Abstract
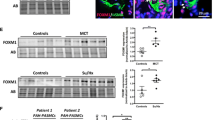
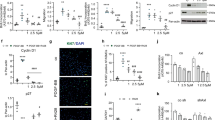
Pulmonary hypertension (PH) is characterized by increased proliferation and apoptosis resistance of pulmonary artery smooth muscle cells (PASMCs). Forkhead box O (FoxO) transcription factors are key regulators of cellular proliferation. Here we show that in pulmonary vessels and PASMCs of human and experimental PH lungs, FoxO1 expression is downregulated and FoxO1 is inactivated via phosphorylation and nuclear exclusion. These findings could be reproduced using ex vivo exposure of PASMCs to growth factors and inflammatory cytokines. Pharmacological inhibition and genetic ablation of FoxO1 in smooth muscle cells reproduced PH features in vitro and in vivo. Either pharmacological reconstitution of FoxO1 activity using intravenous or inhaled paclitaxel, or reconstitution of the transcriptional activity of FoxO1 by gene therapy, restored the physiologically quiescent PASMC phenotype in vitro, linked to changes in cell cycle control and bone morphogenic protein receptor type 2 (BMPR2) signaling, and reversed vascular remodeling and right-heart hypertrophy in vivo. Thus, PASMC FoxO1 is a critical integrator of multiple signaling pathways driving PH, and reconstitution of FoxO1 activity offers a potential therapeutic option for PH.






Similar content being viewed by others
References
D'Alonzo, G.E. et al. Survival in patients with primary pulmonary hypertension. Results from a national prospective registry. Ann. Intern. Med. 115, 343–349 (1991).
Seeger, W. & Pullamsetti, S.S. Mechanics and mechanisms of pulmonary hypertension—conference summary and translational perspectives. Pulm. Circ. 3, 128–136 (2013).
Olschewski, H. et al. Inhaled iloprost for severe pulmonary hypertension. N. Engl. J. Med. 347, 322–329 (2002).
Galiè, N. et al. Sildenafil citrate therapy for pulmonary arterial hypertension. N. Engl. J. Med. 353, 2148–2157 (2005).
Rubin, L.J. et al. Bosentan therapy for pulmonary arterial hypertension. N. Engl. J. Med. 346, 896–903 (2002).
Benza, R.L. et al. An evaluation of long-term survival from time of diagnosis in pulmonary arterial hypertension from the REVEAL Registry. Chest 142, 448–456 (2012).
Schermuly, R.T., Ghofrani, H.A., Wilkins, M.R. & Grimminger, F. Mechanisms of disease: pulmonary arterial hypertension. Nat. Rev. Cardiol. 8, 443–455 (2011).
Grimminger, F., Schermuly, R.T. & Ghofrani, H.A. Targeting non-malignant disorders with tyrosine kinase inhibitors. Nat. Rev. Drug Discov. 9, 956–970 (2010).
Schermuly, R.T. et al. Reversal of experimental pulmonary hypertension by PDGF inhibition. J. Clin. Invest. 115, 2811–2821 (2005).
Pullamsetti, S.S. et al. Role of Src tyrosine kinases in experimental pulmonary hypertension. Arterioscler. Thromb. Vasc. Biol. 32, 1354–1365 (2012).
Merklinger, S.L., Jones, P.L., Martinez, E.C. & Rabinovitch, M. Epidermal growth factor receptor blockade mediates smooth muscle cell apoptosis and improves survival in rats with pulmonary hypertension. Circulation 112, 423–431 (2005).
Archer, S.L. et al. Mitochondrial metabolism, redox signaling, and fusion: a mitochondria-ROS-HIF-1α-Kv1.5 O2-sensing pathway at the intersection of pulmonary hypertension and cancer. Am. J. Physiol. Heart Circ. Physiol. 294, H570–H578 (2008).
Gomez-Arroyo, J. et al. Metabolic gene remodeling and mitochondrial dysfunction in failing right ventricular hypertrophy due to pulmonary arterial hypertension. Circ. Heart Fail. 6, 136–144 (2012).
Tuder, R.M., Davis, L.A. & Graham, B.B. Targeting energetic metabolism: a new frontier in the pathogenesis and treatment of pulmonary hypertension. Am. J. Respir. Crit. Care Med. 185, 260–266 (2012).
Ghofrani, H.A., Seeger, W. & Grimminger, F. Imatinib for the treatment of pulmonary arterial hypertension. N. Engl. J. Med. 353, 1412–1413 (2005).
Hoeper, M.M. et al. Imatinib mesylate as add-on therapy for pulmonary arterial hypertension: results of the randomized IMPRES study. Circulation 127, 1128–1138 (2013).
Savai, R. et al. Immune and inflammatory cell involvement in the pathology of idiopathic pulmonary arterial hypertension. Am. J. Respir. Crit. Care Med. 186, 897–908 (2012).
Yeager, M.E. et al. Circulating myeloid-derived suppressor cells are increased and activated in pulmonary hypertension. Chest 141, 944–952 (2012).
Stacher, E. et al. Modern age pathology of pulmonary arterial hypertension. Am. J. Respir. Crit. Care Med. 186, 261–272 (2012).
Price, L.C. et al. Inflammation in pulmonary arterial hypertension. Chest 141, 210–221 (2012).
Kherbeck, N. et al. The role of inflammation and autoimmunity in the pathophysiology of pulmonary arterial hypertension. Clin. Rev. Allergy Immunol. 44, 31–38 (2013).
Rabinovitch, M. Molecular pathogenesis of pulmonary arterial hypertension. J. Clin. Invest. 118, 2372–2379 (2008).
Pullamsetti, S.S. et al. Inhibition of microRNA-17 improves lung and heart function in experimental pulmonary hypertension. Am. J. Respir. Crit. Care Med. 185, 409–419 (2012).
Rabinovitch, M. Molecular pathogenesis of pulmonary arterial hypertension. J. Clin. Invest. 122, 4306–4313 (2012).
Accili, D. & Arden, K.C. FoxOs at the crossroads of cellular metabolism, differentiation, and transformation. Cell 117, 421–426 (2004).
Hosaka, T. et al. Disruption of forkhead transcription factor (FOXO) family members in mice reveals their functional diversification. Proc. Natl. Acad. Sci. USA 101, 2975–2980 (2004).
Tran, H., Brunet, A., Griffith, E.C. & Greenberg, M.E. The many forks in FOXO's road. Sci. STKE 2003, RE5 (2003).
Eijkelenboom, A. & Burgering, B.M. FOXOs: signalling integrators for homeostasis maintenance. Nat. Rev. Mol. Cell Biol. 14, 83–97 (2013).
Calnan, D.R. & Brunet, A. The FoxO code. Oncogene 27, 2276–2288 (2008).
Oellerich, M.F. & Potente, M. FOXOs and sirtuins in vascular growth, maintenance, and aging. Circ. Res. 110, 1238–1251 (2012).
Paik, J.H. FOXOs in the maintenance of vascular homoeostasis. Biochem. Soc. Trans. 34, 731–734 (2006).
Mahajan, S.G. et al. A novel function of FoxO transcription factors in thrombin-stimulated vascular smooth muscle cell proliferation. Thromb. Haemost. 108, 148–158 (2012).
Paik, J.H. et al. FoxOs are lineage-restricted redundant tumor suppressors and regulate endothelial cell homeostasis. Cell 128, 309–323 (2007).
Zhao, Y., Wang, Y. & Zhu, W.G. Applications of post-translational modifications of FoxO family proteins in biological functions. J. Mol. Cell Biol. 3, 276–282 (2011).
Nagashima, T. et al. Discovery of novel forkhead box O1 inhibitors for treating type 2 diabetes: improvement of fasting glycemia in diabetic db/db mice. Mol. Pharmacol. 78, 961–970 (2010).
Van Der Heide, L.P., Hoekman, M.F. & Smidt, M.P. The ins and outs of FoxO shuttling: mechanisms of FoxO translocation and transcriptional regulation. Biochem. J. 380, 297–309 (2004).
Schroeder, F.C., Kau, T.R., Silver, P.A. & Clardy, J. The psammaplysenes, specific inhibitors of FOXO1a nuclear export. J. Nat. Prod. 68, 574–576 (2005).
Kau, T.R. et al. A chemical genetic screen identifies inhibitors of regulated nuclear export of a Forkhead transcription factor in PTEN-deficient tumor cells. Cancer Cell 4, 463–476 (2003).
Scagliotti, G.V. et al. International, randomized, placebo-controlled, double-blind phase III study of motesanib plus carboplatin/paclitaxel in patients with advanced nonsquamous non-small-cell lung cancer: MONET1. J. Clin. Oncol. 30, 2829–2836 (2012).
Goto, T., Takano, M., Hirata, J. & Tsuda, H. The involvement of FOXO1 in cytotoxic stress and drug-resistance induced by paclitaxel in ovarian cancers. Br. J. Cancer 98, 1068–1075 (2008).
Deng, Z. et al. Familial primary pulmonary hypertension (gene PPH1) is caused by mutations in the bone morphogenetic protein receptor-II gene. Am. J. Hum. Genet. 67, 737–744 (2000).
Atkinson, C. et al. Primary pulmonary hypertension is associated with reduced pulmonary vascular expression of type II bone morphogenetic protein receptor. Circulation 105, 1672–1678 (2002).
Yang, J. et al. Id proteins are critical downstream effectors of BMP signaling in human pulmonary arterial smooth muscle cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 305, L312–L321 (2013).
Frid, M.G. et al. Sustained hypoxia leads to the emergence of cells with enhanced growth, migratory, and promitogenic potentials within the distal pulmonary artery wall. Am. J. Physiol. Lung Cell. Mol. Physiol. 297, L1059–L1072 (2009).
Tu, L. et al. Autocrine fibroblast growth factor-2 signaling contributes to altered endothelial phenotype in pulmonary hypertension. Am. J. Respir. Cell Mol. Biol. 45, 311–322 (2011).
Voelkel, N.F. et al. Janus face of vascular endothelial growth factor: the obligatory survival factor for lung vascular endothelium controls precapillary artery remodeling in severe pulmonary hypertension. Crit. Care Med. 30, S251–S256 (2002).
Humbert, M. et al. Increased interleukin-1 and interleukin-6 serum concentrations in severe primary pulmonary hypertension. Am. J. Respir. Crit. Care Med. 151, 1628–1631 (1995).
Dorfmüller, P., Perros, F., Balabanian, K. & Humbert, M. Inflammation in pulmonary arterial hypertension. Eur. Respir. J. 22, 358–363 (2003).
Brunet, A. et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96, 857–868 (1999).
Taraseviciene-Stewart, L. et al. Inhibition of the VEGF receptor 2 combined with chronic hypoxia causes cell death–dependent pulmonary endothelial cell proliferation and severe pulmonary hypertension. FASEB J. 15, 427–438 (2001).
Lang, M. et al. The soluble guanylate cyclase stimulator riociguat ameliorates pulmonary hypertension induced by hypoxia and SU5416 in rats. PLoS ONE 7, e43433 (2012).
Toba, M. et al. Temporal hemodynamic and histological progression in Sugen5416/hypoxia/normoxia-exposed pulmonary arterial hypertensive rats. Am. J. Physiol. Heart Circ. Physiol. 306, H243–H250 (2014).
Piao, L. et al. FOXO1-mediated upregulation of pyruvate dehydrogenase kinase-4 (PDK4) decreases glucose oxidation and impairs right ventricular function in pulmonary hypertension: therapeutic benefits of dichloroacetate. J. Mol. Med. 91, 333–346 (2013).
Milas, L., Saito, Y., Hunter, N., Milross, C.G. & Mason, K.A. Therapeutic potential of paclitaxel-radiation treatment of a murine ovarian carcinoma. Radiother. Oncol. 40, 163–170 (1996).
Zhang, X. et al. Anti-tumor efficacy and biodistribution of intravenous polymeric micellar paclitaxel. Anticancer Drugs 8, 696–701 (1997).
Stabile, E. et al. Drug-eluting balloon for treatment of superficial femoral artery in-stent restenosis. J. Am. Coll. Cardiol. 60, 1739–1742 (2012).
Yin, Y. et al. The potential efficacy of R8-modified paclitaxel-loaded liposomes on pulmonary arterial hypertension. Pharm. Res. 30, 2050–2062 (2013).
Condliffe, R. et al. Connective tissue disease-associated pulmonary arterial hypertension in the modern treatment era. Am. J. Respir. Crit. Care Med. 179, 151–157 (2009).
Heath, D. & Yacoub, M. Lung mast cells in plexogenic pulmonary arteriopathy. J. Clin. Pathol. 44, 1003–1006 (1991).
Keller, C. et al. Alveolar rhabdomyosarcomas in conditional Pax3:Fkhr mice: cooperativity of Ink4a/ARF and Trp53 loss of function. Genes Dev. 18, 2614–2626 (2004).
Weissmann, N. et al. Classical transient receptor potential channel 6 (TRPC6) is essential for hypoxic pulmonary vasoconstriction and alveolar gas exchange. Proc. Natl. Acad. Sci. USA 103, 19093–19098 (2006).
Kojonazarov, B. et al. Effects of multikinase inhibitors on pressure overload-induced right ventricular remodeling. Int. J. Cardiol. 167, 2630–2637 (2013).
Urboniene, D., Haber, I., Fang, Y.H., Thenappan, T. & Archer, S.L. Validation of high-resolution echocardiography and magnetic resonance imaging vs. high-fidelity catheterization in experimental pulmonary hypertension. Am. J. Physiol. Lung Cell. Mol. Physiol. 299, L401–L412 (2010).
Koskenvuo, J.W. et al. A comparison of echocardiography to invasive measurement in the evaluation of pulmonary arterial hypertension in a rat model. Int. J. Cardiovasc. Imaging 26, 509–518 (2010).
Weisel, F.C. et al. Impact of S-adenosylmethionine decarboxylase 1 on pulmonary vascular remodeling. Circulation 129, 1510–1523 (2014).
Fink, L. et al. Real-time quantitative RT-PCR after laser-assisted cell picking. Nat. Med. 4, 1329–1333 (1998).
Fink, L. et al. cDNA array hybridization after laser-assisted microdissection from nonneoplastic tissue. Am. J. Pathol. 160, 81–90 (2002).
Pullamsetti, S.S. et al. cAMP phosphodiesterase inhibitors increases nitric oxide production by modulating dimethylarginine dimethylaminohydrolases. Circulation 123, 1194–1204 (2011).
Lai, Y.J. et al. Role of the prostanoid EP4 receptor in iloprost-mediated vasodilatation in pulmonary hypertension. Am. J. Respir. Crit. Care Med. 178, 188–196 (2008).
Kim, Y.M. et al. Neutrophil elastase is produced by pulmonary artery smooth muscle cells and is linked to neointimal lesions. Am. J. Pathol. 179, 1560–1572 (2011).
Nakae, J. et al. The forkhead transcription factor Foxo1 (Fkhr) confers insulin sensitivity onto glucose-6-phosphatase expression. J. Clin. Invest. 108, 1359–1367 (2001).
Acknowledgements
We thank J. Clardy and R. Kalb from Harvard Medical School for kindly providing Sam A. We thank M. Rabinovitch (Stanford University School of Medicine) for providing the mouse PASMC isolation protocol. We also acknowledge the excellent technical assistance of K. Leib in performing cell culture experiments, U. Eule, C. Vroom and A. Hecker in intact animal experiments and M. Schmoranzer, M. Hoeck and V. Golchert in histological studies. We thank K. Quanz for performing isolated lung experiments. We acknowledge M. Potente for providing genotyping protocol and siRNAs. This work was supported by the Max Planck Society, the Scientific and Economic Excellence in Hesse (LOEWE) Programm and the German Research Foundation through Excellence Cluster 147 Cardio-Pulmonary System (ECCPS), Cluster of Excellence REBIRTH (From Regenerative Biology to Reconstructive Therapy).
Author information
Authors and Affiliations
Contributions
R.S., D.S., H.M.A.-T., B.K., C.M., R.T.S. and S.S.P. acquired the data. R.S., D.S., H.M.A.-T., B.K., C.M., N.W., R.T.S. and S.S.P. analyzed and interpreted the data. R.S., N.W., R.T.S., D.S., W.S. and S.S.P. conceived and designed research. M.R.C. contributed transgenic mice. R.S., W.S., F.G. and S.S.P. drafted the manuscript. R.S., W.S., N.W., R.T.S. and S.S.P. handled the funding and supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–15 and Supplementary Table 1 (PDF 12040 kb)
Rights and permissions
About this article
Cite this article
Savai, R., Al-Tamari, H., Sedding, D. et al. Pro-proliferative and inflammatory signaling converge on FoxO1 transcription factor in pulmonary hypertension. Nat Med 20, 1289–1300 (2014). https://doi.org/10.1038/nm.3695
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3695
- Springer Nature America, Inc.
This article is cited by
-
A novel complement C3 inhibitor CP40-KK protects against experimental pulmonary arterial hypertension via an inflammasome NLRP3 associated pathway
Journal of Translational Medicine (2024)
-
Oxidative Stress and FOXO-1 Relationship in Stage III Periodontitis
Clinical Oral Investigations (2024)
-
Mitochondrial carrier 1 (MTCH1) governs ferroptosis by triggering the FoxO1-GPX4 axis-mediated retrograde signaling in cervical cancer cells
Cell Death & Disease (2023)
-
Nanomedicine-based treatment: An emerging therapeutical strategy for pulmonary hypertension
Nano Research (2023)
-
Growth differentiation factor 11 induces skeletal muscle atrophy via a STAT3-dependent mechanism in pulmonary arterial hypertension
Skeletal Muscle (2022)