Abstract
Activation of the phosphoinositide 3-kinase (PI3K) pathway occurs widely in human cancers. Although somatic mutations in the PI3K pathway genes PIK3CA and PTEN are known to drive PI3K pathway activation and cancer growth, the significance of somatic mutations in other PI3K pathway genes is less clear. Here, we establish the signaling and oncogenic properties of a recurrent somatic mutation in the PI3K p110β isoform that resides within its kinase domain (PIK3CβD1067V). We initially observed PIK3CβD1067V by exome sequencing analysis of an EGFR-mutant non-small cell lung cancer (NSCLC) tumor biopsy from a patient with acquired erlotinib resistance. On the basis of this finding, we hypothesized that PIK3CβD1067V might function as a novel tumor-promoting genetic alteration, and potentially an oncogene, in certain cancers. Consistent with this hypothesis, analysis of additional tumor exome data sets revealed the presence of PIK3CβD1067V at low frequency in other patient tumor samples (including renal cell carcinoma, glioblastoma multiforme, head and neck squamous cell carcinoma, melanoma, thyroid carcinoma and endometrial carcinoma). Functional studies revealed that PIK3CβD1067V promoted PI3K pathway signaling, enhanced cell growth in vitro, and was sufficient for tumor formation in vivo. Pharmacologic inhibition of PIK3Cβ with TGX-221 (isoform-selective p110β inhibitor) specifically suppressed growth in patient-derived renal-cell carcinoma cells with endogenous PIK3CβD1067V and in NIH-3T3 and human EGFR-mutant lung adenocarcinoma cells engineered to express this mutant PI3K. In the EGFR-mutant lung adenocarcinoma cells, expression of PIK3CβD1067V also promoted erlotinib resistance. Our data establish a novel oncogenic form of PI3K, revealing the signaling and oncogenic properties of PIK3CβD1067V and its potential therapeutic relevance in cancer. Our findings provide new insight into the genetic mechanisms underlying PI3K pathway activation in human tumors and indicate that PIK3CβD1067V is a rational therapeutic target in certain cancers.
Similar content being viewed by others
Introduction
The phosphoinositide 3-kinase (PI3K) signaling pathway controls many processes that regulate cancer cell survival and growth.1, 2 Activation of the PI3K pathway signaling occurs widely in human cancers.3, 4 Genetic studies have revealed somatic mutations in key components of the PI3K pathway that promote its activation and, consequently, tumor initiation and progression.5, 6, 7 The most frequent genetic alterations present in human tumors are activating mutations in PIK3CA (encoding p110α) and inactivating mutations in the tumor suppressor PTEN (encoding phosphatase and tensin homolog).8, 9, 10, 11 Beyond somatic mutations in PIK3CA and PTEN, the significance of somatic mutations in other PI3K pathway genes (including the p110β, p110γ or p110δ PI3K catalytic subunits) in tumor cell growth and survival is less well understood. Hence, the full spectrum of potential biological and tumor-promoting properties of oncogenic variants in PI3K components is incompletely characterized.
Here, we establish the signaling and oncogenic properties of a previously uncharacterized, recurrent somatic mutation in the PI3K p110β isoform that resides within its kinase domain (PIK3CβD1067V). We unexpectedly uncovered PIK3CβD1067V by exome sequencing analysis of an EGFR-mutant non-small cell lung cancer (NSCLC) tumor biopsy from a patient with acquired erlotinib resistance. On the basis of this finding, we hypothesized that PIK3CβD1067V might function as a novel tumor-promoting genetic alteration, and potentially an oncogene, in certain cancers. Consistent with this hypothesis, the analysis of additional tumor exome data sets revealed the presence of PIK3CβD1067V in multiple other human tumors (at low frequency). We demonstrate that PIK3CβD1067V promotes PI3K pathway signaling and tumor formation in vivo. We further found that selective inhibition of PIK3CβD1067V suppressed the growth of patient-derived renal and lung cancer cells, as well as of NIH-3T3 cells expressing this mutant PI3K. Expression of PIK3CβD1067V in human EGFR-mutant NSCLC cells promoted resistance to erlotinib. Taken together, our data establish a new oncogenic form of PI3K. The data provide the first evidence, to our knowledge, that oncogenic mutation of p110β PI3K can drive not only PI3K signaling but also tumor formation in vivo and can modulate therapeutic efficacy.
Results and Discussion
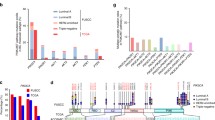
We unexpectedly uncovered PIK3CβD1067V by whole-exome sequencing of a lung adenocarcinoma biopsy specimen obtained from a patient with acquired resistance to first-line treatment with the EGFR kinase inhibitor erlotinib. The patient was a 34-year-old male non-smoker diagnosed with stage IV NSCLC that was found by DNA sequencing (using a clinical assay) to harbor dual activating mutations in EGFR (EGFRG719A/L861Q).12 On the basis of the presence of mutant EGFR, the patient was treated with erlotinib and experienced an initial tumor response (Figure 1a). This initial response was followed by the development of acquired resistance after 11 months of therapy, as is typical for such patients (Figure 1a).13, 14 Unfortunately, the patient succumbed to this drug-resistant disease shortly following this disease progression. Whole-exome sequencing analysis of the patient’s erlotinib-resistant tumor biopsy revealed the presence of the EGFRT790M resistance mutation (Figures 1a and b), which occurs in at least 50% of patients with acquired erlotinib resistance.15 The sequencing analysis did not reveal other genetic events known to promote EGFR inhibitor resistance, such as MET kinase amplification15 (data not shown). However, the whole-exome sequencing analysis revealed a somatic mutation in PIK3Cβ encoding the p110β catalytic isoform of PI3K (PIK3CβD1067V; Figures 1a and b). The mutations in EGFR and the PIK3CβD1067V mutation were confirmed by an independent clinical sequencing assay using DNA from the patient’s resistant tumor biopsy (data not shown, Supplementary Figure S1). There was no evidence of the EGFRT790M or PIK3CβD1067V mutation in the pre-treatment tumor specimen (Figure 1a, data not shown), suggesting emergence during acquired erlotinib resistance. PIK3CβD1067V has not previously been reported in patient tumors with resistance to EGFR inhibitor therapy and is a variant of unknown functional significance. Prompted by the discovery of PIK3CβD1067V in this patient’s tumor biopsy, we hypothesized that PIK3CβD1067V might function as a novel tumor-promoting genetic alteration, and potentially an oncogene, in certain cancers.
D1067V is a recurrent somatic mutation in the kinase domain of PIK3Cβ in patient tumors. (a) CT scans showing erlotinib response and acquired resistance in the EGFR-mutant NSCLC patient. Left panel shows histologic analysis confirming NSCLC in the resistant tumor biopsy. Small cell cancer histologic transformation previously associated with acquired erlotinib resistance in some cases13, 14 was not present in the specimen. Arrows indicate NSCLC nodules. The therapy timeline and genotypes of the pre-treatment and erlotinib-resistant tumor biopsy are shown. (b) Variant frequencies of the somatic mutations in EGFR and PIK3Cβ are shown, expressed as VaF=mutant allele count/(normal allele count+mutant allele count). Sample preparation and exome-deep sequencing and analysis were conducted as previously described,26 with a median fold exome coverage of ~100 ×. The resistant tumor biopsy was exhausted in the molecular analysis and suitable pre-treatment tumor biopsy was not available for exome sequencing. (c) Pan-cancer analysis indicating the presence of PIK3CβD1067V in additional patient tumors (RCC, renal cell carcinoma; GBM, glioblastoma multiforme; HNSCC, head and neck squamous cell carcinoma). (d) Multiple sequence alignment analysis performed using NCI protein Blast (Blastp) showing that D1067 (marked in red) in the C-terminal kinase domain in PIK3Cβ is highly conserved across species.
To test this hypothesis, we first set out to determine whether PIK3CβD1067V is observed in other patient tumors. We analyzed publically available tumor exome data sets (using the cBIO portal http://www.cbioportal.org/ to access The Cancer Genome Atlas datasets16) and found that PIK3CβD1067V/A/Y is present at low frequency in several tumors, including renal cell carcinoma, glioblastoma multiforme, head and neck squamous cell carcinoma, melanoma, thyroid carcinoma and endometrial carcinoma (Figure 1c). This data mining across patient tumors also revealed that mutation of D1067 was the most recurrent missense mutation in the kinase domain of PIK3Cβ, accounting for ~15% (6/41) of the missense mutations in this domain (with the majority, 38, of the other missense mutations occurring only once). Interestingly, these specimens with PIK3CβD1067V/A/Y did not harbor other established oncogenic driver mutations with the exception of BRAFV600E (in two cases: melanoma, thyroid; data not shown), which can cooperate with PI3K pathway alterations to promote oncogenesis.17, 18 Overall, these data suggest that mutation of D1067 in PIK3Cβ might promote tumor growth in certain cancers.
The D1067 residue lies within the kinase domain present at the extreme C-terminus of PI3KCβ (Figure 1d), suggesting that mutation at this site may impact PIK3Cβ activity. In further support of the potential functional impact of mutation of D1067 in PIK3Cβ, we found by multiple sequence alignment analysis that this residue is highly conserved in PIK3Cβ across several species (Figure 1d). On the basis of these collective findings, we investigated the biological and functional properties of PIK3CβD1067V.
We first examined the signaling properties of PIK3CβD1067V in a genetically controlled system we established by engineering stable expression of either PIK3CβD1067V or wild-type PIK3Cβ (PIK3CβWT) in NIH-3T3 cells. These studies revealed that PIK3CβD1067V activated PI3K pathway downstream signaling, as measured by the levels of phosphorylated AKT and S6 kinase, to a greater degree than PIK3CβWT (Figure 2a). These data indicate that PIK3CβD1067V is an activating variant of PIK3Cβ. Expression of either WT or mutant PIK3Cβ did not increase the levels of phosphorylated ERK in these cells (Figure 2a), indicating that PI3KCβ specifically activates PI3K pathway signaling in this system. In addition, we found that expression of PIK3CβD1067V enhanced cell growth in vitro compared with cells expressing PIK3CβWT (or vector-transduced control cells) (Figure 2b). These data establish that expression of PIK3CβD1067V confers a growth advantage in cells in vitro. Together, the findings link hyperactivation of PI3K pathway signaling by PIK3CβD1067V to enhanced cell growth.
Cell signaling and oncogenic properties of PIK3CβD1067V. (a) Effects of the expression of PIK3CβD1067V or PIK3CβWT on the indicated signaling proteins as measured by immunoblot analysis. NIH-3T3 cells were stably transduced with retroviral vectors containing: empty (EV), expressing the wild-type (WT; Addgene #20573-Myr-removed) or mutant (Addgene #20573-mutated by recombinant PCR-Myr-removed) PIK3Cβ gene, as previously described.27 Results represent three independent experiments. (b) Effects of the expression of PIK3CβD1067V or PIK3CβWT on cell growth in the indicated cell lines. Results were derived from CellTiterGLO assays conducted according to the manufacturer’s protocol (n=3, ±s.e.m.). (c, d) Tumor allograft studies assessing the effects of expression of PIK3CβD1067V or PIK3CβWT on tumor growth in immunocompromised mice (n=6 mice/group, 12 tumors/condition±s.e.m.), with d showing representative tumor explants from the indicated cell lines (explants from cells with empty vector (EV) are not shown as minimal or no growth of tumors was observed). Immunoblot, cell viability and tumor explant studies were conducted using established methods.26
On the basis of these findings, we next assessed whether PIK3CβD1067V exhibited oncogenic properties in vivo using a tumor allograft assay in immunocompromised mice. Indeed, we observed that expression of PIK3CβD1067V was sufficient to promote tumor growth in vivo (Figures 2c and d). Notably, expression of PIK3CβWT also promoted tumor growth in vivo, albeit to a lesser extent than PIK3CβD1067V (Figures 2c and d), consistent with previous findings indicating that PIK3CβWT when overexpressed can be oncogenic.19 Together, these data establish the oncogenic properties of PIK3CβD1067V.
Given these observations, we next explored the function of endogenous PIK3CβD1067V in human cancer cells. By mining publically available data sets (Cancer Cell Line Encyclopedia), we identified one patient-derived renal-cell carcinoma cell line with endogenous PIK3CβD1067V (A498 cells, heterozygous PIK3CβD1067V). We investigated whether genetic or pharmacologic inhibition of PIK3CβD1067V would suppress the growth of these cells. We found that knockdown of PIK3Cβ, but not PIK3Cα, in A498 cells decreased growth in vitro (Figure 3a), suggesting a specific requirement for mutant PIK3Cβ in this system. Consistent with these findings, silencing PIK3Cβ, but not PI3KCα, suppressed the levels of phosphorylated AKT in these cells (Figure 3b). Furthermore, pharmacologic inhibition of PIK3Cβ using the p110β isoform selective inhibitor TGX-22120 similarly suppressed growth in A498 cells (Figure 3c). In contrast, treatment with the pan-PI3K inhibitor BKM-120 (which is more potent in vitro against p110α than p110β)21 had less impact on A498 cell growth (Figure 3c). Consistent with these findings, inhibition of PIK3Cβ by TGX-221 suppressed the levels of phosphorylated AKT and the downstream PI3K pathway components S6 and 4EBP1 to a greater degree than BKM-120 in these cells (Figure 3d). Treatment with either TGX-221 or BKM-120 did not substantially impact the levels of phosphorylated ERK in these cells (Figure 3d), providing further evidence linking PIK3Cβ specifically to PI3K pathway signaling. Together, these data suggest that cancer cells with endogenous PIK3CβD1067V might depend on this oncogene for growth and, therefore, be more sensitive to a p110β isoform selective inhibitor than cancer cells with PIK3CβWT.
Biological effects of oncogenic PIK3CβD1067V in patient-derived tumor models. (a) Effects of stable knockdown of PIK3Cβ SIGMA: TRCN0000010024, TRCN0000010025 or PI3KCα SIGMA: TRCN0000196951, TRCN0000196795 by shRNA in the indicated cells as measured by cell viability assays and shown as crystal violet staining, using established methods.26 Results represent three independent experiments. (b) Validation of the effects of each shRNA on target expression and signaling by immunoblot (antibodies obtained from Cell Signaling Technology and used according to the manufacturer’s protocol). Results represent three independent experiments. (c) Effects of treatment with TGX-221 (obtained from SelleckChem) over a dose response of 0.01–1 μM or with BKM-120 (obtained from SelleckChem) over a dose response of 0.01–1 μM in the indicated cells. Cell viability assays conducted as in a. Results represent three independent experiments. (d) Effects of treatment with TGX-221 or BKM-120 in the indicated cells on the indicated proteins as measured by immunoblot analysis. Results represent three independent experiments. (e, f) Effects of treatment with either (e) TGX-221 or (f) BKM-120 on the growth of the indicated cells. Cell viability assays were conducted as in Figure 2 (n=3±s.d.). Inset shows the effects of treatment with TGX-221 or BKM-120 in 786-O cells on the indicated proteins as measured by immunoblot analysis using established methods.26 Results represent three independent experiments. (g, h) Effects of treatment with either (g) TGX-221 or (h) BKM-120 on the growth of the indicated cells. Cell viability assays were conducted as in Figure 2 (n=3±s.d.). Insets show IC50s for the indicated treatments in the indicated cell lines, as previously described26 (IC50s>1000 nM are shown up to 1000 nM).
We examined this hypothesis by investigating TGX-221 or BKM-120 response in 786-O renal-cell carcinoma cells with PIK3CβWT, in addition to the A498 renal-cell carcinoma cells (with PIK3CβD1067V) studied previously. We found that A498 cells were more sensitive to TGX-221 than 786-O cells (Figure 3e). In addition, we observed little difference in sensitivity to the pan-PI3K inhibitor BKM-120 between A498 and 786-O cells (Figure 3f), suggesting a link between sensitivity to p110β isoform selective inhibition and the presence of oncogenic PIK3CβD1067V. We also noted that BKM-120 more effectively suppressed both AKT pathway signaling as measured p-AKT and p-S6 levels (inset in Figures 3e and f) and cell viability (Figures 3e and f) than TGX-221 in 786-O cells with PIK3CβWT. Patient-derived cells, including A498, harbor additional genetic alterations that might contribute to the effect of PI3K inhibitor treatment on cell viability that we observed (Supplementary Figure S2). Therefore, we next used a genetically controlled system to test whether specific expression of PIK3CβD1067V confers increased sensitivity to PI3K inhibitor treatment, and particularly to treatment with TGX-221. We studied NIH-3T3 cells transduced with oncogenic PIK3CβD1067V, PIK3CβWT, or an empty vector control and treated the cells with either the PI3K p110β selective inhibitor TGX-221 or the pan-PI3K inhibitor BKM-120. Expression of mutant PIK3CβD1067V caused increased sensitivity to TGX-221 when compared to expression of PIK3CβWT or the control cells (Figures 3g and h). BKM-120, which has activity against both p110β and p110α, was also more effective in NIH-3T3 cells expressing PIK3CβD1067V compared with those with PIK3CβWT or the control cells, although BKM-120 was generally less effective than TGX-221 likely because it is less potent against p110β than p110α (Figures 3g and h).21 Together, the data show that expression of PIK3CβD1067V is sufficient to confer oncogene dependence and increased sensitivity to pharmacologic p110β PI3K inhibition. As an additional control, we also tested the effects of each form of PIK3Cβ on chemotherapy sensitivity (doxorubicin) in this NIH-3T3 system. Interestingly, we found expression of either PIK3CβD1067V or PIK3CβWT decreased doxorubicin sensitivity in these cells (Supplementary Figure S3). These data suggest that the effects of PIK3CβD1067V on therapeutic efficacy are context-specific and highlight the specificity in enhancement in sensitivity to PI3K inhibitor treatment (in particular TGX-221) caused by oncogenic PIK3CβD1067V expression.
We further tested the impact of PIK3CβD1067V expression on PI3K inhibitor sensitivity in human NSCLC cells (H3255)22 in which we expressed either PIK3CβD1067V or PIK3CβWT and treated the cells with TGX-221 or BKM-120 (Supplementary Figure S3). Consistent with our findings in NIH-3T3 cells, expression of PIK3CβD1067V conferred increased sensitivity to TGX-221 (and to a minor extent to BKM-120), when compared with expression of PIK3CβWT or the control cells in this system (Supplementary Figure S3). Altogether, these observations establish the efficacy of p110β pharmacologic inhibition in cells with oncogenic PIK3Cβ. The findings indicate that PIK3CβD1067V is required for cell growth via activation of PI3K pathway signaling. The data suggest that specific pharmacologic inhibition of PIK3Cβ with a p110β-selective inhibitor may be effective against certain cancers with PIK3CβD1067V.
Given that we initially observed PIK3CβD1067V in a tumor biopsy from an EGFR-mutant NSCLC patient with acquired erlotinib resistance, we next investigated whether PIK3CβD1067V could promote erlotinib resistance. We used the H3255 NSCLC cells, which harbor mutant EGFRL858R, expressing either PIK3CβD1067V or PIK3CβWT to test the effects of each form of PIK3Cβ on erlotinib sensitivity, as well as signaling. First, we found that erlotinib treatment suppressed the levels of phosphorylated EGFR, AKT and S6 in H3255 control cells, as expected (Figure 4a).23 In contrast, expression of PIK3CβD1067V resulted in sustained phosphorylation of AKT and S6 during erlotinib treatment in these cells (Figure 4a). Furthermore, expression of PIK3CβD1067V was sufficient to promote erlotinib resistance in this system (Figures 4b and c). In contrast, expression of PIK3CβWT failed to significantly rescue p-AKT or p-S6 levels upon erlotinib treatment (Figure 4a). Accordingly, only a minor impact on erlotinib sensitivity was observed in H3255 cells expressing PIK3CβWT compared with those with PIK3CβD1067V (Figures 4b and c). Together, these data indicate that the D1067V mutation in PIK3Cβ is critical for resistance to erlotinib in this system. The data suggest that PIK3CβD1067V sustains p-AKT and p-S6 levels during erlotinib exposure to a degree necessary to significantly impact erlotinib efficacy in these cells.
Oncogenic PIK3CβD1067V promotes sustained PI3K pathway signaling and EGFR inhibitor resistance in human EGFR-mutant NSCLC cells. (a) Effects of stable expression of PIK3CβWT and PIK3CβD1067V on signaling in response to erlotinib in the indicated cells, compared with control cells harboring empty vector (EV). Results represent three independent experiments. (b, c) Effects of stable expression of PIK3CβWT and PIK3CβD1067V on cell viability during erlotinib treatment in the indicated cells, shown as (b) crystal violet staining assays or (c) CellTiterGLO cell viability assays with half maximal inhibitory concentration shown (n=3±s.d.), as previously described.26 *indicates P<0.05; ***indicates P<0.01, for differential erlotinib IC50 effects.
Overall, our findings indicate that PIK3CβD1067V differentially modulates therapeutic sensitivity to multiple agents, enhancing sensitivity to PI3K inhibitor treatment whereas diminishing sensitivity to erlotinib or chemotherapy (doxorubicin). With respect to the potential role of PIK3CβD1067V in erlotinib resistance in NSCLC, additional investigations are warranted in properly powered and prospectively acquired patient cohorts in future studies. Interestingly, the erlotinib-resistant patient tumor in which we uncovered PIK3CβD1067V also harbored EGFRT790M, suggesting that EGFR inhibitor resistance may have been multifactorial. PIK3CβD1067V was present at a higher variant frequency than EGFRT790M in the patient’s resistant tumor biopsy (Figure 1b), consistent with a model of multifactorial resistance. This notion is also consistent with other findings showing that EGFRT790M can be present concurrently with other resistance events in erlotinib-resistant patient tumor biopsies24 and with data indicating that ~40% of EGFRT790M positive NSCLC patients fail to respond to investigational EGFR kinase inhibitors with activity against EGFRT790M (http://www.mycancergenome.org/content/disease/lung-cancer/egfr/4/). Our data further highlight the potential relevance of occult co-drivers of resistance in some patients. On the basis of our findings, we propose that examination of PIK3Cβ genetic status as a potential contributor to either tumor progression or therapy resistance should be undertaken in future studies.
In summary, our findings establish a novel oncogenic form of PI3K and offer new insight into the genetic basis of PI3K pathway activation in human cancers. Our data reveal the signaling and oncogenic properties of PIK3CβD1067V, extending recent findings indicating that another somatic variant in PIK3Cβ (PIK3CβE633K) can promote PI3K pathway signaling and cellular transformation (in vitro).25 Together, our findings offer new insight into the oncogenic function of PIK3Cβ and establish for the first time, to our knowledge, that oncogenic mutation of p110β PI3K can drive tumor formation in vivo and modulate therapeutic sensitivity to multiple agents in vitro. Our data also provide rationale for testing p110β isoform-selective inhibitors in cancers with oncogenic PIK3CβD1067V. Importantly, our findings also provide guidance for the interpretation of ongoing and future cancer genome sequencing efforts that may identify PIK3CβD1067V in additional patient tumor specimens by establishing the functional relevance of this mutant PI3K allele in cancer. More broadly, our study highlights the utility of exome-wide genetic analysis of patient tumor biopsies to identify and characterize unanticipated molecular lesions that functionally contribute to tumor growth and progression.
References
Porta C, Paglino C, Mosca A . Targeting PI3K/Akt/mTOR signaling in cancer. Front Oncol 2014; 4: 64.
Engelman JA, Luo J, Cantley LC . The evolution of phosphatidylinositol 3-kinases as regulators of growth and metabolism. Nat Rev Genet 2006; 7: 606–619.
Chalhoub N, Baker SJ . PTEN and the PI3-kinase pathway in cancer. Annu Rev Pathol 2009; 4: 127–150.
Bauer TM, Patel MR, Infante JR . Targeting PI3 kinase in cancer. Pharmacol Ther 2015; 146C: 53–60.
Samuels Y, Wang Z, Bardelli A, Silliman N, Ptak J, Szabo S et al. High frequency of mutations of the PIK3CA gene in human cancers. Science 2004; 304: 554.
Vogt PK, Kang S, Elsliger MA, Gymnopoulos M . Cancer-specific mutations in phosphatidylinositol 3-kinase. Trends Biochem Sci 2007; 32: 342–349.
Kang S, Bader AG, Vogt PK . Phosphatidylinositol 3-kinase mutations identified in human cancer are oncogenic. Proc Natl Acad Sci USA 2005; 102: 802–807.
Janku F, Tsimberidou AM, Garrido-Laguna I, Wang X, Luthra R, Hong DS et al. PIK3CA mutations in patients with advanced cancers treated with PI3K/AKT/mTOR axis inhibitors. Mol Cancer Ther 2011; 10: 558–565.
Karakas B, Bachman KE, Park BH . Mutation of the PIK3CA oncogene in human cancers. Br J Cancer 2006; 94: 455–459.
Hollander MC, Blumenthal GM, Dennis PA . PTEN loss in the continuum of common cancers, rare syndromes and mouse models. Nat Rev Cancer 2011; 11: 289–301.
Carracedo A, Pandolfi PP . The PTEN-PI3K pathway: of feedbacks and cross-talks. Oncogene 2008; 27: 5527–5541.
Wu JY, Yu CJ, Chang YC, Yang CH, Shih JY, Yang PC . Effectiveness of tyrosine kinase inhibitors on "uncommon" epidermal growth factor receptor mutations of unknown clinical significance in non-small cell lung cancer. Clin Cancer Res 2011; 17: 3812–3821.
Kosaka T, Yamaki E, Mogi A, Kuwano H . Mechanisms of resistance to EGFR TKIs and development of a new generation of drugs in non-small-cell lung cancer. J Biomed Biotechnol 2011; 2011: 165214.
Oxnard GR, Arcila ME, Chmielecki J, Ladanyi M, Miller VA, Pao W . New strategies in overcoming acquired resistance to epidermal growth factor receptor tyrosine kinase inhibitors in lung cancer. Clin Cancer Res 2011; 17: 5530–5537.
Tang J, Salama R, Gadgeel SM, Sarkar FH, Ahmad A . Erlotinib resistance in lung cancer: current progress and future perspectives. Front Pharmacol 2013; 4: 15.
Schroeder MP, Gonzalez-Perez A, Lopez-Bigas N . Visualizing multidimensional cancer genomics data. Genome Med 2013; 5: 9.
Deuker MM, Durban VM, Phillips WA, McMahon M . PI3'-kinase inhibition forestalls the onset of MEK1/2 inhibitor resistance in BRAF-mutated melanoma. Cancer Discov 2014; 5: 143–153.
Charles RP, Silva J, Iezza G, Phillips WA, McMahon M . Activating BRAF and PIK3CA mutations cooperate to promote anaplastic thyroid carcinogenesis. Mol Cancer Res 2014; 12: 979–986.
Jia S, Liu Z, Zhang S, Liu P, Zhang L, Lee SH et al. Essential roles of PI(3)K-p110beta in cell growth, metabolism and tumorigenesis. Nature 2008; 454: 776–779.
Jackson SP, Schoenwaelder SM, Goncalves I, Nesbitt WS, Yap CL, Wright CE et al. PI 3-kinase p110beta: a new target for antithrombotic therapy. Nat Med 2005; 11: 507–514.
Burger MT, Pecchi S, Wagman A, Ni ZJ, Knapp M, Hendrickson T et al. Identification of NVP-BKM120 as a potent, selective, orally bioavailable class I PI3 kinase inhibitor for treating cancer. ACS Med Chem Lett 2011; 2: 774–779.
Ling YH, Lin R, Perez-Soler R . Erlotinib induces mitochondrial-mediated apoptosis in human H3255 non-small-cell lung cancer cells with epidermal growth factor receptorL858R mutation through mitochondrial oxidative phosphorylation-dependent activation of BAX and BAK. Mol Pharmacol 2008; 74: 793–806.
Sos ML, Koker M, Weir BA, Heynck S, Rabinovsky R, Zander T et al. PTEN loss contributes to erlotinib resistance in EGFR-mutant lung cancer by activation of Akt and EGFR. Cancer Res 2009; 69: 3256–3261.
Lammers PE, Lovly CM, Horn L . A patient with metastatic lung adenocarcinoma harboring concurrent EGFR L858R, EGFR germline T790M, and PIK3CA mutations: the challenge of interpreting results of comprehensive mutational testing in lung cancer. J Natl Compr Canc Netw 2014; 12: 6–11.
Dbouk HA, Khalil BD, Wu H, Shymanets A, Nurnberg B, Backer JM . Characterization of a tumor-associated activating mutation of the p110beta PI 3-kinase. PloS One 2013; 8: e63833.
Lin L, Asthana S, Chan E, Bandyopadhyay S, Martins MM, Olivas V et al. Mapping the molecular determinants of BRAF oncogene dependence in human lung cancer. Proc Natl Acad Sci USA 2014; 111: E748–E757.
Boehm JS, Zhao JJ, Yao J, Kim SY, Firestein R, Dunn IF et al. Integrative genomic approaches identify IKBKE as a breast cancer oncogene. Cell 2007; 129: 1065–1079.
Acknowledgements
We acknowledge funding support (to TGB) from the following sources: NIH Director's New Innovator Award, Howard Hughes Medical Institute, Doris Duke Charitable Foundation, American Lung Association, National Lung Cancer Partnership, Sidney Kimmel Foundation for Cancer Research and Searle Scholars Program.
Author Contributions
EP designed, conducted and analyzed all experiments; PG, JSJ, MAG, JW, WP and TGB analyzed the patient tumor sequencing data; GH conducted shRNA knockdown experiments; VRO conducted tumor xenografts; RB provided patient tumor samples; TGB designed and analyzed the experiments; EP and TGB wrote the manuscript with input from all authors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
TGB is a consultant to Driver Group, Novartis, Clovis Oncology and Cleave Biosciences, and has received a research grant from Servier; JW is a consultant to Driver Group; PG, JSJ and WP are employees of Driver Group. The remaining authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Pazarentzos, E., Giannikopoulos, P., Hrustanovic, G. et al. Oncogenic activation of the PI3-kinase p110β isoform via the tumor-derived PIK3CβD1067V kinase domain mutation. Oncogene 35, 1198–1205 (2016). https://doi.org/10.1038/onc.2015.173
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.173
- Springer Nature Limited
This article is cited by
-
SUMOylation modulates the stability and function of PI3K-p110β
Cellular and Molecular Life Sciences (2021)
-
Targeting PI3K in cancer: mechanisms and advances in clinical trials
Molecular Cancer (2019)
-
Functional characterization of a novel somatic oncogenic mutation of PIK3CB
Signal Transduction and Targeted Therapy (2017)
-
PDK1 promotes tumor growth and metastasis in a spontaneous breast cancer model
Oncogene (2016)
-
Molecules in medicine mini-review: isoforms of PI3K in biology and disease
Journal of Molecular Medicine (2016)