Key Points
-
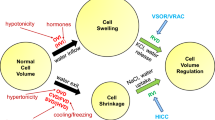
Cells need to adjust their volume in response to external osmotic stress, but also during the execution of cellular functions. These adjustments include changes in metabolism, transepithelial transport, cell division, growth, migration and programmed cell death.
-
Cell volume regulation uses the generation of osmotic gradients across the plasma membrane. These gradients drive water through the membrane, which is facilitated by specialized water channels.
-
Short-term volume regulation depends on plasma membrane channels or transporters that accumulate or release cellular osmolytes — mainly potassium, sodium and chloride, and organic osmolytes such as taurine, glutamate and inositol — in response to cell shrinkage and swelling, respectively. The underlying volume sensors and signalling cascades are complex and generally remain poorly understood.
-
Most volume-regulatory plasma membrane transporters have additional important cellular and organismal functions, linking cell volume to processes such as regulation of cytoplasmic pH, transepithelial transport and the release of signalling molecules.
-
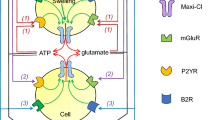
Key players in cell volume regulation are the volume-regulated anion channels (VRACs), which have only recently been discovered to be composed of LRRC8 heteromers. Depending on the particular subunit composition, VRACs not only transport chloride, but also organic osmolytes and even clinically important anticancer drugs, and they have a role in apoptosis.
-
VRAC-mediated release of taurine, glutamate and other metabolites may activate neurotransmitter receptors in the nervous system, suggesting a role for VRACs in astrocyte–neuron communication, systemic volume regulation and pathologies such as stroke.
Abstract
Cells need to regulate their volume to counteract osmotic swelling or shrinkage, as well as during cell division, growth, migration and cell death. Mammalian cells adjust their volume by transporting potassium, sodium, chloride and small organic osmolytes using plasma membrane channels and transporters. This generates osmotic gradients, which drive water in and out of cells. Key players in this process are volume-regulated anion channels (VRACs), the composition of which has recently been identified and shown to encompass LRRC8 heteromers. VRACs also transport metabolites and drugs and function in extracellular signal transduction, apoptosis and anticancer drug resistance.



Similar content being viewed by others
References
Ginzberg, M. B., Kafri, R. & Kirschner, M. On being the right (cell) size. Science 348, 1245075 (2015).
Hoffmann, E. K., Lambert, I. H. & Pedersen, S. F. Physiology of cell volume regulation in vertebrates. Physiol. Rev. 89, 193–277 (2009). This is an exhaustive review of the mechanisms involved in volume regulation and their physiological importance.
Lang, F. et al. Functional significance of cell volume regulatory mechanisms. Physiol. Rev. 78, 247–306 (1998).
Koivusalo, M., Kapus, A. & Grinstein, S. Sensors, transducers, and effectors that regulate cell size and shape. J. Biol. Chem. 284, 6595–6599 (2009).
Okada, Y. Volume expansion-sensing outward-rectifier Cl− channel: fresh start to the molecular identity and volume sensor. Am. J. Physiol. 273, C755–C789 (1997).
Groulx, N., Boudreault, F., Orlov, S. N. & Grygorczyk, R. Membrane reserves and hypotonic cell swelling. J. Membr. Biol. 214, 43–56 (2006).
Nilius, B. et al. Properties of volume-regulated anion channels in mammalian cells. Prog. Biophys. Mol. Biol. 68, 69–119 (1997). This is an excellent, detailed review on the properties of VRACs.
Pedersen, S. F., Klausen, T. K. & Nilius, B. The identification of VRAC (Volume Regulated Anion Channel): an amazing odyssey. Acta Physiol. 213, 868–881 (2015).
Voss, F. K. et al. Identification of LRRC8 heteromers as an essential component of the volume-regulated anion channel VRAC. Science 344, 634–638 (2014). This study shows that LRRC8 heteromers, composed of LRRC8A and at least one other LRRC8 isoform, are indispensable for swelling-activated VRAC currents and taurine efflux, and that subunit composition determines I Cl,vol inactivation.
Qiu, Z. et al. SWELL1, a plasma membrane protein, is an essential component of volume-regulated anion channel. Cell 157, 447–458 (2014). This study identifies LRRC8A (renamed SWELL1) as a crucial VRAC component and shows, using knockdown experiments, that it is important for volume regulation and taurine fluxes.
Planells-Cases, R. et al. Subunit composition of VRAC channels determines substrate specificity and cellular resistance to Pt-based anti-cancer drugs. EMBO J. 34, 2993–3008 (2015). This paper reports, for the first time, that the selectivity of a VRAC depends on its LRRC8 subunit composition (thereby indicating that LRRC8 proteins form its pore) and demonstrates that VRACs have a dual role in cellular drug resistance by mediating cisplatin uptake and facilitating apoptosis.
Nunes, P. et al. Ionic imbalance, in addition to molecular crowding, abates cytoskeletal dynamics and vesicle motility during hypertonic stress. Proc. Natl Acad. Sci. USA 112, E3104–E3113 (2015).
Verkman, A. S. Aquaporins at a glance. J. Cell Sci. 124, 2107–2112 (2011).
Gomes, D. et al. Aquaporins are multifunctional water and solute transporters highly divergent in living organisms. Biochim. Biophys. Acta 1788, 1213–1228 (2009).
Bourque, C. W. Central mechanisms of osmosensation and systemic osmoregulation. Nat. Rev. Neurosci. 9, 519–531 (2008).
Miley, H. E., Sheader, E. A., Brown, P. D. & Best, L. Glucose-induced swelling in rat pancreatic β-cells. J. Physiol. 504, 191–198 (1997).
Schliess, F. & Häussinger, D. Osmosensing and signaling in the regulation of liver function. Contrib. Nephrol. 152, 198–209 (2006).
Best, L., Brown, P. D., Sener, A. & Malaisse, W. J. Electrical activity in pancreatic islet cells: the VRAC hypothesis. Islets 2, 59–64 (2010).
Schwab, A., Fabian, A., Hanley, P. J. & Stock, C. Role of ion channels and transporters in cell migration. Physiol. Rev. 92, 1865–1913 (2012).
Stroka, K. M. et al. Water permeation drives tumor cell migration in confined microenvironments. Cell 157, 611–623 (2014). These experiments with cells migrating in narrow channels, together with mathematical modelling, show that osmolarity-driven asymmetric water transport can support cell migration in an actin- and myosin-independent manner.
Vom Dahl, S., Hallbrucker, C., Lang, F., Gerok, W. & Häussinger, D. Regulation of liver cell volume and proteolysis by glucagon and insulin. Biochem. J. 278, 771–777 (1991).
Maeno, E., Ishizaki, Y., Kanaseki, T., Hazama, A. & Okada, Y. Normotonic cell shrinkage because of disordered volume regulation is an early prerequisite to apoptosis. Proc. Natl Acad. Sci. USA 97, 9487–9492 (2000). This paper shows that AVD precedes caspase induction and other features of apoptosis, and that both AVD and apoptosis can be suppressed by pharmacological inhibitors of Cl− and K+ channels.
Okada, Y. et al. Volume-sensitive chloride channels involved in apoptotic volume decrease and cell death. J. Membr. Biol. 209, 21–29 (2006).
Lang, F. & Hoffmann, E. K. Role of ion transport in control of apoptotic cell death. Compr. Physiol. 2, 2037–2061 (2012).
Hortelano, S., Zeini, M., Castrillo, A., Alvarez, A. M. & Boscá, L. Induction of apoptosis by nitric oxide in macrophages is independent of apoptotic volume decrease. Cell Death Differ. 9, 643–650 (2002).
Wehner, F., Shimizu, T., Sabirov, R. & Okada, Y. Hypertonic activation of a non-selective cation conductance in HeLa cells and its contribution to cell volume regulation. FEBS Lett. 551, 20–24 (2003).
Bortner, C. D. & Cidlowski, J. A. Cell shrinkage and monovalent cation fluxes: role in apoptosis. Arch. Biochem. Biophys. 462, 176–188 (2007).
Poulsen, K. A. et al. Deregulation of apoptotic volume decrease and ionic movements in multidrug-resistant tumor cells: role of chloride channels. Am. J. Physiol. Cell Physiol. 298, C14–C25 (2010).
Lambert, I. H., Kristensen, D. M., Holm, J. B. & Mortensen, O. H. Physiological role of taurine — from organism to organelle. Acta Physiol. 213, 191–212 (2015).
Burg, M. B., Ferraris, J. D. & Dmitrieva, N. I. Cellular response to hyperosmotic stresses. Physiol. Rev. 87, 1441–1474 (2007).
Wehner, F. et al. Hypertonicity-induced cation channels. Acta Physiol. 187, 21–25 (2006).
Numata, T. et al. The ΔC splice-variant of TRPM2 is the hypertonicity-induced cation channel in HeLa cells, and the ecto-enzyme CD38 mediates its activation. J. Physiol. 590, 1121–1138 (2012).
Alessi, D. R. et al. The WNK-SPAK/OSR1 pathway: master regulator of cation-chloride cotransporters. Sci. Signal. 7, re3 (2014).
Alexander, R. T. & Grinstein, S. Na+/H+ exchangers and the regulation of volume. Acta Physiol. 187, 159–167 (2006).
Rotin, D. & Grinstein, S. Impaired cell volume regulation in Na+-H+ exchange-deficient mutants. Am. J. Physiol. 257, C1158–C1165 (1989).
Wakabayashi, S., Hisamitsu, T., Pang, T. & Shigekawa, M. Kinetic dissection of two distinct proton binding sites in Na+/H+ exchangers by measurement of reverse mode reaction. J. Biol. Chem. 278, 43580–43585 (2003).
Dunham, P. B., Kelley, S. J. & Logue, P. J. Extracellular Na+ inhibits Na+/H+ exchange: cell shrinkage reduces the inhibition. Am. J. Physiol. Cell Physiol. 287, C336–C344 (2004).
Stewart, A. K., Kurschat, C. E., Vaughan-Jones, R. D. & Alper, S. L. Putative re-entrant loop 1 of AE2 transmembrane domain has a major role in acute regulation of anion exchange by pH. J. Biol. Chem. 284, 6126–6139 (2009).
Hübner, C. A. et al. Disruption of KCC2 reveals an essential role of K-Cl cotransport already in early synaptic inhibition. Neuron 30, 515–524 (2001).
Mount, D. B. et al. Cloning and characterization of KCC3 and KCC4, new members of the cation-chloride cotransporter gene family. J. Biol. Chem. 274, 16355–16362 (1999).
Mercado, A., Broumand, V., Zandi-Nejad, K., Enck, A. H. & Mount, D. B. A C-terminal domain in KCC2 confers constitutive K+-Cl− cotransport. J. Biol. Chem. 281, 1016–1026 (2006).
Dunham, P. B., Stewart, G. W. & Ellory, J. C. Chloride-activated passive potassium transport in human erythrocytes. Proc. Natl Acad. Sci. USA 77, 1711–1715 (1980).
Rust, M. B. et al. Disruption of erythroid K-Cl cotransporters alters erythrocyte volume and partially rescues erythrocyte dehydration in SAD mice. J. Clin. Invest. 117, 1708–1717 (2007). Using mice with disrupted Kcc1 and Kcc3 genes, this work shows that KCC3 is more important than KCC1 in erythrocyte volume regulation, and that erythrocytes have a larger volume when both isoforms are eliminated.
Brown, F. C. et al. Activation of the erythroid K-Cl cotransporter Kcc1 enhances sickle cell disease pathology in a humanized mouse model. Blood 126, 1683–1670 (2015). An activating mutation in the gene encoding the K+-Cl cotransporter KCC1 identified in a mouse random mutagenesis study is shown here to constitutively decrease erythrocyte volume in mice.
Boettger, T. et al. Loss of K-Cl co-transporter KCC3 causes deafness, neurodegeneration and reduced seizure threshold. EMBO J. 22, 5422–5434 (2003).
Hoffmann, E. K., Holm, N. B. & Lambert, I. H. Functions of volume-sensitive and calcium-activated chloride channels. IUBMB Life 66, 257–267 (2014).
Milenkovic, A. et al. Bestrophin 1 is indispensable for volume regulation in human retinal pigment epithelium cells. Proc. Natl Acad. Sci. USA 112, E2630–E2639 (2015). Using ATP-free intracellular solutions that suppress VRAC currents, these authors suggest that the Ca2+-activated Cl− channel bestrophin 1 is crucial for volume regulation in retinal pigment epithelial cells.
Juul, C. A. et al. Anoctamin 6 differs from VRAC and VSOAC but is involved in apoptosis and supports volume regulation in the presence of Ca2+. Pflügers Arch. 466, 1899–1910 (2014).
Hartzell, H. C., Qu, Z., Yu, K., Xiao, Q. & Chien, L. T. Molecular physiology of bestrophins: multifunctional membrane proteins linked to best disease and other retinopathies. Physiol. Rev. 88, 639–672 (2008).
Pedemonte, N. & Galietta, L. J. Structure and function of TMEM16 proteins (anoctamins). Physiol. Rev. 94, 419–459 (2014).
Stotz, S. C. & Clapham, D. E. Anion-sensitive fluorophore identifies the Drosophila swell-activated chloride channel in a genome-wide RNA interference screen. PLoS ONE 7, e46865 (2012). This paper reports a genome-wide siRNA screen in D. melanogaster cells, which are devoid of canonical VRAC (LRRC8 heteromers), and identifies Bestrophin 1 as a swelling-activated anion channel.
Chien, L. T. & Hartzell, H. C. Rescue of volume-regulated anion current by bestrophin mutants with altered charge selectivity. J. Gen. Physiol. 132, 537–546 (2008). Using ion selectivity-changing mutants to confirm the identity of the channel, this paper demonstrates that the D. melanogaster anion channel Bestrophin 1 can be activated by cell swelling.
Fischmeister, R. & Hartzell, H. C. Volume sensitivity of the bestrophin family of chloride channels. J. Physiol. 562, 477–491 (2005).
Okada, Y. Cell volume-sensitive chloride channels: phenotypic properties and molecular identity. Contrib. Nephrol. 152, 9–24 (2006).
Grunnet, M. et al. KCNQ1 channels sense small changes in cell volume. J. Physiol. 549, 419–427 (2003).
Niemeyer, M. I., Cid, L. P., Barros, L. F. & Sepúlveda, F. V. Modulation of the two-pore domain acid-sensitive K+ channel TASK-2 (KCNK5) by changes in cell volume. J. Biol. Chem. 276, 43166–43174 (2001).
Barrière, H. et al. Role of TASK2 potassium channels regarding volume regulation in primary cultures of mouse proximal tubules. J. Gen. Physiol. 122, 177–190 (2003).
Lolicato, M., Riegelhaupt, P. M., Arrigoni, C., Clark, K. A. & Minor, D. L. Jr. Transmembrane helix straightening and buckling underlies activation of mechanosensitive and thermosensitive K2P channels. Neuron 84, 1198–1212 (2014).
Brohawn, S. G., Su, Z. & MacKinnon, R. Mechanosensitivity is mediated directly by the lipid membrane in TRAAK and TREK1 K+ channels. Proc. Natl Acad. Sci. USA 111, 3614–3619 (2014).
Patel, A. J. et al. A mammalian two pore domain mechano-gated S-like K+ channel. EMBO J. 17, 4283–4290 (1998).
Gründer, S., Thiemann, A., Pusch, M. & Jentsch, T. J. Regions involved in the opening of CIC-2 chloride channel by voltage and cell volume. Nature 360, 759–762 (1992).
Jordt, S. E. & Jentsch, T. J. Molecular dissection of gating in the ClC-2 chloride channel. EMBO J. 16, 1582–1592 (1997).
Sabirov, R. Z., Merzlyak, P. G., Islam, M. R., Okada, T. & Okada, Y. The properties, functions, and pathophysiology of maxi-anion channels. Pflügers Arch. 468, 405–420 (2016).
Thiemann, A., Gründer, S., Pusch, M. & Jentsch, T. J. A chloride channel widely expressed in epithelial and non-epithelial cells. Nature 356, 57–60 (1992).
Hoegg-Beiler, M. B. et al. Disrupting MLC1 and GlialCAM and ClC-2 interactions in leukodystrophy entails glial chloride channel dysfunction. Nat. Commun. 5, 3475 (2014).
Nehrke, K. et al. Loss of hyperpolarization-activated Cl− current in salivary acinar cells from Clcn2 knockout mice. J. Biol. Chem. 277, 23604–23611 (2002).
Cahalan, S. M. et al. Piezo1 links mechanical forces to red blood cell volume. eLife 4, e07370 (2015). This study shows that mechanical activation of the PIEZO1 channel in red blood cells leads to Ca2+ influx that activates the Ca2+-sensitive Gardos (KCa3.1) K+ channel and thereby leads to smaller erythrocyte volume.
Ranade, S. S., Syeda, R. & Patapoutian, A. Mechanically activated ion channels. Neuron 87, 1162–1179 (2015).
Sukharev, S. I., Blount, P., Martinac, B., Blattner, F. R. & Kung, C. A large-conductance mechanosensitive channel in E. coli encoded by mscL alone. Nature 368, 265–268 (1994).
Anishkin, A., Loukin, S. H., Teng, J. & Kung, C. Feeling the hidden mechanical forces in lipid bilayer is an original sense. Proc. Natl Acad. Sci. USA 111, 7898–7905 (2014).
Maingret, F., Fosset, M., Lesage, F., Lazdunski, M. & Honoré, E. TRAAK is a mammalian neuronal mechano-gated K+ channel. J. Biol. Chem. 274, 1381–1387 (1999).
Prager-Khoutorsky, M., Khoutorsky, A. & Bourque, C. W. Unique interweaved microtubule scaffold mediates osmosensory transduction via physical interaction with TRPV1. Neuron 83, 866–878 (2014). This paper shows that the TRPV1 cation channel is activated in osmosensory neurons by cell shrinkage through a pushing force of an attached microtubule scaffold.
Zhang, W. et al. Ankyrin repeats convey force to gate the NOMPC mechanotransduction channel. Cell 162, 1391–1403 (2015).
Tilly, B. C. et al. Activation of the osmo-sensitive chloride conductance involves P21rho and is accompanied by a transient reorganization of the F-actin cytoskeleton. Mol. Biol. Cell 7, 1419–1427 (1996).
Fuster, D., Moe, O. W. & Hilgemann, D. W. Lipid- and mechanosensitivities of sodium/hydrogen exchangers analyzed by electrical methods. Proc. Natl Acad. Sci. USA 101, 10482–10487 (2004).
Nasuhoglu, C. et al. Modulation of cardiac PIP2 by cardioactive hormones and other physiologically relevant interventions. Am. J. Physiol. Cell Physiol. 283, C223–C234 (2002).
Yamamoto, M. et al. Hypertonic stress increases phosphatidylinositol 4,5-bisphosphate levels by activating PIP5KIβ. J. Biol. Chem. 281, 32630–32638 (2006).
Voets, T., Droogmans, G., Raskin, G., Eggermont, J. & Nilius, B. Reduced intracellular ionic strength as the initial trigger for activation of endothelial volume-regulated anion channels. Proc. Natl Acad. Sci. USA 96, 5298–5303 (1999). This work convincingly shows that VRACs can be stimulated by reducing intracellular ionic strength, independent of cell volume changes.
Emma, F., McManus, M. & Strange, K. Intracellular electrolytes regulate the volume set point of the organic osmolyte/anion channel VSOAC. Am. J. Physiol. 272, C1766–C1775 (1997).
Syeda, R. et al. LRRC8 proteins form volume-regulated anion channels that sense ionic strength. Cell 164, 499–511 (2016). Using reconstitution of purified LRRC8 heteromers into lipid bilayers, this paper shows that LRRC8 proteins are sufficient for forming a VRAC channel, and that it can be activated under conditions of low ionic strength.
Rinehart, J. et al. Sites of regulated phosphorylation that control K-Cl cotransporter activity. Cell 138, 525–536 (2009).
de Los Heros, P. et al. The WNK-regulated SPAK/OSR1 kinases directly phosphorylate and inhibit the K+-Cl− co-transporters. Biochem. J. 458, 559–573 (2014).
Melo, Z. et al. N-terminal serine dephosphorylation is required for KCC3 cotransporter full activation by cell swelling. J. Biol. Chem. 288, 31468–31476 (2013).
Gagnon, K. B., England, R. & Delpire, E. Volume sensitivity of cation-Cl-cotransporters is modulated by the interaction of two kinases: Ste20-related proline-alanine-rich kinase and WNK4. Am. J. Physiol. Cell Physiol. 290, C134–C142 (2006).
Piala, A. T. et al. Chloride sensing by WNK1 involves inhibition of autophosphorylation. Sci. Signal. 7, ra41 (2014). This report of the crystal structure of the catalytic domain of the WNK1 kinase reveals that it directly binds to Cl−, and this binding is shown to prevent kinase activation.
Varela, D., Simon, F., Riveros, A., Jørgensen, F. & Stutzin, A. NAD(P)H oxidase-derived H2O2 signals chloride channel activation in cell volume regulation and cell proliferation. J. Biol. Chem. 279, 13301–13304 (2004).
Shimizu, T., Numata, T. & Okada, Y. A role of reactive oxygen species in apoptotic activation of volume-sensitive Cl− channel. Proc. Natl Acad. Sci. USA 101, 6770–6773 (2004). This paper shows that I Cl,vol -like currents are activated by several pro-apoptotic stimuli and provides evidence for a role of reactive oxygen species in the activation of those currents and in apoptosis.
Carton, I. et al. RhoA exerts a permissive effect on volume-regulated anion channels in vascular endothelial cells. Am. J. Physiol. Cell Physiol. 283, C115–C125 (2002).
Lepple-Wienhues, A. et al. The tyrosine kinase p56lck mediates activation of swelling-induced chloride channels in lymphocytes. J. Cell Biol. 141, 281–286 (1998).
Holm, J. B., Grygorczyk, R. & Lambert, I. H. Volume-sensitive release of organic osmolytes in the human lung epithelial cell line A549: role of the 5-lipoxygenase. Am. J. Physiol. Cell Physiol. 305, C48–C60 (2013).
Levitan, I., Christian, A. E., Tulenko, T. N. & Rothblat, G. H. Membrane cholesterol content modulates activation of volume-regulated anion current in bovine endothelial cells. J. Gen. Physiol. 115, 405–416 (2000).
Browe, D. M. & Baumgarten, C. M. EGFR kinase regulates volume-sensitive chloride current elicited by integrin stretch via PI-3K and NADPH oxidase in ventricular myocytes. J. Gen. Physiol. 127, 237–251 (2006).
Grinstein, S., Clarke, C. A., Dupre, A. & Rothstein, A. Volume-induced increase of anion permeability in human lymphocytes. J. Gen. Physiol. 80, 801–823 (1982).
Hoffmann, E. K., Simonsen, L. O. & Lambert, I. H. Volume-induced increase of K+ and Cl− permeabilities in Ehrlich ascites tumor cells. Role of internal Ca2+. J. Membr. Biol. 78, 211–222 (1984).
Hazama, A. & Okada, Y. Ca2+ sensitivity of volume-regulatory K+ and Cl− channels in cultured human epithelial cells. J. Physiol. 402, 687–702 (1988).
Okada, Y., Sato, K. & Numata, T. Pathophysiology and puzzles of the volume-sensitive outwardly rectifying anion channel. J. Physiol. 587, 2141–2149 (2009).
Jackson, P. S., Morrison, R. & Strange, K. The volume-sensitive organic osmolyte-anion channel VSOAC is regulated by nonhydrolytic ATP binding. Am. J. Physiol. 267, C1203–C1209 (1994).
Stutzin, A. et al. Separate taurine and chloride efflux pathways activated during regulatory volume decrease. Am. J. Physiol. 277, C392–C402 (1999).
Lambert, I. H. & Hoffmann, E. K. Cell swelling activates separate taurine and chloride channels in Ehrlich mouse ascites tumor cells. J. Membr. Biol. 142, 289–298 (1994).
Jackson, P. S. & Strange, K. Volume-sensitive anion channels mediate swelling-activated inositol and taurine efflux. Am. J. Physiol. 265, C1489–C1500 (1993).
Akita, T. & Okada, Y. Regulation of bradykinin-induced activation of volume-sensitive outwardly rectifying anion channels by Ca2+ nanodomains in mouse astrocytes. J. Physiol. 589, 3909–3927 (2011).
Mongin, A. A. & Kimelberg, H. K. ATP potently modulates anion channel-mediated excitatory amino acid release from cultured astrocytes. Am. J. Physiol. Cell Physiol. 283, C569–C578 (2002).
Deng, W., Baki, L. & Baumgarten, C. M. Endothelin signalling regulates volume-sensitive Cl− current via NADPH oxidase and mitochondrial reactive oxygen species. Cardiovasc. Res. 88, 93–100 (2010).
Nilius, B. et al. Volume-activated Cl− currents in different mammalian non-excitable cell types. Pflügers Arch. 428, 364–371 (1994). This paper suggests that VRACs are widely, and maybe ubiquitously, expressed in mammalian cells.
Abascal, F. & Zardoya, R. LRRC8 proteins share a common ancestor with pannexins, and may form hexameric channels involved in cell-cell communication. Bioessays 34, 551–560 (2012). This is a bioinformatic analysis showing that LRRC8 proteins are homologous to pannexins and suggesting that LRRC8 proteins form hexameric ion channels.
Lee, C. C., Freinkman, E., Sabatini, D. M. & Ploegh, H. L. The protein synthesis inhibitor blasticidin S enters mammalian cells via leucine-rich repeat-containing protein 8D. J. Biol. Chem. 289, 17124–17131 (2014). This work shows that the cellular uptake of blasticidin depends on LRRC8D , a gene that was previously identified in a genome-wide reporter screen for genes involved in NF-κB activation that used blasticidin as trigger.
Kubota, K. et al. LRRC8 involved in B cell development belongs to a novel family of leucine-rich repeat proteins. FEBS Lett. 564, 147–152 (2004).
Bond, S. R. & Naus, C. C. The pannexins: past and present. Front. Physiol. 5, 58 (2014).
Shennan, D. B. Swelling-induced taurine transport: relationship with chloride channels, anion-exchangers and other swelling-activated transport pathways. Cell Physiol. Biochem. 21, 15–28 (2008).
Min, X. J. et al. Dysfunction of volume-sensitive chloride channels contributes to cisplatin resistance in human lung adenocarcinoma cells. Exp. Biol. Med. 236, 483–491 (2011).
Ise, T. et al. Roles of volume-sensitive Cl− channel in cisplatin-induced apoptosis in human epidermoid cancer cells. J. Membr. Biol. 205, 139–145 (2005).
Sørensen, B. H., Thorsteinsdottir, U. A. & Lambert, I. H. Acquired cisplatin resistance in human ovarian A2780 cancer cells correlates with shift in taurine homeostasis and ability to volume regulate. Am. J. Physiol. Cell Physiol. 307, C1071–C1080 (2014).
Lee, E. L. et al. Impaired activity of volume-sensitive Cl− channel is involved in cisplatin resistance of cancer cells. J. Cell. Physiol. 211, 513–521 (2007). In this work, sensitivity to cisplatin and VRAC currents are restored in parallel in a drug-resistant cell line that displays small VRAC currents by nonspecifically changing gene expression with a histone deacetylate inhibitor.
Hisadome, K. et al. Volume-regulated anion channels serve as an auto/paracrine nucleotide release pathway in aortic endothelial cells. J. Gen. Physiol. 119, 511–520 (2002).
Hyzinski- García, M. C., Rudkouskaya, A. & Mongin, A. A. LRRC8A protein is indispensable for swelling-activated and ATP-induced release of excitatory amino acids in rat astrocytes. J. Physiol. 592, 4855–4862 (2014).
Model, M. A. Possible causes of apoptotic volume decrease: an attempt at quantitative review. Am. J. Physiol. Cell Physiol. 306, C417–C424 (2014).
Inoue, H. & Okada, Y. Roles of volume-sensitive chloride channel in excitotoxic neuronal injury. J. Neurosci. 27, 1445–1455 (2007).
Lai, T. W., Zhang, S. & Wang, Y. T. Excitotoxicity and stroke: identifying novel targets for neuroprotection. Prog. Neurobiol. 115, 157–188 (2014).
Kimelberg, H. K., Goderie, S. K., Higman, S., Pang, S. & Waniewski, R. A. Swelling-induced release of glutamate, aspartate, and taurine from astrocyte cultures. J. Neurosci. 10, 1583–1591 (1990).
Pasantes-Morales, H., Murray, R. A., Sánchez-Olea, R. & Morán, J. Regulatory volume decrease in cultured astrocytes. II. Permeability pathway to amino acids and polyols. Am. J. Physiol. 266, C172–C178 (1994).
Feustel, P. J., Jin, Y. & Kimelberg, H. K. Volume-regulated anion channels are the predominant contributors to release of excitatory amino acids in the ischemic cortical penumbra. Stroke 35, 1164–1168 (2004).
Mongin, A. A. Volume-regulated anion channel-a frenemy within the brain. Pflügers Arch. 468, 421–441 (2015).
Haskew-Layton, R. E., Mongin, A. A. & Kimelberg, H. K. Hydrogen peroxide potentiates volume-sensitive excitatory amino acid release via a mechanism involving Ca2+/calmodulin-dependent protein kinase II. J. Biol. Chem. 280, 3548–3554 (2005).
Phillis, J. W., Song, D. & O'Regan, M. H. Inhibition by anion channel blockers of ischemia-evoked release of excitotoxic and other amino acids from rat cerebral cortex. Brain Res. 758, 9–16 (1997).
Zhang, Y., Zhang, H., Feustel, P. J. & Kimelberg, H. K. DCPIB, a specific inhibitor of volume regulated anion channels (VRACs), reduces infarct size in MCAo and the release of glutamate in the ischemic cortical penumbra. Exp. Neurol. 210, 514–520 (2008). This is one of several studies showing that anion channel inhibitors reduce cerebral infarct size, now using a somewhat more specific VRAC inhibitor that is applied intracisternally.
Inoue, H., Ohtaki, H., Nakamachi, T., Shioda, S. & Okada, Y. Anion channel blockers attenuate delayed neuronal cell death induced by transient forebrain ischemia. J. Neurosci. Res. 85, 1427–1435 (2007).
Hussy, N., Deleuze, C., Pantaloni, A., Desarmenien, M. G. & Moos, F. Agonist action of taurine on glycine receptors in rat supraoptic magnocellular neurones: possible role in osmoregulation. J. Physiol. 502, 609–621 (1997). This seminal paper supports the notion that hypotonicity-induced release of taurine from astrocytes activates glycine receptors on nearby osmosensing neurons and suggests that this plays an important part in systemic volume regulation.
Le-Corronc, H., Rigo, J. M., Branchereau, P. & Legendre, P. GABAA receptor and glycine receptor activation by paracrine/autocrine release of endogenous agonists: more than a simple communication pathway. Mol. Neurobiol. 44, 28–52 (2011).
Hussy, N., Deleuze, C., Desarmenien, M. G. & Moos, F. C. Osmotic regulation of neuronal activity: a new role for taurine and glial cells in a hypothalamic neuroendocrine structure. Prog. Neurobiol. 62, 113–134 (2000).
Choe, K. Y., Olson, J. E. & Bourque, C. W. Taurine release by astrocytes modulates osmosensitive glycine receptor tone and excitability in the adult supraoptic nucleus. J. Neurosci. 32, 12518–12527 (2012).
Prager-Khoutorsky, M. & Bourque, C. W. Mechanical basis of osmosensory transduction in magnocellular neurosecretory neurones of the rat supraoptic nucleus. J. Neuroendocrinol. 27, 507–515 (2015).
Zhang, Z. & Bourque, C. W. Osmometry in osmosensory neurons. Nat. Neurosci. 6, 1021–1022 (2003). This work suggests that osmosensory neurons from the SONfunction as osmometers, because they apparently lack both RVD and RVI.
Sato, K., Numata, T., Saito, T., Ueta, Y. & Okada, Y. V2 receptor-mediated autocrine role of somatodendritic release of AVP in rat vasopressin neurons under hypo-osmotic conditions. Sci. Signal. 4, ra5 (2011). Investigating RVD and I Cl,vol on longer time scales, this work contradicts reference 132 by showing that these neurons slowly activate VRACs and RVD.
Kumar, L. et al. Leucine-rich repeat containing 8A (LRRC8A) is essential for T lymphocyte development and function. J. Exp. Med. 211, 929–942 (2014). Although based on the wrong assumption that LRRC8A is a receptor, this first description of the severe phenotype of constitutive Lrrc8a -knockout mice highlights the importance of VRACs in many tissues.
Sawada, A. et al. A congenital mutation of the novel gene LRRC8 causes agammaglobulinemia in humans. J. Clin. Invest. 112, 1707–1713 (2003).
Krane, C. M. et al. Salivary acinar cells from aquaporin 5-deficient mice have decreased membrane water permeability and altered cell volume regulation. J. Biol. Chem. 276, 23413–23420 (2001).
Zeuthen, T. Water-transporting proteins. J. Membr. Biol. 234, 57–73 (2010).
Nilius, B. Is the volume-regulated anion channel VRAC a “water-permeable” channel? Neurochem. Res. 29, 3–8 (2004).
Klein, M., Seeger, P., Schuricht, B., Alper, S. L. & Schwab, A. Polarization of Na+/H+ and Cl−/HCO3− exchangers in migrating renal epithelial cells. J. Gen. Physiol. 115, 599–608 (2000).
Denker, S. P. & Barber, D. L. Cell migration requires both ion translocation and cytoskeletal anchoring by the Na-H exchanger NHE1. J. Cell Biol. 159, 1087–1096 (2002).
Saadoun, S., Papadopoulos, M. C., Hara-Chikuma, M. & Verkman, A. S. Impairment of angiogenesis and cell migration by targeted aquaporin-1 gene disruption. Nature 434, 786–792 (2005).
Papadopoulos, M. C., Saadoun, S. & Verkman, A. S. Aquaporins and cell migration. Pflügers Arch. 456, 693–700 (2008).
Huang, X. et al. EAG2 potassium channel with evolutionarily conserved function as a brain tumor target. Nat. Neurosci. 18, 1236–1246 (2015).
Schneider, L. et al. H-ras transformation sensitizes volume-activated anion channels and increases migratory activity of NIH3T3 fibroblasts. Pflügers Arch. 455, 1055–1062 (2008).
Charras, G. T., Yarrow, J. C., Horton, M. A., Mahadevan, L. & Mitchison, T. J. Non-equilibration of hydrostatic pressure in blebbing cells. Nature 435, 365–369 (2005).
Orlov, S. N., Platonova, A. A., Hamet, P. & Grygorczyk, R. Cell volume and monovalent ion transporters: their role in cell death machinery triggering and progression. Am. J. Physiol. Cell Physiol. 305, C361–C372 (2013).
McGlasson, L., Best, L. & Brown, P. D. The glucokinase activator GKA50 causes an increase in cell volume and activation of volume-regulated anion channels in rat pancreatic β-cells. Mol. Cell. Endocrinol. 342, 48–53 (2011).
Catalán, M. A. et al. A fluid secretion pathway unmasked by acinar-specific Tmem16A gene ablation in the adult mouse salivary gland. Proc. Natl Acad. Sci. USA 112, 2263–2268 (2015).
Acknowledgements
The author thanks Rosa Planells-Cases for help with figures, and members of his laboratory for critical reading of the manuscript. Work on VRACs is supported in the author's laboratory by the European Research Council Advanced Grant (FP/2007-2013) 294435 'Cytovolion' and the Deutsche Forschungsgemeinschaft (Exc 257 'Neurocure' and JE164/12).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Supplementary information
Supplementary information S1 (box)
Methods to measure cell volume (PDF 185 kb)
Glossary
- Osmolytes
-
Dissolved substances that contribute to osmolarity; in biology, the term mainly refers to substances playing a significant part in osmoregulation. Osmolytes can be electrically charged or neutral, as only the concentration of particles matters.
- Hypo- or hypertonic
-
Condition in which the concentration of osmolytes that are not freely membrane permeable is lower or higher, respectively, on the reference side of a membrane, compared with the other side. This concentration difference creates an osmotic pressure.
- Osmolarity
-
Concentration of solute particles per kg of solution, irrespective of their ability to freely cross a given membrane.
- Depolarization
-
A change in the resting voltage of cells (−40 to −80 mV inside versus outside) to more positive values. A change to more negative potentials is called hyperpolarization.
- Electrochemical gradients
-
The passive, diffusive transport of an ion across a membrane through a channel is driven not only by the concentration difference of that ion, but also by the voltage across that membrane. The driving force is given by the difference in electrochemical potential according to the Nernst equation, for example, for a singly charged anion A− by EA = (RT/F)*ln([A−]i / [A−]o), with R being the gas constant, T the absolute temperature, F Faraday's constant, and [A−]i the intracellular and [A−]o the extracellular concentration of the anion.
- Donnan effects
-
Effects on the voltage and concentration differences of charged particles across a semipermeable membrane, caused by the impermeability of the membrane to one ionic species.
- GABA (γ-aminobutyric acid) type A receptor
-
(GABAA receptor). A pentameric anion channel that is opened by the binding of GABA. Channels of this type mediate fast synaptic inhibition in the nervous system by allowing a passive influx of Cl− which hyperpolarizes the membrane. This requires a low intracellular Cl− concentration, as found in most adult neurons.
- Glycine receptor
-
A pentameric anion channel that inhibits neuronal excitability by mediating a hyperpolarizing Cl− influx upon extracellular binding of the amino acid and neurotransmitter glycine.
- Conductance
-
Electrical conductance is the inverse of electrical resistance. For instance, the K+ conductance of a biological membrane is a quantitative measure of the current mediated by K+ channels divided by the driving force (given by potential and concentration differences across the membrane). It depends on the number of open K+ channels and how readily they allow the passage of K+ ions.
- Outward rectification
-
A channel is called outwardly rectifying if its currents are larger at inside-positive than at inside-negative voltages, which corresponds to an outward transport of positive charge. For anion channels this corresponds to an inward transport of anions.
- Channel inactivation
-
The spontaneous closure of a channel at constant voltage.
- Astrocytes
-
A major class of glial (non-neuronal) cells in the central nervous system, which have important functions, for instance, in buffering extracellular ion concentrations, in removal of neurotransmitters and in neuronal metabolism.
Rights and permissions
About this article
Cite this article
Jentsch, T. VRACs and other ion channels and transporters in the regulation of cell volume and beyond. Nat Rev Mol Cell Biol 17, 293–307 (2016). https://doi.org/10.1038/nrm.2016.29
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm.2016.29
- Springer Nature Limited
This article is cited by
-
Structure of a volume-regulated heteromeric LRRC8A/C channel
Nature Structural & Molecular Biology (2023)
-
Structural basis for assembly and lipid-mediated gating of LRRC8A:C volume-regulated anion channels
Nature Structural & Molecular Biology (2023)
-
Disruption of ER ion homeostasis maintained by an ER anion channel CLCC1 contributes to ALS-like pathologies
Cell Research (2023)
-
QCM sensor provides insight into the role of pivotal ions in cellular regulatory volume decrease
Analytical and Bioanalytical Chemistry (2023)
-
Developmental RNA-Seq transcriptomics of haploid germ cells and spermatozoa uncovers novel pathways associated with teleost spermiogenesis
Scientific Reports (2022)