Abstract
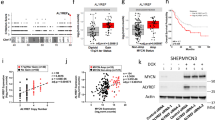
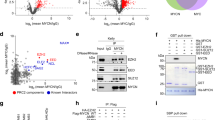
The MYCN proto-oncogene is amplified in a number of advanced-stage human tumors, such as neuroblastomas. Similar to other members of the MYC family of oncoproteins, MYCN (also known as N-Myc) is a transcription factor, and its stability and activity are tightly controlled by ubiquitination-dependent proteasome degradation1,2,3,4. Although numerous studies have demonstrated that N-Myc is a driver of neuroblastoma tumorigenesis, therapies that directly suppress N-Myc activity in human tumors are limited. Here we have identified ubiquitin-specific protease 7 (USP7; also known as HAUSP)5,6,7 as a regulator of N-Myc function in neuroblastoma. HAUSP interacts with N-Myc, and HAUSP expression induces deubiquitination and subsequent stabilization of N-Myc. Conversely, RNA interference (RNAi)-mediated knockdown of USP7 in neuroblastoma cancer cell lines, or genetic ablation of Usp7 in the mouse brain, destabilizes N-Myc, which leads to inhibition of N-Myc function. Notably, HAUSP is more abundant in patients with neuroblastoma who have poorer prognosis, and HAUSP expression substantially correlates with N-Myc transcriptional activity. Furthermore, small-molecule inhibitors of HAUSP's deubiquitinase activity markedly suppress the growth of MYCN-amplified human neuroblastoma cell lines in xenograft mouse models. Taken together, our findings demonstrate a crucial role of HAUSP in regulating N-Myc function in vivo and suggest that HAUSP inhibition is a potential therapy for MYCN-amplified tumors.




Similar content being viewed by others
References
Dang, C.V. MYC on the path to cancer. Cell 149, 22–35 (2012).
Conacci-Sorrell, M., McFerrin, L. & Eisenman, R.N. An overview of MYC and its interactome. Cold Spring Harb. Perspect. Med. 4, a014357 (2014).
Popov, N. et al. The ubiquitin-specific protease USP28 is required for MYC stability. Nat. Cell Biol. 9, 765–774 (2007).
Otto, T. et al. Stabilization of N-Myc is a critical function of aurora A in human neuroblastoma. Cancer Cell 15, 67–78 (2009).
Li, M. et al. Deubiquitination of p53 by HAUSP is an important pathway for p53 stabilization. Nature 416, 648–653 (2002).
Li, M., Brooks, C.L., Kon, N. & Gu, W. A dynamic role of HAUSP in the p53–Mdm2 pathway. Mol. Cell 13, 879–886 (2004).
Hu, M. et al. Crystal structure of a UBP family deubiquitinating enzyme in isolation and in complex with ubiquitin aldehyde. Cell 111, 1041–1054 (2002).
Nicholson, B. & Suresh Kumar, K.G. The multifaceted roles of USP7: new therapeutic opportunities. Cell Biochem. Biophys. 60, 61–68 (2011).
van der Horst, A. et al. FOXO4 transcriptional activity is regulated by monoubiquitination and USP7 (HAUSP). Nat. Cell Biol. 8, 1064–1073 (2006).
Song, M.S. et al. The deubiquitinylation and localization of PTEN are regulated by a HAUSP–PML network. Nature 455, 813–817 (2008).
Du, Z. et al. DNMT1 stability is regulated by proteins coordinating deubiquitination and acetylation-driven ubiquitination. Sci. Signal. 3, ra80 (2010).
Faesen, A.C. et al. Mechanism of USP7 (HAUSP) activation by its C-terminal ubiquitin-like domain and allosteric regulation by GMP synthetase. Mol. Cell 44, 147–159 (2011).
Lee, H.R. et al. Bilateral inhibition of HAUSP deubiquitinase by a viral interferon regulatory factor protein. Nat. Struct. Mol. Biol. 18, 1336–1344 (2011).
Ma, H. et al. M phase phosphorylation of the epigenetic regulator UHRF1 regulates its physical association with the deubiquitylase USP7 and stability. Proc. Natl. Acad. Sci. USA 109, 4828–4833 (2012).
Colleran, A. et al. Deubiquitination of NF-κB by ubiquitin-specific protease 7 promotes transcription. Proc. Natl. Acad. Sci. USA 110, 618–623 (2013).
Cheng, C., Niu, C., Yang, Y., Wang, Y. & Lu, M. Expression of HAUSP in gliomas correlates with disease progression and survival of patients. Oncol. Rep. 29, 1730–1736 (2013).
Pfoh, R., Lacdao, I.K. & Saridakis, V. Deubiquitinases and the new therapeutic opportunities offered to cancer. Endocr. Relat. Cancer 22, T35–T54 (2015).
Kon, N. et al. Roles of HAUSP-mediated p53 regulation in central nervous system development. Cell Death Differ. 18, 1366–1375 (2011).
Eilers, M. & Eisenman, R.N. Myc's broad reach. Genes Dev. 22, 2755–2766 (2008).
Dang, C.V. MYC, metabolism, cell growth and tumorigenesis. Cold Spring Harb. Perspect. Med. 3, a014217 (2013).
Charron, J. et al. Embryonic lethality in mice homozygous for a targeted disruption of the N-myc gene. Genes Dev. 6, 12A, 2248–2257 (1992).
Stanton, B.R., Perkins, A.S., Tessarollo, L., Sassoon, D.A. & Parada, L.F. Loss of N-myc function results in embryonic lethality and failure of the epithelial component of the embryo to develop. Genes Dev. 6, 12A, 2235–2247 (1992).
Sawai, S. et al. Defects of embryonic organogenesis resulting from targeted disruption of the N-myc gene in the mouse. Development 117, 1445–1455 (1993).
Knoepfler, P.S., Cheng, P.F. & Eisenman, R.N. N-myc is essential during neurogenesis for the rapid expansion of progenitor cell populations and the inhibition of neuronal differentiation. Genes Dev. 16, 2699–2712 (2002).
Arvanitis, C. & Felsher, D.W. Conditional transgenic models define how MYC initiates and maintains tumorigenesis. Semin. Cancer Biol. 16, 313–317 (2006).
Colland, F. et al. Small-molecule inhibitor of USP7 (HAUSP) ubiquitin protease stabilizes and activates p53 in cells. Mol. Cancer Ther. 8, 2286–2295 (2009).
Altun, M. et al. Activity-based chemical proteomics accelerates inhibitor development for deubiquitylating enzymes. Chem. Biol. 18, 1401–1412 (2011).
Reverdy, C. et al. Discovery of specific inhibitors of human USP7 (HAUSP) deubiquitinating enzyme. Chem. Biol. 19, 467–477 (2012).
Chauhan, D. et al. A small-molecule inhibitor of ubiquitin-specific protease 7 induces apoptosis in multiple myeloma cells and overcomes bortezomib resistance. Cancer Cell 22, 345–358 (2012).
Pugh, T.J. et al. The genetic landscape of high-risk neuroblastoma. Nat. Genet. 45, 279–284 (2013).
Valentijn, L.J. et al. Functional MYCN signature predicts outcome of neuroblastoma irrespective of MYCN amplification. Proc. Natl. Acad. Sci. USA 109, 19190–19195 (2012).
Kim, E.S. et al. Potent VEGF blockade causes regression of coopted vessels in a model of neuroblastoma. Proc. Natl. Acad. Sci. USA 99, 11399–11404 (2002).
Choi, S.H., Wright, J.B., Gerber, S.A. & Cole, M.D. Myc protein is stabilized by suppression of a novel E3 ligase complex in cancer cells. Genes Dev. 24, 1236–1241 (2010).
Sjostrom, S.K., Finn, G., Hahn, W.C., Rowitch, D.H. & Kenney, A.M. The Cdk1 complex plays a prime role in regulating N-myc phosphorylation and turnover in neural precursors. Dev. Cell 9, 327–338 (2005).
Welcker, M. et al. The Fbw7 tumor suppressor regulates glycogen synthase kinase 3 phosphorylation–dependent c-Myc protein degradation. Proc. Natl. Acad. Sci. USA 101, 9085–9090 (2004).
Zhao, X. et al. The HECT-domain ubiquitin ligase Huwe1 controls neural differentiation and proliferation by destabilizing the N-Myc oncoprotein. Nat. Cell Biol. 10, 643–653 (2008).
Brockmann, M. et al. Small-molecule inhibitors of aurora-A induce proteasomal degradation of N-myc in childhood neuroblastoma. Cancer Cell 24, 75–89 (2013).
Gustafson, W.C. et al. Drugging MYCN through an allosteric transition in aurora kinase A. Cancer Cell 26, 414–427 (2014).
Cummins, J.M. & Vogelstein, B. HAUSP is required for p53 destabilization. Cell Cycle 3, 689–692 (2004).
Kon, N. et al. Inactivation of HAUSP in vivo modulates p53 function. Oncogene 29, 1270–1279 (2010).
Rader, J. et al. Dual CDK4–CDK6 inhibition induces cell-cycle arrest and senescence in neuroblastoma. Clin. Cancer Res. 19, 6173–6182 (2013).
Alvarez, M.J. et al. Functional characterization of somatic mutations in cancer using network-based inference of protein activity. Nat. Genet. 48, 838–847 (2016).
Acknowledgements
The authors would like to thank J. Maris (Children's Hospital of Philadelphia) for providing cell lines. This work was supported by the National Cancer Institute, US National Institutes of Health (NIH) (grant no. 5R01CA193890, 5RO1CA190477, 5RO1CA085533 and 2P01CA080058; all to W.G.) and was partially supported by the NIH cancer biology training grant T32-CA09503 (O.T.). The content of this paper is solely the responsibility of the authors and does not necessarily represent the official views of the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
The whole project was conceived and designed by O.T. and W.G.; experiments were performed mainly by O.T., D.L. and C.D.; bioinformatic analysis was performed by G.L. and A.C.; and some of the experiments were performed with help from D.B., N.K., C.C., H.S. and D.J.Y. The paper was written by O.T. and W.G.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Table 1 (PDF 2595 kb)
Rights and permissions
About this article
Cite this article
Tavana, O., Li, D., Dai, C. et al. HAUSP deubiquitinates and stabilizes N-Myc in neuroblastoma. Nat Med 22, 1180–1186 (2016). https://doi.org/10.1038/nm.4180
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4180
- Springer Nature America, Inc.
This article is cited by
-
Ubiquitin specific peptidase 38 epigenetically regulates KLF transcription factor 5 to augment malignant progression of lung adenocarcinoma
Oncogene (2024)
-
USP43 stabilizes c-Myc to promote glycolysis and metastasis in bladder cancer
Cell Death & Disease (2024)
-
CRISPR/Cas9-based genome-wide screening of the deubiquitinase subfamily identifies USP3 as a protein stabilizer of REST blocking neuronal differentiation and promotes neuroblastoma tumorigenesis
Journal of Experimental & Clinical Cancer Research (2023)
-
UBE4B interacts with the ITCH E3 ubiquitin ligase to induce Ku70 and c-FLIPL polyubiquitination and enhanced neuroblastoma apoptosis
Cell Death & Disease (2023)
-
Deficiency of BAP1 inhibits neuroblastoma tumorigenesis through destabilization of MYCN
Cell Death & Disease (2023)