Abstract
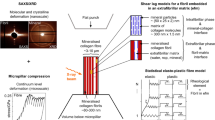
In human bone, an amorphous mineral serves as a precursor to the formation of a highly substituted nanocrystalline apatite. However, the precise role of this amorphous mineral remains unknown. Here, we show by using transmission electron microscopy that 100–300 nm amorphous calcium phosphate regions are present in the disordered phase of trabecular bone. Nanomechanical experiments on cylindrical samples, with diameters between 250 nm and 3,000 nm, of the bone’s ordered and disordered phases revealed a transition from plastic deformation to brittle failure and at least a factor-of-2 higher strength in the smaller samples. We postulate that this transition in failure mechanism is caused by the suppression of extrafibrillar shearing in the smaller samples, and that the emergent smaller-is-stronger size effect is related to the sample-size scaling of the distribution of flaws. Our findings should help in the understanding of the multi-scale nature of bone and provide insights into the biomineralization process.




Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.References
Ritchie, R. O. The conflicts between strength and toughness. Nature Mater. 10, 817–822 (2011).
Chen, P. Y., McKittrick, J. & Meyers, M. A. Biological materials: functional adaptations and bioinspired designs. Prog. Mater. Sci. 57, 1492–1704 (2012).
Wegst, U. G. K., Bai, H., Saiz, E., Tomsia, A. P. & Ritchie, R. O. Bioinspired structural materials. Nature Mater. 14, 23–36 (2014).
Ortiz, C. & Boyce, M. M. C. Bioinspired structural materials. Science 14, 1053–1054 (2008).
Currey, J. D. Hierarchies in biomineral structures. Science 309, 253–254 (2005).
Gao, H., Ji, B., Jager, I. L., Arzt, E. & Fratzl, P. Materials become insensitive to flaws at nanoscale: lessons from nature. Proc. Natl Acad. Sci. USA 100, 5597–5600 (2003).
Meyers, M. A., McKittrick, J. & Chen, P.-Y. Structural biological materials: critical mechanics–materials connections. Science 339, 773–779 (2013).
Pasteris, J. D. et al. Lack of OH in nanocrystalline apatite as a function of degree of atomic order: implications for bone and biomaterials. Biomaterials 25, 229–238 (2004).
Fantner, G. E. et al. Sacrificial bonds and hidden length dissipate energy as mineralized fibrils separate during bone fracture. Nature Mater. 4, 612–616 (2005).
Weiner, S., Traub, W. & Wagner, H. D. Lamellar bone: structure-function relations. J. Struct. Biol. 126, 241–255 (1999).
Gibson, L. J. & Asbhy, M. F. Cellular Solids: Structure and Properties (Cambridge Univ. Press, 1997).
Gibson, L. J. Biomechanics of cellular solids. J. Biomech. 38, 377–399 (2005).
Gal, A., Weiner, S. & Addadi, L. A perspective on underlying crystal growth mechanisms in biomineralization: solution mediated growth versus nanosphere particle accretion. CrystEngComm 17, 2606–2615 (2015).
Mahamid, J., Sharir, A., Addadi, L. & Weiner, S. Amorphous calcium phosphate is a major component of the forming fin bones of zebrafish: indications for an amorphous precursor phase. Proc. Natl Acad. Sci. USA 105, 12748–12753 (2008).
Mahamid, J. et al. Mapping amorphous calcium phosphate transformation into crystalline mineral from the cell to the bone in zebrafish fin rays. Proc. Natl Acad. Sci. USA 107, 6316–6321 (2010).
Wu, Y. et al. Nuclear magnetic resonance spin-spin relaxation of the crystals of bone, dental enamel, and synthetic hydroxyapatites. J. Bone Miner. Res. 17, 472–480 (2002).
Wang, Y. et al. Water-mediated structuring of bone apatite. Nature Mater. 12, 1144–1153 (2013).
Zimmermann, E. A. & Ritchie, R. O. Bone as a structural material. Adv. Healthc. Mater. 4, 1287–1304 (2015).
Jäger, I. & Fratzl, P. Mineralized collagen fibrils: a mechanical model with a staggered arrangement of mineral particles. Biophys. J. 79, 1737–1746 (2000).
Nair, A. K., Gautieri, A., Chang, S.-W. & Buehler, M. J. Molecular mechanics of mineralized collagen fibrils in bone. Nature Commun. 4, 1724 (2013).
Greer, J. R., Oliver, W. C. & Nix, W. D. Size dependence of mechanical properties of gold at the micron scale in the absence of strain gradients. Acta Mater. 53, 1821–1830 (2005).
Greer, J. R. & De Hosson, J. T. M. Plasticity in small-sized metallic systems: intrinsic versus extrinsic size effect. Prog. Mater. Sci. 56, 654–724 (2011).
Boskey, A. L. & Posner, A. S. Formation of hydroxyapatite at low supersaturation. J. Phys. Chem. 80, 40–45 (1976).
Hassenkam, T. et al. High-resolution AFM imaging of intact and fractured trabecular bone. Bone 35, 4–10 (2004).
Reznikov, N., Almany-Magal, R., Shahar, R. & Weiner, S. Three-dimensional imaging of collagen fibril organization in rat circumferential lamellar bone using a dual beam electron microscope reveals ordered and disordered sub-lamellar structures. Bone 52, 676–683 (2013).
Reznikov, N., Shahar, R. & Weiner, S. Three-dimensional structure of human lamellar bone: the presence of two different materials and new insights into the hierarchical organization. Bone 59, 93–104 (2014).
Reznikov, N., Chase, H., Brumfeld, V., Shahar, R. & Weiner, S. The 3D structure of the collagen fibril network in human trabecular bone: relation to trabecular organization. Bone 71, 189–195 (2015).
Currey, J. D. Bones: Structure and Mechanics (Princeton Univ. Review, 1984).
Nassif, N. et al. Self-assembled collagen-apatite matrix with bone-like hierarchy. Chem. Mater. 22, 3307–3309 (2010).
Giraud-Guille, M. M. Liquid crystallinity in condensed type I collagen solutions. A clue to the packing of collagen in extracellular matrices. J. Mol. Biol. 224, 861–873 (1992).
Wang, Y. et al. The predominant role of collagen in the nucleation, growth, structure and orientation of bone apatite. Nature Mater. 11, 724–733 (2012).
Nudelman, F. et al. The role of collagen in bone apatite formation in the presence of hydroxyapatite nucleation inhibitors. Nature Mater. 9, 1004–1009 (2010).
Gupta, H. S. et al. Nanoscale deformation mechanisms in bone. Nano Lett. 5, 2108–2111 (2005).
Gupta, H. S. et al. Fibrillar level fracture in bone beyond the yield point. Int. J. Fract. 139, 425–436 (2006).
Gupta, H. S. et al. Cooperative deformation of mineral and collagen in bone at the nanoscale. Proc. Natl Acad. Sci. USA 103, 17741–17746 (2006).
Schwiedrzik, J. et al. In situ micropillar compression reveals superior strength and ductility but an absence of damage in lamellar bone. Nature Mater. 13, 1–8 (2014).
Faingold, A. et al. The effect of hydration on mechanical anisotropy, topography and fibril organization of the osteonal lamellae. J. Biomech. 47, 367–372 (2014).
Inglis, C. E. Stresses in a plate due to the presence of cracks and sharp corners. Trans. I. N.A. 55, 219–241 (1913).
Hansma, P. K. et al. Sacrificial bonds in the interfibrillar matrix of bone. J. Musculoskelet. Neuronal Interact. 5, 313–315 (2005).
Thompson, J. B. et al. Bone indentation recovery time correlates with bond reforming time. Nature 414, 773–776 (2001).
Currey, J. D. The structure and mechanics of bone. J. Mater. Sci. 47, 41–54 (2012).
Chen, P. Y., Toroian, D., Price, P. A. & McKittrick, J. Minerals form a continuum phase in mature cancellous bone. Calcif. Tissue Int. 88, 351–361 (2011).
Alexander, B. et al. The nanometre-scale physiology of bone: steric modelling and scanning transmission electron microscopy of collagen-mineral structure. J. R. Soc. Interface 9, 1774–1786 (2012).
Acknowledgements
We thank M. Jett for help acquiring bone, C.-L. Guo for help with sample preparation and C. Garland for help with TEM. The authors are grateful for the financial support of the Institute for Collaborative Biotechnologies through grant W911NF-09-0001 from the US Army Research Office and the National Science Foundation through O.A.T.’s Graduate Research Fellowship Program (NSF GFRP). The content of the information does not necessarily reflect the position or the policy of the Government, and no official endorsement should be inferred.
Author information
Authors and Affiliations
Contributions
J.R.G. and O.A.T. designed the experiments. O.A.T. performed the experiments, analysed the data, and developed the model. O.A.T. and J.R.G. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1661 kb)
Supplementary Information
Supplementary movie 1 (AVI 12766 kb)
Supplementary Information
Supplementary movie 2 (AVI 9752 kb)
Rights and permissions
About this article
Cite this article
Tertuliano, O., Greer, J. The nanocomposite nature of bone drives its strength and damage resistance. Nature Mater 15, 1195–1202 (2016). https://doi.org/10.1038/nmat4719
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat4719
- Springer Nature Limited
This article is cited by
-
Glycerol solutions of highly concentrated biomineral counter-ions towards water-responsive mineralization: Demonstration on bacterial cellulose and its application in hard tissue repair
Nano Research (2023)
-
Bone architecture, bone material properties, and bone turnover in non-osteoporotic post-menopausal women with fragility fracture
Osteoporosis International (2022)
-
Cobalt-containing calcium phosphate induces resorption of biomineralized collagen by human osteoclasts
Biomaterials Research (2021)
-
Advances in biomineralization-inspired materials for hard tissue repair
International Journal of Oral Science (2021)
-
Aggravated stress fluctuation and mechanical size effects of nanoscale lamellar bone pillars
NPG Asia Materials (2021)