Behav Dev. 2010;33:1–6.
Kim P. How stress can influence brain adaptations to motherhood. Front Neuroendocrinol. 2020;60:100875.
Morgan JK, Guo C, Moses-Kolko EL, Phillips ML, Stepp SD, Hipwell AE. Postpartum depressive symptoms moderate the link between mothers’ neural response to positive faces in reward and social regions and observed caregiving. Soc Cogn Affect Neurosci. 2017;12:1605–13.
Thurgood S, Avery DM, Williamson, Lloyda. Postpartum depression (PPD). Am J Clin Med. 2009;6:17–22.
Evans GW. The environment of childhood poverty. Am Psychol. 2004;59:77–92.
Farah MJ. The neuroscience of socioeconomic status: correlates, causes, and consequences. Neuron. 2017;96:56–71.
Hackman DA, Farah MJ, Meaney MJ. Socioeconomic status and the brain: mechanistic insights from human and animal research. Nat Rev Neurosci. 2010;11:651–9.
Devenish B, Hooley M, Mellor D. The pathways between socioeconomic status and adolescent outcomes: a systematic review. Am J Community Psychol. 2017;59:219–38.
Larson CP. Poverty during pregnancy: Its effects on child health outcomes. Paediatr Child Health. 2007;12:673–7.
McLoyd VC. Socioeconomic disadvantage and child development. Am Psychol. 1998;53:185–204.
Koster A, Bosma H, Kempen GI, Penninx BW, Beekman AT, Deeg DJ, et al. Socioeconomic differences in incident depression in older adults: the role of psychosocial factors, physical health status, and behavioral factors. J Psychosom Res. 2006;61:619–27.
McLaughlin KA, Breslau J, Green JG, Lakoma MD, Sampson NA, Zaslavsky AM, et al. Childhood socio-economic status and the onset, persistence, and severity of DSM-IV mental disorders in a US national sample. Soc Sci Med. 2011;73:1088–96.
Odgers CL. Income inequality and the developing child: Is it all relative? Am Psychol. 2015;70:722–31.
Bradley RH, Corwyn RF. Socioeconomic status and child development. Annu Rev Psychol. 2002;53:371–99.
Brooks-Gunn J, Duncan GJ. The effects of poverty on children. Future Child. 1997;7:55–71.
Perry RE, Finegood ED, Braren SH, Dejoseph ML, Putrino DF, Wilson DA, et al. Developing a neurobehavioral animal model of poverty: Drawing cross-species connections between environments of scarcity-adversity, parenting quality, and infant outcome. Dev Psychopathol. 2019a;31:399–418.
Walker CD, Bath KG, Joels M, Korosi A, Larauche M, Lucassen PJ, et al. Chronic early life stress induced by limited bedding and nesting (LBN) material in rodents: critical considerations of methodology, outcomes and translational potential. Stress. 2017;20:421–48.
Ivy AS, Brunson KL, Sandman C, Baram TZ. Dysfunctional nurturing behavior in rat dams with limited access to nesting material: a clinically relevant model for early-life stress. Neuroscience. 2008;154:1132–42.
Bolton JL, Short AK, Simeone KA, Daglian J, Baram TZ. Programming of stress-sensitive neurons and circuits by early-life experiences. Front Behav Neurosci. 2019;13:30.
Rincon-Cortes M, Sullivan RM. Early life trauma and attachment: immediate and enduring effects on neurobehavioral and stress axis development. Front Endocrinol (Lausanne). 2014;5:33.
Bolton JL, Molet J, Regev L, Chen Y, Rismanchi N, Haddad E, et al. Anhedonia following early-life adversity involves aberrant interaction of reward and anxiety circuits and is reversed by partial silencing of amygdala corticotropin-releasing hormone gene. Biol Psychiatry. 2018;83:137–47.
Bolton JL, Ruiz CM, Rismanchi N, Sanchez GA, Castillo E, Huang J, et al. Early-life adversity facilitates acquisition of cocaine self-administration and induces persistent anhedonia. Neurobiol Stress. 2018;8:57–67.
Perry RE, Rincon-Cortes M, Braren SH, Brandes-Aitken AN, Opendak M, Pollonini G, et al. Corticosterone administration targeting a hypo-reactive HPA axis rescues a socially-avoidant phenotype in scarcity-adversity reared rats. Dev Cogn Neurosci. 2019;40:100716.
Raineki C, Cortes MR, Belnoue L, Sullivan RM. Effects of early-life abuse differ across development: infant social behavior deficits are followed by adolescent depressive-like behaviors mediated by the amygdala. J Neurosci. 2012;32:7758–65.
Raineki C, Sarro E, Rincon-Cortes M, Perry R, Boggs J, Holman CJ, et al. Paradoxical neurobehavioral rescue by memories of early-life abuse: the safety signal value of odors learned during abusive attachment. Neuropsychopharmacology. 2015;40:906–14.
Rincon-Cortes M, Sullivan RM. Emergence of social behavior deficit, blunted corticolimbic activity and adult depression-like behavior in a rodent model of maternal maltreatment. Transl Psychiatry. 2016;6:e930.
Yan CG, Rincon-Cortes M, Raineki C, Sarro E, Colcombe S, Guilfoyle DN, et al. Aberrant development of intrinsic brain activity in a rat model of caregiver maltreatment of offspring. Transl Psychiatry. 2017;7:e1005.
Finegood ED, Blair C, Granger DA, Hibel LC, Mills-Koonce R, Family Life Project Key I. Psychobiological influences on maternal sensitivity in the context of adversity. Dev Psychol. 2016;52:1073–87.
Huston AC, Bentley AC. Human development in societal context. Annu Rev Psychol. 2010;61:411–437. C411.
Champagne FA, Chretien P, Stevenson CW, Zhang TY, Gratton A, Meaney MJ. Variations in nucleus accumbens dopamine associated with individual differences in maternal behavior in the rat. J Neurosci. 2004;24:4113–23.
Fleming AS, Korsmit M, Deller M. Rat pups are potent reinforcers ot the maternal animal: effects of experience, parity, hormones and dopamine function. Psychobiology. 1994;22:44–53.
Hansen S, Bergvall AH, Nyiredi S. Interaction with pups enhances dopamine release in the ventral striatum of maternal rats: a microdialysis study. Pharm Biochem Behav. 1993;45:673–6.
Rincon-Cortes M, Grace AA. Adaptations in reward-related behaviors and mesolimbic dopamine function during motherhood and the postpartum period. Front Neuroendocrinol. 2020;57:100839.
Grieb ZA, Vitale EM, Morrell JI, Lonstein JS, Pereira M. Decreased mesolimbic dopaminergic signaling underlies the waning of maternal caregiving across the postpartum period in rats. Psychopharmacology (Berl). 2020;237:1107–19.
Numan M. Motivational systems and the neural circuitry of maternal behavior in the rat. Dev Psychobiol. 2007;49:12–21.
Pereira M, Morrell JI. Functional mapping of the neural circuitry of rat maternal motivation: effects of site-specific transient neural inactivation. J Neuroendocrinol. 2011;23:1020–35.
Numan M, Stolzenberg DS. Medial preoptic area interactions with dopamine neural systems in the control of the onset and maintenance of maternal behavior in rats. Front Neuroendocrinol. 2009;30:46–64.
Keller SM, Nowak A, Roth TL. Female pups receive more maltreatment from stressed dams. Dev Psychobiol. 2019;61:824–31.
Birnie MT, Kooiker CL, Short AK, Bolton JL, Chen Y, Baram TZ. Plasticity of the reward circuitry after early-life adversity: mechanisms and significance. Biol Psychiatry. 2020;87:875–84.
Drury SS, Sanchez MM, Gonzalez A. When mothering goes awry: challenges and opportunities for utilizing evidence across rodent, nonhuman primate and human studies to better define the biological consequences of negative early caregiving. Horm Behav. 2016;77:182–92.
Orso R, Creutzberg KC, Wearick-Silva LE, Wendt Viola T, Tractenberg SG, Benetti F, et al. How early life stress impact maternal care: a systematic review of rodent studies. Front Behav Neurosci. 2019;13:197.
Rincon-Cortes M, Grace AA. Sex-dependent effects of stress on immobility behavior and VTA dopamine neuron activity: modulation by ketamine. Int J Neuropsychopharmacol. 2017;20:823–32.
Rincon-Cortes M, Grace AA. Postpartum changes in affect-related behavior and VTA dopamine neuron activity in rats. Prog Neuropsychopharmacol Biol Psychiatry. 2020;97:109768.
Rincon-Cortes M, Grace AA. Early pup removal leads to social dysfunction and dopamine deficit in late postpartum rats: prevention by social support. Front Glob Women’s Health. 2021;2:.694808.
Lee DY, Guttilla M, Fung KD, McFeron S, Yan J, Ranaldi R. Rostral-caudal differences in the effects of intra-VTA muscimol on cocaine self-administration. Pharm Biochem Behav. 2007;86:542–9.
Paxinos G, Watson C. The Rat Brain in Stereotaxic Coordinates, 6th edn. Academic Press, 456 pp, 2006.
Moreines JL, Owrutsky ZL, Grace AA. Involvement of Infralimbic Prefrontal Cortex but not Lateral Habenula in Dopamine Attenuation After Chronic Mild Stress. Neuropsychopharmacology. 2017;42:904–13.
Valenti O, Gill KM, Grace AA. Different stressors produce excitation or inhibition of mesolimbic dopamine neuron activity: response alteration by stress pre-exposure. Eur J Neurosci. 2012;35:1312–21.
Ungless MA, Grace AA. Are you or aren’t you? Challenges associated with physiologically identifying dopamine neurons. Trends Neurosci. 2012;35:422–30.
Kotch JB, Browne DC, Ringwalt CL, Stewart PW, Ruina E, Holt K, et al. Risk of child abuse or neglect in a cohort of low-income children. Child Abus Negl. 1995;19:1115–30.
Webster-Stratton C. Stress: a potential disruptor of parent perceptions and family interactions. J Clin Child Psychol. 1990;19:302–12.
Lewin M, Lopachin J, Delorme J, Opendak M, Sullivan RM, Wilson DA. Early life trauma has lifelong consequences for sleep and behavior. Sci Rep. 2019;9:16701.
Raineki C, Opendak M, Sarro E, Showler A, Bui K, McEwen BS, et al. During infant maltreatment, stress targets hippocampus, but stress with mother present targets amygdala and social behavior. Proc Natl Acad Sci USA. 2019;116:22821–32.
Murgatroyd CA, Nephew BC. Effects of early life social stress on maternal behavior and neuroendocrinology. Psychoneuroendocrinology. 2013;38:219–28.
Commons KG, Cholanians AB, Babb JA, Ehlinger DG. The rodent forced swim test measures stress-coping strategy, not depression-like behavior. ACS Chem Neurosci. 2017;8:955–60.
Molendijk ML, de Kloet ER. Immobility in the forced swim test is adaptive and does not reflect depression. Psychoneuroendocrinology. 2015;62:389–91.
Molendijk ML, de Kloet ER. Coping with the forced swim stressor: current state-of-the-art. Behav Brain Res. 2019;364:1–10.
Haim A, Sherer M, Leuner B. Gestational stress induces persistent depressive-like behavior and structural modifications within the postpartum nucleus accumbens. Eur J Neurosci. 2014;40:3766–73.
Smith JW, Seckl JR, Evans AT, Costall B, Smythe JW. Gestational stress induces post-partum depression-like behaviour and alters maternal care in rats. Psychoneuroendocrinology. 2004;29:227–44.
Galea LA, Wide JK, Barr AM. Estradiol alleviates depressive-like symptoms in a novel animal model of post-partum depression. Behav Brain Res. 2001;122:1–9.
Melon L, Hammond R, Lewis M, Maguire J. A novel, synthetic, neuroactive steroid is effective at decreasing depression-like behaviors and improving maternal care in preclinical models of postpartum depression. Front Endocrinol (Lausanne). 2018;9:703.
Rincon-Cortes M, Grace AA. Antidepressant effects of ketamine on depression-related phenotypes and dopamine dysfunction in rodent models of stress. Behav Brain Res. 2020;379:112367.
Rincon-Cortes M, Herman JP, Lupien S, Maguire J, Shansky RM. Stress: influence of sex, reproductive status and gender. Neurobiol. Stress. 2019;10:100155.
Fernandez JW, Grizzell JA, Philpot RM, Wecker L. Postpartum depression in rats: differences in swim test immobility, sucrose preference and nurturing behaviors. Behav Brain Res. 2014;272:75–82.
Lavi-Avnon Y, Shayit M, Yadid G, Overstreet HD, Weller A. Immobility in the swim test and observations of maternal behavior in lactating Flinders sensitive line rats. Behav Brain Res. 2005;161:155–63.
Maguire J, Mody I. GABA(A)R plasticity during pregnancy: relevance to postpartum depression. Neuron. 2008;59:207–13.
Maguire J, Mody I. Behavioral deficits in juveniles mediated by maternal stress hormones in mice. Neural Plast. 2016;2016:2762518.
Belujon P, Grace AA. Restoring mood balance in depression: ketamine reverses deficit in dopamine-dependent synaptic plasticity. Biol Psychiatry. 2014;76:927–36.
Chang CH, Grace AA. Amygdala-ventral pallidum pathway decreases dopamine activity after chronic mild stress in rats. Biol Psychiatry. 2014;76:223–30.
Belujon P, Grace AA. Dopamine system dysregulation in major depressive disorders. Int J Neuropsychopharmacol. 2017;20:1036–46.
Gomes FV, Zhu X, Grace AA. The pathophysiological impact of stress on the dopamine system is dependent on the state of the critical period of vulnerability. Mol Psychiatry. 2020;25:3278–91.
Douma EH, de Kloet ER. Stress-induced plasticity and functioning of ventral tegmental dopamine neurons. Neurosci Biobehav Rev. 2019;108:48–77.
Kaufling J. Alterations and adaptation of ventral tegmental area dopaminergic neurons in animal models of depression. Cell Tissue Res. 2019;377:59–71.
Grace AA. Dysregulation of the dopamine system in the pathophysiology of schizophrenia and depression. Nat Rev Neurosci. 2016;17:524–32.
Gaffori O, Le Moal M. Disruption of maternal behavior and appearance of cannibalism after ventral mesencephalic tegmentum lesions. Physiol Behav. 1979;23:317–23.
Numan M, Stolzenberg DS, Dellevigne AA, Correnti CM, Numan MJ. Temporary inactivation of ventral tegmental area neurons with either muscimol or baclofen reversibly disrupts maternal behavior in rats through different underlying mechanisms. Behav Neurosci. 2009;123:740–51.
Hansen S, Harthon C, Wallin E, Lofberg L, Svensson K. Mesotelencephalic dopamine system and reproductive behavior in the female rat: effects of ventral tegmental 6-hydroxydopamine lesions on maternal and sexual responsiveness. Behav Neurosci. 1991;105:588–98.
Byrnes EM, Rigero BA, Bridges RS. Dopamine antagonists during parturition disrupt maternal care and the retention of maternal behavior in rats. Pharm Biochem Behav. 2002;73:869–75.
Giordano AL, Johnson AE, Rosenblatt JS. Haloperidol-induced disruption of retrieval behavior and reversal with apomorphine in lactating rats. Physiol Behav. 1990;48:211–4.
Silva MR, Bernardi MM, Felicio LF. Effects of dopamine receptor antagonists on ongoing maternal behavior in rats. Pharm Biochem Behav. 2001;68:461–8.
Henshaw C. Mood disturbance in the early puerperium: a review. Arch Women’s Ment Health. 2003;6:S33–42.
Kendell RE, McGuire RJ, Connor Y, Cox JL. Mood changes in the first three weeks after childbirth. J Affect Disord. 1981;3:317–26.
Yalom ID, Lunde DT, Moos RH, Hamburg DA. “Postpartum blues” syndrome. A description and related variables. Arch Gen Psychiatry. 1968;18:16–27.
Molet J, Heins K, Zhuo X, Mei YT, Regev L, Baram TZ, et al. Fragmentation and high entropy of neonatal experience predict adolescent emotional outcome. Transl Psychiatry. 2016;6:e702.
Klinger K, Gomes FV, Rincon-Cortes M, Grace AA. Female rats are resistant to the long-lasting neurobehavioral changes induced by adolescent stress exposure. Eur Neuropsychopharmacol. 2019;29:1127–37.
Acknowledgements
We would like to thank Nicole MacMurdo for technical assistance with histology.
Funding
This work was supported by a Ford Foundation Postdoctoral Fellowship to MRC and NIMH-R01-MH057440 to AAG.
Author information
Authors and Affiliations
Contributions
Millie Rincón-Cortés: Conceptualization, methodology, software, validation, formal analysis, investigation, resources, writing—original draft, writing—review and editing, visualization, supervision, project administration, funding acquisition. Antony A. Grace: Resources, writing—review and editing, supervision, funding acquisition.
Corresponding author
Ethics declarations
Competing interests
Dr. Rincón-Cortés declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. Dr. Grace received consultant fees from Lundbeck, Pfizer, Otsuka, Asubio, Autofony, Alkermes, Concert, and Janssen, and is on the advisory board for Alkermes, Newron, and Takeda.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Rincón-Cortés, M., Grace, A.A. Postpartum scarcity-adversity disrupts maternal behavior and induces a hypodopaminergic state in the rat dam and adult female offspring. Neuropsychopharmacol. 47, 488–496 (2022). https://doi.org/10.1038/s41386-021-01210-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-021-01210-3
- Springer Nature Switzerland AG
This article is cited by
-
Early life stress, prenatal secondhand smoke exposure, and the development of internalizing symptoms across childhood
Environmental Health (2023)
-
Urocortin-3 neurons in the perifornical area are critical mediators of chronic stress on female infant-directed behavior
Molecular Psychiatry (2023)
-
Manifestations of Intergenerational Trauma During the Perinatal Period Among Adolescent Mothers in Lima, Peru: A Qualitative Analysis
Journal of Child & Adolescent Trauma (2023)