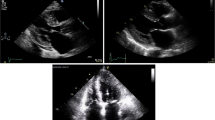
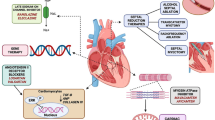
Key Points
-
Although the ACC Foundation (ACCF)/AHA and ESC guidelines show widespread concordance, the ESC definition of hypertrophic cardiomyopathy (HCM) includes nonsarcomeric disease states associated with load-independent left ventricular hypertrophy (LVH)
-
Relatives of patients with HCM should be offered screening; autosomal dominant inheritance is typical, and a range of electrocardiographic and imaging features are recognized to precede the development of LVH
-
Prognostic assessment at baseline and follow-up is recommended for all patients with HCM; despite recognizing many of the same predictors of sudden death, the ACCF/AHA and ESC adopt distinct approaches to risk profiling
-
Specialist HCM centres promote implementation of best-practice guidelines by offering facilities and skills for investigations such as exercise stress echocardiography, cardiovascular magnetic resonance (CMR), and genetic analysis, in addition to high-volume programmes for invasive septal reduction
-
Patients referred to specialist HCM centres also benefit from multidisciplinary support systems and clinical experience in interpreting equivocal clinical findings, adjudicating borderline risk profiles, and managing complex cases
-
Advances in the evaluation of HCM include CMR-based T1 mapping for demonstrating diffuse and interstitial fibrosis; in the therapeutic arena, several putative substrate-modulating agents are progressing from preclinical to clinical trials
Abstract
Hypertrophic cardiomyopathy (HCM) is the most common inherited cardiovascular disorder, affecting 1 in 500 individuals worldwide. Existing epidemiological studies might have underestimated the prevalence of HCM, however, owing to limited inclusion of individuals with early, incomplete phenotypic expression. Clinical manifestations of HCM include diastolic dysfunction, left ventricular outflow tract obstruction, ischaemia, atrial fibrillation, abnormal vascular responses and, in 5% of patients, progression to a 'burnt-out' phase characterized by systolic impairment. Disease-related mortality is most often attributable to sudden cardiac death, heart failure, and embolic stroke. The majority of individuals with HCM, however, have normal or near-normal life expectancy, owing in part to contemporary management strategies including family screening, risk stratification, thromboembolic prophylaxis, and implantation of cardioverter–defibrillators. The clinical guidelines for HCM issued by the ACC Foundation/AHA and the ESC facilitate evaluation and management of the disease. In this Review, we aim to assist clinicians in navigating the guidelines by highlighting important updates, current gaps in knowledge, differences in the recommendations, and challenges in implementing them, including aids and pitfalls in clinical and pathological evaluation. We also discuss the advances in genetics, imaging, and molecular research that will underpin future developments in diagnosis and therapy for HCM.









Similar content being viewed by others
References
Maron, B. J. et al. Hypertrophic cardiomyopathy in adulthood associated with low cardiovascular mortality with contemporary management strategies. J. Am. Coll. Cardiol. 65, 1915–1928 (2015).
Maron, B. J. et al. Hypertrophic cardiomyopathy in children, adolescents, and young adults associated with low cardiovascular mortality with contemporary management strategies. Circulation 133, 62–73 (2016).
Colan, S. D. et al. Epidemiology and cause-specific outcome of hypertrophic cardiomyopathy in children: findings from the Pediatric Cardiomyopathy Registry. Circulation 115, 773–781 (2007).
McKenna, W. J. & Sen-Chowdhry, S. From Teare to the present day: a fifty year odyssey in hypertrophic cardiomyopathy, a paradigm for the logic of the discovery process. Rev. Esp. Cardiol. 61, 1239–1244 (2008).
Sen-Chowdhry, S. & McKenna, W. J. Standing on the shoulders of giants: J. A. P. Paré and the birth of cardiovascular genetics. Can. J. Cardiol. 31, 1305–1308 (2015).
Maron, B. J. et al. Asymetric septal hypertrophy in childhood. Circulation 53, 9–19 (1976).
McKenna, W. et al. Prognosis in hypertrophic cardiomyopathy: role of age and clinical, electrocardiographic and hemodynamic features. Am. J. Cardiol. 47, 532–538 (1981).
Ziółkowska, L., Turska-Kmiec, A., Petryka, J. & Kawalec, W. Predictors of long-term outcome in children with hypertrophic cardiomyopathy. Pediatr. Cardiol. 37, 448–458 (2015).
Spirito, P. et al. Clinical course and prognosis of hypertrophic cardiomyopathy in an outpatient population. N. Engl. J. Med. 320, 749–755 (1989).
Maron, B. J. et al. Clinical course of hypertrophic cardiomyopathy in a regional United States cohort. JAMA 281, 650–655 (1999).
Elliott, P. M. et al. Historical trends in reported survival rates in patients with hypertrophic cardiomyopathy. Heart 92, 785–791 2006).
Ellenbogen, K. A. et al. Are implantable cardioverter defibrillator shocks a surrogate for sudden cardiac death in patients with nonischemic cardiomyopathy? Circulation 113, 776–782 (2006).
Maron, B. J. et al. American College of Cardiology/European Society of Cardiology clinical expert consensus document on hypertrophic cardiomyopathy: a report of the American College of Cardiology foundation task force on clinical expert consensus documents and the European Society of Cardiology committee for practice guidelines. J. Am. Coll. Cardiol. 42, 1687–1713 (2003).
Gersh, B. J. et al. 2011 ACCF/AHA Guideline for the Diagnosis and Treatment of Hypertrophic Cardiomyopathy : a report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines developed in collaboration with the American Association for Thoracic Surgery, American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Failure Society of America, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J. Am. Coll. Cardiol. 58, e212–e260 (2011).
Elliott, P. M. et al. 2014 ESC guidelines on diagnosis and management of hypertrophic cardiomyopathy: the task force for the diagnosis and management of hypertrophic cardiomyopathy of the European Society of Cardiology (ESC). Eur. Heart J. 35, 2733–2779 (2014).
Elliott, P. M. et al. Left ventricular outflow tract obstruction and sudden death risk in patients with hypertrophic cardiomyopathy. Eur. Heart J. 27, 1933–1941 (2006).
Thaman, R. et al. Reversal of inappropriate peripheral vascular responses in hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 46, 883–892 (2005).
Ammirati, E. et al. Pharmacological treatment of hypertrophic cardiomyopathy: current practice and novel perspectives. Eur. J. Heart Fail. http://dx.doi.org/10.1002/ejhf.541 (2016).
Corrado, D. et al. Trends in sudden cardiovascular death in young competitive athletes after implementation of a preparticipation screening program. JAMA 296, 1593–1601 (2006).
Richards, S. et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 17, 405–424 (2015).
Biagini, E. et al. Dilated-hypokinetic evolution of hypertrophic cardiomyopathy: prevalence, incidence, risk factors, and prognostic implications in pediatric and adult patients. J. Am. Coll. Cardiol. 46, 1543–1550 (2005).
Levine, R. A. et al. Papillary muscle displacement causes systolic anterior motion of the mitral valve. Experimental validation and insights into the mechanism of subaortic obstruction. Circulation 91, 1189–1195 (1995).
Elliott, P. et al. Prevalence of Anderson-Fabry disease in patients with hypertrophic cardiomyopathy: the European Anderson-Fabry Disease survey. Heart 97, 1957–1960 (2011).
Sachdev, B. et al. Prevalence of Anderson-Fabry disease in male patients with late onset hypertrophic cardiomyopathy. Circulation 105, 1407–1411 (2002).
Monserrat, L. et al. Prevalence of fabry disease in a cohort of 508 unrelated patients with hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 50, 2399–2403 (2007).
Vitale, G., Pivonello, R., Lombardi, G. & Colao, A. Cardiac abnormalities in acromegaly. Pathophysiology and implications for management. Treat. Endocrinol. 3, 309–318 (2004).
Kampmann, C. in Fabry Disease: Perspectives from 5 Years of FOSCh. 37 (eds Mehta, A., Beck, M. & Sunder-Plassmann, G.) (Oxford: Oxford PharmaGenesis, 2006).
Ramaswami, U. Update on role of agalsidase alfa in management of Fabry disease. Drug Des. Devel. Ther. 5, 155–173 (2011).
Mariotti, C. et al. Idebenone treatment in Friedreich patients: one-year-long randomized placebo-controlled trial. Neurology 60, 1676–1679 (2003).
Maron, B. J. et al. Clinical outcome and phenotypic expression in LAMP2 cardiomyopathy. JAMA 301, 1253–1259 (2009).
Feriozzi, S., Schwarting, A., Sunder-Plassmann, G., West, M. & Cybulla, M. Agalsidase alfa slows the decline in renal function in patients with Fabry disease. Am. J. Nephrol. 29, 353–361 (2009).
Murphy, R. T. et al. Adenosine monophosphate-activated protein kinase disease mimicks hypertrophic cardiomyopathy and Wolff-Parkinson-White syndrome: natural history. J. Am. Coll. Cardiol. 45, 922–930 (2005).
Richards, D. B. et al. Therapeutic clearance of amyloid by antibodies to serum amyloid p component. N. Engl. J. Med. 373, 1106–1114 (2015).
Hawkins, P. N. et al. Evolving landscape in the management of transthyretin amyloidosis. Ann. Med. 47, 625–638 (2015).
Gutgesell, H. P., Speer, M. E. & Rosenberg, H. S. Characterization of the cardiomyopathy in infants of diabetic mothers. Circulation 61, 441–450 (1980).
Atkison, P. et al. Hypertrophic cardiomyopathy associated with tacrolimus in paediatric transplant patients. Lancet 345, 894–896 (1995).
Yang, C. et al. Very early treatment for infantile-onset Pompe disease contributes to better outcomes. J. Pediatr. 169, 174.e1–180.e1 (2016).
Güngör, D. et al. Quality of life and participation in daily life of adults with Pompe disease receiving enzyme replacement therapy: 10 years of international follow-up. J. Inherit. Metab. Dis. 39, 253–260 (2016).
McKenna, W. J., Stewart, J. T., Nihoyannopoulos, P., McGinty, F. & Davies, M. J. Hypertrophic cardiomyopathy without hypertrophy: two families with myocardial disarray in the absence of increased myocardial mass. Br. Heart J. 63, 287–290 (1990).
Varnava, A. et al. A new mutation of the cardiac troponin T gene causing familial hypertrophic cardiomyopathy without left ventricular hypertrophy. Heart 82, 621–624 (1999).
Varnava, A. M. et al. Hypertrophic cardiomyopathy: histopathological features of sudden death in cardiac troponin T disease. Circulation 104, 1380–1384 (2001).
Pasquale, F. et al. Long-term outcomes in hypertrophic cardiomyopathy caused by mutations in the cardiac troponin T gene. Circ. Cardiovasc. Genet. 5, 10–17 (2012).
Coppini, R. et al. Clinical phenotype and outcome of hypertrophic cardiomyopathy associated with thin-filament gene mutations. J. Am. Coll. Cardiol. 64, 2589–2600 (2014).
Sheppard, M. N. et al. A detailed pathologic examination of heart tissue from three older patients with Anderson-Fabry disease on enzyme replacement therapy. Cardiovasc. Pathol. 19, 293–301 (2010).
Burch, M. et al. Myocardial disarray in Noonan syndrome. Br. Heart J. 68, 586–588 (1992).
Arad, M. et al. Constitutively active AMP kinase mutations cause glycogen storage disease mimicking hypertrophic cardiomyopathy. J. Clin. Invest. 109, 357–362 (2002).
Varnava, A. M., Elliott, P. M., Mahon, N., Davies, M. J. & McKenna, W. J. Relation between myocyte disarray and outcome in hypertrophic cardiomyopathy. Am. J. Cardiol. 88, 275–279 (2001).
Mogensen, J. et al. Frequency and clinical expression of cardiac troponin I mutations in 748 consecutive families with hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 44, 2315–2325 (2004).
Mogensen, J. et al. Idiopathic restrictive cardiomyopathy is part of the clinical expression of cardiac troponin I mutations. J. Clin. Invest. 111, 209–216 (2003).
Sen-Chowdhry, S., Syrris, P. & McKenna, W. J. Genetics of restrictive cardiomyopathy. Heart Fail. Clin. 6, 179–186 (2010).
Sen-Chowdhry, S., Tomé Esteban, M. T. & McKenna, W. J. Insights and challenges in hypertrophic cardiomyopathy, 2012. Herzschrittmacherther. Elektrophysiol. 23, 174–185 (2012).
Ashrafian, H., Redwood, C., Blair, E. & Watkins, H. Hypertrophic cardiomyopathy:a paradigm for myocardial energy depletion. Trends Genet. 19, 263–268 (2003).
Rickers, C. et al. Utility of cardiac magnetic resonance imaging in the diagnosis of hypertrophic cardiomyopathy. Circulation 112, 855–861 (2005).
Flett, A. S. et al. Diagnosis of apical hypertrophic cardiomyopathy: T-wave inversion and relative but not absolute apical left ventricular hypertrophy. Int. J. Cardiol. 183, 143–148 (2015).
Moon, J. C. C., Fisher, N. G., McKenna, W. J. & Pennell, D. J. Detection of apical hypertrophic cardiomyopathy by cardiovascular magnetic resonance in patients with non-diagnostic echocardiography. Heart 90, 645–649 (2004).
Maron, M. S. et al. Prevalence, clinical significance, and natural history of left ventricular apical aneurysms in hypertrophic cardiomyopathy. Circulation 118, 1541–1549 (2008).
Maron, M. S. et al. Right ventricular involvement in hypertrophic cardiomyopathy. Am. J. Cardiol. 100, 1293–1298 (2007).
Rosca, M. et al. Right ventricular remodeling, its correlates, and its clinical impact in hypertrophic cardiomyopathy. J. Am. Soc. Echocardiogr. 28, 1329–1338 (2015).
Fikrle, M. et al. The diagnostic performance of cardiac magnetic resonance in detection of myocardial involvement in AL amyloidosis. Clin. Physiol. Funct. Imaging 36, 218–224 (2014).
Fontana, M. et al. Prognostic value of late gadolinium enhancement cardiovascular magnetic resonance in cardiac amyloidosis. Circulation 132, 1570–1579 (2015).
Kozor, R. et al. Cardiac involvement in genotype-positive Fabry disease patients assessed by cardiovascular MR. Heart 102, 298–302 (2016).
Flett, A. S. et al. Evaluation of techniques for the quantification of myocardial scar of differing etiology using cardiac magnetic resonance. JACC Cardiovasc. Imaging 4, 150–156 (2011).
Burt, J. R., Zimmerman, S. L., Kamel, I. R., Halushka M. & Bluemke, D. A. Myocardial T1 mapping: techniques and potential applications. Radiographics 34, 377–395 (2014).
Miller, C. A. et al. Comprehensive validation of cardiovascular magnetic resonance techniques for the assessment of myocardial extracellular volume. Circ. Cardiovasc. Imaging 6, 373–383 (2013).
Kammerlander, A. A. et al. T1 Mapping by CMR imaging: from histological validation to clinical implication. JACC Cardiovasc. Imaging 9, 14–23 (2016).
Moon, J. C. C. et al. Gadolinium enhanced cardiovascular magnetic resonance in Anderson-Fabry disease. Evidence for a disease specific abnormality of the myocardial interstitium. Eur. Heart J. 24, 2151–2155 (2003).
Vucicevic, D. et al. The incremental value of magnetic resonance imaging for identification of apical pouch in patients with apical variant of hypertrophic cardiomyopathy. Echocardiography 33, 572–578 (2016).
Maron, M. S. et al. Hypertrophic cardiomyopathy phenotype revisited after 50 years with cardiovascular magnetic resonance. J. Am. Coll. Cardiol. 54, 220–228 (2009).
Kawel, N. et al. Normal left ventricular myocardial thickness for middle-aged and older subjects with steady-state free precession cardiac magnetic resonance: the multi-ethnic study of atherosclerosis. Circ. Cardiovasc. Imaging 5, 500–508 (2012).
Colan, S. D. Hypertrophic cardiomyopathy in childhood. Heart Fail. Clin. 6, 433–444 (2010).
Lipshultz, S. E. et al. Risk stratification at diagnosis for children with hypertrophic cardiomyopathy: an analysis of data from the Pediatric Cardiomyopathy Registry. Lancet 382, 1889–1897 (2013).
Mohamed, S. et al. Tyrosinemia type 1: a rare and forgotten cause of reversible hypertrophic cardiomyopathy in infancy. BMC Res. Notes 6, 362 (2013).
Morita, H. et al. Shared genetic causes of cardiac hypertrophy in children and adults. N. Engl. J. Med. 358, 1899–1908 (2008).
Kaski, J. P. et al. Prevalence of sarcomere protein gene mutations in preadolescent children with hypertrophic cardiomyopathy. Circ. Cardiovasc. Genet. 2, 436–441 (2009).
Maron, B. J. et al. Prevention of sudden cardiac death with implantable cardioverter-defibrillators in children and adolescents with hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 61, 1527–1535 (2013).
Nugent, A. W. et al. Clinical features and outcomes of childhood hypertrophic cardiomyopathy: results from a national population-based study. Circulation 112, 1332–1338 (2005).
Decker, J. A. et al. Risk factors and mode of death in isolated hypertrophic cardiomyopathy in children. J. Am. Coll. Cardiol. 54, 250–254 (2009).
Biagini, E. et al. Prognostic implications of the Doppler restrictive filling pattern in hypertrophic cardiomyopathy. Am. J. Cardiol. 104, 1727–1731 (2009).
Maddukuri, P. V. et al. What is the best approach for the assessment of left atrial size? Comparison of various unidimensional and two-dimensional parameters with three-dimensional echocardiographically determined left atrial volume. J. Am. Soc. Echocardiogr. 19, 1026–1032 (2006).
Tsang, T. S. M. et al. Prediction of cardiovascular outcomes with left atrial size: is volume superior to area or diameter? J. Am. Coll. Cardiol. 47, 1018–1023 (2006).
Lang, R. M. et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 28, 1–39.e14 (2015).
Guttmann, O. P. et al. Prediction of thrombo-embolic risk in patients with hypertrophic cardiomyopathy (HCM Risk-CVA). Eur. J. Heart Fail. 17, 837–845 (2015).
O'Mahony, C. et al. A novel clinical risk prediction model for sudden cardiac death in hypertrophic cardiomyopathy (HCM risk-SCD). Eur. Heart J. 35, 2010–2020 (2014).
Guttmann, O. P., Rahman, M. S., O'Mahony, C., Anastasakis, A. & Elliott, P. M. Atrial fibrillation and thromboembolism in patients with hypertrophic cardiomyopathy: systematic review. Heart 100, 465–472 (2014).
Maron, B. J. et al. Left atrial remodeling in hypertrophic cardiomyopathy and susceptibility markers for atrial fibrillation identified by cardiovascular magnetic resonance. Am. J. Cardiol. 113, 1394–1400 (2014).
Nistri, S. et al. Prognostic significance of left atrial size in patients with hypertrophic cardiomyopathy (from the Italian Registry for Hypertrophic Cardiomyopathy). Am. J. Cardiol. 98, 960–965 (2006).
Kramer, C. M. et al. Regional heterogeneity of function in hypertrophic cardiomyopathy. Circulation 90, 186–194 (1994).
Serri, K. et al. Global and regional myocardial function quantification by two-dimensional strain: application in hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 47, 1175–1181 (2006).
Urbano-Moral, J. A., Rowin, E. J., Maron, M. S., Crean, A. & Pandian, N. G. Investigation of global and regional myocardial mechanics with 3-dimensional speckle tracking echocardiography and relations to hypertrophy and fibrosis in hypertrophic cardiomyopathy. Circ. Cardiovasc. Imaging 7, 11–19 (2014).
Maciver, D. H. A new method for quantification of left ventricular systolic function using a corrected ejection fraction. Eur. J. Echocardiogr. 12, 228–234 (2011).
Maron, M. S. et al. Effect of left ventricular outflow tract obstruction on clinical outcome in hypertrophic cardiomyopathy. N. Engl. J. Med. 348, 295–303 (2003).
Maron, M. S. et al. Hypertrophic cardiomyopathy is predominantly a disease of left ventricular outflow tract obstruction. Circulation 114, 2232–2239 (2006).
Sherrid, M. V., Wever-Pinzon, O., Shah, A. & Chaudhry, F. A. Reflections of inflections in hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 54, 212–219 (2009).
Sherrid, M. V., Gunsburg, D. Z. & Pearle, G. Mid-systolic drop in left ventricular ejection velocity in obstructive hypertrophic cardiomyopathy—the lobster claw abnormality. J. Am. Soc. Echocardiogr. 10, 707–712 (1997).
Nishimura, R. A. & Ommen, S. R. Hypertrophic cardiomyopathy: the search for obstruction. Circulation 114, 2200–2202 (2006).
Aurigemma, G. et al. Abnormal left ventricular intracavitary flow acceleration in patients undergoing aortic valve replacement for aortic stenosis. A marker for high postoperative morbidity and mortality. Circulation 86, 926–936 (1992).
Ingles, J., Burns, C., Barratt, A. & Semsarian, C. Application of genetic testing in hypertrophic cardiomyopathy for preclinical disease detection. Circ. Cardiovasc. Genet. 8, 852–859 (2015).
Bottillo, I. et al. Molecular analysis of sarcomeric and non-sarcomeric genes in patients with hypertrophic cardiomyopathy. Gene 577, 227–235 (2016).
Lopes, L. R. et al. Use of high-throughput targeted exome-sequencing to screen for copy number variation in hypertrophic cardiomyopathy. Eur. J. Med. Genet. 58, 611–616 (2015).
Pan, S. et al. Cardiac structural and sarcomere genes associated with cardiomyopathy exhibit marked intolerance of genetic variation. Circ. Cardiovasc. Genet. 5, 602–610 (2012).
Lopes, L. R. et al. Genetic complexity in hypertrophic cardiomyopathy revealed by high-throughput sequencing. J. Med. Genet. 50, 228–239 (2013).
Richard, P. et al. Hypertrophic cardiomyopathy: distribution of disease genes, spectrum of mutations, and implications for a molecular diagnosis strategy. Circulation 107, 2227–2232 (2003).
Girolami, F. et al. Clinical features and outcome of hypertrophic cardiomyopathy associated with triple sarcomere protein gene mutations. J. Am. Coll. Cardiol. 55, 1444–1453 (2010).
Maron, B. J., Maron, M. S. & Semsarian, C. Double or compound sarcomere mutations in hypertrophic cardiomyopathy: a potential link to sudden death in the absence of conventional risk factors. Heart Rhythm 9, 57–63 (2012).
Ortlepp, J. R. et al. Genetic polymorphisms in the renin-angiotensin-aldosterone system associated with expression of left ventricular hypertrophy in hypertrophic cardiomyopathy: a study of five polymorphic genes in a family with a disease causing mutation in the myosin binding protein C gene. Heart 87, 270–275 (2002).
Kaufman, B. D. et al. RAAS gene polymorphisms influence progression of pediatric hypertrophic cardiomyopathy. Hum. Genet. 122, 515–523 (2007).
Lopes, L. R. et al. Novel genotype-phenotype associations demonstrated by high-throughput sequencing in patients with hypertrophic cardiomyopathy. Heart 101, 294–301 (2015).
Friedrich, F. W. et al. A new polymorphism in human calmodulin III gene promoter is a potential modifier gene for familial hypertrophic cardiomyopathy. Eur. Heart J. 30, 1648–1655 (2009).
Bick, A. G. et al. Burden of rare sarcomere gene variants in the Framingham and Jackson Heart Study cohorts. Am. J. Hum. Genet. 91, 513–519 (2012).
Semsarian, C., Ingles, J., Maron, M. S. & Maron, B. J. New perspectives on the prevalence of hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 65, 1249–1254 (2015).
Charron, P. et al. Penetrance of familial hypertrophic cardiomyopathy. Genet. Couns. 8, 107–114 (1997).
Elliott, P. M., D'Cruz, L. & McKenna, W. J. Late-onset hypertrophic cardiomyopathy caused by a mutation in the cardiac troponin T gene. N. Engl. J. Med. 341, 1855–1856 (1999).
Andersen, P. S. et al. A novel myosin essential light chain mutation causes hypertrophic cardiomyopathy with late onset and low expressivity. Biochem. Res. Int. 2012, 685108 (2012).
Niimura, H. et al. Mutations in the gene for cardiac myosin-binding protein C and late-onset familial hypertrophic cardiomyopathy. N. Engl. J. Med. 338, 1248–1257 (1998).
McKenna, W. J., Spirito, P., Desnos, M., Dubourg, O. & Komajda, M. Experience from clinical genetics in hypertrophic cardiomyopathy: proposal for new diagnostic criteria in adult members of affected families. Heart 77, 130–132 (1997).
Kauer, F. et al. Diastolic abnormalities in normal phenotype hypertrophic cardiomyopathy gene carriers: a study using speckle tracking echocardiography. Echocardiography 30, 558–563 (2013).
Captur, G. et al. Abnormal cardiac formation in hypertrophic cardiomyopathy: fractal analysis of trabeculae and preclinical gene expression. Circ. Cardiovasc. Genet. 7, 241–248 (2014).
Deva, D. P. et al. Deep basal inferoseptal crypts occur more commonly in patients with hypertrophic cardiomyopathy due to disease-causing myofilament mutations. Radiology 269, 68–76 (2013).
Brouwer, W. P. et al. Multiple myocardial crypts on modified long-axis view are a specific finding in pre-hypertrophic HCM mutation carriers. Eur. Heart J. Cardiovasc. Imaging 13, 292–297 (2012).
Captur, G. et al. Prediction of sarcomere mutations in subclinical hypertrophic cardiomyopathy. Circ. Cardiovasc. Imaging 7, 863–871 (2014).
Petryka, J., Baksi, A. J., Prasad, S. K., Pennell, D. J. & Kilner, P. J. Prevalence of inferobasal myocardial crypts among patients referred for cardiovascular magnetic resonance. Circ. Cardiovasc. Imaging 7, 259–264 (2014).
Basso, C., Marra, M. P. & Thiene, G. Myocardial clefts, crypts, or crevices: once again, you see only what you look for. Circ. Cardiovasc. Imaging 7, 217–219 (2014).
Maron, M. S. et al. Mitral valve abnormalities identified by cardiovascular magnetic resonance represent a primary phenotypic expression of hypertrophic cardiomyopathy. Circulation 124, 40–47 (2011).
Reant, P. et al. Abnormal septal convexity into the left ventricle occurs in subclinical hypertrophic cardiomyopathy. J. Cardiovasc. Magn. Reson. 17, 64 (2015).
Ho, C. Y. et al. Myocardial fibrosis as an early manifestation of hypertrophic cardiomyopathy. N. Engl. J. Med. 363, 552–563 (2010).
Ho, C. Y. et al. T1 measurements identify extracellular volume expansion in hypertrophic cardiomyopathy sarcomere mutation carriers with and without left ventricular hypertrophy. Circ. Cardiovasc. Imaging 6, 415–422 (2013).
Lopes, L. R. et al. Left ventricular outflow tract obstruction as a primary phenotypic expression of hypertrophic cardiomyopathy in mutation carriers without hypertrophy. Int. J. Cardiol. 176, 1264–1267 (2014).
Ranasinghe, I., Ayoub, C., Cheruvu, C., Freedman, S. B. & Yiannikas, J. Isolated hypertrophy of the basal ventricular septum: characteristics of patients with and without outflow tract obstruction. Int. J. Cardiol. 173, 487–493 (2014).
Kelshiker, M. A., Mayet, J., Unsworth, B. & Okonko, D. O. Basal septal hypertrophy. Curr. Cardiol. Rev. 9, 325–330 (2013).
Binder, J. et al. Echocardiography-guided genetic testing in hypertrophic cardiomyopathy: septal morphological features predict the presence of myofilament mutations. Mayo Clin. Proc. 81, 459–467 (2006).
Maron, B. J. et al. Relationship of race to sudden cardiac death in competitive athletes with hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 41, 974–980 (2003).
Papadakis, M. et al. The prevalence, distribution, and clinical outcomes of electrocardiographic repolarization patterns in male athletes of African/Afro-Caribbean origin. Eur. Heart J. 32, 2304–2313 (2011).
Sheikh, N. et al. Cardiac adaptation to exercise in adolescent athletes of African ethnicity: an emergent elite athletic population. Br. J. Sports Med. 47, 585–592 (2013).
ten Cate, F. J. et al. Long-term outcome of alcohol septal ablation in patients with obstructive hypertrophic cardiomyopathy: a word of caution. Circ. Heart Fail. 3, 362–369 (2010).
Saumarez, R. C. et al. Paced ventricular electrogram fractionation predicts sudden cardiac death in hypertrophic cardiomyopathy. Eur. Heart J. 29, 1653–1661 (2008).
Maron, B. J. et al. Implantable cardioverter-defibrillators and prevention of sudden cardiac death in hypertrophic cardiomyopathy. JAMA 298, 405–412 (2007).
O'Mahony, C. et al. A validation study of the 2003 American College of Cardiology/European Society of Cardiology and 2011 American College of Cardiology Foundation/American Heart Association risk stratification and treatment algorithms for sudden cardiac death in patients with hypertrophic cardiomyopathy. Heart 99, 534–541 (2013).
Vriesendorp, P. A. et al. Validation of the 2014 European Society of Cardiology guidelines risk prediction model for the primary prevention of sudden cardiac death in hypertrophic cardiomyopathy. Circ. Arrhythm. Electrophysiol. 8, 829–835 (2015).
Ruiz-Salas, A. et al. Comparison of the new risk prediction model (HCM Risk-SCD) and classic risk factors for sudden death in patients with hypertrophic cardiomyopathy and defibrillator. Europace 18, 773–777 (2015).
Maron, B. J. et al. Independent assessment of the European Society of Cardiology sudden death risk model for hypertrophic cardiomyopathy. Am. J. Cardiol. 116, 757–764 (2015).
Ommen, S. R. et al. Long-term effects of surgical septal myectomy on survival in patients with obstructive hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 46, 470–476 (2005).
Olivotto, I. et al. Prognostic value of systemic blood pressure response during exercise in a community-based patient population with hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 33, 2044–2051 (1999).
Maron, B. J. & Nishimura, R. A. Surgical septal myectomy versus alcohol septal ablation: assessing the status of the controversy in 2014. Circulation 130, 1617–1624 (2014).
Kim, L. et al. Hospital volume outcomes after septal myectomy and alcohol septal ablation for treatment of obstructive hypertrophic cardiomyopathy: US Nationwide Inpatient Database, 2003–2011. JAMA Cardiol. 1, 324–332 (2016).
Ommen, S. R. & Nishimura, R. A. Hypertrophic cardiomyopathy — one case per year? A clarion call to do what is right. JAMA Cardiol. 1, 333–334 (2016).
Parry, D. J. et al. Short and medium term outcomes of surgery for patients with hypertrophic obstructive cardiomyopathy. Ann. Thorac. Surg. 99, 1213–1219 (2015).
Fifer, M. A. Controversies in cardiovascular medicine. Most fully informed patients choose septal ablation over septal myectomy. Circulation 116, 207–216; discussion 216 (2007).
Smedira, N. G. et al. Current effectiveness and risks of isolated septal myectomy for hypertrophic obstructive cardiomyopathy. Ann. Thorac. Surg. 85, 127–133 (2008).
Mohr, F. W., Seeburger, J. & Misfeld, M. Keynote lecture — transmitral hypertrophic obstructive cardiomyopathy (HOCM) repair. Ann. Cardiothorac. Surg. 2, 729–732 (2013).
Gutermann, H. et al. Myectomy and mitral repair through the left atrium in hypertrophic obstructive cardiomyopathy: the preferred approach for contemporary surgical candidates? J. Thorac. Cardiovasc. Surg. 147, 1833–1836 (2014).
Liebregts, M. et al. Systematic review and meta-analysis of long-term outcomes after septal reduction therapy in patients with hypertrophic cardiomyopathy. JACC Heart Fail. 3, 896–905 (2015).
Vatasescu, R. et al. Biventricular / left ventricular pacing in hypertrophic obstructive cardiomyopathy: an overview. Indian Pacing Electrophysiol. J. 12, 114–123 (2012).
Slade, A. K. et al. DDD pacing in hypertrophic cardiomyopathy: a multicentre clinical experience. Heart 75, 44–49 (1996).
Nishimura, R. A. et al. Dual-chamber pacing for hypertrophic cardiomyopathy: a randomized, double-blind, crossover trial. J. Am. Coll. Cardiol. 29, 435–441 (1997).
Kappenberger, L. et al. Pacing in hypertrophic obstructive cardiomyopathy. A randomized crossover study. Eur. Heart J. 18, 1249–1256 (1997).
Maron, B. J. et al. Assessment of permanent dual-chamber pacing as a treatment for drug-refractory symptomatic patients with obstructive hypertrophic cardiomyopathy. A randomized, double-blind, crossover study (M-PATHY). Circulation 99, 2927–2933 (1999).
Kappenberger, L. J. et al. Clinical progress after randomized on/off pacemaker treatment for hypertrophic obstructive cardiomyopathy. Europace 1, 77–84 (1999).
Berruezo, A. et al. Biventricular pacing in hypertrophic obstructive cardiomyopathy: a pilot study. Heart Rhythm 8, 221–227 (2011).
Giraldeau, G. et al. Dyssynchronization reduces dynamic obstruction without affecting systolic function in patients with hypertrophic obstructive cardiomyopathy: a pilot study. Int. J. Cardiovasc. Imaging 32, 1179–1188 (2016).
Lenarczyk, R. et al. Effect of cardiac resynchronization on gradient reduction in patients with obstructive hypertrophic cardiomyopathy: preliminary study. Pacing Clin. Electrophysiol. 34, 1544–1552 (2011).
Schaff, H. V., Dearani, J. A., Ommen, S. R., Sorajja, P. & Nishimura, R. A. Expanding the indications for septal myectomy in patients with hypertrophic cardiomyopathy: results of operation in patients with latent obstruction. J. Thorac. Cardiovasc. Surg. 143, 303–309 (2012).
Minakata, K., Dearani, J. A., O'Leary, P. W. & Danielson, G. K. Septal myectomy for obstructive hypertrophic cardiomyopathy in pediatric patients: early and late results. Ann. Thorac. Surg. 80, 1424–1429; discussion 1429–1430 (2005).
Altarabsheh, S. E. et al. Outcome of septal myectomy for obstructive hypertrophic cardiomyopathy in children and young adults. Ann. Thorac. Surg. 95, 663–669; discussion 669 (2013).
Minakata, K. et al. Mechanisms for recurrent left ventricular outflow tract obstruction after septal myectomy for obstructive hypertrophic cardiomyopathy. Ann. Thorac. Surg. 80, 851–856 (2005).
McLeod, C. J. et al. Surgical septal myectomy decreases the risk for appropriate implantable cardioverter defibrillator discharge in obstructive hypertrophic cardiomyopathy. Eur. Heart J. 28, 2583–2588 (2007).
Efthimiadis, G. K. et al. Can septal myectomy prevent sudden cardiac death in hypertrophic obstructive cardiomyopathy? Eur. Heart J. 28, 2177; author reply 2177–2178 (2007).
Nishimura, R. A. & Ommen, S. R. Septal reduction therapy for obstructive hypertrophic cardiomyopathy and sudden death: what statistics cannot tell you. Circ. Cardiovasc. Interv. 3, 91–93 (2010).
Agarwal, S. et al. Updated meta-analysis of septal alcohol ablation versus myectomy for hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 55, 823–834 (2010).
Singh, K., Qutub, M., Carson, K., Hibbert, B. & Glover, C. A meta analysis of current status of alcohol septal ablation and surgical myectomy for obstructive hypertrophic cardiomyopathy. Catheter. Cardiovasc. Interv. 88, 107–115 (2015).
Leonardi, R. A., Kransdorf, E. P., Simel, D. L. & Wang, A. Meta-analyses of septal reduction therapies for obstructive hypertrophic cardiomyopathy: comparative rates of overall mortality and sudden cardiac death after treatment. Circ. Cardiovasc. Interv. 3, 97–104 (2010).
Kaple, R. K. et al. Mitral valve abnormalities in hypertrophic cardiomyopathy: echocardiographic features and surgical outcomes. Ann. Thorac. Surg. 85, 1527–1535, 1535.e1–1535.e2 (2008).
Murat Tuzcu, E. & Kapadia, S. R. Percutaneous mitral valve repair and replacement: a new landmark for structural heart interventions. Eur. Heart J. 37, 826–828 (2016).
Schäfer, U. et al. Targeting systolic anterior motion and left ventricular outflow tract obstruction in hypertrophic obstructed cardiomyopathy with a mitraclip. EuroIntervention 11, 942–947 (2015).
Khalpey, Z., Korovin, L., Chitwood, W. R. J. & Poston, R. Robot-assisted septal myectomy for hypertrophic cardiomyopathy with left ventricular outflow tract obstruction. J. Thorac. Cardiovasc. Surg. 147, 1708–1709 (2014).
Kim, H. R., Yoo, J. S. & Lee, J. W. Minimally invasive trans-mitral septal myectomy to treat hypertrophic obstructive cardiomyopathy. Korean J. Thorac. Cardiovasc. Surg. 48, 419–421 (2015).
Spirito, P. et al. Infective endocarditis in hypertrophic cardiomyopathy: prevalence, incidence, and indications for antibiotic prophylaxis. Circulation 99, 2132–2137 (1999).
Maron, B. J. & Lever, H. In defense of antimicrobial prophylaxis for prevention of infective endocarditis in patients with hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 54, 2339–2340; author reply 2340 (2009).
Zhao, D. et al. Outcomes of catheter ablation of atrial fibrillation in patients with hypertrophic cardiomyopathy: a systematic review and meta-analysis. Europace 18, 508–520 (2015).
Ha, H. S. K. et al. Catheter ablation for atrial fibrillation in hypertrophic cardiomyopathy patients: a systematic review. J. Interv. Card. Electrophysiol. 44, 161–170 (2015).
Francia, P. et al. Eligibility for the subcutaneous implantable cardioverter-defibrillator in patients with hypertrophic cardiomyopathy. J. Cardiovasc. Electrophysiol. 26, 893–899 (2015).
Maurizi, N. et al. Prevalence of subcutaneous implantable cardioverter-defibrillator candidacy based on template ECG screening in patients with hypertrophic cardiomyopathy. Heart Rhythm 13, 457–463 (2016).
Frommeyer, G. et al. Long-term follow-up of subcutaneous ICD systems in patients with hypertrophic cardiomyopathy: a single-center experience. Clin. Res. Cardiol. 105, 89–93 (2016).
Olde Nordkamp, L. R. A. et al. Inappropriate shocks in the subcutaneous ICD: Incidence, predictors and management. Int. J. Cardiol. 195, 126–133 (2015).
Shimada, Y. J. et al. Effects of losartan on left ventricular hypertrophy and fibrosis in patients with nonobstructive hypertrophic cardiomyopathy. JACC Heart Fail. 1, 480–487 (2013).
Axelsson, A. et al. Efficacy and safety of the angiotensin II receptor blocker losartan for hypertrophic cardiomyopathy: the INHERIT randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 3, 123–131 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01912534 (2016).
Barajas-Martínez, H., Hu, D., Goodrow, R. J. J., Joyce, F. & Antzelevitch, C. Electrophysiologic characteristics and pharmacologic response of human cardiomyocytes isolated from a patient with hypertrophic cardiomyopathy. Pacing Clin. Electrophysiol. 36, 1512–1515 (2013).
Coppini, R. et al. Late sodium current inhibition reverses electromechanical dysfunction in human hypertrophic cardiomyopathy. Circulation 127, 575–584 (2013).
EU Clinical Trials Register. Clinicaltrialsregister.eu https://www.clinicaltrialsregister.eu/ctr-search/trial/2011-004507-20/DE (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02291237 (2016).
Spudich, J. A. Hypertrophic and dilated cardiomyopathy: four decades of basic research on muscle lead to potential therapeutic approaches to these devastating genetic diseases. Biophys. J. 106, 1236–1249 (2014).
Alves, M. L., Gaffin, R. D. & Wolska, B. M. Rescue of familial cardiomyopathies by modifications at the level of sarcomere and Ca2+ fluxes. J. Mol. Cell. Cardiol. 48, 834–842 (2010).
Semsarian, C. et al. The L-type calcium channel inhibitor diltiazem prevents cardiomyopathy in a mouse model. J. Clin. Invest. 109, 1013–1020 (2002).
Ho, C. Y. et al. Diltiazem treatment for pre-clinical hypertrophic cardiomyopathy sarcomere mutation carriers: a pilot randomized trial to modify disease expression. JACC Heart Fail. 3, 180–188 (2015).
Kirschner, S. E. et al. Hypertrophic cardiomyopathy-related β-myosin mutations cause highly variable calcium sensitivity with functional imbalances among individual muscle cells. Am. J. Physiol. Heart Circ. Physiol. 288, H1242–H1251 (2005).
Sung, J., Sivaramakrishnan, S., Dunn, A. R. & Spudich, J. A. Single-molecule dual-beam optical trap analysis of protein structure and function. Methods Enzymol. 475, 321–375 (2010).
Brenner, B., Seebohm, B., Tripathi, S., Montag, J. & Kraft, T. Familial hypertrophic cardiomyopathy: functional variance among individual cardiomyocytes as a trigger of FHC-phenotype development. Front. Physiol. 5, 392 (2014).
Nattel, S. & Carlsson, L. Innovative approaches to anti-arrhythmic drug therapy. Nat. Rev. Drug Discov. 5, 1034–1049 (2006).
Spudich, J. A. The myosin mesa and a possible unifying hypothesis for the molecular basis of human hypertrophic cardiomyopathy. Biochem. Soc. Trans. 43, 64–72 (2015).
Green, E. M. et al. A small-molecule inhibitor of sarcomere contractility suppresses hypertrophic cardiomyopathy in mice. Science 351, 617–621 (2016).
MyoKardia Inc. MyoKardia provides update on two phase 1 trials of MYK-461 for the treatment of hypertrophic cardiomyopathy. http://investors.myokardia.com/phoenix.zhtml?c=254211&p=irol-newsArticle&ID=2097088 (2015).
Abozguia, K. et al. Metabolic modulator perhexiline corrects energy deficiency and improves exercise capacity in symptomatic hypertrophic cardiomyopathy. Circulation 122, 1562–1569 (2010).
Horowitz, J. D. & Chirkov, Y. Y. Perhexiline and hypertrophic cardiomyopathy: a new horizon for metabolic modulation. Circulation 122, 1547–1549 (2010).
US National Library of Medicine. ClinicalTrials.gov http://clinicaltrials.gov/ct2/show/NCT02431221 (2015).
Lombardi, R. et al. Resolution of established cardiac hypertrophy and fibrosis and prevention of systolic dysfunction in a transgenic rabbit model of human cardiomyopathy through thiol-sensitive mechanisms. Circulation 119, 1398–1407 (2009).
Wilder, T., Ryba, D. M., Wieczorek, D. F., Wolska, B. M. & Solaro, R. J. N-acetylcysteine reverses diastolic dysfunction and hypertrophy in familial hypertrophic cardiomyopathy. Am. J. Physiol. Heart Circ. Physiol. 309, H1720–1730 (2015).
US National Library of Medicine. ClinicalTrials.gov http://clinicaltrials.gov/ct2/show/NCT01537926? (2015).
Geske, J. B., Sorajja, P., Ommen, S. R. & Nishimura, R. A. Left ventricular outflow tract gradient variability in hypertrophic cardiomyopathy. Clin. Cardiol. 32, 397–402 (2009).
Acknowledgements
S.S.-C. is supported by the BHF Intermediate Clinical Research Fellowship (FS/10/011/27881). W.J.M. is supported by the Fondation Leducq Transatlantic Networks of Excellence Program.
Author information
Authors and Affiliations
Contributions
All the authors researched data for the article and reviewed and edited the manuscript before submission. D.J., J.C.M., and W.J.M. substantially contributed to discussion of content. S.S.-C. and D.J. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Sen-Chowdhry, S., Jacoby, D., Moon, J. et al. Update on hypertrophic cardiomyopathy and a guide to the guidelines. Nat Rev Cardiol 13, 651–675 (2016). https://doi.org/10.1038/nrcardio.2016.140
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2016.140
- Springer Nature Limited
This article is cited by
-
Predictive value of left atrial strain analysis in adverse clinical events in patients with hypertrophic cardiomyopathy: a CMR study
BMC Cardiovascular Disorders (2023)
-
Evaluation of myocardial strain in patients with subclinical hypertrophic cardiomyopathy and subclinical Hypertensive Heart Disease using Cardiac magnetic resonance feature tracking
The International Journal of Cardiovascular Imaging (2023)
-
Metabolic characterization of hypertrophic cardiomyopathy in human heart
Nature Cardiovascular Research (2022)
-
A deleterious mutation in the ALMS1 gene in a naturally occurring model of hypertrophic cardiomyopathy in the Sphynx cat
Orphanet Journal of Rare Diseases (2021)
-
GDF15: emerging biology and therapeutic applications for obesity and cardiometabolic disease
Nature Reviews Endocrinology (2021)