Key Points
-
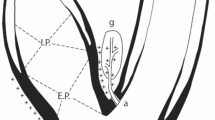
Arterial baroreceptors are pressure-sensitive nerve endings in the walls of the carotid sinuses and aortic arch that sense the increases and decreases in blood pressure
-
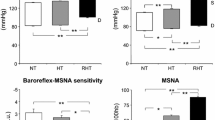
Resistant hypertension is accompanied by sympathetic overactivity and partial attenuation of arterial baroreflex function, caused largely by stiffening of the great vessels in which the baroreceptor nerve endings are embedded
-
Electrical field stimulation of the carotid baroreceptor axons (carotid baroreflex activation therapy) has been shown to mediate acute reductions in sympathetic activity and blood pressure in patients with resistant hypertension
-
In an initial phase III trial on continuous carotid baroreflex activation therapy for resistant hypertension, the prototype implantable device was associated with equivocal efficacy and surgically-induced facial nerve palsy
-
In subsequent phase I and IIa trials with a miniaturized second-generation system, unilateral carotid stimulation produced reductions in blood pressure equivalent to bilateral stimulation, with no major adverse events
-
A new phase III trial for the use of this second-generation system for resistant hypertension has been registered
Abstract
Arterial baroreceptors are mechanosensitive sensory nerve endings in the walls of the carotid sinuses and aortic arch that buffer the increases and decreases in arterial blood pressure. Electrical field stimulation of the carotid sinus, known as carotid baroreflex activation therapy, holds promise as a novel device-based intervention to supplement, but not replace, drug therapy for patients with resistant hypertension. Acute electrical field stimulation of even one carotid sinus can cause a sufficiently large reflex decrease in blood pressure to overcome offsetting reflexes from the contralateral carotid baroreceptors and aortic baroreceptors that are not paced. However, the initial phase III Rheos Pivotal Trial on continuous carotid baroreceptor pacing for resistant hypertension with the first-generation baroreceptor pacemaker yielded equivocal data on efficacy and adverse effects due to facial nerve injury during surgical implantation. A miniaturized second-generation pacing electrode has seemingly overcome the safety issue, and early results with the new device suggest efficacy of unilateral carotid sinus stimulation in heart failure. A phase III trial of this new device for resistant hypertension has been registered.









Similar content being viewed by others
References
Mancia, G. & Mark, A. L. Arterial baroreflexes in humans. Comprehensive Physiology [online], (2011).
Martin, E. A. & Victor, R. G. Premise, promise, and potential limitations of invasive devices to treat hypertension. Curr. Cardiol. Rep. 13, 86–92 (2011).
Bakris, G. L. Interventional cardiology: Indications for renal denervation: a balanced approach? Nat. Rev. Cardiol. 10, 434–436 (2013).
Bohm, M., Linz, D., Urban, D., Mahfoud, F. & Ukena, C. Renal sympathetic denervation: applications in hypertension and beyond. Nat. Rev. Cardiol. 10, 465–476 (2013).
Mearns, B. M. Hypertension: further EnligHTNment on renal sympathetic denervation. Nat. Rev. Cardiol. 10, 490 (2013).
Wang, Y. Ethnicity and sympathetic tone: predictors of the blood pressure response to renal denervation? Nat. Rev. Cardiol. 11, 638 (2014).
Bronk, D. W. & Stella, G. The response to steady pressures of single end organs in the isolated carotid sinus. Am. J. Physiol. 110, 708–714 (1935).
Brown, A. M. Receptors under pressure. An update on baroreceptors. Circ. Res. 46, 1–10 (1980).
Heymans, C. & van den Heuvel-Heymans, G. New aspects of blood pressure regulation. Circulation 4, 581–586 (1951).
Hajduczok, G., Chapleau, M. W., Ferlic, R. J., Mao, H. Z. & Abboud, F. M. Gadolinium inhibits mechanoelectrical transduction in rabbit carotid baroreceptors. Implication of stretch-activated channels. J. Clin. Invest. 94, 2392–2396 (1994).
Chen, H. I., Chapleau, M. W., McDowell, T. S. & Abboud, F. M. Prostaglandins contribute to activation of baroreceptors in rabbits. Possible paracrine influence of endothelium. Circ. Res. 67, 1394–1404 (1990).
Snitsarev, V., Whiteis, C. A., Chapleau, M. W. & Abboud, F. M. Neuronal prostacyclin is an autocrine regulator of arterial baroreceptor activity. Hypertension 46, 540–546 (2005).
Aljadhey, H. et al. Comparative effects of non-steroidal anti-inflammatory drugs (NSAIDs) on blood pressure in patients with hypertension. BMC Cardiovasc. Disord. 12, 93 (2012).
Li, Z. et al. Nitric oxide as an autocrine regulator of sodium currents in baroreceptor neurons. Neuron 20, 1039–1049 (1998).
Andresen, M. C., Doyle, M. W., Jin, Y. H. & Bailey, T. W. Cellular mechanisms of baroreceptor integration at the nucleus tractus solitarius. Ann. N. Y. Acad. Sci. 940, 132–141 (2001).
Reis, D. J. The brain and hypertension: reflections on 35 years of inquiry into the neurobiology of the circulation. Circulation 70, III31–III45 (1984).
Ohta, H., Li, X. & Talman, W. T. Release of glutamate in the nucleus tractus solitarii in response to baroreflex activation in rats. Neuroscience 74, 29–37 (1996).
Ohta, H. & Talman, W. T. Both NMDA and non-NMDA receptors in the NTS participate in the baroreceptor reflex in rats. Am. J. Physiol. 267, R1065–R1070 (1994).
Yin, M., Lee, C. C., Ohta, H. & Talman, W. T. Hemodynamic effects elicited by stimulation of the nucleus tractus solitarii. Hypertension 23, I73–I77 (1994).
Lin, L. H. & Talman, W. T. Vesicular glutamate transporters and neuronal nitric oxide synthase colocalize in aortic depressor afferent neurons. J. Chem. Neuroanat. 32, 54–64 (2006).
Talman, W. T. & Dragon, D. N. Transmission of arterial baroreflex signals depends on neuronal nitric oxide synthase. Hypertension 43, 820–824 (2004).
Madden, C. J. & Sved, A. F. Rostral ventrolateral medulla C1 neurons and cardiovascular regulation. Cell. Mol. Neurobiol. 23, 739–749 (2003).
Bristow, J. D., Honour, A. J., Pickering, T. G. & Sleight, P. Cardiovascular and respiratory changes during sleep in normal and hypertensive subjects. Cardiovasc. Res. 3, 476–485 (1969).
Eckberg, D. L., Drabinsky, M. & Braunwald, E. Defective cardiac parasympathetic control in patients with heart disease. N. Engl. J. Med. 285, 877–883 (1971).
Korner, P. I., West, M. J., Shaw, J. & Uther, J. B. “Steady-state” properties of the baroreceptor-heart rate reflex in essential hypertension in man. Clin. Exp. Pharmacol. Physiol. 1, 65–76 (1974).
Mancia, G. et al. Circulatory reflexes from carotid and extracarotid baroreceptor areas in man. Circ. Res. 41, 309–315 (1977).
Sundlof, G. & Wallin, B. G. Human muscle nerve sympathetic activity at rest. Relationship to blood pressure and age. J. Physiol. 274, 621–637 (1978).
Aksamit, T. R., Floras, J. S., Victor, R. G. & Aylward. P. E. Paroxysmal hypertension due to sinoaortic baroreceptor denervation in humans. Hypertension 9, 309–314 (1987).
Heusser, K., Tank, J., Luft, F. C. & Jordan, J. Baroreflex failure. Hypertension 45, 834–839 (2005).
Jacobsen, T. N. et al. Relative contributions of cardiopulmonary and sinoaortic baroreflexes in causing sympathetic activation in the human skeletal muscle circulation during orthostatic stress. Circ. Res. 73, 367–378 (1993).
Norcliffe-Kaufmann, L., Axelrod, F. & Kaufmann, H. Afferent baroreflex failure in familial dysautonomia. Neurology 75, 1904–1911 (2010).
Anderson, S. L. et al. Familial dysautonomia is caused by mutations of the IKAP gene. Am. J. Hum. Genet. 68, 753–758 (2001).
Abashidze, A., Gold, V., Anavi, Y., Greenspan, H. & Weil, M. Involvement of IKAP in peripheral target innervation and in specific JNK and NGF signaling in developing PNS neurons. PLoS ONE 9, e113428 (2014).
Sanders, J. S., Ferguson, D. W. & Mark, A. L. Arterial baroreflex control of sympathetic nerve activity during elevation of blood pressure in normal man: dominance of aortic baroreflexes. Circulation 77, 279–288 (1988).
Sanders, J. S., Mark, A. L. & Ferguson, D. W. Importance of aortic baroreflex in regulation of sympathetic responses during hypotension. Evidence from direct sympathetic nerve recordings in humans. Circulation 79, 83–92 (1989).
Kaplan, N. M. & Victor, R. G. Kaplan's Clinical Hypertension, 11th edn (Wolters Kluwer, 2014).
Mancia, G. et al. 2013 ESH/ESC guidelines for the management of arterial hypertension: the Task Force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur. Heart J. 34, 2159–2219 (2013).
Fontil, V. et al. Physician underutilization of effective medications for resistant hypertension at office visits in the United States: NAMCS 2006–2010. J. Gen. Intern. Med. 29, 468–476 (2014).
Khanna, R. R., Victor, R. G., Bibbins-Domingo, K., Shapiro, M. F. & Pletcher, M. J. Missed opportunities for treatment of uncontrolled hypertension at physician office visits in the United States, 2005–2009. Arch. Intern. Med. 172, 1344–1345 (2012).
Jaffe, M. G., Lee, G. A., Young, J. D., Sidney, S. & Go, A. S. Improved blood pressure control associated with a large-scale hypertension program. JAMA 310, 699–705 (2013).
Egan, B. M., Zhao, Y., Axon, R. N., Brzezinski, W. A. & Ferdinand, K. C. Uncontrolled and apparent treatment resistant hypertension in the United States, 1988 to 2008. Circulation 124, 1046–1058 (2011).
Egan, B. M. et al. Prevalence of optimal treatment regimens in patients with apparent treatment-resistant hypertension based on office blood pressure in a community-based practice network. Hypertension 62, 691–697 (2013).
Weitzman, D., Chodick, G., Shalev, V., Grossman, C. & Grossman, E. Prevalence and factors associated with resistant hypertension in a large health maintenance organization in Israel. Hypertension 64, 501–507 (2014).
Calhoun, D. A. et al. Resistant hypertension: diagnosis, evaluation, and treatment. A scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Hypertension 51, 1403–1419 (2008).
Vongpatanasin, W. Resistant hypertension: a review of diagnosis and management. JAMA 311, 2216–2224 (2014).
Vongpatanasin, W., Kario, K., Atlas, S. A. & Victor, R. G. Central sympatholytic drugs. J. Clin. Hypertens. (Greenwich) 13, 658–661 (2011).
Guyenet, P. G. The sympathetic control of blood pressure. Nat. Rev. Neurosci. 7, 335–346 (2006).
Harrison, D. G. The immune system in hypertension. Trans. Am. Clin. Climatol. Assoc. 125, 130–138 (2014).
Sander, M., Hansen, J. & Victor, R. G. The sympathetic nervous system is involved in the maintenance but not initiation of the hypertension induced by Nω-nitro-L-arginine methyl ester. Hypertension 30, 64–70 (1997).
Grimson, K. S., Orgain, E. S. Anderson, B., Broome, R. A. & Longino, F. H. Results of treatment of patients with hypertension by total thoracic and partial to total lumbar sympathectomy, splanchnicectomy and celiac ganglionectomy. Ann. Surg. 129, 850–871 (1949).
Darr, R. et al. Pheochromocytoma—update on disease management. Ther. Adv. Endocrinol. Metab. 3, 11–26 (2012).
Guyton, A. C. Blood pressure control—special role of the kidneys and body fluids. Science 252, 1813–1816 (1991).
Lifton, R. P., Wilson, F. H., Choate, K. A. & Geller, D. S. Salt and blood pressure: new insight from human genetic studies. Cold Spring Harb. Symp. Quant. Biol. 67, 445–450 (2002).
Schlaich, M. P. et al. Sympathetic augmentation in hypertension: role of nerve firing, norepinephrine reuptake, and Angiotensin neuromodulation. Hypertension 43, 169–175 (2004).
Esler, M. Sympathetic nervous system moves toward center stage in cardiovascular medicine: from Thomas Willis to resistant hypertension. Hypertension 63, e25–e32 (2014).
DiBona, G. F. Physiology in perspective: the wisdom of the body. Neural control of the kidney. Am. J. Physiol. Regul. Integr. Comp. Physiol. 289, R633–R641 (2005).
Grassi, G. et al. Sympathetic and baroreflex cardiovascular control in hypertension-related left ventricular dysfunction. Hypertension 53, 205–209 (2009).
Grassi, G. et al. Marked sympathetic activation and baroreflex dysfunction in true resistant hypertension. Int. J. Cardiol. 177, 1020–1025 (2014).
Mancia, G. & Grassi, G. The autonomic nervous system and hypertension. Circ. Res. 114, 1804–1814 (2014).
Esler, M. Renal denervation for treatment of drug-resistant hypertension. Trends Cardiovasc. Med. 25, 107–115 (2015).
Mancia, G., Ludbrook, J., Ferrari, A., Gregorini, L. & Zanchetti, A. Baroreceptor reflexes in human hypertension. Circ. Res. 43, 170–177 (1978).
Guo, G. B., Thames, M. D. & Abboud, F. M. Arterial baroreflexes in renal hypertensive rabbits. Selectivity and redundancy of baroreceptor influence on heart rate, vascular resistance, and lumbar sympathetic nerve activity. Circ. Res. 53, 223–234 (1983).
Thrasher, T. N. Unloading arterial baroreceptors causes neurogenic hypertension. Am. J. Physiol. Regul. I ntegr. Comp. Physiol. 282, R1044–R1053 (2002).
Thrasher, T. N. Effects of chronic baroreceptor unloading on blood pressure in the dog. A m. J. Physiol. Regul. Integr. Comp. Physiol. 288, R863–R871 (2005).
Thrasher, T. N. Arterial baroreceptor input contributes to long-term control of blood pressure. Curr. Hypertens. Rep. 8, 249–254 (2006).
Carlsten, A., Folkow, B., Grimby, G., Hamberger, C. A. & Thulesius, O. Cardiovascular effects of direct stimulation of the carotid sinus nerve in man. Acta Physiol. Scand. 44, 138–145 (1958).
Bilgutay, A. M., Bigultay, I. & Lillehei, C. W. In Baroreceptors and Hypertension (ed. Kedzi, P.) 425–437 (Pergamon, 1967).
Bilgutay, A. M. & Lillehei, C. W. Surgical treatment of hypertension with reference to baropacing. Am. J. Cardiol. 17, 663–667 (1966).
Scheffers, I. J., Kroon, A. A. & de Leeuw, P. W. Carotid baroreflex activation: past, present, and future. Curr. Hypertens. Rep. 12, 61–66 (2010).
Braunwald, E. & Sobel, B. E. Braunwald NS: treatment of paroxysmal supraventricular trachycardia by electrical stimulation of the carotid-sinus nerves. N. Engl. J. Med. 281, 885–887 (1969).
Braunwald, E., Epstein, S. E., Glick, G., Wechsler, A. S. & Braunwald, N. S. Relief of angina pectoris by electrical stimulation of the carotid-sinus nerves. N. Engl. J. Med. 277, 1278–1283 (1967).
[No authors listed] Effects of treatment on morbidity in hypertension. Results in patients with diastolic blood pressures averaging 115 through 129 mm Hg. JAMA 202, 1028–1034 (1967).
Cowley, A. W. Jr, Liard, J. F. & Guyton, A. C. Role of baroreceptor reflex in daily control of arterial blood pressure and other variables in dogs. Circ. Res. 32, 564–576 (1973).
Lohmeier, T. E., Irwin, E. D., Rossing, M. A., Serdar, D. J. & Kieval, R. S. Prolonged activation of the baroreflex produces sustained hypotension. Hypertension 43, 306–311 (2004).
Tordoir, J. H. et al. An implantable carotid sinus baroreflex activating system: surgical technique and short-term outcome from a multi-center feasibility trial for the treatment of resistant hypertension. Eur. J. Vasc. Endovasc. Surg. 33, 414–421 (2007).
Brest, A. N., Wiener, L. & Bachrach, B. Bilateral carotid sinus nerve stimulation in the treatment of hypertension. Am. J. Cardiol. 29, 821–825 (1972).
Parsonnet, V., Rothfeld, E. L., Raman, K. V. & Myers, G. H. Electrical stimulation of the carotid sinus nerve. Surg. Clin. North Am. 49, 589–596 (1969).
Schmidli, J. et al. Acute device-based blood pressure reduction: electrical activation of the carotid baroreflex in patients undergoing elective carotid surgery. Vascular 15, 63–69 (2007).
Mohaupt, M. G., Schmidli, J. & Luft, F. C. Management of uncontrollable hypertension with a carotid sinus stimulation device. Hypertension 50, 825–828 (2007).
Wustmann, K. et al. Effects of chronic baroreceptor stimulation on the autonomic cardiovascular regulation in patients with drug-resistant arterial hypertension. Hypertension 54, 530–536 (2009).
Goldstein, D. S., Bentho, O., Park, M. Y. & Sharabi, Y. Low-frequency power of heart rate variability is not a measure of cardiac sympathetic tone but may be a measure of modulation of cardiac autonomic outflows by baroreflexes. Exp. Physiol. 96, 1255–1261 (2011).
Vongpatanasin, W., Taylor, J. A. & Victor, R. G. Effects of cocaine on heart rate variability in healthy subjects. Am. J. Cardiol. 93, 385–388 (2004).
Heusser, K. et al. Carotid baroreceptor stimulation, sympathetic activity, baroreflex function, and blood pressure in hypertensive patients. Hypertension 55, 619–626 (2010).
Scheffers, I. J. et al. Novel baroreflex activation therapy in resistant hypertension: results of a European multi-center feasibility study. J. Am. Coll. Cardiol. 56, 1254–1258 (2010).
Bisognano, J. D., Kaufman, C. L., Bach, D. S., Lovett, E. G. & de Leeuw, P. Improved cardiac structure and function with chronic treatment using an implantable device in resistant hypertension: results from European and United States trials of the Rheos system. J. Am. Coll. Cardiol. 57, 1787–1788 (2011).
Bisognano, J. D. et al. Baroreflex activation therapy lowers blood pressure in patients with resistant hypertension: results from the double-blind, randomized, placebo-controlled Rheos Pivotal Trial. J. Am. Coll. Cardiol. 58, 765–773 (2011).
Alnima, T., de Leeuw, P. W., Tan, F. E. & Kroon, A. A. Renal responses to long-term carotid baroreflex activation therapy in patients with drug-resistant hypertension. Hypertension 61, 1334–1339 (2013).
Bakris, G. L. et al. Baroreflex activation therapy provides durable benefit in patients with resistant hypertension: results of long-term follow-up in the Rheos Pivotal Trial. J. Am. Soc. Hypertens. 6, 152–158 (2012).
Bakris, G., Nadim, M., Haller, H., Lovett, E. & Bisognano, J. Baroreflex activation therapy safely reduces blood pressure for at least five years in a large resistant hypertension cohort [abstract]. J. Am. Soc. Hypertens. 8, e9 (2014).
Patel, H. C. et al. Magnitude of blood pressure reduction in the placebo arms of modern hypertension trials: implications for trials of renal denervation. Hypertension 65, 401–406 (2015).
Chapman, N. et al. Effect of spironolactone on blood pressure in subjects with resistant hypertension. Hypertension 49, 839–845 (2007).
de Leeuw, P. W. et al. Bilateral or unilateral stimulation for baroreflex activation therapy. Hypertension 65, 187–192 (2015).
Sagawa, K. & Watanabe, K. Summation of bilateral carotid sinus signals in the barostatic reflex. Am. J. Physiol. 209, 1278–1286 (1965).
Diedrich, A. et al. Lateralization of expression of neural sympathetic activity to the vessels and effects of carotid baroreceptor stimulation. Am. J. Physio l. Heart Circ. Physiol. 296, H1758–H1765 (2009).
Furlan, R. et al. Effects of unilateral and bilateral carotid baroreflex stimulation on cardiac and neural sympathetic discharge oscillatory patterns. Circulation 108, 717–723 (2003).
Tafil-Klawe, M., Raschke, F. & Hildebrandt, G. Functional asymmetry in carotid sinus cardiac reflexes in humans. Eur. J. Appl. Physiol. Occup. Physiol. 60, 402–405 (1990).
Alnima, T., de Leeuw, P. W. & Kroon, A. A. Baroreflex activation therapy for the treatment of drug-resistant hypertension: new developments. Cardiol. Res. Pract. 12, 1–7 (2012).
Hoppe, U. C. et al. Minimally invasive system for baroreflex activation therapy chronically lowers blood pressure with pacemaker-like safety profile: results from the Barostim neo trial. J. Am. Soc. Hypertens. 6, 270–276 (2012).
Borisenko, O., Beige, J., Lovett, E. G., Hoppe, U. C. & Bjessmo, S. Cost-effectiveness of Barostim therapy for the treatment of resistant hypertension in European settings. J. Hypertens. 32, 681–692 (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2015).
CVRx®. CVRx® rece ives CE mark approva l and introduces new implantable device for hypertension, the Barostim neo™ [online], (2011).
CVRx®. CVRx® granted CE mar king of the Barostim neo System™ for conditional MRI compatibility [online], (2014).
CVRx®. CVRx® granted humanitarian device exem ption approval from FDA for Barostim neo legacy™ hypertension therapy device [online], (2014).
CVRx®. CVRx® receives CE m ark approval of the Barostim neo System™ for the treatment of heart failure [online], (2014).
Halbach, M., Hickethier, T., Madershahian, N. & Muller-Ehmsen, J. Baroreflex activation therapy: a new treatment option for heart failure with reduced ejection fraction. Expert Rev. Cardiovasc. Ther. 12, 1465–1469 (2014).
Madershahian, N. et al. Baroreflex activation therapy in patients with pre-existing implantable cardioverter-defibrillator: compatible, complementary therapies. Europace 16, 861–865 (2014).
Gronda, E. et al. Chronic baroreflex activation effects on sympathetic nerve traffic, baroreflex function, and cardiac haemodynamics in heart failure: a proof-of-concept study. Eur. J. Heart Fail. 16, 977–983 (2014).
Barrett, E. J., Wang, H., Upchurch, C. T. & Liu, Z. Insulin regulates its own delivery to skeletal muscle by feed-forward actions on the vasculature. Am. J. Physiol. Endocrinol. Metab. 301, E252–E263 (2011).
Jamerson, K. A., Julius, S., Gudbrandsson, T., Andersson, O. & Brant, D. O. Reflex sympathetic activation induces acute insulin resistance in the human forearm. Hypertension 21, 618–623 (1993).
Haenni, A. & Lithell, H. Moxonidine improves insulin sensitivity in insulin-resistant hypertensives. J. Hypertens. Suppl. 17, S29–S35 (1999).
May, M. et al. Limited acute influences of electrical baroreceptor activation on insulin sensitivity and glucose delivery: a randomized, double-blind, crossover clinical study. Diabetes 63, 2833–2837 (2014).
Converse, R. L. Jr et al. Sympathetic overactivity in patients with chronic renal failure. N. Engl. J. Med. 327, 1912–1918 (1992).
Ligtenberg, G. et al. Reduction of sympathetic hyperactivity by enalapril in patients with chronic renal failure. N. Engl. J. Med. 340, 1321–1328 (1999).
Wallbach, M. et al. Impact of baroreflex activation therapy on renal function—a pilot study. Am. J. Nephrol. 40, 371–380 (2014).
Page, I. H. A syndrome simulating diencephalic stimulation occurring in patients with essential hypertension. Am. J. Med. Sci. 190, 9–14 (1935).
Eisenhofer, G., Sharabi, Y. & Pacak, K. Unexplained symptomatic paroxysmal hypertension in pseudopheochromocytoma: a stress response disorder? Ann. N. Y. Acad. Sci. 1148, 469–478 (2008).
Kuchel, O., Buu, N. T., Larochelle, P., Hamet, P. & Genest, J. Jr. Episodic dopamine discharge in paroxysmal hypertension. Page's syndrome revisited. Arch. Intern. Med. 146, 1315–1320 (1986).
Pickering, T. G. & Clemow, L. Paroxysmal hypertension: the role of stress and psychological factors. J. Clin. Hypertens. (Greenwich) 10, 575–581 (2008).
Hering, H. E. Die Karotissinus Reflex auf Herz und Gefasse [German] 1–150 (Steinkopff, 1927).
Koch, E. & Mies, H. Chronischer arterieller hochdruck durch experimentelle dauerausschaltung der blutdruckzügler [German]. Krankheitsforschung 7, 241–256 (1929).
Volhard, F. On the pathogenesis of essential hypertension and hypertrophy [German]. Schweiz Med. Wochenschr. 78, 1189 (1948).
Lown, B. & Levine, S. A. The carotid sinus. Clinical value of its stimulation. Circulation 23, 766–789 (1961).
Solti, F. et al. Baropacing of the carotid sinus nerve for treatment of “intractable” hypertension. Z. Kardiol. 64, 368–374 (1975).
Bristow, J. D., Honour, A. J., Pickering, G. W., Sleight, P. & Smyth, H. S. Diminished baroreflex sensitivity in high blood pressure. Circulation 39, 48–54 (1969).
Lu, Y. et al. The ion channel ASIC2 is required for baroreceptor and autonomic control of the circulation. Neuron 64, 885–897 (2009).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
R.V. declares that he is an Advisory Board member for Medtronic, Northwind, and TrophyNOD.
Rights and permissions
About this article
Cite this article
Victor, R. Carotid baroreflex activation therapy for resistant hypertension. Nat Rev Cardiol 12, 451–463 (2015). https://doi.org/10.1038/nrcardio.2015.96
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2015.96
- Springer Nature Limited
This article is cited by
-
Piezo1 channel activation facilitates baroreflex afferent neurotransmission with subsequent blood pressure reduction in control and hypertension rats
Acta Pharmacologica Sinica (2024)
-
Efficacy observation of pushing Qiaogong point with different intensities for treating stage I essential hypertension
Journal of Acupuncture and Tuina Science (2023)
-
Noninvasive sub-organ ultrasound stimulation for targeted neuromodulation
Nature Communications (2019)
-
Modulation of Sympathetic Overactivity to Treat Resistant Hypertension
Current Hypertension Reports (2018)
-
Endovascular Baroreflex Amplification for Resistant Hypertension
Current Hypertension Reports (2018)