Abstract
While non-suicidal self-injury (NSSI) occurs in the general population at a surprisingly high rate, with higher rates among certain clinical populations, its etiology is not well-understood. Consequently, the DSM-5 lists NSSI as requiring further research. This study utilizes a translational model of naturally-occurring NSSI to assess the role of early parental neglect and variation in the serotonin transporter genotype (5-HTT) in the etiology of NSSI. Subjects (N = 161) were rhesus macaques (Macaca mulatta) reared in one of three conditions (mother-reared (MR), peer-reared (PR), or surrogate peer-reared (SPR)), and classified as NSSI (n = 18) or non-NSSI (n = 143). Subjects were genotyped for 5-HTT and their behaviors were recorded during an ecologically-meaningful, stress-evoking, intruder paradigm. Two weeks prior to testing, blood samples were obtained and assayed for plasma cortisol and adrenocorticotropic hormone (ACTH) concentrations. NSSI subjects were more likely to be SPR, paralleling human studies showing that individuals that exhibit NSSI tend to have experienced abuse or neglect early in life. Results also indicated that variation in the 5-HTT genotype differentiated the NSSI subjects. NSSI subjects that were homozygous for the L allele exhibited high plasma ACTH and high rates of stress-induced stereotypies; whereas NSSI subjects with the s allele exhibited impulsive behaviors, including frequently approaching the potentially dangerous intruder, high rates of aggressive vocal threats, and more activity. These results suggest that there may be different 5-HTT genotype-mediated NSSI typologies and that both early experiences and variation in the 5-HTT genotype may be important factors in understanding the etiology of NSSI.
Similar content being viewed by others
Introduction
Non-suicidal self-injury (NSSI) is a cluster of behaviors that involve the infliction of non-lethal harm to oneself [1]. NSSI tends to co-occur with certain clinical diagnoses [2, 3] and may escalate to suicidal ideation [4] and attempts [5]. The etiology of NSSI is not well understood and the DSM-5 lists it as requiring further study [6].
One of the most frequently cited risk factors for NSSI is aberrant early experiences, including abuse or neglect [7, 8]. The majority of those that experience early maltreatment, however, do not go on to exhibit NSSI [9]. Thus, elucidating why NSSI occurs in some individuals and not in others is an important step toward understanding its etiology.
At a more proximal level, some posit that NSSI is an aberrant means of reducing arousal [10], and studies suggest that self-injury brings temporary relief through tension and anxiety reduction [11]. Other studies show that individuals with NSSI tend to be more impulsive when compared to their non-NSSI peers [11, 12]. While impulsivity appears to be an important factor in humans with NSSI, researchers have generally not found a relationship between temperamental impulsivity and self-injury in nonhuman primates [13], although one study suggests that deficits in extinction learning among self-injuring monkeys evinces impulse-control deficits [14].
Given the role of anxiety and impulsivity in NSSI [15, 16], the present study investigates the role of the serotonin transporter genotype (5-HTT) in naturally-occurring NSSI. The 5-HTT genotype is a known predictor of a variety of stress- and impulse-control-mediated psychopathological outcomes in humans [17, 18] and nonhuman primates [19, 20]. Studies show that individuals possessing the short (s) allele often exhibit phenotypic susceptibility to stress and anxiety [21] and low levels of central serotonin are associated with anxious and impulsive behaviors [22, 23]. Studies in humans [24, 25] and nonhuman primates [26, 27] show that some self-injuring individuals respond to medications that modulate the serotonin system. Such findings indicate that inherent variation in the serotonin system may play a role in the etiology of NSSI for some. Others, however, do not respond to serotonin-related pharmacological strategies, suggesting a complex relationship between central serotonin and NSSI and that other systems are involved.
Anxiolytics reduce the rates of self-injury in some human populations, particularly those with intellectual disabilities [28]. In nonhuman primates, one study showed that benzodiazepines reduced rates of self-injury in some, but increased self-injury in others [29], with benzodiazepine-responders spending more time in a single housing and receiving more minor veterinary procedures. Another study in nonhuman primates shows that anxiogenic drugs increase the frequency of self-injury in some subjects, which the authors suggest is evidence of a dysregulated GABA system, perhaps as a consequence of GABA receptor genotype variation [30]. As anxiolytic drugs tend to reduce the activation of the hypo-thalamic-pituitary-adrenal (HPA) axis during stressful events [31], some suggest that NSSI is a self-soothing mechanism [32]. As further evidence of this, studies show that self-biting lowers the heart rate among self-injuring nonhuman primates [13] and that, for humans with a history of self-injury, imagining self-cutting leads to heart rate reductions [33]. Nevertheless, many do not respond to benzodiazepines, and in some individuals with NSSI, rates of self-injury may even increase following benzodiazepine administration [34].
There are limitations to studying NSSI in human populations. For example, research in humans tends to rely on retrospective memories and self-reports of the frequency of NSSI (for example, see [35]), which may be subject to underreporting and recall biases, and in humans, early life histories cannot be controlled. Rhesus monkey (Macaca mulatta) models provide an alternative because the frequency and severity of self-injury can be directly observed, and early rearing conditions can be closely-controlled. Furthermore, they possess marked genetic similarities to humans [36], including an orthologous serotonin transporter genotype [37]. They also show a high degree of similarity in the central nervous system (CNS) and the HPA axis [38]. Like humans, NSSI occurs naturally in the rhesus monkey. Rhesus monkeys engage in self-injury (~14–25% in captive settings; often termed SIB), typically in the form of self-biting [39], and rates are comparable to the percentages of humans that engage in NSSI [40]. While suicide has not been identified in rhesus monkeys, because this paper is intended to model human NSSI, the term NSSI is used to refer to self-injurious behavior in rhesus monkeys. As in humans, studies attribute NSSI in nonhuman primates to early parental neglect and limited social interactions [39, 41, 42]. Studies show that early maternal absence and limited social interactions not only increases the risk for NSSI but leads to CNS aberrations, including a dysregulated serotonin system [43] and HPA axis functioning [44, 45]. These factors make a rhesus monkey model of NSSI ideally suited for studying the etiology of NSSI.
The purpose of the present study is twofold: (1) to investigate the relationship between early parental neglect and NSSI status and (2) to investigate the role of 5-HTT genotype variation in the biobehavioral response to an ecologically meaningful stimulus among subjects with NSSI.
Materials and methods
Subjects
Subjects (N = 161) were rhesus monkeys (100 females, 61 males; Mage = 4.67 ± 0.19 years) tested at the National Institute of Health Animal Center between 2002 and 2003. Subjects were part of a larger research program designed to study variables associated with variation in alcohol intake (cf. [46, 47]). Of the 161 subjects in this study, n = 18 subjects (10 females, 8 males) were identified by direct observation as engaging in NSSI in the form of self-biting on one or more occasions, while the remaining n = 143 subjects were not observed to self-bite.
As infants, subjects were reared in one of three different rearing conditions (n = 75) mother-reared (MR), n = 37 peer-reared (PR), n = 49 surrogate-peer-reared (SPR) (see [45]). Briefly, the MR animals were reared by their mothers in social settings (with 7–10 adult females, 1–2 adult males, and other similarly-aged infants). The PR animals were separated from their mothers following birth and hand-reared in the nursery for the first 30 days of life and were thereafter housed with 2–3 same-aged peers. The SPR animals were reared in a nursery for the first months of life with an inanimate surrogate and access to 2–3 same-aged, similarly-reared peers for 2 h/day, 5 days/week. When subjects from each condition were ~7 months old, each cohort was permanently relocated into a larger social group of peers, receiving identical treatment until they were 3 years old, when the males and females were separated into separate groups of 8–12 monkeys so that breeding could be controlled. They remained in their respective isosexual groups for the present study. Rhesus monkeys live in troops that are governed by strict social dominance hierarchies. At the time of the study, dominance data was available for n = 135 of the subjects. Of these, n = 35 were high-ranking non-NSSI, n = 45 were middle-ranking non-NSSI, and n = 40 were low-ranking non-NSSI subjects. There were n = 7 high-ranking NSSI, n = 3 middle-ranking NSSI, and n = 5 low-ranking NSSI subjects. Protocols for the use of experimental animals were approved by the Institutional Animal Care and Use Committee of the National Institute of Alcohol Abuse and Alcoholism.
Intruder challenge test
When subjects were ~4 years old, they underwent a slightly-modified version of the standardized intruder challenge test, a stress-inducing experimental paradigm assessing the response to the territorial challenge of a same-age and same-sex unfamiliar monkey (the intruder), allowing researchers to measure stress and impulsivity in a standardized fashion while controlling for age and sex [48, 49]. Pilot testing indicated that it was difficult to maintain experimental control and accurately code the response of subjects to the intruder when the whole homecage group was tested at once. Thus, three same-age test subjects, living together in the same isosexual social group, were randomly selected and isolated from the larger group into the outdoor portion (264CM × 300CM × 244CM) of their homecage enclosure. None of the trios were comprised of more than one NSSI subject and the rearing condition of each the members of each trio was randomly distributed. Because the subjects from each homecage were all the same age and sex, the unfamiliar intruder could be matched for age, sex, and relative body size.
The intruder was separated from their social group and acclimated to a holding cage with an open mesh side (76CM × 63CM × 91CM) for a period of 30 min. Following segregation into the outdoor area of their living quarters, the test subjects were acclimated for a period of 10 min. The intruder holding cage was rolled directly in front of the outdoor portion of the test subjects’ homecage enclosures, with the open mesh side facing into the enclosure. Test subjects could see, approach, and interact with the unfamiliar intruder through the mesh of the enclosure and transfer cage, limited touch was possible, but full contact was prevented to avoid injury (no animals were injured during the test). Each of the three test subjects’ behaviors was recorded by separate observers for 30 min using handheld computers equipped with Observer™ software (Noldus, Leesburg, Virginia). All observers were trained by senior authors (JDH) and (SGL), each of whom had 10+ years of experience with the scoring system and achieved interobserver reliability (r > 0.85) using regression and an established criterion of deviating less than 10% on any behavior. Raters were blind to subjects’ genotype and NSSI status at the time of data collection. The recorded behaviors were based on a behavioral ethogram developed and used extensively by the senior author’s laboratory (see Table 1 for a detailed ethogram).
Hormone sampling and genotyping
Two weeks prior to the intruder challenge test, 3–4 ml of blood was obtained using EDTA coated vacutainers and a 22-g needle to assess levels of plasma cortisol and adrenocorticotropic hormone (ACTH). All subjects were captured and injected with ketamine anesthesia (15 mg/kg, intramuscular) within 8 min of entering their homecage, and samples were obtained within 5 min of injection. Samples were placed on wet ice and centrifuged at 4 °C for 20 min, after which plasma was aliquoted and immediately frozen in liquid nitrogen. Samples were stored at −70 °C until they were assayed for concentrations of plasma cortisol and ACTH.
A subset of subjects (n = 143) was genotyped for 5-HTT. Genotyping procedures have been described in detail elsewhere (see [50]). Briefly, DNA was isolated from whole blood collected in infancy from the femoral vein after subjects were sedated (ketamine anesthesia, 15 mg/kg, intramuscular). The short (s) (398-bp) and long (L) (419-bp) alleles of the rh5-HTTLPR were identified by direct visualization following ethidium-bromide staining (n = 100 animals (47 MR, 23 PR, and 30 SPR) were homozygous for the L allele, n = 40 (26 MR, 4 PR, and 10 SPR) were heterozygous, n = 3 (2 MR and 1 SPR) were homozygous for the s allele). Preliminary analyses showed no difference between the heterozygotes and subjects homozygous for the s allele on the variables of interest. Therefore, subjects that were homozygous for the s allele were combined with the heterozygotes, as has been done in other studies [51]. Genotype frequency did not deviate from Hardy–Weinberg equilibrium (x2(2, N = 143) = 0.19, p = 0.66). See Table 2 for the distribution of the genotypes.
Data analysis
Preliminary t-tests showed no difference between the time to capture or time to obtain blood samples for NSSI and non-NSSI subjects (t(155) = −0.71, p = 0.48) or for LL subjects or subjects with an s allele (t(137) = −0.76, p = 0.45). With the exception of cortisol, preliminary ANOVAs showed no effects of sex on variables of interest (p > 0.05), so sex was excluded from further analyses. A preliminary chi-square (x2) test of independence showed no difference between NSSI and non-NSSI subjects in social dominance rank x2(N = 135, df = 2) = 2.46, (p = 0.29).
To protect against inflating the Type I error rate, a two-way MANOVA was performed, with 5-HTT genotype and NSSI status as independent variables and plasma cortisol and ACTH and each of the nine behaviors as dependent variables. Then, follow-up ANOVAs were conducted, with 5-HTT genotype and NSSI status as independent variables and plasma cortisol or ACTH concentrations or each of the observed behaviors as the dependent variables. All analyses were conducted in SPSS, version 26.
Results
Rearing condition
Results from the x2 test showed that there was a significant relationship between NSSI status and early rearing condition (x2(2, N = 161) = 26.79, p = 0.00001), with SPR subjects more likely to exhibit NSSI than the MR or PR subjects (see Table 3).
Two-way MANOVA
Results of the MANOVA showed a statistically significant effect of 5-HTT genotype (F(11,94) = 3.07, p = 0.001, Wilks’ Λ = 0.74, ηp2 = 0.26) and a statistically significant effect of NSSI status (F(11,94) = 2.52, p = 0.008, Wilks’ Λ = 0.77, ηp2 = 0.123). There was also a statistically significant 5-HTT-genotype-by-NSSI-status interaction (F(11,94) = 2.93, p = 0.004, Wilks’ Λ = 0.75, ηp2 = 0.26).
Follow-up ANOVAs
Plasma ACTH and cortisol concentrations. There was a significant main effect of 5-HTT genotype on ACTH concentrations (F(1,121) = 4.25, p = 0.04), with homozygous LL subjects exhibiting higher ACTH concentrations (M = 69.70 ± 43.26 pmol/mL) when compared to subjects with an s allele (M = 63.84 ± 33.52 pmol/mL).
There was also a significant 5-HTT-genotype-by-NSSI-status interaction on ACTH concentrations (F(1,11) = 4.10, p = 0.04), with homozygous LL/NSSI subjects exhibiting the highest ACTH concentrations (M = 97.95 ± 49.63 pmol/mL), when compared to NSSI subjects with an s allele (M = 44.38 ± 48.30 pmol/mL), or to LL/non-NSSI subjects (M = 67.01 ± 41.95 pmol/mL) or to subjects with an s allele (M = 66.52 ± 31.18 pmol/mL) (see Fig. 1).
There was a significant sex-by-NSSI-status interaction for both plasma cortisol (F(1,144) = 7.63, p = 0.006) and ACTH (F(1,124) = 4.09, p = 0.04), with females that exhibited NSSI exhibiting higher cortisol and ACTH concentrations, when compared to NSSI males and non-NSSI subjects (cortisol: NSSI females: M = 26.00 ± 8.33 pmol/mL, NSSI males: M = 18.93 ± 4.56 pmol/mL; non-NSSI females: M = 24.22 ± 6.87 pmol/mL, non-NSSI males: M = 21.93 ± 6.31 pmol/mL; ACTH: NSSI females: M = 82.02 ± 59.23 pmol/mL, NSSI males: M = 50.52 ± 25.96 pmol/mL; non-NSSI females: M = 66.79 ± 40.86 pmol/mL, non-NSSI males: M = 54.40 ± 16.75 pmol/mL). There were no other cortisol effects.
Impulsivity
There was a significant main effect of 5-HTT genotype on the frequency of approaching the intruder (F(1,139) = 13.17, p < 0.0001), with subjects with an s allele approaching the intruder more often (M = 21.65 ± 16.80 approaches) when compared to homozygous LL subjects (M = 15.43 ± 14.23 approaches).
There was a statistically significant main effect of NSSI status on the frequency of approaching the intruder (F(1,139) = 6.18, p = 0.01), with NSSI subjects exhibiting a higher frequency of approaching the intruder (M = 23.23 ± 22.69 approaches) when compared to non-NSSI subjects (M = 16.71 ± 14.30 approaches).
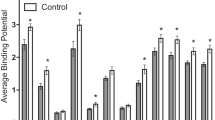
There was also a significant 5-HTT-genotype-by-NSSI-status interaction on the frequency of approaching the intruder (F(1,139) = 7.84, p = 0.006), with NSSI subjects with an s allele approaching the intruder three times more often (M = 43.75 ± 25.97 approaches), when compared to homozygous LL/NSSI subjects (M = 14.11 ± 14.67 approaches), or the LL/non-NSSI subjects (M = 15.56 ± 14.26 approaches) or to subjects with an s allele (M = 19.38 ± 14.22 approaches) (see Fig. 2a).
a There was a significant 5-HTT-genotype-by-NSSI-status interaction, with NSSI subjects with an s allele exhibiting the highest rates of approaching the intruder (F(1,139) = 13.17, p < 0.0001). b There was a significant 5-HTT-genotype-by-NSSI-status interaction, with NSSI subjects with an s allele spending the most time active (F(1,139) = 7.98, p = 0.005). c There was a significant 5-HTT-genotype-by-NSSI-status interaction, with homozygous NSSI subjects exhibiting the most time in stereotypies (F(1,139) = 5.01, p = 0.03). d There was a significant 5-HTT-genotype-by-NSSI-status interaction, with NSSI subjects with an s allele exhibiting the most threat vocalizations (F(1,139) = 11.55, p = 0.001). Black bars indicate homozygous subjects; white bars indicate subjects with an s allele.
Activity
There was a significant main effect of 5-HTT genotype on time spent active (F(1,139) = 17.27, p < 0.0001), with subjects with an s allele spending more time active (M = 362.63 ± 257.52 s) when compared to homozygous LL subjects (M = 263.94 ± 200.17 s).
There was also a significant main effect of NSSI status on time spent active (F(1,139) = 8.33, p = 0.005), with NSSI subjects spending more time active (M = 403.30 ± 311.63 s) when compared to non-NSSI subjects (M = 282.65 ± 210.34 s).
There was also a significant 5-HTT-genotype-by-NSSI-status interaction on the seconds spent active (F(1,139) = 7.98, p = 0.005), with NSSI subjects with an s allele spending more time active (M = 708.30 ± 288.37 s), when compared to homozygous LL/NSSI subjects (M = 267.74 ± 217.50 s), or to LL/non-NSSI subjects (M = 263.57 ± 199.67 s) or to subjects with an s allele (M = 327.18 ± 229.92 s) (see Fig. 2b).
Stereotypic behavior
There was a significant 5-HTT-genotype-by-NSSI-status interaction on seconds spent in stereotypic behavior (F(1,139) = 5.01, p = 0.03), with homozygous LL NSSI subjects exhibiting more time in of stereotypic behaviors (M = 280.84 ± 444.11 s), when compared to NSSI subjects with an s allele (M = 20.95 ± 24.59 s) or to LL/non-NSSI subjects (M = 39.16 ± 190.86 s) or to subjects with an s allele (M = 81.82 ± 200.19 s) (see Fig. 2c).
Threat vocalizations
There was a significant main effect of 5-HTT genotype on the frequency of threat vocalizations (F(1,136) = 12.99, p < 0.0001), with subjects with an s allele exhibiting more threats (M = 20.12 ± 34.81 threats) when compared to homozygous LL subjects (M = 15.03 ± 36.08 threats).
There was also a significant main effect of NSSI status on the frequency of threat vocalizations (F(1,139) = 5.74, p = 0.02), with NSSI subjects exhibiting more threats (M = 23.77 ± 41.83 threats) when compared to non-NSSI subjects (M = 15.84 ± 35.09 threats).
There was a significant 5-HTT-genotype-by-NSSI-status interaction on threat frequency (F(1,139) = 11.55, p = 0.001), with NSSI subjects with an s allele exhibiting the most threat vocalizations (M = 67.25 ± 57.57 threats), when compared to homozygous LL/NSSI subjects (M = 4.44 ± 3.97 threats) or to LL/non-NSSI subjects (M = 16.08 ± 37.66 threats) or to subjects with an s allele (M = 15.28 ± 28.64 threats) (see Fig. 2d).
There were no significant relationships found between NSSI status or 5-HTT genotype and aggression, latency to approach the intruder, environmental exploration, social contact with conspecifics, or behavioral withdrawal (p > 0.05).
Discussion
Results showed that the majority of NSSI subjects experienced limited social experience (83% of NSSI subjects were SPR). To the extent that these limited social experiences are comparabl to early parental neglect in humans, these findings are consistent with studies showing that aberrant early experiences, including child abuse and neglect, are risk factors for NSSI in human [52] and nonhuman primates [13, 39, 41]. The results also showed evidence for two 5-HTT genotype-mediated typological variations in the stress-response of individuals that engage in NSSI. Homozygous LL subjects showed tendencies for overactive HPA axes and hyper-responsiveness to stress, as evidenced by high ACTH concentrations and stereotypies; whereas subjects with an s allele showed evidence of impulse control deficits, exhibiting longer overall activity and high rates of aggressive threat vocalizations and more instances of approaching the unfamiliar, potentially dangerous, intruder.
Studies show SPR subjects tend to have a variety of behavioral pathologies, such as rocking, stereotypies, and self-biting [13, 42]. One interpretation of these results is that SPR subjects may engage in NSSI as a maladaptive self-soothing mechanism. This interpretation may not be complete, however, as 34 of the 49 SPR subjects did not engage in NSSI, indicating that factors other than early neglect likely contribute to the etiology of NSSI. It is of note, that the PR subjects were no more likely to exhibit NSSI than the MR subjects, replicating an earlier finding [42]. While both PR and SPR subjects were deprived of maternal care, PR subjects had continual social interactions with their agemates, whereas SPR subjects had only limited social interactions. Studies show that such limited interactions reduce the ability of SPR subjects to develop social competence when compared to PR and MR subjects [53]. Given these differences, NSSI may develop as a consequence of limited social interactions, although this explanation is speculative. What is clear, however, is that NSSI is not simply the result of parental absence; nor is it the result of early maltreatment, but rather is, at least in part, a consequence of limited social experiences, an interpretation that parallels work in humans showing that NSSI is more frequent in individuals who experience peer-rejection and loneliness [54, 55].
There were two 5-HTT genotype-mediated responses to the intruder challenge test among NSSI subjects. In the first, homozygous LL/NSSI subjects exhibited high rates of stereotypies. Stereotypies are non-adaptive, repetitive behaviors [56], tending to occur among subjects that experienced early maternal deprivation [57]. It is of note that the homozygous LL/NSSI subjects spent 13 times more time in stereotypies than subjects with an s allele, suggesting a trait-like maladaptive response to stress. For homozygous LL/NSSI subjects, one cannot rule out the possibility that NSSI is an extension of such stereotypies. Homozygous LL/NSSI subjects, however, exhibited significantly higher ACTH concentrations, when compared to NSSI subjects with an s allele. While speculative, homozygous LL/NSSI subjects may attempt to reduce arousal by engaging in self-soothing techniques, like stereotypies and NSSI, an interpretation corroborated by other work suggesting that NSSI decreases arousal (i.e., decreases heart rate) in monkeys [13] and humans [33].
In contrast, NSSI subjects with an s allele exhibited more frequent approaches and more threat vocalizations directed toward the unfamiliar intruder and more time in general activity. Studies suggest that the s allele is associated with high rates of impulsivity [58, 59]. In line with this, on average, NSSI subjects with an s allele approached the intruder three times more often than homozygous LL/NSSI subjects. Approaching an unfamiliar conspecific in the context of the intruder challenge test is a high-risk behavior and may indicate impaired impulse control. While CNS serotonin activity is not reported in this manuscript, other studies in nonhuman primates show that, on average, NSSI subjects administered fluoxetine show reductions in rates of self-injury [60], suggesting that the serotonin system may be involved in NSSI. NSSI subjects with an s allele also exhibited lower ACTH concentrations and less time in stereotypies than homozygous LL/NSSI subjects, an indication that their NSSI was not driven by anxiety or arousal. While speculative, these impulse control deficits may lead to an inability to successfully inhibit NSSI tendencies, an interpretation consistent with human research suggesting that NSSI individuals tend to exhibit impulsivity [12].
Conclusions
Taken together, these 5-HTT genotype-mediated response typologies among the NSSI subjects suggest that some individuals with NSSI may exhibit NSSI due to an inability to inhibit impulsive behavior, while others may exhibit NSSI in an attempt to cope with high levels of stress and anxiety. While other studies show that a dysregulated serotonin system may play a role in the etiology of NSSI [61], this study parses out important genotype-mediated nuances that may better explain the differences between subjects.
It is clear that early experiences are, in part, foundational in the development of NSSI. The results of this study also suggest that the proposed 5-HTT genotype-mediated typologies may be useful in determining appropriate NSSI treatment modalities, and could explain why some individuals that self-injure respond to serotonin-specific treatments [24, 25], while others respond to anxiolytics [28]. While this study has sample sizes comparable to others investigating NSSI in nonhuman primates [14, 26, 29], one limitation to the interpretation of these results is the small number of NSSI subjects with the s allele, which tempers somewhat the generalization of the findings. As such, this study provides evidence for a promising area of future study. Future larger-scale studies of self-injury in nonhuman primates should attempt to replicate these results with a larger sample size of subjects possessing an s allele. Nevertheless, these results are compelling and suggest a novel way of dichotomizing human populations with NSSI. While this study focuses on the 5-HTT genotype, it is likely that other systems play a role in the etiology of NSSI. For example, NSSI leads to the release of endogenous opiates [62], and some studies show that blocking opioid release reduces NSSI [63]. It would be of interest to investigate the μ-opioid genotype in future studies.
An important step in providing better treatment for individuals with NSSI is understanding variables that are relevant to its etiology. These findings provide evidence for factors that are important in the development of NSSI. That SPR subjects were at greatest risk for developing NSSI suggests that early life events, in particular limited social experiences, are an important underlying risk factor in the development of NSSI. The results of this study also suggest that there may be different NSSI typologies, modulated by 5-HTT genotype variation. To the extent that these results generalize to humans, they suggest that 5-HTT genotype variation may play an important role in determining which NSSI treatment modalities may be most effective.
Funding and disclosure
This work was funded by the intramural programs of the NIAAA and the NICHD, and by mentoring grants from BYU. The authors have no conflicts of interest to disclose.
References
Nock MK. Why do people hurt themselves? New insights into the nature and functions of self-injury. Curr Dir Psychol Sci. 2009;18:78–83.
Sharp C, Green KL, Yaroslavsky I, Venta A, Zanarini MC, Pettit J. The incremental validity of borderline personality disorder relative to major depressive disorder for suicidal ideation and deliberate self-harm in adolescents. J Pers Disord. 2012;26:927–38.
Weintraub MJ, Van de Loo MM, Gitlin MJ, Miklowitz DJ. Self-harm, affective traits, and psychosocial functioning in adults with depressive and bipolar disorders. J Nerv Ment Dis. 2017;205:896.
Whitlock J, Muehlenkamp J, Eckenrode J, Purington A, Abrams GB, Barreira P, et al. Nonsuicidal self-injury as a gateway to suicide in young adults. J Adolesc Health. 2013;52:486–92.
Hawton K, Bergen H, Cooper J, Turnbull P, Waters K, Ness J, et al. Suicide following self-harm: findings from the multicentre study of self-harm in England, 2000–2012. J Affect Disord. 2015;175:147–51.
American Psychiatric Association. Diagnostic and statistical manual of mental disorders. Washington, DC: American Psychiatric Association; 2013.
Auerbach RP, Kim JC, Chango JM, Spiro WJ, Cha C, Gold J, et al. Adolescent nonsuicidal self-injury: Examining the role of child abuse, comorbidity, and disinhibition. Psychiatry Res. 2014;220:579–84.
Cipriano A, Cella S, Cotrufo P. Nonsuicidal self-injury: a systematic review. Front Psychol. 2017;8:1946.
Santens T, Claes L, Diamond GS, Bosmans G. Depressive symptoms and self-harm among youngsters referred to child welfare: the role of trust in caregiver support and communication. Child Abus Negl. 2018;77:155–67.
Nixon MK, Cloutier PF, Aggarwal S. Affect regulation and addictive aspects of repetitive self-injury in hospitalized adolescents. J Am Acad Child Adolesc Psychiatry. 2002;41:1333–41.
Cassels M, Wilkinson P. Non-suicidal self-injury in adolescence. Paediatr Child Health. 2016;26:554–8.
Ross S, Heath NL, Toste JR. Non‐suicidal self‐injury and eating pathology in high school students. Am J Orthopsychiatry. 2009;79:83–92.
Novak MA. Self‐injurious behavior in rhesus monkeys: New insights into its etiology, physiology, and treatment. Am J Primatol. 2003;59:3–19.
Lutz C, Tiefenbacher S, Meyer J, Novak M. Extinction deficits in male rhesus macaques with a history of self‐injurious behavior. Am J Primatol. 2004;63:41–48.
Kaniuka AR, Kelliher-Rabon J, Chang EC, Sirois FM, Hirsch JK. Symptoms of anxiety and depression and suicidal behavior in college students: Conditional indirect effects of non-suicidal self-injury and self-compassion. J Coll Stud Psychother. 2020;34:316–38.
McHugh CM, Lee RSC, Hermens DF, Corderoy A, Large M, Hickie IB. Impulsivity in the self-harm and suicidal behavior of young people: a systematic review and meta-analysis. J Psychiatr Res. 2019;116:51–60.
Caspi A, Sugden K, Moffitt TE, Taylor A, Craig IW, Harrington H, et al. Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science 2003;301:386–9.
Mandelli L, Serretti A, Marino E, Pirovano A, Calati R, Colombo C. Interaction between serotonin transporter gene, catechol-O-methyltransferase gene and stressful life events in mood disorders. Int J Neuropsychopharmacol. 2007;10:437–47.
Barr CS, Newman TK, Becker ML, Champoux M, Lesch KP, Suomi SJ, et al. Serotonin transporter gene variation is associated with alcohol sensitivity in rhesus macaques exposed to early-life stress. Alcohol. 2003;27:812–7.
Kinnally EL, Karere GM, Lyons LA, Mendoza SP, Mason WA, Capitanio JP. Serotonin pathway gene-gene and gene-environment interactions influence behavioral stress response in infant rhesus macaques. Dev Psychopathol. 2010;22:35–44.
Hankin BL, Barrocas AL, Young JF, Haberstick B, Smolen A. 5-HTTLPR× interpersonal stress interaction and nonsuicidal self-injury in general community sample of youth. Psychiatry Res. 2015;225:609–12.
Bourgeois M. Serotonin, impulsivity and suicide. Hum Psychopharmacol. 1991;6:S31–S36.
Petersen IT, Bates JE, Goodnight JA, Dodge KA, Lansford JE, Pettit GS, et al. Interaction between serotonin transporter polymorphism (5-HTTLPR) and stressful life events in adolescents’ trajectories of anxious/depressed symptoms. Dev Psychol. 2012;48:1463.
Goodyer I, Dubicka B, Wilkinson P, Kelvin R, Roberts C, Byford S, et al. Selective serotonin reuptake inhibitors (SSRIs) and routine specialist care with and without cognitive behaviour therapy in adolescents with major depression: randomised controlled trial. BMJ 2007;335:142.
Lucchelli JP, Bertschy G. Low-dose fluoxetine in four children with autistic spectrum disorder improves self-injurious behavior, ADHD-like symptoms, and irritability. Case Rep Psychiatry. 2018;2018:6278501. https://doi.org/10.1155/2018/6278501.
Fontenot MB, Padgett EE, Dupuy AM, Lynch CR, De Petrillo PB, Higley JD. The effects of fluoxetine and buspirone on self-injurious and stereotypic behavior in adult male rhesus macaques. Comp Med. 2005;55:67–74.
Weld KP, Mench JA, Woodward RA, Bolesta MS, Suomi SJ, Higley JD. Effect of tryptophan treatment on self-biting and central nervous system serotonin metabolism in rhesus monkeys (Macaca mulatta). Neuropsychopharmacology 1999;19:314–21.
Ricketts RW, Goza AB, Ellis CR, Singh YN, Chambers S, Singh NN, et al. Clinical effects of buspirone on intractable self-injury in adults with mental retardation. J Am Acad Child Adolesc Psychiatry. 1993;33:270–6.
Tiefenbacher S, Fahey MA, Rowlett JK, Meyer JS, Pouliot AL, Jones BM, et al. The efficacy of diazepam treatment for the management of acute wounding episodes in captive rhesus macaques. Comp Med. 2005;55:387–92.
Major CA, Kelly BJ, Novak MA, Davenport MD, Stonemetz KM, Meyer JS. The anxiogenic drug FG7142 increases self-injurious behavior in male rhesus monkeys (Macaca mulatta). Life Sci. 2009;85:753–8.
Rohrer T, Von Richthofen V, Schulz C, Beyer J, Lehnert H. The stress-, but not corticotropin-releasing hormone-induced activation of the pituitary-adrenal axis in man is blocked by alprazolam. Horm Metab Res. 1994;26:200–6.
Cawood CD, Huprich SK. Late adolescent nonsuicidal self-injury: the roles of coping style, self-esteem, and personality pathology. J Pers Disord. 2011;25:765–81.
Haines J, Williams CL, Brain KL, Wilson GV. The psychophysiology of self-mutilation. J Abnorm Psychol. 1995;104:471.
Brent DA, Emslie GJ, Clarke GN, Asarnow J, Spirito A, Ritz L, et al. Predictors of spontaneous and systematically assessed suicidal adverse events in the treatment of SSRI-resistant depression in adolescents (TORDIA) study. Am J Psychiatry. 2009;166:418–26.
Wang SB, Pisetsky EM, Skutch JM, Fruzzetti AE, Haynos AF. Restrictive eating and nonsuicidal self-injury in a nonclinical sample: co-occurrence and associations with emotion dysregulation and interpersonal problems. Compr Psychiatry. 2018;82:128–32.
Gibbs RA, Rogers J, Katze MG, Bumgarner R, Weinstock GM, Mardis ER, et al. Evolutionary and biomedical insights from the rhesus macaque genome. Science 2007;316:222–34.
Bennett AJ, Lesch KP, Heils A, Long JC, Lorenz JG, Shoaf SE, et al. Early experience and serotonin transporter gene variation interact toi influence primate CNS function. Mol Psychiatry. 2002;7:118–22.
Phillips KA, Bales KL, Capitanio JP, Conley A, Czoty PW, ‘t Hart BA, et al. Why primate models matter. Am J Primatol. 2014;76:801–27.
Lutz C, Well A, Novak M. Stereotypic and self‐injurious behavior in rhesus macaques: a survey and retrospective analysis of environment and early experience. Am J Primatol. 2003;60:1–15.
Muehlenkamp JJ, Claes L, Havertape L, Plener PL. International prevalence of adolescent non-suicidal self-injury and deliberate self-harm. Child Adolesc Psychiatry Ment Health. 2012;6:10.
Rommeck I, Anderson K, Heagerty A, Cameron A, McCowan B. Risk factors and remediation of self-injurious and self-abuse behavior in rhesus macaques. J Appl Anim Welf Sci. 2009;12:61–72.
Lutz CK, Davis EB, Ruggiero AM, Suomi SJ. Early predictors of self‐biting in socially‐housed rhesus macaques (Macaca mulatta). Am J Primatol. 2007;69:584–90.
Huggins KN, Mathews TA, Locke JL, Szeliga KT, Friedman DP, Bennett AJ, et al. Effects of early life stress on drinking and serotonin system activity in rhesus macaques: 5-hydroxyindoleacetic acid in cerebrospinal fluid predicts brain tissue levels. Alcohol 2012;46:371–6.
Feng X, Wang L, Yang S, Qin D, Wang J, Li C, et al. Maternal separation produces lasting changes in cortisol and behavior in rhesus monkeys. Proc Natl Acad Sci USA. 2011;108:14312–7.
Shannon C, Champoux M, Suomi SJ. Rearing condition and plasma cortisol in rhesus monkey infants. Am J Primatol. 1998;46:311–21.
Wood EK, Kruger R, Cash E, Lindell SG, Schwandt ML, Barr CS, et al. Early life temperamental anxiety is associated with excessive alcohol intake in adolescence: A rhesus monkey (Macaca mulatta) model. Addict Biol. 2020;25:e12825.
Wood EK, Champoux M, Lindell SG, Barr CS, Suomi SJ, Higley JD. Neonatal temperament and neuromotor differences are predictive of adolescent alcohol intake in rhesus monkeys (Macaca mulatta). Am J Primatol. 2020;82:e23043.
Fairbanks LA. Individual differences in response to a stranger: social impulsivity as a dimension of temperament in vervet monkeys (Cercopithecus aethiops sabaeus). J Comp Psychol. 2001;115:22–28.
Schwandt ML, Lindell SG, Sjöberg RL, Chisholm KL, Higley JD, Suomi SJ, et al. Gene–environment interactions and response to social intrusion in male and female rhesus macaques. Biol Psychiatry. 2010;67:323–30.
Lesch KP, Bengel D, Heils A, Sabol SZ, Greenberg BD, Petri S, et al. Association of anxiety-related traits with a polymorphism in the serotonin transporter gene regulatory region. Science 1996;274:1527–31.
Kinnally EL, Lyons LA, Abel K, Mendoza S, Capitanio JP. Effects of early experience and genotype on serotonin transporter regulation in infant rhesus macaques. Genes Brain Behav. 2008;7:481–6.
Brown RC, Heines S, Witt A, Braehler E, Fegert JM, Harsch D, et al. The impact of child maltreatment on non-suicidal self-injury: data from a representative sample of the general population. BMC Psychiatry. 2018;18:1–8.
Bastian ML, Sponberg AC, Sponberg AC, Suomi SJ, Higley JD. Long-term effects of infant rearing condition on the acquisition of dominance rank in juvenile and adult rhesus macaques (Macaca mulatta). Dev Psychobiol. 2003;42:44–51.
Gandhi A, Luyckx K, Goossens L, Maitra S, Claes L. Association between non-suicidal self-injury, parents and peers related loneliness, and attitude towards aloneness in Flemish adolescents: an empirical note. Psychol Belgica. 2018;58:3.
Victor SE, Hipwell AE, Stepp SD, Scott LN. Parent and peer relationships as longitudinal predictors of adolescent non-suicidal self-injury onset. Child Adolesc Psychiatry Ment Health. 2019;13:1.
Mason GJ. Stereotypies: a critical review. Anim Behav. 1991;41:1015–37.
Zhang B, Nelson EE. Effects of adverse rearing experience on organization of brain and behavior in non-human primates. Int J Child Adolesc Health. 2011;4:309.
Walderhaug E, Herman AI, Magnusson A, Morgan MJ, Landrø NI. The short (S) allele of the serotonin transporter polymorphism and acute tryptophan depletion both increase impulsivity in men. Neurosci Lett. 2010;473:208–11.
Cha J, Guffanti G, Gingrich J, Talati A, Wickramaratne P, Weissman M, et al. Effects of serotonin transporter gene variation on impulsivity mediated by default mode network: a family study of depression. Cereb Cortex. 2018;28:1911–21.
Fontenot MB, Musso MW, McFatter RM, Anderson GM. Dose-finding study of fluoxetine and venlafaxine for the treatment of self-injurious and stereotypic behavior in rhesus macaques (Macaca mulatta). J Am Assoc Lab Anim Sci. 2009;48:176–84.
McCloskey MS, Ben-Zeev D, Lee R, Berman ME, Coccaro EF. Acute tryptophan depletion and self-injurious behavior in aggressive patients and healthy volunteers. Psychopharmacology. 2009;203:53.
Bresin K, Gordon KH. Endogenous opioids and nonsuicidal self-injury: a mechanism of affect regulation. Neurosci Biobehav Rev. 2013;37:374–83.
Roth AS, Ostroff RB, Hoffman RE. Naltrexone as a treatment for repetitive self-injurious behavior: an open-label trial. J Clin Psychiatry. 2016;57:233–7.
Acknowledgements
The authors would like to thank the many post-docs, graduate and undergraduate students, as well as the animal care staff, for their contributions to the data collection that made this project possible.
Author information
Authors and Affiliations
Contributions
EKW, RK, JPD, SMD, and JDH were responsible for the study concept and design. SGL, CSB, MLS, DG, SJS, and JDH contributed to the acquisition of the data. EKW, RK, and JDH conducted the analyses and interpreted the findings. EKW, RK, and JDH drafted the initial manuscript. JCH critically revised the manuscript for important intellectual content. EKW, RK, JPD, SMD, JNH, LN, SGL, CSB, MLS, DG, SJS, JCH, and JDH reviewed the manuscript and approved the final version for publication.
Corresponding authors
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wood, E.K., Kruger, R., Day, J.P. et al. A nonhuman primate model of human non-suicidal self-injury: serotonin-transporter genotype-mediated typologies. Neuropsychopharmacol. 47, 1256–1262 (2022). https://doi.org/10.1038/s41386-021-00994-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-021-00994-8
- Springer Nature Switzerland AG