Key Points
-
The levels of copeptin in the circulation correlate with those of arginine vasopressin (AVP)
-
Currently available techniques for the measurement of copeptin have advantages over those for AVP in terms of ex vivo stability of the marker, and the ease and speed of measurement
-
In the differential diagnosis of polyuria–polydipsia syndrome, measurement of copeptin has great potential, enabling direct identification of patients with nephrogenic diabetes insipidus, without prior thirsting
-
Osmotically stimulated copeptin levels can differentiate between patients with primary polydipsia and those with partial central diabetes insipidus
-
In the differential diagnosis of hyponatraemia, low copeptin levels identify primary polydipsia, and the ratio of copeptin to urinary sodium could distinguish between AVP-dependent forms of hyponatraemia
Abstract
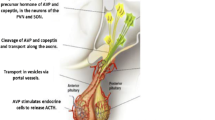
Copeptin and arginine vasopressin (AVP) are derived from a common precursor molecule and have equimolar secretion and response to osmotic, haemodynamic and stress-related stimuli. Plasma concentrations of copeptin and AVP in relation to serum osmolality are highly correlated. The physiological functions of AVP with respect to homeostasis of fluid balance, vascular tonus and regulation of the endocrine stress response are well known, but the exact function of copeptin is undetermined. Quantification of AVP can be difficult, but copeptin is stable in plasma and can be easily measured with a sandwich immunoassay. For this reason, copeptin has emerged as a promising marker for the diagnosis of AVP-dependent fluid disorders. Copeptin measurements can enable differentiation between various conditions within the polyuria–polydipsia syndrome. In the absence of prior fluid deprivation, baseline copeptin levels >20 pmol/l identify patients with nephrogenic diabetes insipidus. Conversely, copeptin levels measured upon osmotic stimulation differentiate primary polydipsia from partial central diabetes insipidus. In patients with hyponatraemia, low levels of copeptin together with low urine osmolality identify patients with primary polydipsia, and the ratio of copeptin to urinary sodium can distinguish the syndrome of inappropriate antidiuretic hormone secretion from other AVP-dependent forms of hyponatraemia.



Similar content being viewed by others
References
Kluge, M., Riedl, S., Erhart-Hofmann, B., Hartmann, J. & Waldhauser, F. Improved extraction procedure and RIA for determination of arginine8-vasopressin in plasma: role of premeasurement sample treatment and reference values in children. Clin. Chem. 45, 98–103 (1999).
Balanescu, S. et al. Correlation of plasma copeptin and vasopressin concentrations in hypo-, iso-, and hyperosmolar states. J. Clin. Endocrinol. Metab. 96, 1046–1052 (2011).
Morgenthaler, N. G., Struck, J., Alonso, C. & Bergmann, A. Assay for the measurement of copeptin, a stable peptide derived from the precursor of vasopressin. Clin. Chem. 52, 112–119 (2006).
Land, H., Schütz, G., Schmale, H. & Richter, D. Nucleotide sequence of cloned cDNA encoding bovine arginine vasopressin–neurophysin II precursor. Nature 295, 299–303 (1982).
Levy, B., Chauvet, M. T., Chauvet, J. & Acher, R. Ontogeny of bovine neurohypophysial hormone precursors. II. Foetal copeptin, the third domain of the vasopressin precursor. Int. J. Pept. Protein Res. 27, 320–324 (1986).
Land, H. et al. Deduced amino acid sequence from the bovine oxytocin–neurophysin I precursor cDNA. Nature 302, 342–344 (1983).
Acher, R., Chauvet, J. & Rouille, Y. Dynamic processing of neuropeptides: sequential conformation shaping of neurohypophysial preprohormones during intraneuronal secretory transport. J. Mol. Neurosci. 18, 223–228 (2002).
de Bree, F. M. & Burbach, J. P. Structure–function relationships of the vasopressin prohormone domains. Cell. Mol. Neurobiol. 18, 173–191 (1998).
Robertson, G. L. Physiology of ADH secretion. Kidney Int. Suppl. 21, S20–S26 (1987).
Ciura, S. & Bourque, C. W. Transient receptor potential vanilloid 1 is required for intrinsic osmoreception in organum vasculosum lamina terminalis neurons and for normal thirst responses to systemic hyperosmolality. J. Neurosci. 26, 9069–9075 (2006).
Vandesande, F. & Dierickx, K. Identification of the vasopressin producing and of the oxytocin producing neurons in the hypothalamic magnocellular neurosecretroy system of the rat. Cell Tissue Res. 164, 153–162 (1975).
Gillies, G. E., Linton, E. A. & Lowry, P. J. Corticotropin releasing activity of the new CRF is potentiated several times by vasopressin. Nature 299, 355–357 (1982).
Rivier, C. & Vale, W. Modulation of stress-induced ACTH release by corticotropin-releasing factor, catecholamines and vasopressin. Nature 305, 325–327 (1983).
Son, S. J. et al. Dendritic peptide release mediates interpopulation crosstalk between neurosecretory and preautonomic networks. Neuron 78, 1036–1049 (2013).
Bourque, C. W. Central mechanisms of osmosensation and systemic osmoregulation. Nat. Rev. Neurosci. 9, 519–531 (2008).
Toney, G. M. & Stocker, S. D. Hyperosmotic activation of CNS sympathetic drive: implications for cardiovascular disease. J. Physiol. 588, 3375–3384 (2010).
Robertson, G. L. The regulation of vasopressin function in health and disease. Rec. Prog. Horm. Res. 33, 333–385 (1976).
LaRochelle, F. T. Jr, North, W. G. & Stern, P. A new extraction of arginine vasopressin from blood: the use of octadecasilyl-silica. Pflugers Arch. 387, 79–81 (1980).
Morgenthaler, N. G. et al. Copeptin, a stable peptide of the arginine vasopressin precursor, is elevated in hemorrhagic and septic shock. Shock 28, 219–226 (2007).
Roussel, R. et al. Comparison between copeptin and vasopressin in a population from the community and in people with chronic kidney disease. J. Clin. Endocrinol. Metab. 99, 4656–4663 (2014).
Nathanson, M. H. et al. Mechanisms of subcellular cytosolic Ca2+ signaling evoked by stimulation of the vasopressin V1a receptor. J. Biol. Chem. 267, 23282–23289 (1992).
Jard, S., Elands, J., Schmidt, A. & Barberis, C. in Progress in Endocrinology (eds Imura, H. & Shizume, K.) 1183–1188 (Elsevier, 1988).
Baylis, P. H. Osmoregulation and control of vasopressin secretion in healthy humans. Am. J. Physiol. 253, R671–R678 (1987).
Nagy, G., Mulchahey, J. J., Smyth, D. G. & Neill, J. D. The glycopeptide moiety of vasopressin-neurophysin precursor is neurohypophysial prolactin releasing factor. Biochem. Biophys. Res. Commun. 151, 524–529 (1988).
Hyde, J. F., North, W. G. & Ben-Jonathan, N. The vasopressin-associated glycopeptide is not a prolactin-releasing factor: studies with lactating Brattleboro rats. Endocrinology 125, 35–40 (1989).
Barat, C., Simpson, L. & Breslow, E. Properties of human vasopressin precursor constructs: inefficient monomer folding in the absence of copeptin as a potential contributor to diabetes insipidus. Biochemistry 43, 8191–8203 (2004).
Parodi, A. J. Protein glucosylation and its role in protein folding. Annu. Rev. Biochem. 69, 69–93 (2000).
Schrag, J. D., Procopio, D. O., Cygler, M., Thomas, D. Y. & Bergeron, J. J. Lectin control of protein folding and sorting in the secretory pathway. Trends Biochem. Sci. 28, 49–57 (2003).
Zittema, D. et al. Vasopressin, copeptin, and renal concentrating capacity in patients with autosomal dominant polycystic kidney disease without renal impairment. Clin. J. Am. Soc. Nephrol. 7, 906–913 (2012).
Zittema, D. et al. Kidney function and plasma copeptin levels in healthy kidney donors and autosomal dominant polycystic kidney disease patients. Clin. J. Am. Soc. Nephrol. 9, 1553–1562 (2014).
Timper, K. et al. Diagnostic accuracy of copeptin in the differential diagnosis of the polyuria-polydipsia syndrome: a prospective multicenter study. J. Clin. Endocrinol. Metab. 100, 2268–2274 (2015).
Fenske, W. et al. Copeptin in the differential diagnosis of the polydipsia-polyuria syndrome — revisiting the direct and indirect water deprivation tests. J. Clin. Endocrinol. Metab. 96, 1506–1515 (2011).
Husain, M. K., Fernando, N., Shapiro, M., Kagan, A. & Glick, S. M. Radioimmunoassay of arginine vasopressin in human plasma. J. Clin. Endocrinol. Metab. 37, 616–625 (1973).
Robertson, G. L., Mahr, E. A., Athar, S. & Sinha, T. Development and clinical application of a new method for the radioimmunoassay of arginine vasopressin in human plasma. J. Clin. Invest. 52, 2340–2352 (1973).
Gerbes, A. L., Witthaut, R., Samson, W. K., Schnizer, W. & Vollmar, A. M. A highly sensitive and rapid radioimmunoassay for the determination of arginine8-vasopressin. Eur. J. Clin. Chem. Clin. Biochem. 30, 229–233 (1992).
Van de Heijning, B. J., Koekkoek-van den Herik, I., Iványi, T. & Van Wimersma Greidanus, T. B. Solid-phase extraction of plasma vasopressin: evaluation, validation and application. J. Chromatogr. 565, 159–171 (1991).
Wun, T. Vasopressin and platelets: a concise review. Platelets 8, 15–22 (1997).
Preibisz, J. J., Sealey, J. E., Laragh, J. H., Cody, R. J. & Weksler, B. B. Plasma and platelet vasopressin in essential hypertension and congestive heart failure. Hypertension 5, I129–I138 (1983).
Jane Ellis, M., Livesey, J. H. & Evans, M. J. Hormone stability in human whole blood. Clin. Biochem. 36, 109–112 (2003).
Baumann, G. & Dingman, J. F. Distribution, blood transport, and degradation of antidiuretic hormone in man. J. Clin. Invest. 57, 1109–1116 (1976).
Bhandari, S. S. et al. Gender and renal function influence plasma levels of copeptin in healthy individuals. Clin. Sci. (Lond.) 116, 257–263 (2009).
US National Library of Science. ClinicalTrials.gov[online], (2015).
Walti, C., Siegenthaler, J. & Christ-Crain, M. Copeptin levels are independent of ingested nutrient type after standardised meal administration — the CoMEAL study. Biomarkers 19, 557–562 (2014).
Puder, J. J. et al. Menstrual cycle symptoms are associated with changes in low-grade inflammation. Eur. J. Clin. Invest. 36, 58–64 (2006).
Katan, M. et al. Copeptin: a novel, independent prognostic marker in patients with ischemic stroke. Ann. Neurol. 66, 799–808 (2009).
Reichlin, T. et al. Incremental value of copeptin for rapid rule out of acute myocardial infarction. J. Am. Coll. Cardiol. 54, 60–68 (2009).
Katan, M. & Christ-Crain, M. The stress hormone copeptin: a new prognostic biomarker in acute illness. Swiss Med. Wkly 140, w13101 (2010).
Siegenthaler, J., Walti, C., Urwyler, S. A., Schuetz, P. & Christ-Crain, M. Copeptin concentrations during psychological stress: the PsyCo study. Eur. J. Endocrinol. 171, 737–742 (2014).
Urwyler, S. A., Schuetz, P., Sailer, C. & Christ-Crain, M. Copeptin as a stress marker prior and after a written examination — the CoEXAM study. Stress 18, 134–137 (2015).
Fenske, W. & Allolio, B. Current state and future perspectives in the diagnosis of diabetes insipidus: a clinical review. J. Clin. Endocrinol. Metab. 97, 3426–3437 (2012).
Carter, A. C. & Robbins, J. The use of hypertonic saline infusions in the differential diagnosis of diabetes insipidus and psychogenic polydipsia. J. Clin. Endocrinol. Metab. 7, 753–766 (1947).
Barlow, E. D. & De Wardener, H. E. Compulsive water drinking. Q. J. Med. 28, 235–258 (1959).
Dashe, A. M., Cramm, R. E., Crist, C. A., Habener, J. F. & Solomon, D. H. A water deprivation test for the differential diagnosis of polyuria. JAMA 185, 699–703 (1963).
Miller, M., Dalakos, T., Moses, A. M., Fellerman, H. & Streeten, D. H. Recognition of partial defects in antidiuretic hormone secretion. Ann. Intern. Med. 73, 721–729 (1970).
Dies, F., Rangel, S. & Rivera, A. Differential diagnosis between diabetes insipidus and compulsive polydipsia. Ann. Intern. Med. 54, 710–725 (1961).
Robertson, G. L. Diabetes insipidus. Endocrinol. Metab. Clin. North Am. 24, 549–572 (1995).
Robertson, G. L. in Diabetes Insipidus in Man. (eds Czernichow, P. & Robinson, A. G.) 176–189 (Karger, 1985).
Milles, J. J., Spruce, B. & Baylis, P. H. A comparison of diagnostic methods to differentiate diabetes insipidus from primary polyuria: a review of 21 patients. Acta Endocrinol. (Copenh.) 104, 410–416 (1983).
Verbalis, J. G. Disorders of body water homeostasis. Best Pract. Res. Clin. Endocrinol. Metab. 17, 471–503 (2003).
Cadnapaphornchai, M. A. et al. Effect of primary polydipsia on aquaporin and sodium transporter abundance. Am. J. Physiol. Renal Physiol. 285, F965–F971 (2003).
Berliner, R. W. & Davidson, D. G. Production of hypertonic urine in the absence of pituitary antidiuretic hormone. J. Clin. Invest. 36, 1416–1427 (1957).
Harrington, A. R. & Valtin, H. Impaired urinary concentration after vasopressin and its gradual correction in hypothalamic diabetes insipidus. J. Clin. Invest. 47, 502–510 (1968).
Block, L. H., Furrer, J., Locher, R. A., Siegenthaler, W. & Vetter, W. Changes in tissue sensitivity to vasopressin in hereditary hypothalamic diabetes insipidus. Klin. Wochenschr. 59, 831–836 (1981).
Gellai, M., Edwards, B. R. & Valtin, H. Urinary concentrating ability during dehydration in the absence of vasopressin. Am. J. Physiol. 237, F100–F104 (1979).
Baylis, P. H. & Robertson, G. L. Plasma vasopressin response to hypertonic saline infusion to assess posterior pituitary function. J. R. Soc. Med. 73, 255–260 (1980).
Baylis, P. H., Gaskill, M. B. & Robertson, G. L. Vasopressin secretion in primary polydipsia and cranial diabetes insipidus. Q. J. Med. 50, 345–358 (1981).
Zerbe, R. L. & Robertson, G. L. A comparison of plasma vasopressin measurements with a standard indirect test in the differential diagnosis of polyuria. N. Engl. J. Med. 305, 1539–1546 (1981).
Baylis, P. H. Diabetes insipidus. J. R. Coll. Physicians Lond. 32, 108–111 (1998).
Czaczkes, J. W. & Kleeman, C. R. The effect of various states of hydration and the plasma concentration on the turnover of antidiuretic hormone in mammals. J. Clin. Invest. 43, 1649–1658 (1964).
Katan, M. et al. Anterior and posterior pituitary function testing with simultaneous insulin tolerance test and a novel copeptin assay. J. Clin. Endocrinol. Metab. 92, 2640–2643 (2007).
Winzeler, B. et al. Postoperative copeptin concentration predicts diabetes insipidus after pituitary surgery. J. Clin. Endocrinol. Metab. 100, 2275–2282 (2015).
Timper, K. et al. Diagnostic accuracy of copeptin in the differential diagnosis of the polyuria-polydipsia syndrome: a prospective multicenter study. J. Clin. Endocrinol. Metab. 100, 2268–2274 (2015).
Smith, D. et al. Baroregulation of vasopressin release in adipsic diabetes insipidus. J. Clin. Endocrinol. Metab. 87, 4564–4568 (2002).
US National Library of Medicine. ClinicalTrials.gov[online], (2013).
Kumar, S. & Berl, T. Sodium. Lancet 352, 220–228 (1998).
Upadhyay, A., Jaber, B. L. & Madias, N. E. Incidence and prevalence of hyponatremia. Am. J. Med. 119, S30–S35 (2006).
Fenske, W. et al. Value of fractional uric acid excretion in differential diagnosis of hyponatremic patients on diuretics. J. Clin. Endocrinol. Metab. 93, 2991–2997 (2008).
Fenske, W. et al. Copeptin in the differential diagnosis of hyponatremia. J. Clin. Endocrinol. Metab. 94, 123–129 (2009).
Tang, W. W., Kaptein, E. M., Feinstein, E. I. & Massry, S. G. Hyponatremia in hospitalized patients with the acquired immunodeficiency syndrome (AIDS) and the AIDS-related complex. Am. J. Med. 94, 169–174 (1993).
Verbalis, J. G., Goldsmith, S. R., Greenberg, A., Schrier, R. W. & Sterns, R. H. Hyponatremia treatment guidelines 2007: expert panel recommendations. Am. J. Med. 120, S1–S21 (2007).
Chung, H. M., Kluge, R., Schrier, R. W. & Anderson, R. J. Clinical assessment of extracellular fluid volume in hyponatremia. Am. J. Med. 83, 905–908 (1987).
Verbalis, J. G. et al. Diagnosis, evaluation, and treatment of hyponatremia: expert panel recommendations. Am. J. Med. 126, S1–S42 (2013).
Schrier, R. W. et al. Tolvaptan, a selective oral vasopressin V2-receptor antagonist, for hyponatremia. N. Engl. J. Med. 355, 2099–2112 (2006).
Renneboog, B., Musch, W., Vandemergel, X., Manto, M. U. & Decaux, G. Mild chronic hyponatremia is associated with falls, unsteadiness, and attention deficits. Am. J. Med. 119, 71.e1–71.e8 (2006).
Verbalis, J. G. et al. Hyponatremia-induced osteoporosis. J. Bone Miner. Res. 25, 554–563 (2010).
Fenske, W., Maier, S. K., Blechschmidt, A., Allolio, B. & Störk, S. Utility and limitations of the traditional diagnostic approach to hyponatremia: a diagnostic study. Am. J. Med. 123, 652–657 (2010).
Boursier, G. et al. CT-pro-AVP as a tool for assessment of intravascular volume depletion in severe hyponatremia. Clin. Biochem. 48, 640–645 (2015).
Nigro, N. et al. Copeptin for the differential diagnosis and therapy management of hyponatremia in hospitalized patients — 'the Co-MED-Study'. Endocr. Abstr. 35, 173 (2014).
Zerbe, R., Stropes, L. & Robertson, G. Vasopressin function in the syndrome of inappropriate antidiuresis. Annu. Rev. Med. 31, 315–327 (1980).
Fenske, W. K. et al. A copeptin-based classification of the osmoregulatory defects in the syndrome of inappropriate antidiuresis. J. Am. Soc. Nephrol. 25, 2376–2383 (2014).
Soupart, A. et al. Successful long-term treatment of hyponatremia in syndrome of inappropriate antidiuretic hormone secretion with satavaptan (SR121463B), an orally active nonpeptide vasopressin V2-receptor antagonist. Clin. J. Am. Soc. Nephrol. 1, 1154–1160 (2006).
Robertson, G. L. Vaptans for the treatment of hyponatremia. Nat. Rev. Endocrinol. 7, 151–161 (2011).
Author information
Authors and Affiliations
Contributions
Both authors contributed to researching data, discussing content and writing the article, and to review and editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M. C.-C. and W. F. have received speaking honoraria from Thermo Fisher AG, the manufacturer of the Copeptin Assay.
Related links
FURTHER INFORMATION
Rights and permissions
About this article
Cite this article
Christ-Crain, M., Fenske, W. Copeptin in the diagnosis of vasopressin-dependent disorders of fluid homeostasis. Nat Rev Endocrinol 12, 168–176 (2016). https://doi.org/10.1038/nrendo.2015.224
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2015.224
- Springer Nature Limited
This article is cited by
-
Long-term health outcomes associated with hydration status
Nature Reviews Nephrology (2024)
-
Copeptin: a novel prognostic biomarker in trauma: a review article
Journal of Health, Population and Nutrition (2023)
-
Copeptin and the prognosis of patients with coronary artery disease: a meta-analysis
Irish Journal of Medical Science (1971 -) (2023)
-
Establishment and Clinical Application in Stroke of a Serum Copeptin Time-Resolved Fluorescence Immunoassay
Journal of Fluorescence (2023)
-
Test diagnostici per il diabete insipido: un aggiornamento
L'Endocrinologo (2023)