Abstract
Background/objectives
We have recently proposed and validated a simple and accurate method to estimate whole-body fat percentage in adults, the relative fat mass (RFM), derived from the ratio of height to waist circumference. We aimed to identify RFM cutoffs to diagnose obesity based on the association between RFM and all-cause mortality.
Subjects/methods
We used data from adult participants (≥20 years of age, n = 43,793) of the National Health and Nutrition Examination Survey (NHANES) 1999–2014 linked with death certificate records from the National Death Index. Optimal RFM cutoffs were determined using receiver-operating characteristic analysis (the Youden’s index and the Euclidean minimum distance to the coordinate (0,1)).
Results
Final dataset for analyses comprised 31,008 adults. During a median follow-up of 8.3 years (IQR, 7.6–13.7), 2,517 deaths occurred. Youden and Euclidean optimal cutoffs of baseline RFM for all-cause mortality were 40.8% and 41.6% for women, and 30.9% and 28.9% for men, respectively. Similar cutoffs were obtained using measured whole-body fat percentage by dual energy X-ray absorptiometry. Adjusting for age, BMI category, ethnicity, education level, and smoking status, the hazard ratio for mortality using Cox proportional hazard regression was 1.41 (95% CI, 1.02–1.95) among women who had an RFM of 40.0–44.9% compared with women who had an RFM <35% (P = 0.035). Among men, the hazard ratio was 1.57 (95% CI, 1.07–2.30) among those with an RFM of 30.0–34.9% compared with men who had an RFM <25% (P = 0.020). Similar adjusted hazard ratios for same RFM categories were obtained in our validation population (NHANES III, n = 12,650, median follow-up: 23.3 years): 1.42 (95% CI, 1.01–2.00) among women (P = 0.043) and 1.50 (95% CI, 1.07–2.10) among men (P = 0.021).
Conclusions
We suggest rounded RFM cutoffs of 40% for women and 30% for men to diagnose obesity and identify individuals at higher risk of death.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
High body fat percentage is associated with mortality [1,2,3,4,5,6,7]. To date, direct assessment of body fat percentage requires the use of sophisticated and costly equipment, rarely available in a clinical setting. To address this limitation, we recently proposed and validated an alternative simple and inexpensive method to estimate whole-body fat percentage in adults, the relative fat mass (RFM) [8]. RFM was derived from the ratio of height to waist circumference. RFM better predicted and estimated whole-body fat percentage than did the body mass index (BMI) among women and men and showed a relatively good accuracy which was consistent across Mexican-Americans, European-Americans, and African-Americans. RFM also improved the misclassification of arbitrary-defined body fat excess [8].
To date, there is still controversy regarding the optimal cutoffs to diagnose obesity based on body fat percentage [9,10,11]. In the 1990s, body fat percentage cutoffs of ~35 among women and ~25 among men were suggested for the diagnosis of obesity [12, 13], although the foundation for such recommendations remains unclear. Later, healthy ranges for body fat percentage have also been proposed on the basis of their corresponding thresholds of body mass index (BMI, body weight in kilograms divided by the square of the height in meters) for underweight, overweight and obesity [14]. However, different cutoffs were proposed across age categories and ethnic groups, resulting in little practicality compared with the BMI. In addition, the findings were derived from non-representative samples and included cohorts from different countries.
Cutoffs using corresponding BMI thresholds have also been proposed for Vietnamese individuals [15]. Others have used the 60th percentile of body fat percentage to define obesity [16]. The approach of matching body fat percentage to corresponding BMI thresholds could bias the estimates due to two known limitations of BMI. First, BMI does not account for sex-related differences in body composition. Second, BMI does not distinguish between fat mass and lean mass. Consequently, there is an urgent need to establish cutoffs of body fat percentage based on its association with mortality to better identify adult individuals with obesity.
The aim of the present study was to identify RFM cutoffs to diagnose obesity based on the association between RFM, a surrogate of whole-body fat percentage, and all-cause mortality in a representative sample of the adult population of the United States.
Methods
Study population
We included for analyses data from 43,793 adult individuals (≥20 years old) who participated in the National Health and Nutrition Examination Survey (NHANES) 1999–2014. Our findings were validated using data from 18,825 adults who participated in the NHANES 1988–1994 (NHANES III) (https://wwwn.cdc.gov/nchs/nhanes/Default.aspx). NHANES is an ongoing program designed to study the health and nutritional status of the non-institutionalized population of the United States. NHANES examines a nationally representative sample using a multistage, probability sampling design [17, 18]. The unweighted response rates for the populations studied were >68% and >70% for NHANES 1999–2014 and NHANES III, respectively (https://wwwn.cdc.gov/nchs/nhanes/ResponseRates.aspx). Baseline data on demographics and physical examination were linked with the National Death Index mortality follow-up data [19]. Observations with incomplete data on body weight, height, waist circumference, age, ethnicity, education level, smoking status, and vital status were excluded. Women who were pregnant at baseline were also excluded. The presence of an existing chronic condition has been reported to affect the relationship between BMI and all-cause mortality as confounding by a preexisting disease may increase the risk of mortality [20, 21]. Thus, to minimize the possibility of reverse causation, individuals with heart disease (congestive heart failure, coronary artery disease, angina, and myocardial infarction), stroke, cancer, and renal failure at baseline were also excluded. It has been argued that early mortality exclusion could control for confounding [22]. Thus, we performed sensitivity analysis to assess the association between RFM and mortality among participants with events during the first two years of follow-up.
Individuals with stage 5 chronic kidney disease (if estimated glomerular filtration rate <15 mL/min/1.73 m2) [23] or self-reported dialysis were also excluded from analyses. Glomerular filtration rate was estimated using the Modification of Diet in Renal Disease Study equation based on serum creatinine [24]. Serum creatinine values were standardized for NHANES 1999–2000 and NHANES 2005–2006 using recommended equations [25, 26].
The present study did not require approval or exemption from the Cedars-Sinai Medical Center Institutional Review Board as it involved the analysis of publicly available de-identified data only.
Mortality data
NHANES 1999–2014 and NHANES III are independent cohorts with different lengths of follow-up for incident mortality. Participants had their vital status and cause of death ascertained through probabilistic matching to the death certificates from the National Death Index recorded up to December 31, 2015. Detailed description of the methodology of linking NHANES with mortality data has been published elsewhere [27]. We used information on all-cause mortality and mortality where the underlying cause was heart disease, stroke, or cancer. Diabetes mortality included those deaths where the underlying cause was diabetes or when diabetes was a contributor among multiple causes of death. This approach was used to face the challenge of identifying all deaths attributable to diabetes since most cases die as a result of diabetes complications and comorbidities [28].
Baseline anthropometric measurements
Waist circumference was measured at the level of the uppermost lateral border of the right ilium during standing position, recorded to the nearest 0.1 cm. Body weight was measured with an electronic scale. Height was measured with a stadiometer [29, 30].
Dual energy X-ray absorptiometry (DXA)
Whole-body fat percentage was assessed using the well accepted method DXA [31]. DXA data were available for participants of the NHANES 1999–2006 only. DXA data were used to determine the cutoffs of measured whole-body fat percentage for all-cause mortality. DXA scans were performed using a Hologic QDR 4500 A fan-beam densitometer (Hologic, Inc., Bedford, Massachusetts). A multiple imputation approach (five imputations) was applied to replace missing DXA data [32]. Body fat percentage was calculated as the ratio of DXA whole-body fat mass (g) to DXA whole-body total mass (g), multiplied by 100 [8].
RFM cutoffs for mortality
RFM, a surrogate of whole-body fat percentage, was calculated as follows: 64 − (20 × height/waist circumference) + (12 × sex); sex equals 0 for men and 1 for women. Height and waist circumference are measured in the same units [8]. Given the existing argument as to which method is best to obtain optimal diagnostic cutoffs [33], baseline RFM cutoffs to predict mortality among women and men were obtained using two widely used methods: the Youden’s index [34] and the Euclidean minimum distance from the receiver-operating-characteristic curve analysis to the coordinates (0,1) [35]. All analyses were performed separately for women and men taking into consideration the well-established sex-related differences in body fat percentage [8, 14]. Cutoffs values of RFM for all-cause mortality were compared with those obtained using DXA-measured body fat percentage.
RFM ability to discriminate obesity and diabetes
Analysis of the receiver-operating characteristic (ROC) area under the curve was used to determine the discriminative accuracy of RFM for obesity among women and men. Accuracy was expressed as the C-statistic. Sensitivity, specificity, the positive predictive value and negative predictive value were calculated to assess the screening and diagnostic utility of RFM for obesity. Obesity is a major risk factor for diabetes [36]. Thus, we were also interested in examining the utility of RFM to discriminate diabetes mortality.
Hazard ratio for all-cause mortality
We used Cox proportional hazards regression to estimate hazard ratios and 95% confidence intervals (CI) for an association between baseline RFM and all-cause mortality. Taking into account the relative even distribution in terms of number of deaths, RFM was categorized as follows: <35.0, 35.0–39.9, 40.0–44.9, and ≥45.0 for women and <25.0, 25.0–29.9, 30.0–34.9, and ≥35.0 for men. RFM reference categories for women (<35.0) and men (<25.0) were chosen based on their association with the lowest mortality. In the multivariate analysis, hazard ratios were adjusted for age, BMI category (underweight: <18.5; normal weight: 18.5–24.9; overweight: 25.0–29.9; obese: ≥30), ethnicity (Mexican-Americans, European-Americans, African-Americans and other ethnicity), education level (less than 9th grade, 9–11th grade, and 12 or more grade), and smoking status: never (less than 100 cigarettes in life), former (if smoked at least 100 cigarettes in life but currently does not smoke), and current smoker (if smoked at least 100 cigarettes in life and currently smokes).
Statistical analysis
Clusters, strata and probability weights were used for all analyses to account for the NHANES complex design [17]. Variance estimates for descriptive variables were obtained using Taylor series linearization. We used the well accepted method of bootstrapping with 1000 replicates to obtain CI for the C-statistic to account for the complexity of the survey design [37]. C-statistic involving DXA data was calculated using imputation 1. Optimal cutoffs derived from DXA-measured body fat percentage to predict all-cause mortality were calculated for each imputation and then averaged. All analyses were performed using Stata 14 for Windows (StataCorp LP, College Station, TX). P < 0.05 was considered as statistically significant.
Results
NHANES 1999–2014 sample selection
NHANES 1999–2014 surveyed 82,091 participants (Fig. 1). We excluded individuals younger than 20 years (n = 38,298). We also excluded participants with missing information on body weight (n = 2,860), height (n = 2,828), waist circumference (n = 4,483), education level (n = 102), smoking status (n = 61) or mortality (n = 73), for a total of 4,797 observations. Participants who were pregnant at baseline (n = 1,263) and those who had heart disease, stroke, cancer or renal failure at baseline (n = 6,725) were also excluded. Final dataset for analysis comprised 31,008 adults. During a median follow-up of 8.3 years (IQR, 7.6–13.7) for a total of 253,433 person-years, 2,517 deaths occurred (1,396 deaths among women). Baseline characteristics of the participants are shown in Table 1.
NHANES III sample selection
NHANES III surveyed 33,994 participants (Fig. 1). We excluded individuals younger than 20 years (n = 15,169). We also excluded participants with missing information on body weight (n = 1,849), height (n = 1,834), waist circumference (n = 3,094), smoking status (n = 18), education level (n = 210), or mortality (n = 20), for a total of 3,213 observations. Participants who were pregnant at baseline (n = 274) and those who had heart disease, stroke, cancer, or renal failure at baseline (n = 2,688) were also excluded. Final dataset for analysis comprised 12,650 adult individuals. During a median follow-up of 23.3 years (IQR, 21.7–25.2) for a total of 260,095 person-years, 4,000 deaths occurred (2,088 deaths among women). Baseline characteristics of the participants are shown in Supplementary Table 1.
RFM cutoffs for mortality
In the NHANES 1999–2014 (8.3-year follow-up), using the maximum Youden’s index, optimal baseline RFM cutoffs for all-cause mortality among women and men were 40.8% and 30.9%, respectively. Based on the minimum distance to the coordinates (0,1), optimal baseline RFM cutoffs for all-cause mortality among women and men were 41.6% and 28.9%, respectively. Based on the highest Youden’s index for mortality attributed to different causes, RFM cutoffs ranged from 38.0 to 41.3% among women and from 28.4 to 32.5% among men (Supplementary Table 2). Based on the minimum distance to coordinate (0,1) for mortality attributed to different causes, RFM cutoffs ranged from 41.1 to 42.3% among women and from 28.6 to 30.9% among men (Supplementary Table 2).
Sensitivity analysis excluding participants with a follow-up <1 year produced identical RFM thresholds for all-cause mortality among men and women when using the maximum Youden’s index or the minimum distance to the coordinates (0,1). Exclusion of participants with a follow-up <2 years resulted in a lower Youden-based threshold for all-cause mortality among women only (39.8%) but thresholds based on the minimum distance to the coordinates (0,1) were identical.
Given the relatively variable thresholds to predict all-cause mortality using the Youden index and the Euclidean minimum distance to the coordinates (0,1), we suggest rounded even integer cutoffs to diagnose obesity that could be practical in the clinic: a cutoff of 40% for women and 30% for men. Among the adult population studied (NHANES 1999–2014), excluding those with heart disease, stroke, cancer, renal failure, and pregnant individuals at baseline, the age-adjusted prevalence of obesity by sex was as follows: 55.8% of women had an RFM ≥40% and 38.4% of men had an RFM ≥30%. Conversely, 34.4% of women and 31.0% of men had a BMI ≥30. In our validation population NHANES III (23.3-year follow-up), 43.6% of women had an RFM ≥40% and 26.6% of men had an RFM ≥30% at baseline, whereas 24.9% of women and 19.3% of men had a BMI ≥30. That is, from the period 1988–1994 to 1999–2014 there was a 28.0% and 43.9% increase in the prevalence of RFM-defined obesity among women and men, respectively, and a 37.9% and 60.3% increase in the prevalence of BMI-defined obesity among women and men, respectively.
Optimal RFM cutoffs to predict all-cause mortality using DXA-measured body fat percentage were 39.5% for women (n = 6,633) and 30.3% for men (n = 6,674) using Youden’s index, and 40.4% for women and 29.0% for men using the minimum distance to the coordinates (0,1). Thus, the RFM cutoffs derived from NHANES 1999–2014 were consistent with those obtained using DXA-measured body fat percentage derived from NHANES 1999–2006. Characteristics of the participants of the NHANES 1999–2006 are shown in Supplementary Table 3.
Usefulness of RFM as a screening/diagnostic test for obesity
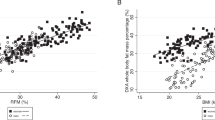
Using the optimal Youden-based cutoffs of DXA-measured body fat percentage to predict all-cause mortality as the reference to diagnose obesity, an RFM cutoff of 40% for women had a sensitivity of 81.3% (95% CI, 79.5–83.1%) and a specificity of 83.7% (95% CI, 82.0–85.3%) to discriminate DXA-defined obesity. An RFM cutoff of 30% for men had a sensitivity of 77.1% (95% CI, 74.8–79.4%) and a specificity of 87.1% (95% CI, 85.8–88.5%) to discriminate DXA-defined obesity. The positive predictive value and negative predictive value for RFM were 85.3% (95% CI, 83.9–86.7%) and 79.4% (95% CI, 77.4–81.3%) among women, respectively, and 74.2% (95% CI, 72.1–76.3%) and 88.8% (95% CI, 87.8–89.9%) among men, respectively. ROC analysis showed RFM had a high accuracy to discriminate adult individuals with DXA-defined obesity among women (C-statistic: 0.91 (95%CI, 0.91–0.92)) and men (C-statistic: 0.91 (95%CI, 0.91–0.92)) (Fig. 2).
Hazard ratio for all-cause mortality
In the univariate analysis, compared with women who had an RFM of <35.0%, women with an RFM of 35.0–39.9% had a hazard ratio for all-cause mortality of 1.68 (95% CI, 1.27–2.22; P < 0.001), 2.47 (95% CI, 1.95–3.14; P < 0.001) for women with an RFM of 40.0–44.9%, and 2.89 (95% CI, 2.26–3.70; P < 0.001) for women with an RFM of 45.0% or higher (Fig. 3a). Compared with men who had an RFM of <25.0%, men with an RFM of 25.0–29.9% had a hazard ratio for all-cause mortality of 1.24 (95% CI, 1.03–1.49; P = 0.024), 1.90 (95% CI, 1.52–2.37; P < 0.001) for men with an RFM of 30.0–34.9%, and 2.84 (95% CI, 2.20–3.66; P < 0.001) for men with an RFM of 35.0% or higher (Fig. 3c).
Estimated adjusted hazard ratios for death according to RFM are shown for women (a) and men (c) who participated in NHANES 1999–2014. In the validation dataset (NHANES III), hazard ratios for death are shown for women and men in b and d, respectively. Vertical lines represent 95% confidence intervals. Estimates were adjusted for age, BMI category, ethnicity, education level, and smoking status. RFM was calculated as follows: 64 − (20 × height/waist circumference) + (12 × sex); sex equals 0 for men and 1 for women. Height and waist circumference are measured in the same units (inches, meters, or centimeters).
In the multivariate analysis, adjusting for age, BMI category, ethnicity, education level, and smoking status, the association between RFM and all-cause mortality was significant at higher RFM ranges. The hazard ratio for all-cause mortality was 1.19 (95% CI, 0.89–1.60; P = 0.23) among women who had an RFM of 35.0–39.9%, 1.41 (95% CI, 1.02–1.95; P = 0.035) among women who had an RFM of 40.0–44.9%, and 1.72 (95% CI, 1.16–2.55; P = 0.007) among women who had an RFM of 45.0% or higher (Fig. 3a). Among men, the hazard ratio was 1.09 (95% CI, 0.85–1.41; P = 0.482) among men with an RFM of 25.0– 29.9%, 1.57 (95% CI, 1.07–2.30; P = 0.020) among those with an RFM of 30.0–34.9%, and 2.78 (95% CI, 1.80–4.30; P < 0.001) among those with an RFM of 35% or higher (Fig. 3c).
In the validation population, multivariate analysis showed that compared with women who had an RFM of <35.0%, women with an RFM of 35.0–39.9% had a hazard ratio for all-cause mortality of 1.32 (95% CI, 1.03–1.70; P = 0.030), 1.42 (95% CI, 1.01–2.00; P = 0.043) for women with an RFM of 40.0–44.9%, and 2.02 (95% CI, 1.31–3.10; P = 0.002) for women with an RFM of 45.0% or higher (Fig. 3b). Compared with men who had an RFM of <25.0%, men with an RFM of 25.0–29.9% had a hazard ratio of 1.18 (95% CI, 0.94–1.47; P = 0.142), 1.50 (95% CI, 1.07–2.10; P = 0.021) for men with an RFM of 30.0–34.9%, and 2.52 (95% CI, 1.61–3.95; P < 0.001) for men with an RFM of 35.0% or higher (Fig. 3d).
Usefulness of RFM to discriminate diabetes mortality
In a subpopulation excluding individuals with baseline diabetes (self-reported or diagnosed by fasting glucose ≥126 mg/dL and/or glycated hemoglobin A1C ≥6.5% at examination), RFM had better discrimination than the BMI for diabetes mortality (where the underlying cause was diabetes or diabetes was a contributor among multiple causes of death) among women (n = 14,810; C-statistic: 0.71 (95% CI, 0.64–0.78) vs. 0.62 (95% CI, 0.54–0.70); P < 0.001) and men (n = 14,336; C-statistic: 0.75 (95% CI, 0.67–0.82) vs. 0.63 (95% CI, 0.54–0.72); P < 0.001). In the validation population, RFM also had better discrimination than the BMI for diabetes mortality among women (n = 5916; C-statistic: 0.77 (95% CI, 0.68–0.85) vs. 0.71 (95% CI, 0.60–0.82); P = 0.024) and men (n = 5421; C-statistic: 0.80 (95% CI, 0.73–0.88) vs. 0.74 (95% CI, 0.65–0.83); P = 0.001).
Discussion
Numerous studies [1,2,3,4,5,6,7], with few exceptions [38, 39], have shown an association between whole-body fat percentage and mortality, hence the importance of an accurate assessment of body adiposity. In the present study, we identified RFM cutoffs to diagnose obesity based on the association between RFM and all-cause mortality. We suggest the following rounded thresholds of baseline RFM to diagnose obesity and predict risk of death: 40% for women and 30% for men. Adjusting for age, BMI category, ethnicity, education level, and smoking status, our cox proportional regression multivariate analyses suggest that women with an RFM of 40% (that is, 40% body fat) or higher and men with an RFM of 30% (30% body fat) or higher, respectively, have ~50% higher risk of death compared with women with an RFM of <35% and men with an RFM of <25%. More striking, women with an RFM of 45% or higher have near double the risk of death, whereas men with an RFM of 35% or higher have more than 2.5 times higher risk (Fig. 3).
A major strength of our study is that the suggested RFM cutoffs were derived using measured data from a representative sample of the United States adult population (more than 31,000 female and male individuals). Our findings were validated in another large representative sample of the United States adult population (more than 12,500 female and male individuals). Previous studies have proposed healthy references for body fat percentage or cutoffs to diagnose obesity [14, 15, 40, 41]. However, those studies were conducted in smaller and non-representative cohorts. Thus, the generalizability of previous published cutoffs to the United States population or to an ethnically mixed population have remained unclear. In addition, the suggested reference values were generated using corresponding BMI thresholds or were based on its association with cardiovascular risk factors but not with mortality. Another major strength of the present study is that we also evaluated and validated the association of RFM categories with all-cause mortality while adjusting for important potential confounders, including BMI (from measured body weight and height) and smoking status.
The RFM cutoffs to diagnose obesity obtained using NHANES 1999–2014 and those derived from DXA-measured body fat percentage in NHANES 1999–2006 are in contrast to the body fat percentage cutoffs of ~35% among women and ~25% among men generally used by other investigators to define obesity [12, 13, 42, 43]. Previous studies citing those recommended cutoffs have provided little or no information on how the cutoffs were generated.
Cutoffs of body fat percentage to define obesity have been proposed for specific ethnic groups. In a study conducted in a cohort of 1,217 Vietnamese adults enlisted from an urban setting the corresponding cutoffs of body fat percentage of a BMI of 30 were 40% for women and 30% for men [15], as in the present study. However, in contrast to the previous study, our RFM cutoffs were derived based on the association between RFM and all-cause mortality in an ethnically mixed population. In a study of healthy adult Koreans, body fat percentage cutoffs of 37% for women and 21% for men to define obesity were proposed based on its association with obesity-related cardiovascular disease risk factors [40]. The cutoffs from the latter study substantially differ from our suggested RFM cutoffs. Since NHANES 1999–2014 did not oversample Asian individuals, we were not able to estimate RFM cutoffs in Asian-American individuals to have a more comparable population with the two previous studies.
In a report from the WHO, the corresponding estimated cutoffs of body fat percentage in European individuals, using BMI thresholds, were 41% in women and 28% in men for obesity, and 35% in women and 22% in men for overweight [44]. However, the characteristics of the study population were not provided. Higher cutoffs among women have also been reported among Australians of European ethnicity [41]. The corresponding cutoffs of body fat percentage to define obesity using BMI thresholds were in the range of 43.4–44.6% for women and 27.5–31.1% for men [41]. Our sensitivity analysis in European-American individuals identified cutoffs of 41.0% among women and 30.9% among men. Besides ethnicity, other possible explanations for the discrepancies between studies include the size of the populations studied, their representativeness, the age distribution of the population, and whether individuals with major chronic conditions at baseline were excluded or not.
Our findings indicate higher risk of death with higher body fat percentage as determined by adjusted hazard ratios and all-cause mortality, which are consistent with those from previous studies [1,2,3,4,5,6,7]. However, the suggested RFM cutoffs to define obesity should be used in the context of the presence of other risk factors (e.g., hypertension, hyperlipidemia, diabetes, smoking, coronary artery disease) to assess the overall risk of mortality. Although RFM was independently associated with mortality in NHANES 1999–2014 and NHANES III, our findings will require further validation to determine the impact of therapeutic intervention, including lifestyle changes, on morbidity and mortality.
It should be noted that ROC analysis will yield identical C-statistics for RFM and waist-to-height ratio as both indices use reciprocal ratios. Previous studies have shown an association between waist-to-height ratio and incident and prevalent diabetes [45]. Whether waist-to-height ratio is better than BMI in discriminating incident or prevalent diabetes is still a matter of debate [45, 46]. However, two recent meta-analyses [47, 48] involving together more than 800,000 adults of variable ethnicity suggest that waist-to-height ratio is superior to BMI in predicting diabetes. In contrast, the discriminative accuracy of waist-to-height ratio for diabetes mortality has remained unknown. In the present study, RFM had better discrimination than the BMI for diabetes mortality among women (C-statistic: 0.71 vs. 0.62) and men (C-statistic: 0.75 vs. 0.63), which was validated in the participants of the NHANES III: 0.77 vs. 0.71 among women and 0.80 vs. 0.74 among men. Our findings suggest a clinical utility of RFM to predict risk of death related to diabetes.
The present study has some important limitations. First, we used a single-point (baseline) measurement of anthropometric variables to calculate RFM. Second, the relatively small number of deaths in the populations studied did not allow us to obtain reliable estimates to compare cutoffs among ethnic groups or to examine the risk of mortality in individuals with a very low RFM. Third, we used DXA as the reference method to evaluate the discriminative accuracy of RFM to diagnose obesity based on actual body adiposity. However, although the four-compartment method is the goal standard to assess body adiposity [49], its use is less common than DXA and data availability is limited to much smaller populations. Fourth, RFM cutoffs to define obesity are sex-specific. Conversely, one of the advantages of BMI is that the determination of the overweight or obesity categories is the same for both men and women. However, we and others have shown that a single BMI cutoff of 30 for women and men results in a high misclassification of women who have high body fat percentage [8, 50]. RFM does account for sex-related differences in body adiposity, which has been shown to improve obesity misclassification [8]. Finally, our analyses did not control for physical activity or diet. Although information on these variables is available for NHANES, it is self-reported. The reliability of data on self-reported physical activity and diet is questionable [51]. Diet recall has a strong bias towards underestimation [52] and in many cases data are not physiologically plausible [53]. Conversely, physical activity is over-reported [54].
In conclusion, we suggest rounded RFM cutoffs of 40% for women and 30% for men to diagnose obesity and identify individuals at higher risk of death in those with a median follow-up of up to 23 years.
References
Heitmann BL, Erikson H, Ellsinger BM, Mikkelsen KL, Larsson B. Mortality associated with body fat, fat-free mass and body mass index among 60-year-old swedish men-a 22-year follow-up. The study of men born in 1913. Int J Obes Relat Metab Disord. 2000;24:33–37.
Ortega FB, Sui X, Lavie CJ, Blair SN. Body mass index, the most widely used but also widely criticized index: would a criterion standard measure of total body fat be a better predictor of cardiovascular disease mortality? Mayo Clin Proc. 2016;91:443–455.
Padwal R, Leslie WD, Lix LM, Majumdar SR. Relationship among body fat percentage, body mass index, and all-cause mortality: a cohort study. Ann Intern Med. 2016;164:532–541.
Zong G, Zhang Z, Yang Q, Wu H, Hu FB, Sun Q. Total and regional adiposity measured by dual-energy X-ray absorptiometry and mortality in NHANES 1999-2006. Obesity (Silver Spring). 2016;24:2414–21.
Dong B, Peng Y, Wang Z, Adegbija O, Hu J, Ma J, et al. Joint association between body fat and its distribution with all-cause mortality: a data linkage cohort study based on NHANES (1988–2011). PLoS ONE. 2018;13:e0193368.
Lahmann PH, Lissner L, Gullberg B, Berglund G. A prospective study of adiposity and all-cause mortality: the Malmo Diet and Cancer Study. Obes Res. 2002;10:361–9.
Jenkins DA, Bowden J, Robinson HA, Sattar N, Loos RJF, Rutter MK, et al. Adiposity-mortality relationships in type 2 diabetes, coronary heart disease, and cancer subgroups in the UK Biobank, and their modification by smoking. Diabetes Care. 2018;41:1878–86.
Woolcott OO, Bergman RN. Relative fat mass (RFM) as a new estimator of whole-body fat percentage—a cross-sectional study in American adult individuals. Sci Rep. 2018;8:10980.
Sui X, LaMonte MJ, Laditka JN, Hardin JW, Chase N, Hooker SP, et al. Cardiorespiratory fitness and adiposity as mortality predictors in older adults. J Am Med Assoc. 2007;298:2507–16.
Lee CD, Blair SN, Jackson AS. Cardiorespiratory fitness, body composition, and all-cause and cardiovascular disease mortality in men. Am J Clin Nutr. 1999;69:373–80.
Bray GA. Fat distribution and body weight. Obes Res. 1993;1:203–5.
Lobman TG, Houtkooper L, Going SB. Body fat measurement goes high-tech: not all are created equal. ACSM's Health Fit J. 1997;1:30–35.
Deurenberg P, Yap M, van Staveren WA. Body mass index and percent body fat: a meta analysis among different ethnic groups. Int J Obes Relat Metab Disord. 1998;22:1164–71.
Gallagher D, Heymsfield SB, Heo M, Jebb SA, Murgatroyd PR, Sakamoto Y. Healthy percentage body fat ranges: an approach for developing guidelines based on body mass index. Am J Clin Nutr. 2000;72:694–701.
Ho-Pham LT, Lai TQ, Nguyen MT, Nguyen TV. Relationship between body mass index and percent body fat in Vietnamese: implications for the diagnosis of obesity. PLoS ONE. 2015;10:e0127198.
Bahat G, Kilic C, Topcu Y, Aydin K, Karan MA. Fat percentage cutoff values to define obesity and prevalence of sarcopenic obesity in community-dwelling older adults in Turkey. Aging Male 2018. https://doi.org/10.1080/13685538.2018.1530208. [e-pub ahead of print].
Johnson CL, Paulose-Ram R, Ogden CL, Carroll MD, Kruszon-Moran D, Dohrmann SM, et al. National health and nutrition examination survey: analytic guidelines, 1999–2010. Vital Health Stat 2. 2013:1–24.
Mohadjer L, Montaquila J, Waksberg J et al. National Health and Nutrition Examination Survey III: Weighting and estimation methodology: executive summary. Rockville, MD, 1996.
National Center for Health Statistics, Office of Analysis and Epidemiology. Public-use linked mortality file, 2015. Hyattsville, Maryland: NCHS. 2015. https://www.cdcgov/nchs/data-linkage/mortality-publichtm. Accessed 07 Sept 2018.
Berrington de Gonzalez A, Hartge P, Cerhan JR, Flint AJ, Hannan L, MacInnis RJ, et al. Body-mass index and mortality among 1.46 million white adults. N Engl J Med. 2010;363:2211–9.
Singh PN, Wang X. Simulation study of the effect of the early mortality exclusion on confounding of the exposure-mortality relation by preexisting disease. Am J Epidemiol. 2001;154:963–71.
Allison DB, Heo M, Flanders DW, Faith MS, Williamson DF. Examination of "early mortality exclusion" as an approach to control for confounding by occult disease in epidemiologic studies of mortality risk factors. Am J Epidemiol. 1997;146:672–80.
Webster AC, Nagler EV, Morton RL, Masson P. Chronic kidney disease. Lancet. 2017;389:1238–52.
Levey AS, Coresh J, Greene T, Stevens LA, Zhang YL, Hendriksen S, et al. Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann Intern Med. 2006;145:247–54.
National Health and Nutrition Examination Survey 1999–2000 data documentation, codebook, and frequencies standard biochemistry profile & hormones (LAB18). https://wwwn.cdc.gov/Nchs/Nhanes/1999-2000/LAB18.htm. Accessed 18 February 2019.
National Health and Nutrition Examination Survey 2005–2006 data documentation, codebook, and frequencies standard biochemistry profile (BIOPRO_D). https://wwwn.cdc.gov/Nchs/Nhanes/2005-2006/BIOPRO_D.htm. Accessed 18 February 2019.
Ingram DD, Lochner KA, Cox CS. Mortality experience of the 1986–2000 National Health Interview Survey Linked Mortality Files participants. Vital Health Stat 2. 2008:1–37.
Jacobs E, Hoyer A, Brinks R, Kuss O, Rathmann W. Burden of mortality attributable to diagnosed diabetes: a nationwide analysis based on claims data from 65 million people in Germany. Diabetes Care. 2017;40:1703–9.
National Health and Nutrition Examination Survey (NHANES). Anthropometry procedures manual. 2007. https://www.cdc.gov/nchs/data/nhanes/nhanes_07_08/manual_an.pdf. Accessed 16 May 2016.
National Health and Nutrition Examination Survey III: Body measurements (anthropometry). Rockville, MD, 1988.
Kelly TL, Wilson KE, Heymsfield SB. Dual energy X-Ray absorptiometry body composition reference values from NHANES. PLoS ONE. 2009;4:e7038.
National Health and Nutrition Examination Survey: Technical documentation for the 1999–2004. Dual Energy X-Ray Absorptiometry (DXA) multiple imputation data files; 2008. https://wwwn.cdc.gov/nchs/data/nhanes/dxa/dxa_techdoc.pdf. Accessed 17 May 2016.
Perkins NJ, Schisterman EF. The inconsistency of "optimal" cutpoints obtained using two criteria based on the receiver operating characteristic curve. Am J Epidemiol. 2006;163:670–675.
Youden WJ. Index for rating diagnostic tests. Cancer. 1950;3:32–35.
Wolfowitz J. The minimum distance method. Ann Math Stat. 1957;28:75–88.
Bray GA, Heisel WE, Afshin A, Jensen MD, Dietz WH, Long M, et al. The science of obesity management: an endocrine society scientific statement. Endocr Rev. 2018;39:79–132.
Haukoos JS, Lewis RJ. Advanced statistics: bootstrapping confidence intervals for statistics with "difficult" distributions. Acad Emerg Med. 2005;12:360–5.
Simpson JA, MacInnis RJ, Peeters A, Hopper JL, Giles GG, English DR. A comparison of adiposity measures as predictors of all-cause mortality: the Melbourne Collaborative Cohort Study. Obesity (Silver Spring). 2007;15:994–1003.
Myint PK, Kwok CS, Luben RN, Wareham NJ, Khaw KT. Body fat percentage, body mass index and waist-to-hip ratio as predictors of mortality and cardiovascular disease. Heart. 2014;100:1613–9.
Kim CH, Park HS, Park M, Kim H, Kim C. Optimal cutoffs of percentage body fat for predicting obesity-related cardiovascular disease risk factors in Korean adults. Am J Clin Nutr. 2011;94:34–39.
Pasco JA, Holloway KL, Dobbins AG, Kotowicz MA, Williams LJ, Brennan SL. Body mass index and measures of body fat for defining obesity and underweight: a cross-sectional, population-based study. BMC Obes. 2014;1:9.
Oreopoulos A, Lavie CJ, Snitker S, Romero-Corral A. More on body fat cutoff points–Reply–I. Mayo Clin Proc. 2011;86:584–5.
AACE/ACE Obesity Task Force. AACE/ACE position statement on the prevention, diagnosis, and treatment of obesity. Endocr Pract. 1998;4:297–350.
WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363:157–63.
Qiao Q, Nyamdorj R. Is the association of type II diabetes with waist circumference or waist-to-hip ratio stronger than that with body mass index? Eur J Clin Nutr. 2010;64:30–34.
Lee CMY, Woodward M, Pandeya N, Adams R, Barrett-Connor E, Boyko EJ, et al. Comparison of relationships between four common anthropometric measures and incident diabetes. Diabetes Res Clin Pract. 2017;132:36–44.
Ashwell M, Gunn P, Gibson S. Waist-to-height ratio is a better screening tool than waist circumference and BMI for adult cardiometabolic risk factors: systematic review and meta-analysis. Obes Rev. 2012;13:275–86.
Savva SC, Lamnisos D, Kafatos AG. Predicting cardiometabolic risk: waist-to-height ratio or BMI. A meta-analysis. Diabetes Metab Syndr Obes. 2013;6:403–19.
Van Der Ploeg GE, Withers RT, Laforgia J. Percent body fat via DEXA: comparison with a four-compartment model. J Appl Physiol (1985). 2003;94:499–506.
Romero-Corral A, Somers VK, Sierra-Johnson J, Thomas RJ, Collazo-Clavell ML, Korinek J, et al. Accuracy of body mass index in diagnosing obesity in the adult general population. Int J Obes (Lond). 2008;32:959–66.
Dhurandhar NV, Schoeller D, Brown AW, Heymsfield SB, Thomas D, Sorensen TI, et al. Energy balance measurement: when something is not better than nothing. Int J Obes (Lond). 2015;39:1109–13.
Black AE, Goldberg GR, Jebb SA, Livingstone MB, Cole TJ, Prentice AM. Critical evaluation of energy intake data using fundamental principles of energy physiology: 2. Evaluating the results of published surveys. Eur J Clin Nutr. 1991;45:583–99.
Archer E, Hand GA, Blair SN. Validity of U.S. nutritional surveillance: National Health and Nutrition Examination Survey caloric energy intake data, 1971–2010. PLoS ONE. 2013;8:e76632.
Walsh MC, Hunter GR, Sirikul B, Gower BA. Comparison of self-reported with objectively assessed energy expenditure in black and white women before and after weight loss. Am J Clin Nutr. 2004;79:1013–9.
Acknowledgements
We thank the Centers for Disease Control and Prevention (CDC) and the National Center for Health Statistics (NCHS) for providing access to the NHANES datasets. This study was self-funded. The author’s responsibilities were as follows: OOW designed the research, conducted the research, performed the statistical analysis, and wrote the paper. RNB contributed with the design of the study and revised the final draft. OOW takes full responsibility for the work as a whole.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Woolcott, O.O., Bergman, R.N. Defining cutoffs to diagnose obesity using the relative fat mass (RFM): Association with mortality in NHANES 1999–2014. Int J Obes 44, 1301–1310 (2020). https://doi.org/10.1038/s41366-019-0516-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-019-0516-8
- Springer Nature Limited
This article is cited by
-
Relative fat mass as an estimator of body fat percentage in Chilean adults
European Journal of Clinical Nutrition (2024)
-
Relative fat mass and prediction of incident atrial fibrillation, heart failure and coronary artery disease in the general population
International Journal of Obesity (2023)
-
Body mass index, waist circumference, waist-to-hip ratio, and body fat in relation to health care use in the Canadian Longitudinal Study on Aging
International Journal of Obesity (2021)