Abstract
Aspirin is widely used in preventing early onset preeclampsia in women who are identified as being high risk. Although the benefit of aspirin is increasingly evident and acknowledged, there remains many unanswered questions with regards to its optimal application in pregnancy. The issues mainly centre around the relatively modest risk reduction that is observed with the use of aspirin prophylactically. We aim to explore the reasons behind the conservative rate of benefit and aim to explore factors that are likely to influence the outcomes with the use of aspirin.
Similar content being viewed by others
Introduction
Preeclampsia is a pregnancy related disorder with a global annual incidence of 2–8% of all pregnancies [1]. Although the general incidence of preeclampsia remains static, the incidence of maternal and fetal morbidity, particularly in developing countries, is increasing and remains the second commonest cause of maternal mortality internationally [1,2,3]. It is widely accepted that impaired placental invasion in the first trimester has a key role in the pathophysiology of early-onset preeclampsia [4, 5]. This is best summarized in a two-stage model (Fig. 1). Stage one describes impaired cytotrophoblast invasion and pseudovasculogenesis of the maternal uterine artery which subsequently leads to impaired utero-placental arterial flow. Stage two of the disease process occurs in the second or third trimester where there is ischaemic and oxidative stress of the hypoxic placenta. The resulting oxidative stress and angiogenic imbalance leads to maternal endothelial dysfunction. This is resultant from an increase in soluble fms-like tyrosine kinase-1 production and reduction in placental growth factor (PlGF) (Fig. 1) [4, 6].
Illustration of the two-stage pathophysiology of preeclampsia. Legends for Fig. 1: HELLP syndrome haemolysis, elevated liver enzymes, low platelet count syndrome, IUGR intrauterine growth retardation, NK cells natural killer cells, PlGF placental growth factor, sFlt-1 soluble fms-like tyrosine kinase-1
The current strategy in managing established preeclampsia focuses on expectant management of the pregnancy—where clinician attempt to balance the risks to the mother, while minimizing the fetus’s risk of iatrogenic prematurity. Although there is no targeted therapy that addresses the pathophysiology of established preeclampsia at present, current research suggest that this will be an option in the near future [7,8,9,10]. Until then, the emphasis is placed on the prevention of preeclampsia, which involves early identification and administration of prophylactic interventions in high-risk women.
The evidence on the benefit of aspirin in preventing preeclampsia continues to grow; however, many unanswered questions remain regarding its clinical application [11,12,13]. To begin with, risk stratification and selection of women who will benefit from prophylactic aspirin remains an area of ongoing debate with varying recommendations [14,15,16]. In addition, the optimal timing of initiating aspirin remains unclear. Previous data indicated a 17–20% risk reduction of early-onset preeclampsia with commencement of low-dose aspirin before 16 weeks of gestation, with a lack of benefit if started later in gestation [13, 17]. There are, however, contradictory data, suggesting benefit even when commenced after 16 weeks of gestation [12]. More recently, a risk reduction of ~60% (odds ratio 0.38; 95% confidence interval (CI), 0.20–0.74, p < 0.004) of preeclampsia was demonstrated when 150 mg of aspirin was commenced between 11 and 14 weeks of gestation [11]. Potential explanations for the observed heterogenous effects include aspirin non-adherence or resistance, varying doses and gestations at which aspirin has been used. The evidence on aspirin non-adherence and resistance is increasingly prominent in cardiology studies, however, has not been adequately examined in pregnant women [18, 19]. There also remains a difference of opinion amongst clinicians on the potential chronotherapeutic effect of aspirin [20]. These variations result in inconsistencies with clinical application of the evidence and therefore needs further exploration to provide clinicians with clarity on the optimal use of aspirin.
The why: the beginning of the use of aspirin in preeclampsia
Almost 40 years ago, aspirin was avoided in pregnancy. Shortly after, however, aspirin’s inhibitory action on cyclo-oxygenase and its effect on thromboxane (TXA2) and prostacyclin (PGI2) was discovered [21]. The understanding of the TXA2/PGI2 imbalance in the pathophysiology of preeclampsia led to the use of aspirin in managing preeclampsia, for the first time, in 1978 [22,23,24]. Subsequently, aspirin was shown to have a prophylactic role in preventing preeclampsia. In 1985, when Beaufils et al. [25] used a combination of aspirin (150 mg) and dipyridamole (300 mg) in the first trimester, a significant reduction in the rate of preeclampsia in the treatment group compared to controls (0/48 vs 6/45 p < 0.05) was observed. This was further noted in 1991 when the EPREDA trial proposed an added benefit of aspirin (150 mg daily) in reducing the rate of fetal growth retardation in comparison with placebo (20 [13%] vs. 19 [26%]; p < 0.02) [26]. Following this, however, the evidence for the use of prophylactic aspirin had been contradictory, with studies demonstrating equivocal or a lack of benefit in preventing preeclampsia [27,28,29]. These studies were confounded by usage of varying doses of aspirin, heterogenous inclusion criteria and different timing of aspirin initiation. More recently, aspirin has once again re-emerged as a promising prophylactic agent [11, 13, 30]. Between 2007 and 2010, Bujold et al. [13] and Askie et al. [30] demonstrated a benefit in the use of aspirin when commenced before 16 weeks of gestation (Risk reduction 0.47: 95% CI 0.34–0.65). These studies led to more recent studies that have supported the use of aspirin [11, 12, 16]. Although these studies have demonstrated variable outcomes, they have, collectively, provided useful safety data by demonstrating a lack of association between aspirin and complications in both the mother and fetus [13, 27, 28, 31].
The how: mechanism of aspirin as a prophylactic therapy in preeclampsia
Aspirin is a non-selective, irreversible cyclooxygenase (COX) inhibitor (Fig. 2). It prevents the production of TXA2 in platelets by acetylating a serine residue at position 529 of the COX-1 isoform. This results in an antithrombotic effect with a decrease in the TXA2/PGI2 ratio [21]. Early studies on the mechanism of aspirin’s action in preeclampsia demonstrated an aspirin-induced decrease of TXA2 concentration and mediation of the unbalanced TXA/PGI2 ratio [32, 33]. Roberts et al. [34] noted a dose-mediated response with prostacyclin production in nonpregnant female and male volunteers. Their study demonstrated that 50 mg of aspirin daily reduced TXA2 production in 95% of their volunteers without an alteration in prostacyclin production. The use of 100–300 mg of aspirin a day, however, completely inhibited TXA2 production and partially inhibited prostacyclin production [34]. When examined in pregnant women, Sibai et al. [35] demonstrated a dose response inhibition of TXA2 production with a 98% reduction after 1 week of 80 mg aspirin therapy. It was then hypothesized that aspirin improves placental blood flow and minimizes risk of placental thrombosis by reducing TXA2 and PGI2. Studies that examined the correlation between TXA2/PGI2 levels and maternal uterine artery pulsatile indexes (PI) demonstrated that aspirin reduced platelet aggregation and inhibited vasoconstriction when an enhanced uterine blood flow was noted [36]. Aspirin, however, has only demonstrated a modest benefit in the prevention of preeclampsia despite the postulated modulation of the TXA2/PGI2 imbalance [37, 38]. One explanation for this variation is that the TXA2/PGI2 imbalance is not the only pathologic pathway of preeclampsia. There may also be a wide inter-patient variation in the degree of aspirin effect in addition to a gestation specific effect. This is partially answered in the observation that the preventative effect of aspirin with early onset preeclampsia is greater than late onset preeclampsia. This suggests that the effect of aspirin may be on placental development and less so on maternal biochemical dysregulation [16, 17].
Mechanism of action of aspirin and aspirin-triggered lipoxin (ATL). Legends for Fig. 2: 15 (R) HETE 15-hydroxyeicosatetraenoic acid, COX cyclooxygenase, ERK extracellular-signal-regulated kinase, IL 10 interleukin 10, IL8 interleukin 8, IL 6 interleukin 6, IL 1β interleukin 1β, MMP 3 matrix metalloproteinase-3, TNF-α tumour necrosis factor ɑ
In 1989, Claria et al. [39] described the generation of a 15-epi-Lipoxin A4, better known as aspirin-triggered lipoxins (ATLs). ATLs are the epimeric form of endogenous lipoxin A4, which is a potent immunomodulator and antioxidant. It can block the generation of reactive oxygen species in endothelial cells, inhibit nuclear factor-κB cells (NF-κB) activation and tumour necrosis factor-ɑ (TNF-α) secretion in activated T cells (Fig. 2) [40,41,42,43]. Use of exogenous ATL has demonstrated positive outcomes in experimental models of inflammatory conditions such as peritonitis [44], colitis [45], ischaemic reperfusion injury [44], and angiogenesis [46]. The generation of endogenous ATL with aspirin consumption was first confirmed by Fiorucci et al. [47]. This was further demonstrated by Chiang et al. [48] who showed a rise in ATL levels with low-dose aspirin (81 mg aspirin group (0.25 ± 0.63 ng/ml, P = 0.04). No effect on ATL was noted with the use of high dose aspirin (325 mg group (0.16 ± 0.71 ng/ml) and 650 mg group (0.01 ± 0.75 ng/ml, P = 0.96) [48].
More recently, ATL has been shown to have a role in reproductive medicine. Cell culture studies have demonstrated that exogenous ATL has the potential to reverse the inflammatory process observed in preeclampsia by upregulating interleukin-10 (IL10) and nitric oxide, whereas downregulating the generation of TNF-α [49, 50]. Zhangye Xu et al. [51] demonstrated that lipoxin A4, an endogenous lipoxin, is reduced by twofold in women with established preeclampsia. This was also demonstrated in a lipopolysaccharide induced preeclampsia rat model [51]. Furthermore, low first trimester lipoxin A4 has also been proposed as a potential screening biomarker in identifying women who are at risk of intrauterine growth dysregulation [52]. Lipa et al. [52] demonstrated decreased values of lipoxin A4 concentrations, both in small for gestational age and large for gestational age groups, when compared with average gestation for age (68.91 ± 33.72 and 68.30 ± 23.49 vs. 102.13 ± 121.9, respectively) [52]. Given the lipoxin A4 dysregulation in high-risk women, increasing understanding of ATL may provide an alternative mechanism by which aspirin modulates preeclampsia risk.
The who: risk stratifying women for prophylactic aspirin therapy
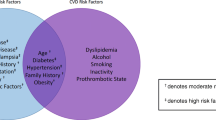
Identifying women who will benefit from the use of prophylactic aspirin is challenging. The current clinical guidelines suggest risk stratifying women based on clinical history (Table 1) [23, 24]. A recent meta-analysis demonstrated that clinical history alone has a detection rate of 37% for early-onset preeclampsia and 28.9% of late-onset preeclampsia [53]. In nulliparous women, clinical history alone detects 37% of preeclampsia for a false positive rate of 5% [54]. Prior history of preeclampsia was the second strongest predictor factor, which is not applicable to nulliparous women [55]. Mean arterial pressure (MAP) compared with systolic or diastolic reading alone has been discussed as another clinical predictor of preeclampsia. A prospective screening study utilising MAP (expressed as log multiple of the median) measured between 11 + 0 and 13 + 6 weeks gestation and maternal variables demonstrated a detection rate of preeclampsia of 62.5% for a false-positive rate of 10% [56, 57]. A predictive testing model incorporating maternal factors and second trimester uterine artery pulsitivity index (PI) demonstrated a detection rate of early and late preeclampsia 100% and 56.4%, respectively, for a false-positive rate of 10% [56]. More recently, the combined utility of pregnancy associated plasma protein A, PlGF, MAP, and clinical history has been shown to be sensitive, when used in combination, detecting 96% of cases of preeclampsia requiring delivery before 34 weeks and 54% of all cases of preeclampsia with a false-positive rate of 10% [58].
Although the utility of screening algorithms has shown to be effective in identifying women at risk, particularly nulliparous women, its use is often limited by accessibility to the required resources. Therefore, many centres rely on clinical history for this purpose. In keeping with this, the current guidelines recommend the use of clinical history in risk stratifying women [14, 15, 59]. This, however, may change based on the accessibility and cost- benefit analysis of the new screening markers.
The when: does timing of intervention and chronotherapy matter?
The optimal timing of the initiation of aspirin therapy is still unclear. Some studies have suggested greater benefit with prophylactic aspirin when initiated before 16 weeks of gestation [13, 17]. Meta-analysis by Bujold et al. [13] demonstrated that commencing low-dose aspirin before 16 weeks of gestation significantly reduced the rate of preeclampsia (RR 0.47, 95% CI 0.34–0.65), intrauterine growth retardation (IUGR) (RR 0.44, 95% CI 0.30–0.65) and severe preeclampsia (RR 0.09, 95% CI 0.02–0.37) [13]. The observed benefit was lost when aspirin was commenced after 16 weeks of gestation (preeclampsia: RR 0.81, 95% CI 0.63–1.03; IUGR: RR 0.98, 95% CI 0.87–1.10) [13]. Contrary to this, another meta-analysis suggested that the 16 weeks gestation cutoff was not as relevant [12]. Meher et al. [12] conducted an individual participant dataset analysis of the PARIS Collaboration dataset and suggested the outcomes with the use of aspirin was consistent when commenced before or after 16 weeks of gestation (RR) 0.90 (95% CI 0.79–1.03) for < 16 weeks and RR 0.90 (95% CI 0.83–0.98) for ≥ 16 weeks (interaction test p = 0.98) [12, 30]. Their analysis of the PARIS Collaboration data, however, only demonstrated a very modest overall preeclampsia reduction of ~10% with the use of prophylactic aspirin [12]. The current guidelines suggest initiating aspirin before 16 weeks of gestation [14, 15, 59]; however, recent studies suggest that a greater benefit maybe observed with earlier initiation of aspirin [11, 16, 60]. This, however, has not been assessed with a prospective randomized trial.
A similarly controversial issue is the chronotherapy of aspirin. Cardiology studies have demonstrated greater nocturnal blood pressure regulation with the use of nocturnal aspirin [61,62,63]. Hermida et al. [63] noted that a significant blood pressure reduction was observed in males and non-pregnant females with untreated grade 1 hypertension (systolic blood pressure (SBP) 140–159 mm Hg, diastolic blood pressure (DBP) 80–99 mm Hg), who received nocturnal aspirin in comparison with daytime aspirin (decrease of 7.2/4.9 mm Hg in SBP/DBP; p = 0.001). The reduction in nocturnal blood pressure mean was double in non-dippers (11.0/7.1 mm Hg) compared with dippers (5.5/3.3 mm Hg; p = 0.001) [63]. Although the exact mechanism for this is unclear, some studies have suggested that a decrease in the renin and aldosterone pressor system could constitute a biologically plausible explanation for better blood pressure regulation when aspirin is ingested at bedtime [64,65,66]. Snoep et al. [65] found that low-dose aspirin (100 mg) when ingested at bedtime, compared with awakening, significantly diminished not only the 24 h level of plasma renin activity (RR 14% (95% CI 5–22) p = 0.003) but also the 24 h urine excretion of cortisol (RR 16% (95% CI 5–30) p = 0.05), dopamine (RR 14% (95% CI 1–28) p = 0.04) and norepinephrine (RR of 40% (95% CI − 1 to 88) p = 0.02) [65].This study was undertaken in non-pregnant females and males with a two of three male predominance. The clinical effect of chronotherapy of aspirin on primary and secondary cardiovascular outcomes is still unknown and is subject to ongoing research (CARING) ClinicalTrials.gov Identifier: NCT00725127.
Translating this to high-risk pregnant women, some studies have suggested a chronotherapeutic role of aspirin in preventing preeclampsia [20, 67]. In a prospective, randomized, double-blind, placebo-controlled, chronotherapy trial, 350 high-risk pregnant women were randomly assigned to 1 of the 6 groups defined according to treatment (placebo or aspirin, 100 mg/day) and time of treatment: upon awakening, 8 h after awakening, or at bedtime [20]. Ayala et al. [20] noted that ingesting 100 mg of aspirin daily at bedtime starting at ≤ 16 weeks of gestation decreased SBP on an average of 12.4/8.1 mm Hg (measured in 24 h SBP/DBP midline estimating statistics of rhythm) compared with ingesting aspirin upon awakening (p < 0.001) [20]. In addition, a lower rate of preeclampsia and IUGR was observed in the group of women who ingested aspirin at bedtime and in the group who ingested aspirin 8 h after awakening in comparison with those who ingested aspirin upon awakening (hazard ratio 0.19, 95% CI 0.10–0.39 p < 0.001) [20]. Although significant, this was not the primary outcome of the study. The mechanism for this, however, remains unclear, as it has not been adequately studied in pregnancy.
In keeping with this, more recent studies have utilised nocturnal aspirin in their studies [11, 16]. Although the current guidelines do not specify the utility of chronotherapy, this is likely to change as the evidence for this strengthens.
Aspirin resistance vs non-adherence
In 2009, the Working Group on Thrombosis of European Society of Cardiology categorized aspirin resistance as either clinical resistance or laboratory resistance [18]. Clinical resistance was described as the occurrence of a cardiovascular event despite aspirin therapy, whereas laboratory resistance or non-responsiveness was described as insufficient platelet inhibition activity. The relatively modest benefit of prophylactic aspirin in some studies in preventing preeclampsia has raised the question of clinical aspirin resistance among high-risk pregnant women. The actual incidence of clinical aspirin resistance in the pregnant cohort is unknown. Approximately 30% of pregnant women demonstrate a lack of laboratory response to aspirin at doses < 100 mg based on platelet function analyser (PFA-100) [68]. However, biochemical aspirin non-responsiveness, based on PFA-100, with low-dose aspirin (81 mg daily) is overcome in pregnant women with higher doses of aspirin (162 mg daily) [68]. A similar study which used Verifynow, a point-of-care assay in patients with coronary artery disease, demonstrated better inhibitory activity with higher doses of aspirin. Daily aspirin doses of < 100 mg were associated with increased prevalence of biochemical aspirin resistance compared with 150 mg and 300 mg daily (30.2% vs. 16.7% vs. 0%, P = 0.0062) [69]. Early studies of prophylactic aspirin in high-risk women utilised an aspirin dose range of 50–150 mg. The first study to suggest a prophylactic role of aspirin in preeclampsia utilised 150 mg of aspirin concurrent with dipyridamole [25]. However, Benigni et al. [70] demonstrated a prophylactic role with the use of only 60 mg daily. The common factor in both these studies was the early (before the end of the first trimester) use of aspirin. The current clinical guidelines recommend the use of low-dose aspirin with a dose range from 81–150 mg [14, 15, 59]. Although most centres use aspirin 100 mg daily, there has been a recent shift to 150 mg daily based on more recent clinical studies [11, 16]. There, however, is a need for stronger evidence to support the use of 150 mg aspirin over 100 mg aspirin in the prevention of preeclampsia in addition to more data on its safety profile.
Poor adherence has been identified as the most common reason for clinical and laboratory aspirin resistance [71, 72]. Non-adherence with prescribed medications among pregnant women is reported to vary from 20% to 80% [73,74,75]. Non-adherence to aspirin in pregnant women was found to vary from 20% to 45% [76, 77]. Recent data suggest that high-risk women will have to be at least 90% adherent with their prescribed aspirin to demonstrate a benefit from it; however, most studies have utilised self-reporting questionnaires to assess adherence, which have not been validated with biochemical assessment [76, 77]. The utility of biochemical measure of aspirin adherence was recently examined by Navaratnam et al. [72] with the use of PFA100 and serum salicylate. They demonstrated that in women with a variable response or lack of response in PFA 100 levels, aspirin non-adherence or variable adherence was more likely than aspirin resistance (41% vs. 0%) [72].
Other factors that may contribute toward aspirin resistance include differences in aspirin preparation: enteric coated vs. non-enteric coated, the effect of smoking, and the presence of gene polymorphism or cellular dysregulation (Fig. 3). The consensus on the recommended preparation of aspirin to be used in pregnancy remains unclear. Enteric-coated aspirin slows and reduces systemic absorption by ~20% in healthy non-pregnant volunteers [78]. Others have demonstrated that it does not alter absorption [79, 80]. These factors, however, have not been examined in the pregnant population and its influence on the clinical efficacy of aspirin in the high-risk pregnancy population remains unknown.
Factors that influences aspirin resistance. Legends for Fig. 3 GP glycoprotein, vWF Von Willebrand factor
Conclusion
The use of aspirin in preventing preeclampsia has evolved over the last 40 years. Although the evidence supporting the use of aspirin in the obstetric setting has re-emerged, many unanswered questions remain in optimizing the use of aspirin in preventing preeclampsia. The key questions that warrant clarification to assist clinicians are the mechanism of action, optimal dose of aspirin, and the chronotherapy of aspirin. Clinicians should also pay more attention to patients’ adherence as recent evidence suggests an underestimation of aspirin non-adherence among high-risk pregnant women.
References
Duley L. The global impact of pre-eclampsia and eclampsia. Semin Perinatol. 2009;33:130–7.
Jeyabalan A. Epidemiology of preeclampsia: impact of obesity. Nutr Rev. 2013;71(Suppl 1):S18–25.
Thornton C, Dahlen H, Korda A, Hennessy A. The incidence of preeclampsia and eclampsia and associated maternal mortality in Australia from population-linked datasets: 2000-2008. Am J Obstet Gynecol. 2013;208:476 e1–5.
Maynard SE, Min JY, Merchan J, Lim KH, Li J, Mondal S, et al. Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J Clin Invest. 2003;111:649–58.
Levine RJ, Lam C, Qian C, Yu KF, Maynard SE, Sachs BP, et al. Soluble endoglin and other circulating antiangiogenic factors in preeclampsia. N Engl J Med. 2006;355:992–1005.
Maynard SE, Venkatesha S, Thadhani R, Karumanchi SA. Soluble Fms-like tyrosine kinase 1 and endothelial dysfunction in the pathogenesis of preeclampsia. Pediatr Res. 2005;57(5 Pt 2):1R–7R.
Makris A, Yeung KR, Lim SM, Sunderland N, Heffernan S, Thompson JF, et al. Placental growth factor reduces blood pressure in a uteroplacental ischemia model of preeclampsia in nonhuman primates. Hypertension. 2016;67:1263–72.
Onda K, Tong S, Beard S, Binder N, Muto M, Senadheera SN, et al. Proton pump inhibitors decrease soluble fms-like tyrosine kinase-1 and soluble endoglin secretion, decrease hypertension, and rescue endothelial dysfunction. Hypertension. 2017;69:457–68.
Thadhani R, Hagmann H, Schaarschmidt W, Roth B, Cingoez T, Karumanchi SA, et al. Removal of soluble Fms-like tyrosine kinase-1 by dextran sulfate apheresis in preeclampsia. J Am Soc Nephrol. 2016;27:903–13.
Wester-Rosenlof L, Casslen V, Axelsson J, Edstrom-Hagerwall A, Gram M, Holmqvist M, et al. A1M/alpha1-microglobulin protects from heme-induced placental and renal damage in a pregnant sheep model of preeclampsia. PLoS ONE. 2014;9:e86353.
Rolnik DL, Wright D, Poon LC, O’Gorman N, Syngelaki A, de Paco Matallana C, et al. Aspirin versus placebo in pregnancies at high risk for preterm preeclampsia. N Engl J Med. 2017;377:613–22.
Meher S, Duley L, Hunter K, Askie L. Antiplatelet therapy before or after 16 weeks’ gestation for preventing preeclampsia: an individual participant data meta-analysis. Am J Obstet Gynecol. 2017;216:121–8 e2.
Bujold E, Roberge S, Lacasse Y, Bureau M, Audibert F, Marcoux S, et al. Prevention of preeclampsia and intrauterine growth restriction with aspirin started in early pregnancy: a meta-analysis. Obstet Gynecol. 2010;116(2 Pt 1):402–14.
Lowe SA, Bowyer L, Lust K, McMahon LP, Morton M, North RA, et al. SOMANZ guidelines for the management of hypertensive disorders of pregnancy 2014. Aust N Z J Obstet Gynaecol. 2015;55:e1–29.
Redman CW. Hypertension in pregnancy: the NICE guidelines. Heart. 2011;97:1967–9.
Park F, Russo K, Williams P, Pelosi M, Puddephatt R, Walter M, et al. Prediction and prevention of early-onset pre-eclampsia: impact of aspirin after first-trimester screening. Ultrasound Obstet Gynecol. 2015;46:419–23.
Roberge S, Nicolaides K, Demers S, Hyett J, Chaillet N, Bujold E. The role of aspirin dose on the prevention of preeclampsia and fetal growth restriction: systematic review and meta-analysis. Am J Obstet Gynecol. 2017;216:110–20 e6.
Kuliczkowski W, Witkowski A, Polonski L, Watala C, Filipiak K, Budaj A, et al. Interindividual variability in the response to oral antiplatelet drugs: a position paper of the Working Group on antiplatelet drugs resistance appointed by the Section of Cardiovascular Interventions of the Polish Cardiac Society, endorsed by the Working Group on Thrombosis of the European Society of Cardiology. Eur Heart J. 2009;30:426–35.
Kuliczkowski W, Halawa B, Korolko B, Mazurek W. Aspirin resistance in ischaemic heart disease. Kardiol Pol. 2005;62:14–25.
Ayala DE, Ucieda R, Hermida RC. Chronotherapy with low-dose aspirin for prevention of complications in pregnancy. Chronobiol Int. 2013;30:260–79.
Roth GJ, Majerus PW. The mechanism of the effect of aspirin on human platelets. I. Acetylation of a particulate fraction protein. J Clin Invest. 1975;56:624–32.
Goodlin RC, Haesslein HO, Fleming J. Aspirin for the treatment of recurrent toxaemia. Lancet (Lond, Engl). 1978;2:51.
Walsh SW. Thromboxane production in placentas of women with preeclampsia. Am J Obstet Gynecol. 1989;160:1535–6.
Walsh SW. Physiology of low-dose aspirin therapy for the prevention of preeclampsia. Semin Perinatol. 1990;14:152–70.
Beaufils M, Uzan S, Donsimoni R, Colau JC. Prevention of pre-eclampsia by early antiplatelet therapy. Lancet (Lond, Engl). 1985;1:840–2.
Uzan S, Beaufils M, Breart G, Bazin B, Capitant C, Paris J. Prevention of fetal growth retardation with low-dose aspirin: findings of the EPREDA trial. Lancet (Lond, Engl). 1991;337:1427–31.
CLASP (Collaborative Low-dose Aspirin Study in Pregnancy) Collaborative Group. CLASP: a randomised trial of low-dose aspirin for the prevention and treatment of pre-eclampsia among 9364 pregnant women. Lancet (Lond, Engl). 1994;343:619–29.
Rotchell YE, Cruickshank JK, Gay MP, Griffiths J, Stewart A, Farrell B, et al. Barbados Low Dose Aspirin Study in Pregnancy (BLASP): a randomised trial for the prevention of pre-eclampsia and its complications. Br J Obstet Gynaecol. 1998;105:286–92.
Caritis S, Sibai B, Hauth J, Lindheimer MD, Klebanoff M, Thom E, et al. Low-dose aspirin to prevent preeclampsia in women at high risk. National Institute of Child Health and Human Development Network of Maternal-Fetal Medicine Units. N Engl J Med. 1998;338:701–5.
Askie LM, Duley L, Henderson-Smart DJ, Stewart LA, Group PC. Antiplatelet agents for prevention of pre-eclampsia: a meta-analysis of individual patient data. Lancet (Lond, Engl). 2007;369:1791–8.
Sibai BM, Caritis SN, Thom E, Shaw K, McNellis D. Low-dose aspirin in nulliparous women: safety of continuous epidural block and correlation between bleeding time and maternal-neonatal bleeding complications. National Institute of Child Health and Human Developmental Maternal-Fetal Medicine Network. Am J Obstet Gynecol. 1995;172:1553–7.
Schiff E, Peleg E, Goldenberg M, Rosenthal T, Ruppin E, Tamarkin M, et al. The use of aspirin to prevent pregnancy-induced hypertension and lower the ratio of thromboxane A2 to prostacyclin in relatively high risk pregnancies. N Engl J Med. 1989;321:351–6.
Viinikka L, Hartikainen-Sorri AL, Lumme R, Hiilesmaa V, Ylikorkala O. Low dose aspirin in hypertensive pregnant women: effect on pregnancy outcome and prostacyclin-thromboxane balance in mother and newborn. Br J Obstet Gynaecol. 1993;100:809–15.
Roberts MS, Joyce RM, McLeod LJ, Vial JH, Seville PR. Slow-release aspirin and prostaglandin inhibition. Lancet (Lond, Engl). 1986;1:1153–4.
Sibai BM, Mirro R, Chesney CM, Leffler C. Low-dose aspirin in pregnancy. Obstet Gynecol. 1989;74:551–7.
Haapsamo M, Martikainen H, Rasanen J. Low-dose aspirin reduces uteroplacental vascular impedance in early and mid gestation in IVF and ICSI patients: a randomized, placebo-controlled double-blind study. Ultrasound Obstet Gynecol. 2008;32:687–93.
Coomarasamy A, Papaioannou S, Gee H, Khan KS. Aspirin for the prevention of preeclampsia in women with abnormal uterine artery Doppler: a meta-analysis. Obstet Gynecol. 2001;98(5 Pt 1):861–6.
Vainio M, Riutta A, Koivisto AM, Maenpaa J. Prostacyclin, thromboxane A and the effect of low-dose ASA in pregnancies at high risk for hypertensive disorders. Acta Obstet Gynecol Scand. 2004;83:1119–23.
Claria J, Serhan CN. Aspirin triggers previously undescribed bioactive eicosanoids by human endothelial cell-leukocyte interactions. Proc Natl Acad Sci USA. 1995;92:9475–9.
Goh J, Baird AW, O’Keane C, Watson RW, Cottell D, Bernasconi G, et al. Lipoxin A(4) and aspirin-triggered 15-epi-lipoxin A(4) antagonize TNF-alpha-stimulated neutrophil-enterocyte interactions in vitro and attenuate TNF-alpha-induced chemokine release and colonocyte apoptosis in human intestinal mucosa ex vivo. J Immunol. 2001;167:2772–80.
Ariel A, Chiang N, Arita M, Petasis NA, Serhan CN. Aspirin-triggered lipoxin A4 and B4 analogs block extracellular signal-regulated kinase-dependent TNF-alpha secretion from human T cells. J Immunol. 2003;170:6266–72.
Nascimento-Silva V, Arruda MA, Barja-Fidalgo C, Villela CG, Fierro IM. Novel lipid mediator aspirin-triggered lipoxin A4 induces heme oxygenase-1 in endothelial cells. Am J Physiol Cell Physiol. 2005;289:C557–63.
Mitchell S, Thomas G, Harvey K, Cottell D, Reville K, Berlasconi G, et al. Lipoxins, aspirin-triggered epi-lipoxins, lipoxin stable analogues, and the resolution of inflammation: stimulation of macrophage phagocytosis of apoptotic neutrophils in vivo. J Am Soc Nephrol. 2002;13:2497–507.
Chiang N, Takano T, Arita M, Watanabe S, Serhan CN. A novel rat lipoxin A4 receptor that is conserved in structure and function. Br J Pharmacol. 2003;139:89–98.
Gewirtz AT, Collier-Hyams LS, Young AN, Kucharzik T, Guilford WJ, Parkinson JF, et al. Lipoxin a4 analogs attenuate induction of intestinal epithelial proinflammatory gene expression and reduce the severity of dextran sodium sulfate-induced colitis. J Immunol. 2002;168:5260–7.
Fierro IM, Kutok JL, Serhan CN. Novel lipid mediator regulators of endothelial cell proliferation and migration: aspirin-triggered-15R-lipoxin A(4) and lipoxin A(4). J Pharmacol Exp Ther. 2002;300:385–92.
Fiorucci S, Distrutti E, Mencarelli A, Morelli A, Laufor SA, Cirino G, et al. Evidence that 5-lipoxygenase and acetylated cyclooxygenase 2-derived eicosanoids regulate leukocyte-endothelial adherence in response to aspirin. Br J Pharmacol. 2003;139:1351–9.
Chiang N, Hurwitz S, Ridker PM, Serhan CN. Aspirin has a gender-dependent impact on antiinflammatory 15-epi-lipoxin A4 formation: a randomized human trial. Arterioscler Thromb Vasc Biol. 2006;26:e14–7.
Alvarez AM, Mulla MJ, Chamley LW, Cadavid AP, Abrahams VM. Aspirin-triggered lipoxin prevents antiphospholipid antibody effects on human trophoblast migration and endothelial cell interactions. Arthritis Rheumatol. 2015;67:488–97.
Gil-Villa AM, Norling LV, Serhan CN, Cordero D, Rojas M, Cadavid A. Aspirin triggered-lipoxin A4 reduces the adhesion of human polymorphonuclear neutrophils to endothelial cells initiated by preeclamptic plasma. Prostaglandins Leukot Essent Fat Acids. 2012;87:127–34.
Zhangye XuMD, Feng Zhao MM, Feng Lin MM, Huiqiu Xiang MM, Ni Wang MM, Duyun Ye,MM, Yinping Huang MM. Preeclampsia is associated with a deficiency of lipoxin A4, an endogenous anti-inflammatory mediator. Fertil Steril. 2014;102:282–90.e4.
Lipa M, Bomba-Opon D, Lipa J, Bartnik P, Bartoszewicz Z, Wielgos M. Lipoxin A4 (LXA4) as a potential first trimester biochemical marker of intrauterine growth disorders. J Matern Fetal Neonatal Med. 2017;30:2495–7.
Poon LC, Kametas NA, Chelemen T, Leal A, Nicolaides KH. Maternal risk factors for hypertensive disorders in pregnancy: a multivariate approach. J Hum Hypertens. 2010;24:104–10.
North RA, McCowan LM, Dekker GA, Poston L, Chan EH, Stewart AW, et al. Clinical risk prediction for pre-eclampsia in nulliparous women: development of model in international prospective cohort. BMJ. 2011;342:d1875.
Visintin C, Mugglestone MA, Almerie MQ, Nherera LM, James D, Walkinshaw S, et al. Management of hypertensive disorders during pregnancy: summary of NICE guidance. BMJ. 2010;341:c2207.
Onwudiwe N, Yu CK, Poon LC, Spiliopoulos I, Nicolaides KH. Prediction of pre-eclampsia by a combination of maternal history, uterine artery Doppler and mean arterial pressure. Ultrasound Obstet Gynecol. 2008;32:877–83.
Poon LC, Kametas NA, Valencia C, Chelemen T, Nicolaides KH. Hypertensive disorders in pregnancy: screening by systolic diastolic and mean arterial pressure at 11-13 weeks. Hypertens Pregnancy. 2011;30:93–107.
Akolekar R, Syngelaki A, Poon L, Wright D, Nicolaides KH. Competing risks model in early screening for preeclampsia by biophysical and biochemical markers. Fetal Diagn Ther. 2013;33:8–15.
Tranquilli AL. Introduction to ISSHP new classification of preeclampsia. Pregnancy Hypertens. 2013;3:58–9.
Zvanca ME, Bot M, Radu D, Radu N, Petca A. Impact of early supplementation with low-dose aspirin on functional first trimester parameters in low-risk pregnancies. J Matern Fetal Neonatal Med. 2017:1–6. https://doi.org/10.1080/14767058.2017.1387532
Hermida RC, Ayala DE, Calvo C, Lopez JE. Aspirin administered at bedtime, but not on awakening, has an effect on ambulatory blood pressure in hypertensive patients. J Am Coll Cardiol. 2005;46:975–83.
Hermida RC, Ayala DE, Calvo C, Lopez JE, Fernandez JR, Mojon A, et al. Administration time-dependent effects of aspirin on blood pressure in untreated hypertensive patients. Hypertension. 2003;41:1259–67.
Hermida RC, Ayala DE, Calvo C, Lopez JE, Mojon A, Rodriguez M, et al. Differing administration time-dependent effects of aspirin on blood pressure in dipper and non-dipper hypertensives. Hypertension. 2005;46:1060–8.
Kanzik I, Cakici I, Ersoy S, Ark M, Abacioglu N, Zengil H. Effects of cyclooxygenase and lipoxygenase inhibitors on digoxin-induced arrhythmias and haemodynamics in guinea-pigs. Pharmacol Res. 1992;26:305–16.
Snoep JD, Hovens MM, Pasha SM, Frolich M, Pijl H, Tamsma JT, et al. Time-dependent effects of low-dose aspirin on plasma renin activity, aldosterone, cortisol, and catecholamines. Hypertension. 2009;54:1136–42.
Cornelissen G, Halberg F, Prikryl P, Dankova E, Siegelova J, Dusek J. Prophylactic aspirin treatment: the merits of timing. International Womb-to-Tomb Chronome Study Group. JAMA. 1991;266:3128–9.
Hermida RC, Ayala DE, Fernandez JR, Mojon A, Alonso I, Silva I, et al. Administration time-dependent effects of aspirin in women at differing risk for preeclampsia. Hypertension. 1999;34(4 Pt 2):1016–23.
Caron N, Rivard GE, Michon N, Morin F, Pilon D, Moutquin JM, et al. Low-dose ASA response using the PFA-100 in women with high-risk pregnancy. J Obstet Gynaecol Can. 2009;31:1022–7.
Lee PY, Chen WH, Ng W, Cheng X, Kwok JY, Tse HF, et al. Low-dose aspirin increases aspirin resistance in patients with coronary artery disease. Am J Med. 2005;118:723–7.
Benigni A, Gregorini G, Frusca T, Chiabrando C, Ballerini S, Valcamonico A, et al. Effect of low-dose aspirin on fetal and maternal generation of thromboxane by platelets in women at risk for pregnancy-induced hypertension. N Engl J Med. 1989;321:357–62.
Cuisset T, Frere C, Quilici J, Gaborit B, Bali L, Poyet R, et al. Aspirin noncompliance is the major cause of “aspirin resistance” in patients undergoing coronary stenting. Am Heart J. 2009;157:889–93.
Navaratnam K, Alfirevic A, Jorgensen A, Alfirevic Z. Aspirin non-responsiveness in pregnant women at high-risk of pre-eclampsia. Eur J Obstet Gynecol Reprod Biol. 2018;221:144–50.
Lupattelli A, Spigset O, Nordeng H. Adherence to medication for chronic disorders during pregnancy: results from a multinational study. Int J Clin Pharm. 2014;36:145–53.
Julsgaard M, Norgaard M, Hvas CL, Buck D, Christensen LA. Self-reported adherence to medical treatment prior to and during pregnancy among women with ulcerative colitis. Inflamm Bowel Dis. 2011;17:1573–80.
Sawicki E, Stewart K, Wong S, Leung L, Paul E, George J. Medication use for chronic health conditions by pregnant women attending an Australian maternity hospital. Aust N Z J Obstet Gynaecol. 2011;51:333–8.
Abheiden CN, van Reuler AV, Fuijkschot WW, de Vries JI, Thijs A, de Boer MA. Aspirin adherence during high-risk pregnancies, a questionnaire study. Pregnancy Hypertens. 2016;6:350–5.
Wright D, Poon LC, Rolnik DL, Syngelaki A, Delgado JL, Vojtassakova D, et al. Aspirin for evidence-based preeclampsia prevention trial: influence of compliance on beneficial effect of aspirin in prevention of preterm preeclampsia. Am J Obstet Gynecol. 2017;217:685e1–e5.
Cox D, Maree AO, Dooley M, Conroy R, Byrne MF, Fitzgerald DJ. Effect of enteric coating on antiplatelet activity of low-dose aspirin in healthy volunteers. Stroke. 2006;37:2153–8.
Van Hecken A, Juliano ML, Depre M, De Lepeleire I, Arnout J, Dynder A, et al. Effects of enteric-coated, low-dose aspirin on parameters of platelet function. Aliment Pharmacol Ther. 2002;16:1683–8.
Karha J, Rajagopal V, Kottke-Marchant K, Bhatt DL. Lack of effect of enteric coating on aspirin-induced inhibition of platelet aggregation in healthy volunteers. Am Heart J. 2006;151:976 e7–11.
Acknowledgements
We thank Mr Rajeev Wijesekera for his assistance with the design of the illustrations used in this manuscript.
Author contributions
RS, AH, and AM contributed equally and collaboratively in the literature review, proof reading, and preparation of this manuscript.
Funding
None to declare.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Details of ethics approval
Not applicable.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Shanmugalingam, R., Hennessy, A. & Makris, A. Aspirin in the prevention of preeclampsia: the conundrum of how, who and when. J Hum Hypertens 33, 1–9 (2019). https://doi.org/10.1038/s41371-018-0113-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41371-018-0113-7
- Springer Nature Limited
This article is cited by
-
The Involvement of MicroRNAs in SARS-CoV-2 Infection Comorbid with HIV-Associated Preeclampsia
Current Hypertension Reports (2021)