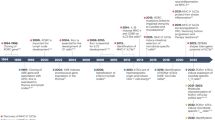
Key Points
-
Understanding the function of dendritic cells (DCs) forms the basis of the characterization of professional antigen-presenting cells (APCs). DCs recognize and respond to danger-associated and pathogen-associated signals; express all of the necessary antigen-processing machinery; localize to the T cell zones of secondary lymphoid organs; and express the necessary co-stimulatory molecules and cytokines to activate naive CD4+ T cells.
-
Almost all myeloid-derived cells — including neutrophils, mast cells, basophils and eosinophils — have been shown to upregulate MHC class II molecules in certain settings. There is evidence that these MHC class II-expressing cells may support ongoing T helper (TH2) cell responses but less compelling data that they can prime naive CD4+ T cells.
-
Innate lymphoid cells are a recently described population of cells that can express MHC class II molecules. Recent data conflict as to whether they drive pro-inflammatory or tolerogenic CD4+ T cell responses. As with many other atypical APCs, their function may be dependent on the anatomical site in which their activities are assessed and the specific assays that are used to measure their function.
-
Many epithelial, endothelial and stromal cell populations can express MHC class II. Expression of MHC class II molecules by these cells may contribute to CD4+ T cell tolerance.
-
Many studies that have explored the antigen-presenting functions of atypical APCs have been conducted in mice. A major limitation of the field is that there are few corroborative studies in humans.
Abstract
Dendritic cells, macrophages and B cells are regarded as the classical antigen-presenting cells of the immune system. However, in recent years, there has been a rapid increase in the number of cell types that are suggested to present antigens on MHC class II molecules to CD4+ T cells. In this Review, we describe the key characteristics that define an antigen-presenting cell by examining the functions of dendritic cells. We then examine the functions of the haematopoietic cells and non-haematopoietic cells that can express MHC class II molecules and that have been suggested to represent 'atypical' antigen-presenting cells. We consider whether any of these cell populations can prime naive CD4+ T cells and, if not, question the effects that they do have on the development of immune responses.


Similar content being viewed by others
References
Lemos, M. P., Fan, L., Lo, D. & Laufer, T. M. CD8α+ and CD11b+ dendritic cell-restricted MHC class II controls Th1 CD4+ T cell immunity. J. Immunol. 171, 5077–5084 (2003).
Lemos, M. P., Esquivel, F., Scott, P. & Laufer, T. M. MHC class II expression restricted to CD8α+ and CD11b+ dendritic cells is sufficient for control of Leishmania major. J. Exp. Med. 199, 725–730 (2004).
Sokol, C. L. et al. Basophils function as antigen-presenting cells for an allergen-induced T helper type 2 response. Nature Immunol. 10, 713–720 (2009).
Perrigoue, J. G. et al. MHC class II-dependent basophil-CD4+ T cell interactions promote TH2 cytokine-dependent immunity. Nature Immunol. 10, 697–705 (2009).
Gregory, G. D., Robbie-Ryan, M., Secor, V. H., Sabatino, J. J. Jr & Brown, M. A. Mast cells are required for optimal autoreactive T cell responses in a murine model of multiple sclerosis. Eur. J. Immunol. 35, 3478–3486 (2005).
Maurer, M. et al. Skin mast cells control T cell-dependent host defense in Leishmania major infections. FASEB J. 20, 2460–2467 (2006).
Frandji, P. et al. Antigen-dependent stimulation by bone marrow-derived mast cells of MHC class II-restricted T cell hybridoma. J. Immunol. 151, 6318–6328 (1993).
Fox, C. C., Jewell, S. D. & Whitacre, C. C. Rat peritoneal mast cells present antigen to a PPD-specific T cell line. Cell. Immunol. 158, 253–264 (1994).
Dimitriadou, V. et al. Expression of functional major histocompatibility complex class II molecules on HMC-1 human mast cells. J. Leukoc. Biol. 64, 791–799 (1998).
Poncet, P., Arock, M. & David, B. MHC class II-dependent activation of CD4+ T cell hybridomas by human mast cells through superantigen presentation. J. Leukoc. Biol. 66, 105–112 (1999).
Frandji, P. et al. Presentation of soluble antigens by mast cells: upregulation by interleukin-4 and granulocyte/macrophage colony-stimulating factor and downregulation by interferon-γ. Cell. Immunol. 163, 37–46 (1995).
Frandji, P. et al. Exogenous and endogenous antigens are differentially presented by mast cells to CD4+ T lymphocytes. Eur. J. Immunol. 26, 2517–2528 (1996).
Skokos, D. et al. Mast cell-derived exosomes induce phenotypic and functional maturation of dendritic cells and elicit specific immune responses in vivo. J. Immunol. 170, 3037–3045 (2003).
Tkaczyk, C. et al. In vitro and in vivo immunostimulatory potential of bone marrow-derived mast cells on B- and T-lymphocyte activation. J. Allergy Clin. Immunol. 105, 134–142 (2000).
Skokos, D. et al. Mast cell-dependent B and T lymphocyte activation is mediated by the secretion of immunologically active exosomes. J. Immunol. 166, 868–876 (2001).
Raposo, G. et al. Accumulation of major histocompatibility complex class II molecules in mast cell secretory granules and their release upon degranulation. Mol. Biol. Cell 8, 2631–2645 (1997).
Nakae, S. et al. Mast cells enhance T cell activation: importance of mast cell costimulatory molecules and secreted TNF. J. Immunol. 176, 2238–2248 (2006).
Kambayashi, T. et al. Indirect involvement of allergen-captured mast cells in antigen presentation. Blood 111, 1489–1496 (2008).
Nakano, N. et al. Notch signaling confers antigen-presenting cell functions on mast cells. J. Allergy Clin. Immunol. 123, 74–81. e1 (2009).
Kambayashi, T. et al. Inducible MHC class II expression by mast cells supports effector and regulatory T cell activation. J. Immunol. 182, 4686–4695 (2009). This study shows that MHC class II expression on mast cells is induced by TLR agonists and IFNγ, and can support the activation of effector T cells and T Reg cells but not that of naive T cells.
Razin, E. et al. Interleukin 3: A differentiation and growth factor for the mouse mast cell that contains chondroitin sulfate E proteoglycan. J. Immunol. 132, 1479–1486 (1984).
Tkaczyk, C., Villa, I., Peronet, R., David, B. & Mecheri, S. FcεRI-mediated antigen endocytosis turns interferon-gamma-treated mouse mast cells from inefficient into potent antigen-presenting cells. Immunology 97, 333–340 (1999).
Yoshimoto, T. et al. Basophils contribute to TH2-IgE responses in vivo via IL-4 production and presentation of peptide-MHC class II complexes to CD4+ T cells. Nature Immunol. 10, 706–712 (2009). References 3, 4 and 23 show that basophils express MHC class II and present antigens to T cells to promote T H 2-type responses.
Hammad, H. et al. Inflammatory dendritic cells—not basophils—are necessary and sufficient for induction of Th2 immunity to inhaled house dust mite allergen. J. Exp. Med. 207, 2097–2111 (2010). This study argues that FcεRI-expressing DCs and not basophils are responsible for antigen presentation in response to HDM allergen.
Phythian-Adams, A. T. et al. CD11c depletion severely disrupts Th2 induction and development in vivo. J. Exp. Med. 207, 2089–2096 (2010).
Gong, J. et al. The antigen presentation function of bone marrow-derived mast cells is spatiotemporally restricted to a subset expressing high levels of cell surface FcεRI and MHC II. BMC Immunol. 11, 34 (2010).
Gaudenzio, N. et al. Cell-cell cooperation at the T helper cell/mast cell immunological synapse. Blood 114, 4979–4988 (2009).
Ito, T. et al. Roles of PU.1 in monocyte- and mast cell-specific gene regulation: PU.1 transactivates CIITA pIV in cooperation with IFN-γ. Int. Immunol. 21, 803–816 (2009).
Lu, L. F. et al. Mast cells are essential intermediaries in regulatory T-cell tolerance. Nature 442, 997–1002 (2006).
Eller, K. et al. IL-9 production by regulatory T cells recruits mast cells that are essential for regulatory T cell-induced immune suppression. J Immunol. 186, 83–91 (2011).
Maurer, D. et al. Fcε receptor I on dendritic cells delivers IgE-bound multivalent antigens into a cathepsin S-dependent pathway of MHC class II presentation. J. Immunol. 161, 2731–2739 (1998).
Shin, J. S., Shelburne, C. P., Jin, C., LeFurgey, E. A. & Abraham, S. N. Harboring of particulate allergens within secretory compartments by mast cells following IgE/FcεRI-lipid raft-mediated phagocytosis. J. Immunol. 177, 5791–5800 (2006).
Tkaczyk, C. et al. Specific antigen targeting to surface IgE and IgG on mouse bone marrow-derived mast cells enhances efficiency of antigen presentation. Immunology 94, 318–324 (1998).
Dvorak, A. M., Newball, H. H., Dvorak, H. F. & Lichtenstein, L. M. Antigen-induced IgE-mediated degranulation of human basophils. Lab Invest. 43, 126–139 (1980).
Ishizaka, T. & Ishizaka, K. Immunological events at the surface of basophil granulocytes and mast cells which induce degranulation. Scand. J. Respir. Dis. Suppl. 98, 13–22 (1977).
Arinobu, Y., Iwasaki, H. & Akashi, K. Origin of basophils and mast cells. Allergol Int. 58, 21–28 (2009).
Arinobu, Y. et al. Developmental checkpoints of the basophil/mast cell lineages in adult murine hematopoiesis. Proc. Natl Acad. Sci. USA 102, 18105–18110 (2005).
Voehringer, D., Shinkai, K. & Locksley, R. M. Type 2 immunity reflects orchestrated recruitment of cells committed to IL-4 production. Immunity 20, 267–277 (2004).
Min, B. et al. Basophils produce IL-4 and accumulate in tissues after infection with a Th2-inducing parasite. J. Exp. Med. 200, 507–517 (2004).
Gessner, A., Mohrs, K. & Mohrs, M. Mast cells, basophils, and eosinophils acquire constitutive IL-4 and IL-13 transcripts during lineage differentiation that are sufficient for rapid cytokine production. J. Immunol. 174, 1063–1072 (2005).
Karasuyama, H., Mukai, K., Tsujimura, Y. & Obata, K. Newly discovered roles for basophils: a neglected minority gains new respect. Nature Rev. Immunol. 9, 9–13 (2009).
Sokol, C. L., Barton, G. M., Farr, A. G. & Medzhitov, R. A mechanism for the initiation of allergen-induced T helper type 2 responses. Nature Immunol. 9, 310–318 (2007).
Tsujimura, Y. et al. Basophils play a pivotal role in immunoglobulin-G-mediated but not immunoglobulin-E-mediated systemic anaphylaxis. Immunity 28, 581–589 (2008).
Le Gros, G., Ben-Sasson, S. Z., Seder, R., Finkelman, F. D. & Paul, W. E. Generation of interleukin 4 (IL-4)-producing cells in vivo and in vitro: IL-2 and IL-4 are required for in vitro generation of IL-4-producing cells. J. Exp. Med. 172, 921–929 (1990).
Swain, S. L., Weinberg, A. D., English, M. & Huston, G. IL-4 directs the development of Th2-like helper effectors. J. Immunol. 145, 3796–3806 (1990).
Else, K. J., Finkelman, F. D., Maliszewski, C. R. & Grencis, R. K. Cytokine-mediated regulation of chronic intestinal helminth infection. J. Exp. Med. 179, 347–351 (1994).
Cohn, L., Homer, R. J., Marinov, A., Rankin, J. & Bottomly, K. Induction of airway mucus production By T helper 2 (Th2) cells: a critical role for interleukin 4 in cell recruitment but not mucus production. J. Exp. Med. 186, 1737–1747 (1997).
Min, B. Th2 immunity: a step closer to completion. Immunol. Cell Biol. 88, 235 (2010).
Hida, S., Tadachi, M., Saito, T. & Taki, S. Negative control of basophil expansion by IRF-2 critical for the regulation of Th1/Th2 balance. Blood 106, 2011–2017 (2005).
Oh, K., Shen, T., Le Gros, G. & Min, B. Induction of Th2 type immunity in a mouse system reveals a novel immunoregulatory role of basophils. Blood 109, 2921–2927 (2007).
Allenspach, E. J., Lemos, M. P., Porrett, P. M., Turka, L. A. & Laufer, T. M. Migratory and lymphoid-resident dendritic cells cooperate to efficiently prime naive CD4 T cells. Immunity 29, 795–806 (2008).
Mack, M. et al. Identification of antigen-capturing cells as basophils. J. Immunol. 174, 735–741 (2005).
Tang, H. et al. The T helper type 2 response to cysteine proteases requires dendritic cell-basophil cooperation via ROS-mediated signaling. Nature Immunol. 11, 608–617 (2010). This study argues that basophils are important for papain-induced T H 2 cell responses not for their APC function but for their effects on DCs.
Merad, M. et al. Langerhans cells renew in the skin throughout life under steady-state conditions. Nature Immunol. 3, 1135–1141 (2002).
Bursch, L. S. et al. Identification of a novel population of Langerin+ dendritic cells. J. Exp. Med. 204, 3147–3156 (2007).
Ginhoux, F. et al. Blood-derived dermal langerin+ dendritic cells survey the skin in the steady state. J. Exp. Med. 204, 3133–3146 (2007).
Poulin, L. F. et al. The dermis contains langerin+ dendritic cells that develop and function independently of epidermal Langerhans cells. J. Exp. Med. 204, 3119–3131 (2007).
Ohnmacht, C. et al. Basophils orchestrate chronic allergic dermatitis and protective immunity against helminths. Immunity 33, 364–374 (2010).
Sullivan, B. M. et al. Genetic analysis of basophil function in vivo. Nature Immunol. 12, 527–535 (2011).
Otsuka, A. et al. Basophils are required for the induction of Th2 immunity to haptens and peptide antigens. Nature Commun. 4, 1739 (2013).
Eckl-Dorna, J. et al. Basophils are not the key antigen-presenting cells in allergic patients. Allergy 67, 601–608 (2012).
Kitzmuller, C. et al. Human blood basophils do not act as antigen-presenting cells for the major birch pollen allergen Bet v 1. Allergy 67, 593–600 (2012). References 61 and 62 were the first studies to propose that basophils might not act as APCs in the setting of human allergy.
Sharma, M. et al. Circulating human basophils lack the features of professional antigen presenting cells. Sci. Rep. 3, 1188 (2013).
Charles, N., Hardwick, D., Daugas, E., Illei, G. G. & Rivera, J. Basophils and the T helper 2 environment can promote the development of lupus nephritis. Nature Med. 16, 701–707 (2010).
Voskamp, A. L., Prickett, S. R., Mackay, F., Rolland, J. M. & O'Hehir, R. E. MHC class II expression in human basophils: induction and lack of functional significance. PLoS ONE 8, e81777 (2013).
Foster, P. S., Hogan, S. P., Ramsay, A. J., Matthaei, K. I. & Young, I. G. Interleukin 5 deficiency abolishes eosinophilia, airways hyperreactivity, and lung damage in a mouse asthma model. J. Exp. Med. 183, 195–201 (1996).
Collins, P. D., Marleau, S., Griffiths-Johnson, D. A., Jose, P. J. & Williams, T. J. Cooperation between interleukin-5 and the chemokine eotaxin to induce eosinophil accumulation in vivo. J. Exp. Med. 182, 1169–1174 (1995).
Hogan, S. P., Koskinen, A. & Foster, P. S. Interleukin-5 and eosinophils induce airway damage and bronchial hyperreactivity during allergic airway inflammation in BALB/c mice. Immunol. Cell Biol. 75, 284–288 (1997).
Sabin, E. A., Kopf, M. A. & Pearce, E. J. Schistosoma mansoni egg-induced early IL-4 production is dependent upon IL-5 and eosinophils. J. Exp. Med. 184, 1871–1878 (1996).
Hogan, S. P. et al. Eosinophils: biological properties and role in health and disease. Clin. Exp. Allergy 38, 709–750 (2008).
Shinkai, K., Mohrs, M. & Locksley, R. M. Helper T cells regulate type-2 innate immunity in vivo. Nature 420, 825–829 (2002).
Jacobsen, E. A. et al. Allergic pulmonary inflammation in mice is dependent on eosinophil-induced recruitment of effector T cells. J. Exp. Med. 205, 699–710 (2008).
Walsh, E. R. et al. Strain-specific requirement for eosinophils in the recruitment of T cells to the lung during the development of allergic asthma. J. Exp. Med. 205, 1285–1292 (2008).
Fulkerson, P. C. et al. A central regulatory role for eosinophils and the eotaxin/CCR3 axis in chronic experimental allergic airway inflammation. Proc. Natl Acad. Sci. USA 103, 16418–16423 (2006).
Hansel, T. T. et al. Sputum eosinophils from asthmatics express ICAM-1 and HLA-DR. Clin. Exp. Immunol. 86, 271–277 (1991). This is the first study to show that sputum but not blood eosinophils from a large proportion of patients with asthma express HLA-DR.
Mengelers, H. J. et al. Immunophenotyping of eosinophils recovered from blood and BAL of allergic asthmatics. Am. J. Respir. Crit. Care Med. 149, 345–351 (1994).
Beninati, W. et al. Pulmonary eosinophils express HLA-DR in chronic eosinophilic pneumonia. J. Allergy Clin. Immunol. 92, 442–449 (1993).
Hansel, T. T. et al. Induction and function of eosinophil intercellular adhesion molecule-1 and HLA-DR. J. Immunol. 149, 2130–2136 (1992).
Mawhorter, S. D., Kazura, J. W. & Boom, W. H. Human eosinophils as antigen-presenting cells: relative efficiency for superantigen- and antigen-induced CD4+ T-cell proliferation. Immunology 81, 584–591 (1994).
Celestin, J. et al. IL-3 induces B7.2 (CD86) expression and costimulatory activity in human eosinophils. J. Immunol. 167, 6097–6104 (2001).
Shi, H. Z., Humbles, A., Gerard, C., Jin, Z. & Weller, P. F. Lymph node trafficking and antigen presentation by endobronchial eosinophils. J. Clin. Invest. 105, 945–953 (2000). This is the first study to show that mouse eosinophils from antigen-sensitized airways express MHC class II and can support T cell activation.
Shi, H. Z. et al. Endobronchial eosinophils preferentially stimulate T helper cell type 2 responses. Allergy 59, 428–435 (2004).
Duez, C. et al. Migration and accumulation of eosinophils toward regional lymph nodes after airway allergen challenge. J. Allergy Clin. Immunol. 114, 820–825 (2004).
van Rijt, L. S. et al. Airway eosinophils accumulate in the mediastinal lymph nodes but lack antigen-presenting potential for naive T cells. J. Immunol. 171, 3372–3378 (2003).
Tamura, N. et al. Requirement of CD80 and CD86 molecules for antigen presentation by eosinophils. Scand. J. Immunol. 44, 229–238 (1996).
Del Pozo, V. et al. Eosinophil as antigen-presenting cell: activation of T cell clones and T cell hybridoma by eosinophils after antigen processing. Eur. J. Immunol. 22, 1919–1925 (1992).
Wang, H. B., Ghiran, I., Matthaei, K. & Weller, P. F. Airway eosinophils: allergic inflammation recruited professional antigen-presenting cells. J. Immunol. 179, 7585–7592 (2007).
Akuthota, P., Melo, R. C., Spencer, L. A. & Weller, P. F. M. H. C. Class II and CD9 in human eosinophils localize to detergent-resistant membrane microdomains. Am. J. Respir. Cell. Mol. Biol. 46, 188–195 (2012).
Handzel, Z. T. et al. Eosinophils bind rhinovirus and activate virus-specific T cells. J. Immunol. 160, 1279–1284 (1998).
MacKenzie, J. R., Mattes, J., Dent, L. A. & Foster, P. S. Eosinophils promote allergic disease of the lung by regulating CD4+ Th2 lymphocyte function. J. Immunol. 167, 3146–3155 (2001).
Padigel, U. M. et al. Eosinophils act as antigen-presenting cells to induce immunity to Strongyloides stercoralis in mice. J. Infect. Dis. 196, 1844–1851 (2007).
Jacobsen, E. A. et al. Eosinophil activities modulate the immune/inflammatory character of allergic respiratory responses in mice. Allergy 69, 315–327 (2014).
Denkers, E. Y., Butcher, B. A., Del Rio, L. & Bennouna, S. Neutrophils, dendritic cells and Toxoplasma. Int. J. Parasitol. 34, 411–421 (2004).
Abi Abdallah, D. S., Egan, C. E., Butcher, B. A. & Denkers, E. Y. Mouse neutrophils are professional antigen-presenting cells programmed to instruct Th1 and Th17 T-cell differentiation. Int. Immunol. 23, 317–326 (2011).
Fanger, N. A. et al. Activation of human T cells by major histocompatability complex class II expressing neutrophils: proliferation in the presence of superantigen, but not tetanus toxoid. Blood 89, 4128–4135 (1997). This study shows that human neutrophils can express MHC class II after stimulation with GM-CSF and IFNγ, and can support superantigen-mediated but not peptide-mediated T cell activation.
Yamamoto, S. et al. Cutting edge: Pseudomonas aeruginosa abolishes established lung transplant tolerance by stimulating B7 expression on neutrophils. J. Immunol. 189, 4221–4225 (2012).
Serbina, N. V., Salazar-Mather, T. P., Biron, C. A., Kuziel, W. A. & Pamer, E. G. TNF/iNOS-producing dendritic cells mediate innate immune defense against bacterial infection. Immunity 19, 59–70 (2003).
Matsushima, H. et al. Neutrophil differentiation into a unique hybrid population exhibiting dual phenotype and functionality of neutrophils and dendritic cells. Blood 121, 1677–1689 (2013).
Hepworth, M. R. & Sonnenberg, G. F. Regulation of the adaptive immune system by innate lymphoid cells. Curr. Opin. Immunol. 27C, 75–82 (2014).
Withers, D. R. et al. Cutting edge: lymphoid tissue inducer cells maintain memory CD4 T cells within secondary lymphoid tissue. J. Immunol. 189, 2094–2098 (2012).
Lane, P. J., Gaspal, F. M., McConnell, F. M., Withers, D. R. & Anderson, G. Lymphoid tissue inducer cells: pivotal cells in the evolution of CD4 immunity and tolerance? Front. Immunol. 3, 24 (2012).
Hepworth, M. R. et al. Innate lymphoid cells regulate CD4+ T-cell responses to intestinal commensal bacteria. Nature 498, 113–117 (2013). This is the first study to suggest that ILC3s in the gut regulate CD4+ T cell responses to commensal bacteria through MHC class II expression.
von Burg, N. et al. Activated group 3 innate lymphoid cells promote T-cell-mediated immune responses. Proc. Natl Acad. Sci. USA 111, 12835–12840 (2014). This study shows that ILC3s stimulated with IL-1β express MHC class II and co-stimulatory molecules, and can stimulate naive CD4+ T cell activation.
Korn, L. L. et al. Conventional CD4+ T cells regulate IL-22-producing intestinal innate lymphoid cells. Mucosal Immunol. 7, 1045–1057 (2014).
Neill, D. R. et al. Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature 464, 1367–1370 (2010).
Mirchandani, A. S. et al. Type 2 innate lymphoid cells drive CD4+ Th2 cell responses. J. Immunol. 192, 2442–2448 (2014).
Oliphant, C. J. et al. MHCII-mediated dialog between group 2 innate lymphoid cells and CD4+ T cells potentiates type 2 immunity and promotes parasitic helminth expulsion. Immunity 41, 283–295 (2014). This is the first study to show that ILC2s express MHC class II and co-stimulatory molecules, and can support peptide-mediated naive CD4+ T cell activation.
Ko, H. S., Fu, S. M., Winchester, R. J., Yu, D. T. & Kunkel, H. G. Ia determinants on stimulated human T lymphocytes. Occurrence on mitogen- and antigen-activated T cells. J. Exp. Med. 150, 246–255 (1979).
Evans, R. L. et al. Peripheral human T cells sensitized in mixed leukocyte culture synthesize and express Ia-like antigens. J. Exp. Med. 148, 1440–1445 (1978).
LaSalle, J. M., Tolentino, P. J., Freeman, G. J., Nadler, L. M. & Hafler, D. A. Early signaling defects in human T cells anergized by T cell presentation of autoantigen. J. Exp. Med. 176, 177–186 (1992). This is the first study to fully characterize the potential of human CD4+ T cells to anergize other CD4+ T cells through antigen presentation.
Costantino, C. M., Spooner, E., Ploegh, H. L. & Hafler, D. A. Class, I. I. MHC self-antigen presentation in human B and T lymphocytes. PLoS ONE 7, e29805 (2012).
Card, C. M., Yu, S. S. & Swartz, M. A. Emerging roles of lymphatic endothelium in regulating adaptive immunity. J. Clin. Invest. 124, 943–952 (2014).
Cohen, J. N. et al. Lymph node-resident lymphatic endothelial cells mediate peripheral tolerance via Aire-independent direct antigen presentation. J. Exp. Med. 207, 681–688 (2010).
Fletcher, A. L. et al. Lymph node fibroblastic reticular cells directly present peripheral tissue antigen under steady-state and inflammatory conditions. J. Exp. Med. 207, 689–697 (2010).
Lee, J. W. et al. Peripheral antigen display by lymph node stroma promotes T cell tolerance to intestinal self. Nature Immunol. 8, 181–190 (2007).
Tewalt, E. F. et al. Lymphatic endothelial cells induce tolerance via PD-L1 and lack of costimulation leading to high-level PD-1 expression on CD8 T cells. Blood 120, 4772–4782 (2012).
Gardner, J. M. et al. Extrathymic Aire-expressing cells are a distinct bone marrow-derived population that induce functional inactivation of CD4+ T cells. Immunity 39, 560–572 (2013). This study suggests that some APCs termed LNSCs are actually a bone marrow-derived population of cells that induce T cell anergy.
Malhotra, D. et al. Transcriptional profiling of stroma from inflamed and resting lymph nodes defines immunological hallmarks. Nature Immunol. 13, 499–510 (2012). This transcriptional analysis dissects which LNSCs can and do express MHC class II that is inducible by inflammatory signals.
Dubrot, J. et al. Lymph node stromal cells acquire peptide-MHCII complexes from dendritic cells and induce antigen-specific CD4+ T cell tolerance. J. Exp. Med. 211, 1153–1166 (2014). This paper suggests that LNSCs express endogenous MHC class II molecules and can also acquire peptide–MHC class II complexes from DCs.
Hughes, C. C., Savage, C. O. & Pober, J. S. Endothelial cells augment T cell interleukin 2 production by a contact-dependent mechanism involving CD2/LFA-3 interaction. J. Exp. Med. 171, 1453–1467 (1990).
Lechler, R. I. & Batchelor, J. R. Restoration of immunogenicity to passenger cell-depleted kidney allografts by the addition of donor strain dendritic cells. J. Exp. Med. 155, 31–41 (1982).
Lakkis, F. G., Arakelov, A., Konieczny, B. T. & Inoue, Y. Immunologic 'ignorance' of vascularized organ transplants in the absence of secondary lymphoid tissue. Nature Med. 6, 686–688 (2000).
Shiao, S. L., McNiff, J. M. & Pober, J. S. Memory T cells and their costimulators in human allograft injury. J. Immunol. 175, 4886–4896 (2005).
Shiao, S. L. et al. Human effector memory CD4+ T cells directly recognize allogeneic endothelial cells in vitro and in vivo. J. Immunol. 179, 4397–4404 (2007). References 123 and 124 are two of many articles from the Pober laboratory examining the function of MHC class II+ endothelial cells as APCs during graft acceptance or rejection. Many of the articles from this laboratory examine the mechanisms for stimulating memory but not naive CD4+ T cells.
Fan, L. et al. Antigen presentation by keratinocytes directs autoimmune skin disease. Proc. Natl Acad. Sci. USA 100, 3386–3391 (2003).
Mulder, D. J. et al. Antigen presentation and MHC class II expression by human esophageal epithelial cells: role in eosinophilic esophagitis. Am. J. Pathol. 178, 744–753 (2011).
Sanderson, I. R., Ouellette, A. J., Carter, E. A., Walker, W. A. & Harmatz, P. R. Differential regulation of B7 mRNA in enterocytes and lymphoid cells. Immunology 79, 434–438 (1993).
Framson, P. E., Cho, D. H., Lee, L. Y. & Hershberg, R. M. Polarized expression and function of the costimulatory molecule CD58 on human intestinal epithelial cells. Gastroenterology 116, 1054–1062 (1999).
Nakazawa, A. et al. Functional expression of costimulatory molecule CD86 on epithelial cells in the inflamed colonic mucosa. Gastroenterology 117, 536–545 (1999).
Kaiserlian, D., Vidal, K. & Revillard, J. P. Murine enterocytes can present soluble antigen to specific class II-restricted CD4+ T cells. Eur. J. Immunol. 19, 1513–1516 (1989).
Buning, J. et al. Antigen targeting to MHC class II-enriched late endosomes in colonic epithelial cells: trafficking of luminal antigens studied in vivo in Crohn's colitis patients. FASEB J. 20, 359–361 (2006).
Bland, P. W. & Warren, L. G. Antigen presentation by epithelial cells of the rat small intestine. I. Kinetics, antigen specificity and blocking by anti-Ia antisera. Immunology 58, 1–7 (1986).
Mayer, L. & Shlien, R. Evidence for function of Ia molecules on gut epithelial cells in man. J. Exp. Med. 166, 1471–1483 (1987). References 130, 132 and 133 are among the first manuscripts to demonstrate that IECs express MHC class II and present protein antigen to CD4+ T cell hybridomas.
Hershberg, R. M. et al. Intestinal epithelial cells use two distinct pathways for HLA class II antigen processing. J. Clin. Invest. 100, 204–215 (1997).
Hershberg, R. M. et al. Highly polarized HLA class II antigen processing and presentation by human intestinal epithelial cells. J. Clin. Invest. 102, 792–803 (1998).
Westendorf, A. M. et al. CD4+Foxp3+ regulatory T cell expansion induced by antigen-driven interaction with intestinal epithelial cells independent of local dendritic cells. Gut 58, 211–219 (2009).
Maggio-Price, L. et al. Lineage targeted MHC-II transgenic mice demonstrate the role of dendritic cells in bacterial-driven colitis. Inflamm. Bowel Dis. 19, 174–184 (2013).
Thelemann, C. et al. Interferon-γ induces expression of MHC class II on intestinal epithelial cells and protects mice from colitis. PLoS ONE 9, e86844 (2014).
Cruickshank, S. M., McVay, L. D., Baumgart, D. C., Felsburg, P. J. & Carding, S. R. Colonic epithelial cell mediated suppression of CD4 T cell activation. Gut 53, 678–684 (2004).
Ward, H. E. & Nicholas, T. E. Alveolar type I and type II cells. Aust. N. Z. J. Med. 14, 731–734 (1984).
Fehrenbach, H. Alveolar epithelial type II cell: defender of the alveolus revisited. Respir. Res. 2, 33–46 (2001).
Strunk, R. C., Eidlen, D. M. & Mason, R. J. Pulmonary alveolar type II epithelial cells synthesize and secrete proteins of the classical and alternative complement pathways. J. Clin. Invest. 81, 1419–1426 (1988).
Gereke, M., Jung, S., Buer, J. & Bruder, D. Alveolar type II epithelial cells present antigen to CD4+ T cells and induce Foxp3+ regulatory T cells. Am. J. Respir. Crit. Care Med. 179, 344–355 (2009).
Kreisel, D. et al. Cutting edge: MHC class II expression by pulmonary nonhematopoietic cells plays a critical role in controlling local inflammatory responses. J. Immunol. 185, 3809–3813 (2010).
Steinman, R. M. Dendritic cells: understanding immunogenicity. Eur. J. Immunol. 37, S53–S60 (2007).
McDevitt, H. O. & Sela, M. Genetic control of the antibody response. I. Demonstration of determinant-specific differences in response to synthetic polypeptide antigens in two strains of inbred mice. J. Exp. Med. 122, 517–531 (1965).
McDevitt, H. O. et al. Genetic control of the immune response. Mapping of the IR-1 locus. J. Exp. Med. 135, 1259–1278 (1972).
Grumet, F. C. & McDevitt, H. O. Genetic control of the immune response. Relationship between the immune response-1 gene(s) and individual H-2 antigenic specificities. Transplantation 13, 171–173 (1972).
Zinkernagel, R. M. & Doherty, P. C. Restriction of in vitro T cell-mediated cytotoxicity in lymphocytic choriomeningitis within a syngeneic or semiallogeneic system. Nature 248, 701–702 (1974).
Banchereau, J. & Steinman, R. M. Dendritic cells and the control of immunity. Nature 392, 245–252 (1998).
Mosier, D. E. A requirement for two cell types for antibody formation in vitro. Science 158, 1573–1575 (1967).
Steinman, R. M. & Cohn, Z. A. Identification of a novel cell type in peripheral lymphoid organs of mice. I. Morphology, quantitation, tissue distribution. J. Exp. Med. 137, 1142–1162 (1973).
Steinman, R. M. & Witmer, M. D. Lymphoid dendritic cells are potent stimulators of the primary mixed leukocyte reaction in mice. Proc. Natl Acad. Sci. USA 75, 5132–5136 (1978).
Blum, J. S., Wearsch, P. A. & Cresswell, P. Pathways of antigen processing. Annu. Rev. Immunol. 31, 443–473 (2013).
Van Niel, G. et al. Intestinal epithelial exosomes carry MHC class II/peptides able to inform the immune system in mice. Gut 52, 1690–1697 (2003). This study suggests that IECs may generate immunologically active exosomes.
Thery, C. et al. Indirect activation of naive CD4+ T cells by dendritic cell-derived exosomes. Nature Immunol. 3, 1156–1162 (2002). This is one of the first articles demonstrating that exosomes may contribute to CD4+ T cell activation.
Thery, C. et al. Molecular characterization of dendritic cell-derived exosomes. Selective accumulation of the heat shock protein Hsc73. J. Cell Biol. 147, 599–610 (1999).
Mallegol, J., van Niel, G. & Heyman, M. Phenotypic and functional characterization of intestinal epithelial exosomes. Blood Cells Mol. Dis. 35, 11–16 (2005).
Mallegol, J. et al. T84-intestinal epithelial exosomes bear MHC class II/peptide complexes potentiating antigen presentation by dendritic cells. Gastroenterology 132, 1866–1876 (2007).
Acknowledgements
Work in the laboratory of T.M.L. is supported by a VA Merit Award. Research in the laboratory of T.K. is supported by funding from the US National Institutes of Health and the American Asthma Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
DATABASES
Glossary
- Experimental autoimmune encephalomyelitis
-
An experimental model for the human disease multiple sclerosis. Autoimmune disease is induced in experimental animals by immunization with myelin or peptides derived from myelin. The animals develop a paralytic disease with inflammation and demyelination in the brain and spinal cord.
- Mixed lymphocyte responses
-
A tissue-culture technique for testing T cell reactivity. The proliferation of one population of T cells — induced by exposure to inactivated MHC-mismatched stimulator cells — is determined by measuring the incorporation of 3H-thymidine into the DNA of dividing cells.
- Macropinocytosis
-
A type of endocytosis (or phagocytosis) that occurs during the engulfment of apoptotic cells. During macropinocytosis, large droplets of fluid are trapped within the membrane protrusions (ruffles) or phagocytic arms.
- Superantigens
-
Proteins that bind to and activate all T cells that express a particular set of Vβ T cell receptor genes.
- Neutrophil extracellular traps
-
(NETs). A set of extracellular fibres produced by activated neutrophils to ensnare invading microorganisms. NETs enhance neutrophil killing of extracellular pathogens, while minimizing damage to host cells.
Rights and permissions
About this article
Cite this article
Kambayashi, T., Laufer, T. Atypical MHC class II-expressing antigen-presenting cells: can anything replace a dendritic cell?. Nat Rev Immunol 14, 719–730 (2014). https://doi.org/10.1038/nri3754
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3754
- Springer Nature Limited
This article is cited by
-
The emerging family of RORγt+ antigen-presenting cells
Nature Reviews Immunology (2024)
-
Synovial microenvironment-influenced mast cells promote the progression of rheumatoid arthritis
Nature Communications (2024)
-
Hypoimmune induced pluripotent stem cells survive long term in fully immunocompetent, allogeneic rhesus macaques
Nature Biotechnology (2024)
-
Acute graft-versus-host disease
Nature Reviews Disease Primers (2023)
-
Expression of mammalian cell entry genes in clinical isolates of M. tuberculosis and the cell entry potential and immunological reactivity of the Rv0590A protein
Medical Microbiology and Immunology (2023)