Key Points
-
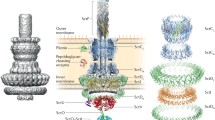
Gram-negative bacteria have evolved a wide array of secretion systems to transport small molecules, proteins and DNA into the extracellular space or target cells. In this Review, we describe insights into the structural and mechanistic features of the six secretion systems (types I–VI) of Gram-negative bacteria, the unique mycobacterial type VII secretion system, the chaperone–usher pathway and the curli biogenesis machinery.
-
These systems are remarkably varied in size, composition and architecture. Double-membrane-spanning secretion systems are composed of many tens of protein subunits and can reach multi-megadalton sizes, whereas outer-membrane-spanning systems are relatively simple and are usually composed of only one type of subunit.
-
These systems can transport folded or unfolded substrates and use various energy sources to power transport, from ATP to proton or entropy gradients.
-
Recent structural and molecular advances have uncovered remarkable structural and functional similarities between secretion systems that have the potential to be exploited for the development of novel antibacterial compounds.
Abstract
Bacteria have evolved a remarkable array of sophisticated nanomachines to export various virulence factors across the bacterial cell envelope. In recent years, considerable progress has been made towards elucidating the structural and molecular mechanisms of the six secretion systems (types I–VI) of Gram-negative bacteria, the unique mycobacterial type VII secretion system, the chaperone–usher pathway and the curli secretion machinery. These advances have greatly enhanced our understanding of the complex mechanisms that these macromolecular structures use to deliver proteins and DNA into the extracellular environment or into target cells. In this Review, we explore the structural and mechanistic relationships between these single- and double-membrane-embedded systems, and we briefly discuss how this knowledge can be exploited for the development of new antimicrobial strategies.




Similar content being viewed by others
References
Gerlach, R. G. & Hensel, M. Protein secretion systems and adhesins: the molecular armory of Gram-negative pathogens. Int. J. Med. Microbiol. 297, 401–415 (2007).
Lycklama a Nijehot, J. A. & Driessen, A. J. M. The bacterial Sec-translocase: structure and mechanism. Phil. Trans. R. Soc. B 367, 1016–1028 (2012).
Palmer, T. & Berks, B. C. The twin-arginine translocation (Tat) protein export pathway. Nature Rev. Microbiol. 10, 483–496 (2012).
Rego, A. T., Chandran, V. & Waksman, G. Two-step and one-step secretion mechanisms in Gram-negative bacteria: contrasting the type IV secretion system and the chaperone–usher pathway of pilus biogenesis. Biochem. J. 425, 475–488 (2010).
Kanonenberg, K., Schwarz, C. K. W. & Schmitt, L. Type I secretion systems — a story of appendices. Res. Microbiol. 164, 596–604 (2013).
Piddock, L. J. V. Multidrug-resistance efflux pumps — not just for resistance. Nature Rev. Microbiol. 4, 629–636 (2006).
Kadaba, N. S., Kaiser, J. T., Johnson, E., Lee, A. & Rees, D. C. The high-affinity E. coli methionine ABC transporter: structure and allosteric regulation. Science 321, 250–253 (2008).
Shintre, C. A. et al. Structures of ABCB10, a human ATP-binding cassette transporter in apo- and nucleotide-bound states. Proc. Natl Acad. Sci. USA 110, 9710–9715 (2013). This article describes the molecular mechanism of ABC transporters.
Murakami, S., Nakashima, R., Yamashita, E., Matsumoto, T. & Yamaguchi, A. Crystal structures of a multidrug transporter reveal a functionally rotating mechanism. Nature 443, 173–179 (2006).
Murakami, S., Nakashima, R., Yamashita, E. & Yamaguchi, A. Crystal structure of bacterial multidrug efflux transporter AcrB. Nature 419, 587–593 (2002).
Yu, E. W., McDermott, G., Zgurskaya, H. I., Nikaido, H. & Koshland, D. E. Structural basis of multiple drug-binding capacity of the AcrB multidrug efflux pump. Science 300, 976–980 (2003).
Seeger, M. A. Structural asymmetry of AcrB trimer suggests a peristaltic pump mechanism. Science 313, 1295–1298 (2006).
Sennhauser, G., Amstutz, P., Briand, C., Storchenegger, O. & Grütter, M. G. Drug export pathway of multidrug exporter AcrB revealed by DARPin inhibitors. PLoS Biol. 5, e7 (2007). This paper describes the molecular mechanism of transport of the RND inner membrane component.
Eicher, T. et al. Coupling of remote alternating-access transport mechanisms for protons and substrates in the multidrug efflux pump AcrB. eLife 3, e03145 (2014).
Balakrishnan, L., Hughes, C. & Koronakis, V. Substrate-triggered recruitment of the TolC channel-tunnel during type I export of hemolysin by Escherichia coli. J. Mol. Biol. 313, 501–510 (2001).
Delepelaire, P. Type I secretion in Gram-negative bacteria. Biochim. Biophys. Acta 1694, 149–161 (2004).
Yum, S. et al. Crystal structure of the periplasmic component of a tripartite macrolide-specific efflux pump. J. Mol. Biol. 387, 1286–1297 (2009).
Koronakis, V., Eswaran, J. & Hughes, C. Structure and function of TolC: the bacterial exit duct for proteins and drugs. Annu. Rev. Biochem. 73, 467–489 (2004).
Bavro, V. N. et al. Assembly and channel opening in a bacterial drug efflux machine. Mol. Cell 30, 114–121 (2008). This article describes the opening mechanism of TolC.
Pei, X. Y. et al. Structures of sequential open states in a symmetrical opening transition of the TolC exit duct. Proc. Natl Acad. Sci. USA 108, 2112–2117 (2011).
Du, D. et al. Structure of the AcrAB–TolC multidrug efflux pump. Nature 509, 512–515 (2014). This article describes the EM structure of a complete RND pump.
Nivaskumar, M. & Francetic, O. Type II secretion system: a magic beanstalk or a protein escalator. Biochim. Biophys. Acta 1843, 1568–1577 (2014).
Korotkov, K. V., Sandkvist, M. & Hol, W. G. The type II secretion system: biogenesis, molecular architecture and mechanism. Nature Rev. Microbiol. 10, 336–351 (2012).
Reichow, S. L., Korotkov, K. V., Hol, W. G. & Gonen, T. Structure of the cholera toxin secretion channel in its closed state. Nature Struct. Mol. Biol. 17, 1226–1232 (2010). This study describes the EM map of a T2SS secretin.
Korotkov, K. V., Pardon, E., Steyaert, J. & Hol, W. G. Crystal structure of the N-terminal domain of the secretin GspD from ETEC determined with the assistance of a nanobody. Structure 17, 255–265 (2009).
Korotkov, K. V., Delarosa, J. R. & Hol, W. G. A dodecameric ring-like structure of the N0 domain of the type II secretin from enterotoxigenic Escherichia coli. J. Struct. Biol. 183, 354–362 (2013).
Gu, S., Rehman, S., Wang, X., Shevchik, V. E. & Pickersgill, R. W. Structural and functional insights into the pilotin-secretin complex of the type II secretion system. PLoS Pathog. 8, e1002531 (2012).
Abendroth, J. et al. The three-dimensional structure of the cytoplasmic domains of EpsF from the type 2 secretion system of Vibrio cholerae. J. Struct. Biol. 166, 303–315 (2009).
Abendroth, J., Bagdasarian, M., Sandkvist, M. & Hol, W. G. The structure of the cytoplasmic domain of EpsL, an inner membrane component of the type II secretion system of Vibrio cholerae: an unusual member of the actin-like ATPase superfamily. J. Mol. Biol. 344, 619–633 (2004).
Abendroth, J., Murphy, P., Sandkvist, M., Bagdasarian, M. & Hol, W. G. The X-ray structure of the type II secretion system complex formed by the N-terminal domain of EpsE and the cytoplasmic domain of EpsL of Vibrio cholerae. J. Mol. Biol. 348, 845–855 (2005).
Abendroth, J., Kreger, A. C. & Hol, W. G. The dimer formed by the periplasmic domain of EpsL from the type 2 secretion system of Vibrio parahaemolyticus. J. Struct. Biol. 168, 313–322 (2009).
Korotkov, K. V. et al. Structural and functional studies on the interaction of GspC and GspD in the type II secretion system. PLoS Pathog. 7, e1002228 (2011).
McLaughlin, L. S., Haft, R. J. & Forest, K. T. Structural insights into the type II secretion nanomachine. Curr. Opin. Struct. Biol. 22, 208–216 (2012).
Lu, C. et al. Hexamers of the type II secretion ATPase GspE from Vibrio cholerae with increased ATPase activity. Structure 21, 1707–1717 (2013).
Gray, M. D., Bagdasarian, M., Hol, W. G. & Sandkvist, M. In vivo cross-linking of EpsG to EpsL suggests a role for EpsL as an ATPase-pseudopilin coupling protein in the type II secretion system of Vibrio cholerae. Mol. Microbiol. 79, 786–798 (2011).
Py, B., Loiseau, L. & Barras, F. An inner membrane platform in the type II secretion machinery of Gram-negative bacteria. EMBO Rep. 2, 244–248 (2001).
Campos, M., Nilges, M., Cisneros, D. A. & Francetic, O. Detailed structural and assembly model of the type II secretion pilus from sparse data. Proc. Natl Acad. Sci. USA 107, 13081–13086 (2010).
Cisneros, D. A., Pehau-Arnaudet, G. & Francetic, O. Heterologous assembly of type IV pili by a type II secretion system reveals the role of minor pilins in assembly initiation. Mol. Microbiol. 86, 805–818 (2012).
Nivaskumar, M. et al. Distinct docking and stabilization steps of the pseudopilus conformational transition path suggest rotational assembly of type IV pilus-like fibers. Structure 22, 685–696 (2014).
Berry, J. L. et al. Structure and assembly of a trans-periplasmic channel for type IV pili in Neisseria meningitidis. PLoS Pathog. 8, e1002923 (2012).
Galan, J. E. & Wolf-Watz, H. Protein delivery into eukaryotic cells by type III secretion machines. Nature 444, 567–573 (2006).
Cornelis, G. R. The type III secretion injectisome. Nature Rev. Microbiol. 4, 811–825 (2006).
Büttner, D. Protein export according to schedule: architecture, assembly, and regulation of type III secretion systems from plant- and animal-pathogenic bacteria. Microbiol. Mol. Biol. Rev. 76, 262–310 (2012).
Kubori, T. et al. Supramolecular structure of the Salmonella typhimurium type III protein secretion system. Science 280, 602–605 (1998). This study reveals and characterizes the first T3SS isolated from S . Typhimurium by EM.
Schraidt, O. & Marlovits, T. C. Three-dimensional model of Salmonella's needle complex at subnanometer resolution. Science 331, 1192–1195 (2011).
Marlovits, T. C. et al. Assembly of the inner rod determines needle length in the type III secretion injectisome. Nature 441, 637–640 (2006).
Marlovits, T. C. et al. Structural insights into the assembly of the type III secretion needle complex. Science 306, 1040–1042 (2004). This paper provides the first structural insights into a T3SS needle complex.
Kawamoto, A. et al. Common and distinct structural features of Salmonella injectisome and flagellar basal body. Sci. Rep. 3, 3369 (2013).
Eichelberg, K., Ginocchio, C. C. & Galan, J. E. Molecular and functional characterization of the Salmonella typhimurium invasion genes invB and invC: homology of InvC to the F0F1 ATPase family of proteins. J. Bacteriol. 176, 4501–4510 (1994).
Akeda, Y. & Galán, J. E. Chaperone release and unfolding of substrates in type III secretion. Nature 437, 911–915 (2005).
Lara-Tejero, M., Kato, J., Wagner, S., Liu, X. & Galán, J. E. A sorting platform determines the order of protein secretion in bacterial type III systems. Science 331, 1188–1191 (2011).
Schraidt, O. et al. Topology and organization of the Salmonella typhimurium type III secretion needle complex components. PLoS Pathog. 6, e1000824 (2010).
Spreter, T. et al. A conserved structural motif mediates formation of the periplasmic rings in the type III secretion system. Nature Struct. Mol. Biol. 16, 468–476 (2009).
Yip, C. K. et al. Structural characterization of the molecular platform for type III secretion system assembly. Nature 435, 702–707 (2005).
Worrall, L. J., Lameignere, E. & Strynadka, N. C. Structural overview of the bacterial injectisome. Curr. Opin. Microbiol. 14, 3–8 (2011).
Abrusci, P. et al. Architecture of the major component of the type III secretion system export apparatus. Nature Struct. Mol. Biol. 20, 99–104 (2013).
Kubori, T., Sukhan, A., Aizawa, S. I. & Galán, J. E. Molecular characterization and assembly of the needle complex of the Salmonella typhimurium type III protein secretion system. Proc. Natl Acad. Sci. USA 97, 10225–10230 (2000).
Kimbrough, T. G. & Miller, S. I. Contribution of Salmonella typhimurium type III secretion components to needle complex formation. Proc. Natl Acad. Sci. USA 97, 11008–11013 (2000).
Poyraz, O. et al. Protein refolding is required for assembly of the type three secretion needle. Nature Struct. Mol. Biol. 17, 788–792 (2010).
Loquet, A. et al. Atomic model of the type III secretion system needle. Nature 486, 276–279 (2012).
Radics, J., Konigsmaier, L. & Marlovits, T. C. Structure of a pathogenic type 3 secretion system in action. Nature Struct. Mol. Biol. 21, 82–87 (2014). This study describes the first three-dimensional snapshot of a T3SS needle complex in the process of substrate secretion.
Blocker, A. J. et al. What's the point of the type III secretion system needle? Proc. Natl Acad. Sci. USA 105, 6507–6513 (2008).
Izore, T., Job, V. & Dessen, A. Biogenesis, regulation, and targeting of the type III secretion system. Structure 19, 603–612 (2011).
Galan, J. E., Lara-Tejero, M., Marlovits, T. C. & Wagner, S. Bacterial type III secretion systems: specialized nanomachines for protein delivery into target cells. Annu. Rev. Microbiol. 68, 415–438 (2014).
Parsot, C., Hamiaux, C. & Page, A. L. The various and varying roles of specific chaperones in type III secretion systems. Curr. Opin. Microbiol. 6, 7–14 (2003).
Hu, B. et al. Visualization of the type III secretion sorting platform of Shigella flexneri. Proc. Natl Acad. Sci. USA 112, 1047–1052 (2015).
Alvarez-Martinez, C. E. & Christie, P. J. Biological diversity of prokaryotic type IV secretion systems. Microbiol. Mol. Biol. Rev. 73, 775–808 (2009).
Christie, P. J., Whitaker, N. & González-Rivera, C. Mechanism and structure of the bacterial type IV secretion systems. Biochim. Biophys. Acta 1843, 1578–1591 (2014).
Trokter, M., Felisberto-Rodrigues, C., Christie, P. J. & Waksman, G. Recent advances in the structural and molecular biology of type IV secretion systems. Curr. Opin. Struct. Biol. 27, 16–23 (2014).
Low, H. H. et al. Structure of a type IV secretion system. Nature 508, 550–553 (2014). This is the first study to describe the overall architecture of a T4SS.
Fronzes, R. et al. Structure of a type IV secretion system core complex. Science 323, 266–268 (2009). This paper describes the assembly, purification and EM structure of the core–OM complex of a T4SS.
Rivera-Calzada, A. et al. Structure of a bacterial type IV secretion core complex at subnanometre resolution. EMBO J. 32, 1195–1204 (2013).
Chandran, V. et al. Structure of the outer membrane complex of a type IV secretion system. Nature 462, 1011–1015 (2009).
Cascales, E. & Christie, P. J. Agrobacterium VirB10, an ATP energy sensor required for type IV secretion. Proc. Natl Acad. Sci. USA 101, 17228–17233 (2004).
Terradot, L. et al. Structures of two core subunits of the bacterial type IV secretion system, VirB8 from Brucella suis and ComB10 from Helicobacter pylori. Proc. Natl Acad. Sci. USA 102, 4596–4601 (2005).
Savvides, S. N. et al. VirB11 ATPases are dynamic hexameric assemblies: new insights into bacterial type IV secretion. EMBO J. 22, 1969–1980 (2003).
Pena, A. et al. The hexameric structure of a conjugative VirB4 protein ATPase provides new insights for a functional and phylogenetic relationship with DNA translocases. J. Biol. Chem. 287, 39925–39932 (2012).
Yeo, H. J., Yuan, Q., Beck, M. R., Baron, C. & Waksman, G. Structural and functional characterization of the VirB5 protein from the type IV secretion system encoded by the conjugative plasmid pKM101. Proc. Natl Acad. Sci. USA 100, 15947–15952 (2003).
Gomis-Rüth, F. X. et al. The bacterial conjugation protein TrwB resembles ring helicases and F1-ATPase. Nature 409, 637–641 (2001).
Wallden, K. et al. Structure of the VirB4 ATPase, alone and bound to the core complex of a type IV secretion system. Proc. Natl Acad. Sci. USA 109, 11348–11353 (2012).
Bradley, D. E. Morphological and serological relationships of conjugative pili. Plasmid 4, 155–169 (1980).
Durrenberger, M. B., Villiger, W. & Bachi, T. Conjugational junctions: morphology of specific contacts in conjugating Escherichia coli bacteria. J. Struct. Biol. 107, 146–156 (1991).
Aly, K. A. & Baron, C. The VirB5 protein localizes to the T-pilus tips in Agrobacterium tumefaciens. Microbiology 153, 3766–3775 (2007).
Cascales, E. & Christie, P. J. Definition of a bacterial type IV secretion pathway for a DNA substrate. Science 304, 1170–1173 (2004). This study defines the translocation pathway for a DNA substrate through the bacterial T4SS.
Ripoll-Rozada, J., Zunzunegui, S., de la Cruz, F., Arechaga, I. & Cabezon, E. Functional interactions of VirB11 traffic ATPases with VirB4 and VirD4 molecular motors in type IV secretion systems. J. Bacteriol. 195, 4195–4201 (2013).
Zoued, A. et al. Architecture and assembly of the Type VI secretion system. Biochim. Biophys. Acta 1843, 1664–1673 (2014).
Ho, B. T., Dong, T. G. & Mekalanos, J. J. A view to a kill: the bacterial type VI secretion system. Cell Host Microbe 15, 9–21 (2014).
Pukatzki, S. et al. Identification of a conserved bacterial protein secretion system in Vibrio cholerae using the Dictyostelium host model system. Proc. Natl Acad. Sci. USA 103, 1528–1533 (2006).
Boyer, F., Fichant, G., Berthod, J., Vandenbrouck, Y. & Attree, I. Dissecting the bacterial type VI secretion system by a genome wide in silico analysis: what can be learned from available microbial genomic resources? BMC Genomics 10, 104 (2009).
Zheng, J. & Leung, K. Y. Dissection of a type VI secretion system in Edwardsiella tarda. Mol. Microbiol. 66, 1192–1206 (2007).
Ma, L. S., Lin, J. S. & Lai, E. M. An IcmF family protein, ImpLM, is an integral inner membrane protein interacting with ImpKL, and its walker a motif is required for type VI secretion system-mediated Hcp secretion in Agrobacterium tumefaciens. J. Bacteriol. 191, 4316–4329 (2009).
Leiman, P. G. et al. Type VI secretion apparatus and phage tail-associated protein complexes share a common evolutionary origin. Proc. Natl Acad. Sci. USA 106, 4154–4159 (2009).
Basler, M., Pilhofer, M., Henderson, G. P., Jensen, G. J. & Mekalanos, J. J. Type VI secretion requires a dynamic contractile phage tail-like structure. Nature 483, 182–186 (2012). This study uses a combination of fluorescence microscopy and electron microscopy to visualize the T6SS in action.
Felisberto-Rodrigues, C. et al. Towards a structural comprehension of bacterial type VI secretion systems: characterization of the TssJ–TssM complex of an Escherichia coli pathovar. PLoS Pathog. 7, e1002386 (2011).
Zoued, A. et al. TssK is a trimeric cytoplasmic protein interacting with components of both phage-like and membrane anchoring complexes of the type VI secretion system. J. Biol. Chem. 288, 27031–27041 (2013).
Shneider, M. M. et al. PAAR-repeat proteins sharpen and diversify the type VI secretion system spike. Nature 500, 350–353 (2013).
Brunet, Y. R., Hénin, J., Celia, H. & Cascales, E. Type VI secretion and bacteriophage tail tubes share a common assembly pathway. EMBO Rep. 15, 315–321 (2014).
Mougous, J. D. et al. A virulence locus of Pseudomonas aeruginosa encodes a protein secretion apparatus. Science 312, 1526–1530 (2006).
Kube, S. et al. Structure of the VipA/B type VI secretion complex suggests a contraction-state-specific recycling mechanism. Cell Rep. 8, 20–30 (2014).
Bönemann, G., Pietrosiuk, A., Diemand, A., Zentgraf, H. & Mogk, A. Remodelling of VipA/VipB tubules by ClpV-mediated threading is crucial for type VI protein secretion. EMBO J. 28, 315–325 (2009).
Kudryashev, M. et al. Structure of the type VI secretion system contractile sheath. Cell 160, 952–962 (2015).
Silverman, J. M. et al. Haemolysin coregulated protein is an exported receptor and chaperone of type VI secretion substrates. Mol. Cell 51, 584–593 (2013).
Basler, M. & Mekalanos, J. J. Type 6 secretion dynamics within and between bacterial cells. Science 337, 815 (2012).
Clemens, D. L., Ge, P., Lee, B. Y., Horwitz, M. A. & Zhou, Z. H. Atomic structure of T6SS reveals interlaced array essential to function. Cell 160, 940–951 (2015).
Leo, J. C., Grin, I. & Linke, D. Type V secretion: mechanism(s) of autotransport through the bacterial outer membrane. Phil. Trans. R. Soc. B 367, 1088–1101 (2012).
Leyton, D. L., Rossiter, A. E. & Henderson, I. R. From self sufficiency to dependence: mechanisms and factors important for autotransporter biogenesis. Nature Rev. Microbiol. 10, 213–225 (2012).
Junker, M., Besingi, R. N. & Clark, P. L. Vectorial transport and folding of an autotransporter virulence protein during outer membrane secretion. Mol. Microbiol. 71, 1323–1332 (2009).
Roman-Hernandez, G., Peterson, J. H. & Bernstein, H. D. Reconstitution of bacterial autotransporter assembly using purified components. eLife 3, e04234 (2014).
Voulhoux, R., Bos, M. P., Geurtsen, J., Mols, M. & Tommassen, J. Role of a highly conserved bacterial protein in outer membrane protein assembly. Science 299, 262–265 (2003).
Wu, T. et al. Identification of a multicomponent complex required for outer membrane biogenesis in Escherichia coli. Cell 121, 235–245 (2005).
Hagan, C. L., Kim, S. & Kahne, D. Reconstitution of outer membrane protein assembly from purified components. Science 328, 890–892 (2010).
Ieva, R., Tian, P., Peterson, J. H. & Bernstein, H. D. Sequential and spatially restricted interactions of assembly factors with an autotransporter β domain. Proc. Natl Acad. Sci. USA 108, E383–E391 (2011).
Noinaj, N., Kuszak, A. J., Balusek, C., Gumbart, J. C. & Buchanan, S. K. Lateral opening and exit pore formation are required for BamA function. Structure 22, 1055–1062 (2014).
Noinaj, N. et al. Structural insight into the biogenesis of β-barrel membrane proteins. Nature 501, 385–390 (2013).
Ieva, R. & Bernstein, H. D. Interaction of an autotransporter passenger domain with BamA during its translocation across the bacterial outer membrane. Proc. Natl Acad. Sci. USA 106, 19120–19125 (2009).
Oomen, C. J. et al. Structure of the translocator domain of a bacterial autotransporter. EMBO J. 23, 1257–1266 (2004).
van den Berg, B. Crystal structure of a full-length autotransporter. J. Mol. Biol. 396, 627–633 (2010). This article defines the first atomic structure of a full-length autotransporter, including both the translocator and passenger domains.
Meng, G., Surana, N. K., St Geme, J. W. & Waksman, G. Structure of the outer membrane translocator domain of the Haemophilus influenzae Hia trimeric autotransporter. EMBO J. 25, 2297–2304 (2006).
Ieva, R., Skillman, K. M. & Bernstein, H. D. Incorporation of a polypeptide segment into the β-domain pore during the assembly of a bacterial autotransporter. Mol. Microbiol. 67, 188–201 (2008).
Skillman, K. M., Barnard, T. J., Peterson, J. H., Ghirlando, R. & Bernstein, H. D. Efficient secretion of a folded protein domain by a monomeric bacterial autotransporter. Mol. Microbiol. 58, 945–958 (2005).
Pavlova, O., Peterson, J. H., Ieva, R. & Bernstein, H. D. Mechanistic link between β barrel assembly and the initiation of autotransporter secretion. Proc. Natl Acad. Sci. USA 110, E938–E947 (2013).
Selkrig, J. et al. Discovery of an archetypal protein transport system in bacterial outer membranes. Nature Struct. Mol. Biol. 19, 506–510 (2012).
Gruss, F. et al. The structural basis of autotransporter translocation by TamA. Nature Struct. Mol. Biol. 20, 1318–1320 (2013).
Wright, K. J., Seed, P. C. & Hultgren, S. J. Development of intracellular bacterial communities of uropathogenic Escherichia coli depends on type 1 pili. Cell. Microbiol. 9, 2230–2241 (2007).
Lillington, J., Geibel, S. & Waksman, G. Reprint of “Biogenesis and adhesion of type 1 and P pili”. Biochim. Biophys. Acta 1850, 554–564 (2015).
Mu, X. Q. & Bullitt, E. Structure and assembly of P-pili: a protruding hinge region used for assembly of a bacterial adhesion filament. Proc. Natl Acad. Sci. USA 103, 9861–9866 (2006).
Hahn, E. et al. Exploring the 3D molecular architecture of Escherichia coli type 1 pili. J. Mol. Biol. 323, 845–857 (2002).
Choudhury, D. et al. X-ray structure of the FimC–FimH chaperone-adhesin complex from uropathogenic Escherichia coli. Science 285, 1061–1066 (1999).
Hultgren, S. J., Normark, S. & Abraham, S. N. Chaperone-assisted assembly and molecular architecture of adhesive pili. Annu. Rev. Microbiol. 45, 383–415 (1991).
Vetsch, M. et al. Pilus chaperones represent a new type of protein-folding catalyst. Nature 431, 329–333 (2004).
Sauer, F. G. et al. Structural basis of chaperone function and pilus biogenesis. Science 285, 1058–1061 (1999).
Phan, G. et al. Crystal structure of the FimD usher bound to its cognate FimC–FimH substrate. Nature 474, 49–53 (2011). This paper provides the first structural insight into the mechanism of pilus assembly.
Geibel, S., Procko, E., Hultgren, S. J., Baker, D. & Waksman, G. Structural and energetic basis of folded-protein transport by the FimD usher. Nature 496, 243–246 (2013).
Remaut, H. et al. Donor-strand exchange in chaperone-assisted pilus assembly proceeds through a concerted β strand displacement mechanism. Mol. Cell 22, 831–842 (2006).
Verger, D., Miller, E., Remaut, H., Waksman, G. & Hultgren, S. Molecular mechanism of P pilus termination in uropathogenic Escherichia coli. EMBO Rep. 7, 1228–1232 (2006).
Olsen, A., Jonsson, A. & Normark, S. Fibronectin binding mediated by a novel class of surface organelles on Escherichia coli. Nature 338, 652–655 (1989).
Hammar, M., Arnqvist, A., Bian, Z., Olsen, A. & Normark, S. Expression of two csg operons is required for production of fibronectin- and Congo red-binding curli polymers in Escherichia coli K-12. Mol. Microbiol. 18, 661–670 (1995).
Hammer, N. D. et al. The C-terminal repeating units of CsgB direct bacterial functional amyloid nucleation. J. Mol. Biol. 422, 376–389 (2012).
Robinson, L. S., Ashman, E. M., Hultgren, S. J. & Chapman, M. R. Secretion of curli fibre subunits is mediated by the outer membrane-localized CsgG protein. Mol. Microbiol. 59, 870–881 (2006).
Wang, X., Smith, D. R., Jones, J. W. & Chapman, M. R. In vitro polymerization of a functional Escherichia coli amyloid protein. J. Biol. Chem. 282, 3713–3719 (2007).
Goyal, P. et al. Structural and mechanistic insights into the bacterial amyloid secretion channel CsgG. Nature 516, 250–253 (2014). This study provides the first structure of the translocation channel in the curli system.
Takagi, F., Koga, N. & Takada, S. How protein thermodynamics and folding mechanisms are altered by the chaperonin cage: molecular simulations. Proc. Natl Acad. Sci. USA 100, 11367–11372 (2003).
Brinker, A. et al. Dual function of protein confinement in chaperonin-assisted protein folding. Cell 107, 223–233 (2001).
Nenninger, A. A., Robinson, L. S. & Hultgren, S. J. Localized and efficient curli nucleation requires the chaperone-like amyloid assembly protein CsgF. Proc. Natl Acad. Sci. USA 106, 900–905 (2009).
Hodgkinson, J. L. et al. Three-dimensional reconstruction of the Shigella T3SS transmembrane regions reveals 12-fold symmetry and novel features throughout. Nature Struct. Mol. Biol. 16, 477–485 (2009).
Leake, M. C. et al. Stoichiometry and turnover in single, functioning membrane protein complexes. Nature 443, 355–358 (2006).
Knowles, T. J., Scott-Tucker, A., Overduin, M. & Henderson, I. R. Membrane protein architects: the role of the BAM complex in outer membrane protein assembly. Nature Rev. Microbiol. 7, 206–214 (2009).
Palomino, C., Marin, E. & Fernandez, L. A. The fimbrial usher FimD follows the SurA–BamB pathway for its assembly in the outer membrane of Escherichia coli. J. Bacteriol. 193, 5222–5230 (2011).
Chen, J., Lu, G., Lin, J., Davidson, A. L. & Quiocho, F. A. A tweezers-like motion of the ATP-binding cassette dimer in an ABC transport cycle. Mol. Cell 12, 651–661 (2003).
Yamagata, A. & Tainer, J. A. Hexameric structures of the archaeal secretion ATPase GspE and implications for a universal secretion mechanism. EMBO J. 26, 878–890 (2007).
Lu, C., Korotkov, K. V. & Hol, W. G. Crystal structure of the full-length ATPase GspE from the Vibrio vulnificus type II secretion system in complex with the cytoplasmic domain of GspL. J. Struct. Biol. 187, 223–235 (2014).
Zarivach, R., Vuckovic, M., Deng, W., Finlay, B. B. & Strynadka, N. C. Structural analysis of a prototypical ATPase from the type III secretion system. Nature Struct. Mol. Biol. 14, 131–137 (2007).
Yeo, H. J., Savvides, S. N., Herr, A. B., Lanka, E. & Waksman, G. Crystal structure of the hexameric traffic ATPase of the Helicobacter pylori type IV secretion system. Mol. Cell 6, 1461–1472 (2000).
Steadman, D., Lo, A., Waksman, G. & Remaut, H. Bacterial surface appendages as targets for novel antibacterial therapeutics. Future Microbiol. 9, 887–900 (2014).
Ruer, S., Pinotsis, N., Steadman, D., Waksman, G. & Remaut, H. Virulence-targeted antibacterials: concept, promise, and susceptibility to resistance mechanisms. Chem. Biol. Drug Des. http://dx.doi.org/10.1111/cbdd.12517 (2015).
Houben, E. N., Korotkov, K. V. & Bitter, W. Take five — type VII secretion systems of Mycobacteria. Biochim. Biophys. Acta 1843, 1707–1716 (2014).
Stanley, S. A., Raghavan, S., Hwang, W. W. & Cox, J. S. Acute infection and macrophage subversion by Mycobacterium tuberculosis require a specialized secretion system. Proc. Natl Acad. Sci. USA 100, 13001–13006 (2003).
Solomonson, M. et al. Structure of the mycosin-1 protease from the mycobacterial ESX-1 protein type VII secretion system. J. Biol. Chem. 288, 17782–17790 (2013).
Korotkova, N. et al. Structure of the Mycobacterium tuberculosis type VII secretion system chaperone EspG5 in complex with PE25–PPE41 dimer. Mol. Microbiol. 94, 367–384 (2014).
Cegelski, L. et al. Small-molecule inhibitors target Escherichia coli amyloid biogenesis and biofilm formation. Nature Chem. Biol. 5, 913–919 (2009).
Duncan, M. C., Linington, R. G. & Auerbuch, V. Chemical inhibitors of the type three secretion system: disarming bacterial pathogens. Antimicrob. Agents Chemother. 56, 5433–5441 (2012).
Paschos, A. et al. An in vivo high-throughput screening approach targeting the type IV secretion system component VirB8 identified inhibitors of Brucella abortus 2308 proliferation. Infect. Immun. 79, 1033–1043 (2011).
Brinton, C. C. Jr. Non-flagellar appendages of bacteria. Nature 183, 782–786 (1959).
d'Enfert, C., Ryter, A. & Pugsley, A. P. Cloning and expression in Escherichia coli of the Klebsiella pneumoniae genes for production, surface localization and secretion of the lipoprotein pullulanase. EMBO J. 6, 3531–3538 (1987).
Galán, J. E. & Curtiss, R. Cloning and molecular characterization of genes whose products allow Salmonella typhimurium to penetrate tissue culture cells. Proc. Natl Acad. Sci. USA 86, 6383–6387 (1989).
Koronakis, V., Sharff, A., Koronakis, E., Luisi, B. & Hughes, C. Crystal structure of the bacterial membrane protein TolC central to multidrug efflux and protein export. Nature 405, 914–919 (2000).
Kuldau, G. A., De Vos, G., Owen, J., McCaffrey, G. & Zambryski, P. The virB operon of Agrobacterium tumefaciens pTiC58 encodes 11 open reading frames. Mol. Gen. Genet. 221, 256–266 (1990).
Ma, D. et al. Molecular cloning and characterization of acrA and acrE genes of Escherichia coli. J. Bacteriol. 175, 6299–6313 (1993).
Welch, R. A., Dellinger, E. P., Minshew, B. & Falkow, S. Haemolysin contributes to virulence of extra-intestinal E. coli infections. Nature 294, 665–667 (1981).
Pohlner, J., Halter, R., Beyreuther, K. & Meyer, T. F. Gene structure and extracellular secretion of Neisseria gonorrhoeae IgA protease. Nature 325, 458–462 (1987).
Korotkov, K. V. & Hol, W. G. Structure of the GspK–GspI–GspJ complex from the enterotoxigenic Escherichia coli type 2 secretion system. Nature Struct. Mol. Biol. 15, 462–468 (2008).
Lederberg, J. & Tatum, E. L. Gene recombination in Escherichia coli. Nature 158, 558 (1946).
Acknowledgements
The authors thank T. Marlovits for providing an EM map with a longer T3SS needle and W. G. J. Hol for the constructed dodecameric ring of the GspD cytoplasmic domains. This work was funded by Wellcome Trust grant 098302 and ERC grant 321630 to G.W. The authors apologize for the omission of some studies owing to space constraints.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Pili
-
Long appendages that are found on the surface of Gram-negative bacteria and are involved in bacterial attachment, motility and inter-bacterial communication.
- Curli
-
Extracellular amyloid-like protein fibres produced by some bacteria, which are involved in adhesion, biofilm formation and surface colonization.
- SecYEG translocon
-
Evolutionarily conserved machinery that transports cytosolic proteins through the cytoplasmic membrane of bacteria and archaea and through the membrane of the endoplasmic reticulum in eukaryotic cells.
- Tat system
-
(Twin-arginine-translocation system). A system that transports fully folded protein substrates across the cytoplasmic membrane of bacteria and archaea, and the thylakoid membrane of plant chloroplasts.
- ATP-binding cassette transporter
-
(ABC transporter). A member of a ubiquitous superfamily of membrane-bound pumps present in all prokaryotes, fungi, plants, yeast and animals. Directional substrate transport across a membrane bilayer is achieved by an ATP-dependent flipping mechanism from an inward- to an outward-facing conformation.
- Pseudopilus
-
A short pilus-like periplasmic structure in the type II secretion system (composed of the GspG, GspH, GspI, GspJ and GspK pseudopilins) that is involved in substrate extrusion through the outer membrane channel.
- Type IV pilus
-
(T4P). A surface appendage used by many pathogenic bacteria for surface motility, biofilm formation and adhesion at the initial stages of infection. T4Ps enable directional crawling (twitching motility) by cycles of repeated extension-adhesion and retraction-release movements driven by ATPases.
- Pilotins
-
Outer membrane lipoproteins that are involved in secretin oligomerization, insertion and correct localization in the bacterial outer membrane.
- Bitopic
-
A transmembrane protein that contains only one transmembrane segment.
- Polytopic
-
A transmembrane protein that crosses the lipid bilayer twice or more.
- Solid-state nuclear magnetic resonance spectroscopy
-
(Solid-state NMR spectroscopy). A powerful atomic-scale characterization technique applicable to systems that cannot be investigated by either solution NMR or X-ray crystallography, ranging from non-crystalline or poorly crystalline assemblies to protein aggregates or fibrils.
- Rosetta modelling
-
A unified software package for computational protein modelling and functional design.
- Conjugative plasmids
-
Self-transmissible plasmids that possess all of the necessary genes for their own mobilization by conjugation.
- ClpV recognition motif
-
A motif present in the amino-terminal domain of TssC that recruits the ATPase ClpV.
- Translocation and assembly module
-
(TAM). A nanomachine composed of two proteins (the outer membrane protein TamA and the inner membrane TamB subunit), which promotes efficient secretion of autotransporters in Proteobacteria.
- Donor strand exchange
-
(DSE). Chaperone-assisted mechanism of type I pilus assembly in which the stabilizing β-strand that is donated by the chaperone is replaced by the amino-terminal extension of the subunit that is next in assembly.
- Amyloids
-
A class of thread-like protein aggregates that self-assemble into insoluble toxic nanofibres. In bacteria, the accumulation of such fibres promotes the formation of a protective biofilm, whereas in humans they are involved in neurodegenerative diseases.
- Entropy gradient
-
An energy gradient produced between two compartments when, in one compartment, the conformational entropy of a peptide or protein is reduced (usually by confinement) while in the other compartment it is not.
- Chaperonins
-
A family of ATP-driven molecular chaperones that form large multisubunit machines to promote protein folding.
Rights and permissions
About this article
Cite this article
Costa, T., Felisberto-Rodrigues, C., Meir, A. et al. Secretion systems in Gram-negative bacteria: structural and mechanistic insights. Nat Rev Microbiol 13, 343–359 (2015). https://doi.org/10.1038/nrmicro3456
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro3456
- Springer Nature Limited
This article is cited by
-
Genomic profiling and characteristics of a C1 degrading heterotrophic fresh-water bacterium Paracoccus sp. strain DMF
Archives of Microbiology (2024)
-
Function of Burkholderia pseudomallei RpoS and RpoN2 in bacterial invasion, intracellular survival, and multinucleated giant cell formation in mouse macrophage cell line
Antonie van Leeuwenhoek (2024)
-
Function and molecular mechanism analysis of CaLasSDE460 effector involved in the pathogenesis of “Candidatus Liberibacter asiaticus” in citrus
Molecular Horticulture (2023)
-
Plant commensal type VII secretion system causes iron leakage from roots to promote colonization
Nature Microbiology (2023)
-
Cytoplasmic contractile injection systems mediate cell death in Streptomyces
Nature Microbiology (2023)