Abstract
Phosphatidic acid (PA), the central intermediate in membrane phospholipid synthesis, is generated by two acyltransferases in a pathway conserved in all life forms. The second step in this pathway is catalyzed by 1-acyl-sn-glycerol-3-phosphate acyltransferase, called PlsC in bacteria. Here we present the crystal structure of PlsC from Thermotoga maritima, revealing an unusual hydrophobic/aromatic N-terminal two-helix motif linked to an acyltransferase αβ-domain that contains the catalytic HX4D motif. PlsC dictates the acyl chain composition of the 2-position of phospholipids, and the acyl chain selectivity 'ruler' is an appropriately placed and closed hydrophobic tunnel. We confirmed this by site-directed mutagenesis and membrane composition analysis of Escherichia coli cells that expressed mutant PlsC. Molecular dynamics (MD) simulations showed that the two-helix motif represents a novel substructure that firmly anchors the protein to one leaflet of the membrane. This binding mode allows the PlsC active site to acylate lysophospholipids within the membrane bilayer by using soluble acyl donors.





Similar content being viewed by others
Accession codes
References
Rock, C.O., Goelz, S.E. & Cronan, J.E. Jr. Phospholipid synthesis in Escherichia coli. Characteristics of fatty acid transfer from acyl-acyl carrier protein to sn-glycerol 3-phosphate. J. Biol. Chem. 256, 736–742 (1981).
Ray, T.K. & Cronan, J.E. Jr. Acylation of glycerol 3-phosphate is the sole pathway of de novo phospholipid synthesis in Escherichia coli. J. Bacteriol. 169, 2896–2898 (1987).
Shindou, H. & Shimizu, T. Acyl-CoA:lysophospholipid acyltransferases. J. Biol. Chem. 284, 1–5 (2009).
Yao, J. & Rock, C.O. Phosphatidic acid synthesis in bacteria. Biochim. Biophys. Acta 1831, 495–502 (2013).
Turnbull, A.P. et al. Crystallization and preliminary X-ray analysis of the glycerol-3-phosphate 1-acyltransferase from squash (Cucurbita moschata). Acta Crystallogr. D Biol. Crystallogr. 57, 451–453 (2001).
Tamada, T. et al. Substrate recognition and selectivity of plant glycerol-3-phosphate acyltransferases (GPATs) from Cucurbita moscata and Spinacea oleracea. Acta Crystallogr. D Biol. Crystallogr. 60, 13–21 (2004).
Albesa-Jové, D. et al. Structural basis for selective recognition of acyl chains by the membrane-associated acyltransferase PatA. Nat. Commun. 7, 10906 (2016).
Shindou, H., Hishikawa, D., Harayama, T., Eto, M. & Shimizu, T. Generation of membrane diversity by lysophospholipid acyltransferases. J. Biochem. 154, 21–28 (2013).
Yamashita, A. et al. Acyltransferases and transacylases that determine the fatty acid composition of glycerolipids and the metabolism of bioactive lipid mediators in mammalian cells and model organisms. Prog. Lipid Res. 53, 18–81 (2014).
Agarwal, A.K. Lysophospholipid acyltransferases: 1-acylglycerol-3-phosphate O-acyltransferases. From discovery to disease. Curr. Opin. Lipidol. 23, 290–302 (2012).
Agarwal, A.K. et al. AGPAT2 is mutated in congenital generalized lipodystrophy linked to chromosome 9q34. Nat. Genet. 31, 21–23 (2002).
Lewin, T.M., Wang, P. & Coleman, R.A. Analysis of amino acid motifs diagnostic for the sn-glycerol-3-phosphate acyltransferase reaction. Biochemistry 38, 5764–5771 (1999).
White, S.P., Scott, D.L., Otwinowski, Z., Gelb, M.H. & Sigler, P.B. Crystal structure of cobra-venom phospholipase A2 in a complex with a transition-state analogue. Science 250, 1560–1563 (1990).
Dessen, A. et al. Crystal structure of human cytosolic phospholipase A2 reveals a novel topology and catalytic mechanism. Cell 97, 349–360 (1999).
Miller, D.J., Jerga, A., Rock, C.O. & White, S.W. Analysis of the Staphylococcus aureus DgkB structure reveals a common catalytic mechanism for the soluble diacylglycerol kinases. Structure 16, 1036–1046 (2008).
Feng, L. et al. Structure of a site-2 protease family intramembrane metalloprotease. Science 318, 1608–1612 (2007).
Chung, B.C. et al. Crystal structure of MraY, an essential membrane enzyme for bacterial cell wall synthesis. Science 341, 1012–1016 (2013).
Liu, X., Yin, Y., Wu, J. & Liu, Z. Structure and mechanism of an intramembrane liponucleotide synthetase central for phospholipid biosynthesis. Nat. Commun. 5, 4244 (2014).
Schmidt, J.A., Yvone, G.M. & Brown, W.J. Membrane topology of human AGPAT3 (LPAAT3). Biochem. Biophys. Res. Commun. 397, 661–667 (2010).
Yamashita, A. et al. Topology of acyltransferase motifs and substrate specificity and accessibility in 1-acyl-sn-glycero-3-phosphate acyltransferase 1. Biochim. Biophys. Acta 1771, 1202–1215 (2007).
Parsons, J.B., Frank, M.W., Subramanian, C., Saenkham, P. & Rock, C.O. Metabolic basis for the differential susceptibility of Gram-positive pathogens to fatty acid synthesis inhibitors. Proc. Natl. Acad. Sci. USA 108, 15378–15383 (2011).
Yao, J., Cherian, P.T., Frank, M.W. & Rock, C.O. Chlamydia trachomatis relies on autonomous phospholipid synthesis for membrane biogenesis. J. Biol. Chem. 290, 18874–18888 (2015).
Zhang, C.L. et al. Carbon isotopic fractionations associated with thermophilic bacteria Thermotoga maritima and Persephonella marina. Environ. Microbiol. 4, 58–64 (2002).
Turnbull, A.P. et al. Analysis of the structure, substrate specificity, and mechanism of squash glycerol-3-phosphate (1)-acyltransferase. Structure 9, 347–353 (2001).
Zhang, Y.M. et al. Identification and analysis of the acyl carrier protein (ACP) docking site on beta-ketoacyl-ACP synthase III. J. Biol. Chem. 276, 8231–8238 (2001).
White, S.W., Zheng, J., Zhang, Y.M. & Rock, C.O. The structural biology of type II fatty acid biosynthesis. Annu. Rev. Biochem. 74, 791–831 (2005).
Masoudi, A., Raetz, C.R., Zhou, P. & Pemble, C.W., IV . Chasing acyl carrier protein through a catalytic cycle of lipid A production. Nature 505, 422–426 (2014).
Nguyen, C. et al. Trapping the dynamic acyl carrier protein in fatty acid biosynthesis. Nature 505, 427–431 (2014).
Emptage, R.P., Daughtry, K.D., Pemble, C.W. IV & Raetz, C.R. Crystal structure of LpxK, the 4′-kinase of lipid A biosynthesis and atypical P-loop kinase functioning at the membrane interface. Proc. Natl. Acad. Sci. USA 109, 12956–12961 (2012).
Viklund, H., Granseth, E. & Elofsson, A. Structural classification and prediction of reentrant regions in α-helical transmembrane proteins: application to complete genomes. J. Mol. Biol. 361, 591–603 (2006).
Pan, J. et al. Molecular structures of fluid phase phosphatidylglycerol bilayers as determined by small angle neutron and X-ray scattering. Biochim. Biophys. Acta 1818, 2135–2148 (2012).
Killian, J.A. & von Heijne, G. How proteins adapt to a membrane-water interface. Trends Biochem. Sci. 25, 429–434 (2000).
von Heijne, G. Membrane-protein topology. Nat. Rev. Mol. Cell Biol. 7, 909–918 (2006).
Hayman, M.W., Fawcett, T. & Slabas, A.R. Kinetic mechanism and order of substrate binding for sn-glycerol-3-phosphate acyltransferase from squash (Cucurbita moschata). FEBS Lett. 514, 281–284 (2002).
Kang, Q., Srividhya, J., Ipe, J. & Pomerening, J.R. Evidence toward a dual phosphatase mechanism that restricts Aurora A (Thr-295) phosphorylation during the early embryonic cell cycle. J. Biol. Chem. 289, 17480–17496 (2014).
Bligh, E.G. & Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37, 911–917 (1959).
Ivanova, P.T., Milne, S.B., Byrne, M.O., Xiang, Y. & Brown, H.A. Glycerophospholipid identification and quantitation by electrospray ionization mass spectrometry. Methods Enzymol. 432, 21–57 (2007).
Krank, J., Murphy, R.C., Barkley, R.M., Duchoslav, E. & McAnoy, A. Qualitative analysis and quantitative assessment of changes in neutral glycerol lipid molecular species within cells. Methods Enzymol. 432, 1–20 (2007).
Kabsch, W. XDS. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
Evans, P.R. & Murshudov, G.N. How good are my data and what is the resolution? Acta Crystallogr. D Biol. Crystallogr. 69, 1204–1214 (2013).
Sheldrick, G.M. Experimental phasing with SHELXC/D/E: combining chain tracing with density modification. Acta Crystallogr. D Biol. Crystallogr. 66, 479–485 (2010).
McCoy, A.J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Emsley, P., Lohkamp, B., Scott, W.G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Terwilliger, T.C. et al. Decision-making in structure solution using Bayesian estimates of map quality: the PHENIX AutoSol wizard. Acta Crystallogr. D Biol. Crystallogr. 65, 582–601 (2009).
Terwilliger, T.C. Maximum-likelihood density modification. Acta Crystallogr. D Biol. Crystallogr. 56, 965–972 (2000).
Terwilliger, T.C. et al. Iterative model building, structure refinement and density modification with the PHENIX AutoBuild wizard. Acta Crystallogr. D Biol. Crystallogr. 64, 61–69 (2008).
Afonine, P.V. et al. Towards automated crystallographic structure refinement with phenix.refine. Acta Crystallogr. D Biol. Crystallogr. 68, 352–367 (2012).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38, 27–8 (1996).
Phillips, J.C. et al. Scalable molecular dynamics with NAMD. J. Comput. Chem. 26, 1781–1802 (2005).
Klauda, J.B. et al. Update of the CHARMM all-atom additive force field for lipids: validation on six lipid types. J. Phys. Chem. B 114, 7830–7843 (2010).
Vanommeslaeghe, K. et al. CHARMM general force field: a force field for drug-like molecules compatible with the CHARMM all-atom additive biological force fields. J. Comput. Chem. 31, 671–690 (2010).
Vanommeslaeghe, K. & MacKerell, A.D. Jr. Automation of the CHARMM General Force Field (CGenFF) I: bond perception and atom typing. J. Chem. Inf. Model. 52, 3144–3154 (2012).
Vanommeslaeghe, K., Raman, E.P. & MacKerell, A.D. Jr. Automation of the CHARMM General Force Field (CGenFF) II: assignment of bonded parameters and partial atomic charges. J. Chem. Inf. Model. 52, 3155–3168 (2012).
Aleksandrov, A. & Field, M. Efficient solvent boundary potential for hybrid potential simulations. Phys. Chem. Chem. Phys. 13, 10503–10509 (2011).
Acknowledgements
We thank M. Frank for mass spectrometry, and M.-K. Yun for crystallography assistance. Diffraction data were collected at Southeast Regional Collaborative Access Team (SERCAT) beamlines 22-ID and 22-BM at the Advanced Photon Source (APS), Argonne National Laboratory (Argonne, Illinois, USA). Supporting SERCAT institutions may be found at http://www.ser-cat.org/members.html. Use of the APS is supported by the US Department of Energy under Contract No. W-31-109-Eng-38. This work was supported by the NIH (grant GM034496 to C.O.R.), the NCI (Cancer Center core grant CA21765 to St. Jude Children's Research Hospital), and the American Lebanese Syrian Associated Charities (ALSAC).
Author information
Authors and Affiliations
Contributions
R.M.R., J.Y., C.O.R. and S.W.W. designed the study. R.M.R., S.G. and S.W.W. determined and interpreted the crystal structures. J.Y. and C.O.R. performed and analyzed the biochemical experiments. G.K. performed the modeling and docking studies. E.W.M. performed the MD simulations. All authors contributed to and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Complementation of strain SM2-1 with constructs expressing PlsC.
E. coli strain SM2-1 (plsC(Ts)) is defective in PlsC activity. This strain is able to grow at 30 °C, but unable to grow at 42 °C without PlsC complementation. (a) Growth of the temperature-sensitive E. coli strain SM2-1 (plsC(Ts)) transformed with plasmids expressing the empty vector (Control) or PlsC from E. coli (EcPlsC), Chlamydia trachomatis (CtPlsC), or T. maritima (TmPlsC) under the permissive growth condition (30 °C). (b) Growth of the same strain series at the non-permissive temperature (42 °C). (c) Growth of the temperature-sensitive E. coli strain SM2-1 (plsC(Ts)) transformed with plasmids expressing the empty vector (Control) or TmPlsC mutants G25A, G25V, G25L, G25M under the permissive growth condition (30 °C). (d) Growth of the same strain series at the non-permissive temperature (42 °C). Four independent colonies were analyzed for each condition.
Supplementary Figure 2 Acyl chain specificity of PlsC variants.
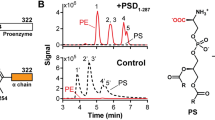
The temperature-sensitive Escherichia coli strain SM2-1 (plsC(Ts)) was transformed with various PlsC constructs and the PE molecular species were determined in the complemented SM2-1 strains to assess acyl chain selectivity. (a) The 2-position acyl chain composition of strain SM2-1 complemented with E. coli PlsC (EcPlsC), Chlamydia trachomatis PlsC (CtPlsC), Thermotoga maritima PlsC (TmPlsC) and the TmPlsC(G25M) mutant. (b) Molecular species profile of strain SM2-1 complemented with the TmPlsC(G25A) mutant. (c) Molecular species profile of strain SM2-1 complemented with the TmPlsC(G25V) mutant. (d) Molecular species profile of strain SM2-1 complemented with the TmPlsC(G25L) mutant. In (b), (c) and (d), the molecular species containing 16:0 in the 2-position are colored red, while molecular species containing 14:0 in the 2-position are colored blue. In these experiments, the phospholipids from complemented SM2-1 cells were selectively hydrolyzed for the 2-position fatty acid using snake venom phospholipase A2. The hydrolyzed fatty acids were then converted into fatty acid methyl esters and analyzed via gas chromatography. Gas chromatography data are from duplicate biological replicates and the mass spectra shown are representative of duplicate biological replicates.
Supplementary Figure 3 Biochemical characterization of TmPlsC.
(a) TmPlsC was overexpressed and purified by affinity chromatography. Activity versus enzyme concentration was determined for the purified TmPlsC using saturating concentrations of 1-hexadecanoyl-sn-glycero-3-phosphate (16:0-LPA) and [14C]16:0-ACP. Inset, SDS-PAGE shows the purity of TmPlsC. Lane 1, standards; lane 2, TmPlsC. (b) TmPlsC catalyzed 1-acyl-sn-glycerol-3-phosphate acyltransferase activity was compared using the [14C]16:0-ACP or [14C]16:0-CoA as the acyl donor and 16:0-LPA. The TmPlsC was able to use both acyl-donors, with higher activity using [14C]16:0-ACP. (c) The specific activity of the purified wild type TmPlsC, TmPlsC-H1 (missing the N-terminal helix α1), and TmPlsC-H12 (missing both N-terminal helices α1 and α2) proteins. The TmPlsC-H1 protein had a greater than 100 fold decrease in activity compared to the wild type, while the TmPlsC-H12 protein was inactive. The data were derived from duplicate experiments. See Supplementary Data Set 1 for additional information.
Supplementary Figure 4 TmPlsC electron density.
(a) The active site locale. (b) The N-terminal two-helix motif. (c) Putative acyl chain within the specificity tunnel. The electron densities are from 2Fo-Fc maps contoured at 1.4 σ in (a) and 1 σ in (b) and (c).
Supplementary Figure 5 The primary, secondary and tertiary structure of TmPlsC.
(a) The amino acid sequence highlighting the N-terminal two-helix motif (orange), and the ab catalytic domain (light blue α-helices and light green β-strands). Yellow, active site residues; magenta, exposed aromatic residues proposed to interact with the lipid bilayer; blue, basic residues proposed to interact with the membrane phospholipids; pink, residues that line the hydrophobic selectivity tunnel. The four conserved signature AGPAT family motifs are boxed and labeled. The figure was prepared using ENDscript 2 (Robert & Gouet, Nucleic Acids Res 42, W320-4 (2014). (b) AGPAT family motifs in the ab domain. Motifs 1 (light green), 3 (magenta) and 4 (teal) are at the active site, and motif 2 (purple) is in a flexible loop.
Supplementary Figure 6 Structural conservation of the ab-acyltransferase catalytic domain.
(a) PlsC. (b) G3P acyltransferase (GPAT). (c) Mycobacterial PatA. (d) Overlay of the three structures. In each figure, the acyltransferase domain is colored light blue (α-helices) and light green (β-strands and loops), and the different additional α-helical subdomains are colored orange (PlsC), brown (GPAT) and pink (PatA). The catalytic histidine (yellow carbon sticks) shows the common location of the active site.
Supplementary Figure 7 The packing of the N-terminal two-helix motif in the TmPlsC crystal.
In the I23 crystal lattice, the two TmPlsC molecules in the asymmetric unit pack around the three-fold axis via their two-helix motifs to create a double ring trimeric assembly. (a) (Side view) and (b) (Top view). Each trimer in the assembly encloses electron density (blue) that is consistent with bound phospholipid molecules that are aligned around the axis in a fashion that approximates how TmPlsC engages one leaflet of the membrane. The three asymmetric units are colored green, magenta and brown. The central electron density is best resolved in assembly 1, and (c) and (d) show the side and top views and the nine refined phospholipid molecules, three for each TmPlsC molecule. (e) Close up of the electron densities shown in (c) and (d). (f) The electron density for the best resolved phospholipid shown in (e). The electron densities are from 2Fo-Fc maps contoured at 1 σ.
Supplementary Figure 8 Diffusion of TmPlsC in the MD simulation box.
The root mean squared deviation of the protein center of mass from the starting position (after equilibration) was calculated in the xy plane (blue circles) showing diffusion in the membrane. The root mean squared vertical displacement of the protein center of mass versus the starting position (red circles) was calculated and then normalized for displacement of the entire membrane in the simulation box. This plot shows that the protein converged on a defined depth in the membrane.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8. (PDF 1088 kb)
Supplementary Data set 1
Supplementary Data Set 1. (PDF 271 kb)
Rights and permissions
About this article
Cite this article
Robertson, R., Yao, J., Gajewski, S. et al. A two-helix motif positions the lysophosphatidic acid acyltransferase active site for catalysis within the membrane bilayer. Nat Struct Mol Biol 24, 666–671 (2017). https://doi.org/10.1038/nsmb.3436
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.3436
- Springer Nature America, Inc.
This article is cited by
-
Acylation of glycerolipids in mycobacteria
Nature Communications (2023)
-
Structure of a eukaryotic cholinephosphotransferase-1 reveals mechanisms of substrate recognition and catalysis
Nature Communications (2023)
-
Identification of an alternative triglyceride biosynthesis pathway
Nature (2023)
-
Structural basis of the acyl-transfer mechanism of human GPAT1
Nature Structural & Molecular Biology (2023)
-
Division of the role and physiological impact of multiple lysophosphatidic acid acyltransferase paralogs
BMC Microbiology (2022)