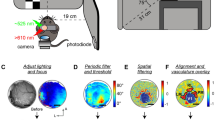
Abstract
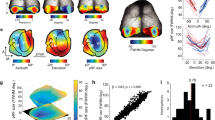
Intrinsic signal optical imaging (ISI) is a rapid and noninvasive method for observing brain activity in vivo over a large area of the cortex. Here we describe our protocol for mapping retinotopy to identify mouse visual cortical areas using ISI. First, surgery is performed to attach a head frame to the mouse skull (∼1 h). The next day, intrinsic activity across the visual cortex is recorded during the presentation of a full-field drifting bar in the horizontal and vertical directions (∼2 h). Horizontal and vertical retinotopic maps are generated by analyzing the response of each pixel during the period of the stimulus. Last, an algorithm uses these retinotopic maps to compute the visual field sign and coverage, and automatically construct visual borders without human input. Compared with conventional retinotopic mapping with episodic presentation of adjacent stimuli, a continuous, periodic stimulus is more resistant to biological artifacts. Furthermore, unlike manual hand-drawn approaches, we present a method for automatically segmenting visual areas, even in the small mouse cortex. This relatively simple procedure and accompanying open-source code can be implemented with minimal surgical and computational experience, and is useful to any laboratory wishing to target visual cortical areas in this increasingly valuable model system.




Similar content being viewed by others
References
Grinvald, A., Lieke, E., Frostig, R.D., Gilbert, C.D. & Wiesel, T.N. Functional architecture of cortex revealed by optical imaging of intrinsic signals. Nature 324, 361–364 (1986).
Malonek, D. et al. Vascular imprints of neuronal activity: relationships between the dynamics of cortical blood flow, oxygenation, and volume changes following sensory stimulation. Proc. Natl. Acad. Sci. USA 94, 14826–14831 (1997).
Attwell, D. & Laughlin, S.B. An energy budget for signaling in the grey matter of the brain. J. Cereb. Blood Flow Metab. 21, 1133–1145 (2001).
Frostig, R.D., Lieke, E.E., Ts'o, D.Y. & Grinvald, A. Cortical functional architecture and local coupling between neuronal activity and the microcirculation revealed by in vivo high-resolution optical imaging of intrinsic signals. Proc. Natl. Acad. Sci. USA 87, 6082–6086 (1990).
Shmuel, A. & Grinvald, A. Functional organization for direction of motion and its relationship to orientation maps in cat area 18. J. Neurosci. 16, 6945–6964 (1996).
Bonhoeffer, T. & Grinvald, A. Iso-orientation domains in cat visual cortex are arranged in pinwheel-like patterns. Nature 353, 429–431 (1991).
Ts'o, D.Y., Frostig, R.D., Lieke, E.E. & Grinvald, A. Functional organization of primate visual cortex revealed by high resolution optical imaging. Science 249, 417–420 (1990).
Garrett, M.E., Nauhaus, I., Marshel, J.H. & Callaway, E.M. Topography and areal organization of mouse visual cortex. J. Neurosci. 34, 12587–12600 (2014).
Sereno, M.I., McDonald, C.T. & Allman, J.M. Analysis of retinotopic maps in extrastriate cortex. Cereb. Cortex 4, 601–620 (1994).
Sereno, M.I. et al. Borders of multiple visual areas in humans revealed by functional magnetic resonance imaging. Science 268, 889–893 (1995).
Wandell, B.A. & Winawer, J. Imaging retinotopic maps in the human brain. Vision Res. 51, 718–737 (2011).
Sereno, M.I. & Allman, J.M. in The Neural Basis of Visual Function 160–172 (Macmillan, 1991).
Allman, J.M. & Kaas, J.H. A representation of the visual field in the caudal third of the middle temporal gyrus of the owl monkey (Aotus trivirgatus). Brain Res. 31, 85–105 (1971).
Kaas, J.H. in Extra-Striate Cortex in Primates 12, 91–119 (Springer, 1997).
Huberman, A.D. & Niell, C.M. What can mice tell us about how vision works? Trends Neurosci. 34, 464–473 (2011).
Niell, C.M. Exploring the next frontier of mouse vision. Neuron 72, 889–892 (2011).
Luo, L., Callaway, E.M. & Svoboda, K. Genetic dissection of neural circuits. Neuron 57, 634–660 (2008).
Marshel, J.H., Garrett, M.E., Nauhaus, I. & Callaway, E.M. Functional specialization of seven mouse visual cortical areas. Neuron 72, 1040–1054 (2011).
Juavinett, A.L. & Callaway, E.M. Pattern and component motion responses in mouse visual cortical areas. Curr. Biol. 25, 1759–1764 (2015).
Glickfeld, L.L., Andermann, M.L., Bonin, V. & Reid, R.C. Cortico-cortical projections in mouse visual cortex are functionally target specific. Nat. Neurosci. 16, 219–226 (2013).
Olcese, U., Iurilli, G. & Medini, P. Cellular and synaptic architecture of multisensory integration in the mouse neocortex. Neuron 1–15 (2013).
Raposo, D., Kaufman, M.T. & Churchland, A.K. A category-free neural population supports evolving demands during decision-making. Nat. Neurosci. 17, 1784–1792 (2014).
Vanni, M.P. & Murphy, T.H. Mesoscale transcranial spontaneous activity mapping in GCaMP3 transgenic mice reveals extensive reciprocal connections between areas of somatomotor cortex. J. Neurosci. 34, 15931–15946 (2014).
Polimeni, J.R., Granquist-Fraser, D., Wood, R.J. & Schwartz, E.L. Physical limits to spatial resolution of optical recording: clarifying the spatial structure of cortical hypercolumns. Proc. Natl. Acad. Sci. 102, 4158–4163 (2005).
Orbach, H.S. & Cohen, L.B. Optical monitoring of activity from many areas of the in vitro and in vivo salamander olfactory bulb: a new method for studying functional organization in the vertebrate central nervous system. J. Neurosci. 3, 2251–2262 (1983).
Das, A. & Gilbert, C.D. Long-range horizontal connections and their role in cortical reorganization revealed by optical recording of cat primary visual cortex. Nature 375, 780–784 (1995).
Kodandaramaiah, S.B. et al. Assembly and operation of the autopatcher for automated intracellular neural recording in vivo. Nat. Protoc. 11, 634–654 (2016).
Goldey, G.J. et al. Removable cranial windows for long-term imaging in awake mice. Nat. Protoc. 9, 2515–2538 (2014).
Holtmaat, A. et al. Long-term, high-resolution imaging in the mouse neocortex through a chronic cranial window. Nat. Protoc. 4, 1128–1144 (2009).
Yang, G., Pan, F., Parkhurst, C.N., Grutzendler, J. & Gan, W.-B. Thinned-skull cranial window technique for long-term imaging of the cortex in live mice. Nat. Protoc. 5, 201–208 (2010).
Goltstein, P.M., Montijn, J.S. & Pennartz, C.M.A. Effects of isoflurane anesthesia on ensemble patterns of Ca2+ activity in mouse V1: reduced direction selectivity independent of increased correlations in cellular activity. PLoS One 10, e0118277 (2015).
Villeneuve, M.Y. & Casanova, C. On the use of isoflurane versus halothane in the study of visual response properties of single cells in the primary visual cortex. J. Neurosci. Methods 129, 19–31 (2003).
Shtoyerman, E., Arieli, A., Slovin, H., Vanzetta, I. & Grinvald, A. Long-term optical imaging and spectroscopy reveal mechanisms underlying the intrinsic signal and stability of cortical maps in V1 of behaving monkeys. J. Neurosci. 20, 8111–8121 (2000).
Kalatsky, V.A. & Stryker, M.P. New paradigm for optical imaging: temporally encoded maps of intrinsic signal. Neuron 38, 529–545 (2003).
Malonek, D. & Grinvald, A. Interactions between electrical activity and cortical microcirculation revealed by imaging spectroscopy: implications for functional brain mapping. Science 272, 551–554 (1996).
Nauhaus, I. & Ringach, D.L. Precise alignment of micromachined electrode arrays with V1 functional maps. J. Neurophysiol. 97, 3781–3789 (2007).
Ratzlaff, E.H. & Grinvald, A. A tandem-lens epifluourescence macroscope: hundred-fold brightness advantage for wide field imaging. J. Neurosci. Methods 36, 127–137 (1991).
Silasi, G., Xiao, D., Vanni, M.P., Chen, A.C.N. & Murphy, T.H. Intact skull chronic windows for mesoscopic wide-field imaging in awake mice. J. Neurosci. Methods 267, 141–149 (2016).
Schuett, S., Bonhoeffer, T. & Hübener, M. Mapping retinotopic structure in mouse visual cortex with optical imaging. J. Neurosci. 22, 6549–6559 (2002).
Bandettini, P.A., Jesmanowicz, A., Wong, E.C. & Hyde, J.S. Processing strategies for time-course data sets in functional mri of the human brain. Magn. Reson. Med. 30, 161–173 (1993).
Engel, S., Glover, G.H. & Wandell, B.A. Retinotopic organization in human visual cortex and the spatial precision of functional MRI. Cereb. Cortex 7, 181–192 (1997).
Oommen, B.S. & Stahl, J.S. Eye orientation during static tilts and its relationship to spontaneous head pitch in the laboratory mouse. Brain Res. 1193, 57–66 (2008).
Acknowledgements
We thank the Callaway laboratory, especially P. Li, T. Ito, and E. Kim, for their help in developing this setup and protocol; D. Ringach (University of California, Los Angeles) for writing the initial version of the image acquisition code; and O. Odoemene (Cold Spring Harbor Laboratory) for encouraging the inception of this article. We would also like to thank the Allen Institute for Brain Science founders, Paul G. Allen and Jody Allen, for their vision, encouragement, and support. This work was supported by National Institutes of Health grants EY022577, MH063912, and EY019005, as well as grants from the Gatsby Charitable Foundation and the Rose Hills Foundation to M.E.G. A.L.J. was supported by the National Science Foundation and the Martinet Foundation.
Author information
Authors and Affiliations
Contributions
A.L.J., I.N., M.E.G., and E.M.C. designed the experiments, developed the protocol, and wrote the manuscript. A.L.J. and M.E.G. collected and analyzed intrinsic signal imaging data. I.N., M.E.G., and A.L.J. wrote MATLAB code. J.Z. wrote Python code.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Imager and Stimulator GUIs
a) Screenshot of Imager GUI for acquisition of intrinsic signal images b) Screenshot of Stimulator GUI, with three windows for stimulus modification and presentation. The ‘Main Window’ contains experiment information, the ‘Looper’ window allows you to choose which parameters to change from trial to trial, and the ‘paramSelect’ window shows a list of parameters that can be changed for each stimulus type.
Supplementary Figure 2 Analyzer File Structure
Diagram showing the structure of the .analyzer file that is saved during acquisition and contains all information about the experiment.
Supplementary information
Supplementary Figures and Text
Supplementary Figures 1 and 2 (PDF 479 kb)
Supplementary Data 1
Head frame 3D drawing file. Design file to allow reproduction of mouse head frame. (ZIP 1 kb)
Rights and permissions
About this article
Cite this article
Juavinett, A., Nauhaus, I., Garrett, M. et al. Automated identification of mouse visual areas with intrinsic signal imaging. Nat Protoc 12, 32–43 (2017). https://doi.org/10.1038/nprot.2016.158
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2016.158
- Springer Nature Limited
This article is cited by
-
Aggregation-induced T1/T2 contrast enhancement for magnetic resonance imaging-guided intraoperative surgery
Science China Chemistry (2024)
-
Parabolic avalanche scaling in the synchronization of cortical cell assemblies
Nature Communications (2023)
-
Enriched environment exposure during development positively impacts the structure and function of the visual cortex in mice
Scientific Reports (2023)
-
Acute head-fixed recordings in awake mice with multiple Neuropixels probes
Nature Protocols (2023)
-
Optogenetics for light control of biological systems
Nature Reviews Methods Primers (2022)