Abstract
Neuron-glia interactions establish functional membrane domains along myelinated axons. These include nodes of Ranvier, paranodal axoglial junctions and juxtaparanodes. Paranodal junctions are the largest vertebrate junctional adhesion complex, and they are essential for rapid saltatory conduction and contribute to assembly and maintenance of nodes. However, the molecular mechanisms underlying paranodal junction assembly are poorly understood. Ankyrins are cytoskeletal scaffolds traditionally associated with Na+ channel clustering in neurons and are important for membrane domain establishment and maintenance in many cell types. Here we show that ankyrin-B, expressed by Schwann cells, and ankyrin-G, expressed by oligodendrocytes, are highly enriched at the glial side of paranodal junctions where they interact with the essential glial junctional component neurofascin 155. Conditional knockout of ankyrins in oligodendrocytes disrupts paranodal junction assembly and delays nerve conduction during early development in mice. Thus, glial ankyrins function as major scaffolds that facilitate early and efficient paranodal junction assembly in the developing CNS.








Similar content being viewed by others
References
Poliak, S. & Peles, E. The local differentiation of myelinated axons at nodes of Ranvier. Nat. Rev. Neurosci. 4, 968–980 (2003).
Salzer, J.L. Polarized domains of myelinated axons. Neuron 40, 297–318 (2003).
Babbs, C.F. & Shi, R. Subtle paranodal injury slows impulse conduction in a mathematical model of myelinated axons. PLoS ONE 8, e67767 (2013).
Chang, K.J. & Rasband, M.N. Excitable domains of myelinated nerves: axon initial segments and nodes of Ranvier. Curr. Top. Membr. 72, 159–192 (2013).
Bennett, V. & Lorenzo, D.N. Spectrin- and ankyrin-based membrane domains and the evolution of vertebrates. Curr. Top. Membr. 72, 1–37 (2013).
Bennett, V. & Healy, J. Membrane domains based on ankyrin and spectrin associated with cell-cell interactions. Cold Spring Harb. Perspect. Biol. 1, a003012 (2009).
Kordeli, E., Lambert, S. & Bennett, V. AnkyrinG. A new ankyrin gene with neural-specific isoforms localized at the axonal initial segment and node of Ranvier. J. Biol. Chem. 270, 2352–2359 (1995).
Dzhashiashvili, Y. et al. Nodes of Ranvier and axon initial segments are ankyrin G-dependent domains that assemble by distinct mechanisms. J. Cell Biol. 177, 857–870 (2007).
Rasband, M.N. The axon initial segment and the maintenance of neuronal polarity. Nat. Rev. Neurosci. 11, 552–562 (2010).
Gasser, A. et al. An ankyrinG-binding motif is necessary and sufficient for targeting Nav1.6 sodium channels to axon initial segments and nodes of Ranvier. J. Neurosci. 32, 7232–7243 (2012).
Chan, W., Kordeli, E. & Bennett, V. 440-kD ankyrinB: structure of the major developmentally regulated domain and selective localization in unmyelinated axons. J. Cell Biol. 123, 1463–1473 (1993).
Galiano, M.R. et al. A distal axonal cytoskeleton forms an intra-axonal boundary that controls axon initial segment assembly. Cell 149, 1125–1139 (2012).
Ogawa, Y. et al. Spectrins and ankyrinB constitute a specialized paranodal cytoskeleton. J. Neurosci. 26, 5230–5239 (2006).
Rasband, M.N. et al. Dependence of nodal sodium channel clustering on paranodal axoglial contact in the developing CNS. J. Neurosci. 19, 7516–7528 (1999).
Scotland, P., Zhou, D., Benveniste, H. & Bennett, V. Nervous system defects of AnkyrinB−/− mice suggest functional overlap between the cell adhesion molecule L1 and 440-kD AnkyrinB in premyelinated axons. J. Cell Biol. 143, 1305–1315 (1998).
Lappe-Siefke, C. et al. Disruption of Cnp1 uncouples oligodendroglial functions in axonal support and myelination. Nat. Genet. 33, 366–374 (2003).
Horresh, I., Bar, V., Kissil, J.L. & Peles, E. Organization of myelinated axons by Caspr and Caspr2 requires the cytoskeletal adapter protein 4.1B. J. Neurosci. 30, 2480–2489 (2010).
Cifuentes-Diaz, C. et al. Protein 4.1B contributes to the organization of peripheral myelinated axons. PLoS ONE 6, e25043 (2011).
Zhang, C., Susuki, K., Zollinger, D.R., Dupree, J.L. & Rasband, M.N. Membrane domain organization of myelinated axons requires βII spectrin. J. Cell Biol. 203, 437–443 (2013).
Rios, J.C. et al. Paranodal interactions regulate expression of sodium channel subtypes and provide a diffusion barrier for the node of Ranvier. J. Neurosci. 23, 7001–7011 (2003).
Hoshi, T. et al. Nodal protrusions, increased Schmidt-Lanterman incisures, and paranodal disorganization are characteristic features of sulfatide-deficient peripheral nerves. Glia 55, 584–594 (2007).
Cahoy, J.D. et al. A transcriptome database for astrocytes, neurons, and oligodendrocytes: a new resource for understanding brain development and function. J. Neurosci. 28, 264–278 (2008).
Susuki, K. et al. Three mechanisms assemble central nervous system nodes of Ranvier. Neuron 78, 469–482 (2013).
Garver, T.D., Ren, Q., Tuvia, S. & Bennett, V. Tyrosine phosphorylation at a site highly conserved in the L1 family of cell adhesion molecules abolishes ankyrin binding and increases lateral mobility of neurofascin. J. Cell Biol. 137, 703–714 (1997).
Tuvia, S., Garver, T.D. & Bennett, V. The phosphorylation state of the FIGQY tyrosine of neurofascin determines ankyrin-binding activity and patterns of cell segregation. Proc. Natl. Acad. Sci. USA 94, 12957–12962 (1997).
Jenkins, S.M. et al. FIGQY phosphorylation defines discrete populations of L1 cell adhesion molecules at sites of cell-cell contact and in migrating neurons. J. Cell Sci. 114, 3823–3835 (2001).
Schafer, D.P., Bansal, R., Hedstrom, K.L., Pfeiffer, S.E. & Rasband, M.N. Does paranode formation and maintenance require partitioning of neurofascin 155 into lipid rafts? J. Neurosci. 24, 3176–3185 (2004).
Bhat, M.A. et al. Axon-glia interactions and the domain organization of myelinated axons requires neurexin IV/Caspr/paranodin. Neuron 30, 369–383 (2001).
Gollan, L., Salomon, D., Salzer, J.L. & Peles, E. Caspr regulates the processing of contactin and inhibits its binding to neurofascin. J. Cell Biol. 163, 1213–1218 (2003).
Zonta, B. et al. Glial and neuronal isoforms of neurofascin have distinct roles in the assembly of nodes of Ranvier in the central nervous system. J. Cell Biol. 181, 1169–1177 (2008).
Kunimoto, M. A neuron-specific isoform of brain ankyrin, 440-kD ankyrinB, is targeted to the axons of rat cerebellar neurons. J. Cell Biol. 131, 1821–1829 (1995).
Cunha, S.R., Le Scouarnec, S., Schott, J.J. & Mohler, P.J. Exon organization and novel alternative splicing of the human ANK2 gene: implications for cardiac function and human cardiac disease. J. Mol. Cell. Cardiol. 45, 724–734 (2008).
Zhou, D. et al. AnkyrinG is required for clustering of voltage-gated Na channels at axon initial segments and for normal action potential firing. J. Cell Biol. 143, 1295–1304 (1998).
Rueckert, E.H. et al. Cis-acting regulation of brain-specific ANK3 gene expression by a genetic variant associated with bipolar disorder. Mol. Psychiatry 18, 922–929 (2013).
Davis, J.Q., Lambert, S. & Bennett, V. Molecular composition of the node of Ranvier: identification of ankyrin-binding cell adhesion molecules neurofascin (mucin+/third FNIII domain-) and NrCAM at nodal axon segments. J. Cell Biol. 135, 1355–1367 (1996).
Tait, S. et al. An oligodendrocyte cell adhesion molecule at the site of assembly of the paranodal axo-glial junction. J. Cell Biol. 150, 657–666 (2000).
Susuki, K. & Rasband, M.N. Spectrin and ankyrin-based cytoskeletons at polarized domains in myelinated axons. Exp. Biol. Med. (Maywood) 233, 394–400 (2008).
Susuki, K. et al. Schwann cell spectrins modulate peripheral nerve myelination. Proc. Natl. Acad. Sci. USA 108, 8009–8014 (2011).
Hammarlund, M., Jorgensen, E.M. & Bastiani, M.J. Axons break in animals lacking β-spectrin. J. Cell Biol. 176, 269–275 (2007).
Feinberg, K. et al. A glial signal consisting of gliomedin and NrCAM clusters axonal Na+ channels during the formation of nodes of Ranvier. Neuron 65, 490–502 (2010).
Eisenbach, M. et al. Differential clustering of Caspr by oligodendrocytes and Schwann cells. J. Neurosci. Res. 87, 3492–3501 (2009).
Einheber, S. et al. The 4.1B cytoskeletal protein regulates the domain organization and sheath thickness of myelinated axons. Glia 61, 240–253 (2013).
Munro, S. Lipid rafts: elusive or illusive? Cell 115, 377–388 (2003).
Zhang, X., Davis, J.Q., Carpenter, S. & Bennett, V. Structural requirements for association of neurofascin with ankyrin. J. Biol. Chem. 273, 30785–30794 (1998).
Ren, Q. & Bennett, V. Palmitoylation of neurofascin at a site in the membrane-spanning domain highly conserved among the L1 family of cell adhesion molecules. J. Neurochem. 70, 1839–1849 (1998).
Shi, L. et al. Whole-genome sequencing in an autism multiplex family. Mol. Autism 4, 8 (2013).
Iqbal, Z. et al. Homozygous and heterozygous disruptions of ANK3: at the crossroads of neurodevelopmental and psychiatric disorders. Hum. Mol. Genet. 22, 1960–1970 (2013).
Yuan, A. et al. ANK3 as a risk gene for schizophrenia: new data in Han Chinese and meta analysis. Am. J. Med. Genet. B. Neuropsychiatr. Genet. 159B, 997–1005 (2012).
Leussis, M.P., Madison, J.M. & Petryshen, T.L. Ankyrin 3: genetic association with bipolar disorder and relevance to disease pathophysiology. Biol. Mood Anxiety Disord. 2, 18 (2012).
Sherman, D.L. et al. Neurofascins are required to establish axonal domains for saltatory conduction. Neuron 48, 737–742 (2005).
Jenkins, P.M. et al. E-cadherin polarity is determined by a multifunction motif mediating lateral membrane retention through ankyrin-G and apical-lateral transcytosis through clathrin. J. Biol. Chem. 288, 14018–14031 (2013).
Yang, Y., Lacas-Gervais, S., Morest, D.K., Solimena, M. & Rasband, M.N. βIV spectrins are essential for membrane stability and the molecular organization of nodes of Ranvier. J. Neurosci. 24, 7230–7240 (2004).
Rhodes, K.J., Keilbaugh, S.A., Barrezueta, N.X., Lopez, K.L. & Trimmer, J.S. Association and colocalization of K+ channel α- and β-subunit polypeptides in rat brain. J. Neurosci. 15, 5360–5371 (1995).
Schafer, D.P., Custer, A.W., Shrager, P. & Rasband, M.N. Early events in node of Ranvier formation during myelination and remyelination in the PNS. Neuron Glia Biol. 2, 69–79 (2006).
Ivanovic, A. et al. The cytoskeletal adapter protein 4.1G organizes the internodes in peripheral myelinated nerves. J. Cell Biol. 196, 337–344 (2012).
Chang, K.J., Susuki, K., Dours-Zimmermann, M.T., Zimmermann, D.R. & Rasband, M.N. Oligodendrocyte myelin glycoprotein does not influence node of Ranvier structure or assembly. J. Neurosci. 30, 14476–14481 (2010).
Susuki, K. et al. Gangliosides contribute to stability of paranodal junctions and ion channel clusters in myelinated nerve fibers. Glia 55, 746–757 (2007).
Radonić, A. et al. Guideline to reference gene selection for quantitative real-time PCR. Biochem. Biophys. Res. Commun. 313, 856–862 (2004).
Acknowledgements
E. Peles (Weizmann Institute of Science) provided NfascF/F mice and 4.1G antibodies. K.-A. Nave (Max Planck Institute of Experimental Medicine) provided Cnp-Cre mice. P.M. Jenkins (Duke University) provided AnkG 480/270 antibodies. This work was supported by grants from the US National Institutes of Health (NS069688 and NS044916 to M.N.R.; HL084583, HL083422 and HL114383 to P.J.M.), the National Multiple Sclerosis Society (M.N.R.) and the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation (M.N.R.). V.B. is an investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
M.N.R. and K.-J.C. conceived the project, designed the experiments and wrote the manuscript. D.R.Z. and K.S. did electrophysiology experiments and analyzed the data. D.R.Z. conducted the electron microscopy experiments. K.-J.C. did all other experiments and analyzed the data. P.J.M. and M.A.M. designed and constructed the Ank2F/F allele. D.L.S., P.J.B., E.C.C. and V.B. provided reagents and mice.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
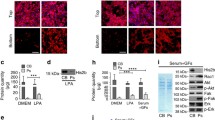
Supplementary Figure 1 Paranodal junction assembly is disrupted in the CNS of AnkB/G-cKO mice.
P8 spinal cord (a,b) and P17 brain (corpus callosum shown in c,d) were immunostained with antibodies as indicated. Scale bars = 10 μm (a,b) and 5 μm (c,d).
Supplementary Figure 2 The summary and implications of this study.
(a) The molecular architecture of nodes of Ranvier and paranodes including membrane proteins and cytoskeletal scaffolds. Paranodal ankyrins are located in myelinating glia and interact with NF155. Schwann cells in the PNS express AnkB (B) whereas oligodendrocytes in the CNS mainly express AnkG (G). The shorter tail of paranodal AnkG symbolizes 190-kDa and 270-kDa isoforms and the longer tail of nodal AnkG designates 480-kDa and 270-kDa isoforms. Cntn, contactin. αII/βII, αII/βII spectrin tetramer. βIV, βIV spectrin. This figure was reprinted from Chang & Rasband, Excitable domains of myelinated nerves: axon initial segments and nodes of Ranvier. Curr. Top. Membr. 72, 159-192 (2013) with permission from Elsevier. (b) The cartoon illustrates the differential timing of formation of nodes (red) and paranodal junctions (green) in the PNS, CNS and AnkB/G-cKO CNS.
Supplementary Figure 4 The full immunoblots of Figs. 3i, 7b, 7c and 8b.
The same membranes were used for stripping and reprobing in a top-to-bottom order except that Fig. 3i Contactin blot was from another immunoblotted membrane. Dotted lines indicate regions of the blots used in each figure.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 and Supplementary Tables 1 and 2 (PDF 1329 kb)
Supplementary Methods Checklist
(PDF 1630 kb)
Rights and permissions
About this article
Cite this article
Chang, KJ., Zollinger, D., Susuki, K. et al. Glial ankyrins facilitate paranodal axoglial junction assembly. Nat Neurosci 17, 1673–1681 (2014). https://doi.org/10.1038/nn.3858
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3858
- Springer Nature America, Inc.
This article is cited by
-
Mapping the expression of an ANK3 isoform associated with bipolar disorder in the human brain
Translational Psychiatry (2022)
-
Roles and mechanisms of ankyrin-G in neuropsychiatric disorders
Experimental & Molecular Medicine (2022)
-
CNS myelination requires VAMP2/3-mediated membrane expansion in oligodendrocytes
Nature Communications (2022)
-
Mechanisms of node of Ranvier assembly
Nature Reviews Neuroscience (2021)
-
ANK3 related neurodevelopmental disorders: expanding the spectrum of heterozygous loss-of-function variants
neurogenetics (2021)