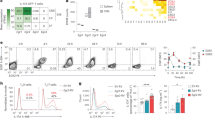
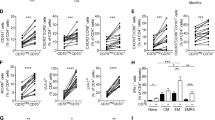
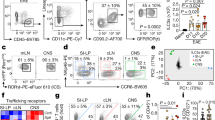
Abstract
The transcription factor T-bet has been associated with increased susceptibility to systemic and organ-specific autoimmunity, but the mechanism by which T-bet expression promotes neuroinflammation remains unknown. In this study, we demonstrate a cardinal role of T-bet-dependent NKp46+ innate lymphoid cells (ILCs) in the initiation of CD4+ TH17-mediated neuroinflammation. Loss of T-bet specifically in NKp46+ ILCs profoundly impaired the ability of myelin-reactive TH17 cells to invade central nervous system (CNS) tissue and protected the mice from autoimmunity. T-bet-dependent NKp46+ ILCs localized in the meninges and acted as chief coordinators of meningeal inflammation by inducing the expression of proinflammatory cytokines, chemokines and matrix metalloproteinases, which together facilitated T cell entry into CNS parenchyma. Our findings uncover a detrimental role of T-bet-dependent NKp46+ ILCs in the development of CNS autoimmune disease.








Similar content being viewed by others
References
McFarland, H.F. & Martin, R. Multiple sclerosis: a complicated picture of autoimmunity. Nat. Immunol. 8, 913–919 (2007).
Park, H. et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat. Immunol. 6, 1133–1141 (2005).
Langrish, C.L. et al. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J. Exp. Med. 201, 233–240 (2005).
McGeachy, M.J. et al. The interleukin 23 receptor is essential for the terminal differentiation of interleukin 17-producing effector T helper cells in vivo. Nat. Immunol. 10, 314–324 (2009).
Cua, D.J. et al. Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature 421, 744–748 (2003).
Schläger, C. et al. Effector T-cell trafficking between the leptomeninges and the cerebrospinal fluid. Nature 530, 349–353 (2016).
Bettelli, E. et al. Loss of T-bet, but not STAT1, prevents the development of experimental autoimmune encephalomyelitis. J. Exp. Med. 200, 79–87 (2004).
Lovett-Racke, A.E. et al. Silencing T-bet defines a critical role in the differentiation of autoreactive T lymphocytes. Immunity 21, 719–731 (2004).
Lazarevic, V., Glimcher, L.H. & Lord, G.M. T-bet: a bridge between innate and adaptive immunity. Nat. Rev. Immunol. 13, 777–789 (2013).
Lazarevic, V. & Glimcher, L.H. T-bet in disease. Nat. Immunol. 12, 597–606 (2011).
Lee, Y. et al. Induction and molecular signature of pathogenic TH17 cells. Nat. Immunol. 13, 991–999 (2012).
Wang, Y. et al. The transcription factors T-bet and Runx are required for the ontogeny of pathogenic interferon-γ-producing T helper 17 cells. Immunity 40, 355–366 (2014).
Yang, Y. et al. T-bet is essential for encephalitogenicity of both Th1 and Th17 cells. J. Exp. Med. 206, 1549–1564 (2009).
Ghoreschi, K. et al. Generation of pathogenic TH17 cells in the absence of TGF-β signalling. Nature 467, 967–971 (2010).
Odoardi, F. et al. T cells become licensed in the lung to enter the central nervous system. Nature 488, 675–679 (2012).
Flügel, A. et al. Migratory activity and functional changes of green fluorescent effector cells before and during experimental autoimmune encephalomyelitis. Immunity 14, 547–560 (2001).
Zhu, J. et al. The transcription factor T-bet is induced by multiple pathways and prevents an endogenous Th2 cell program during Th1 cell responses. Immunity 37, 660–673 (2012).
Greter, M. et al. Dendritic cells permit immune invasion of the CNS in an animal model of multiple sclerosis. Nat. Med. 11, 328–334 (2005).
Kivisäkk, P. et al. Localizing central nervous system immune surveillance: meningeal antigen-presenting cells activate T cells during experimental autoimmune encephalomyelitis. Ann. Neurol. 65, 457–469 (2009).
McMahon, E.J., Bailey, S.L., Castenada, C.V., Waldner, H. & Miller, S.D. Epitope spreading initiates in the CNS in two mouse models of multiple sclerosis. Nat. Med. 11, 335–339 (2005).
Fuchs, A. et al. Intraepithelial type 1 innate lymphoid cells are a unique subset of IL-12- and IL-15-responsive IFN-γ-producing cells. Immunity 38, 769–781 (2013).
Gordon, S.M. et al. The transcription factors T-bet and Eomes control key checkpoints of natural killer cell maturation. Immunity 36, 55–67 (2012).
Klose, C.S.N. et al. Differentiation of type 1 ILCs from a common progenitor to all helper-like innate lymphoid cell lineages. Cell 157, 340–356 (2014).
Klose, C.S. et al. A T-bet gradient controls the fate and function of CCR6-RORγt+ innate lymphoid cells. Nature 494, 261–265 (2013).
Rankin, L.C. et al. The transcription factor T-bet is essential for the development of NKp46+ innate lymphocytes via the Notch pathway. Nat. Immunol. 14, 389–395 (2013).
Sciumé, G. et al. Distinct requirements for T-bet in gut innate lymphoid cells. J. Exp. Med. 209, 2331–2338 (2012).
Townsend, M.J. et al. T-bet regulates the terminal maturation and homeostasis of NK and Valpha14i NKT cells. Immunity 20, 477–494 (2004).
Morandi, B. et al. Role of natural killer cells in the pathogenesis and progression of multiple sclerosis. Pharmacol. Res. 57, 1–5 (2008).
Winkler-Pickett, R. et al. In vivo regulation of experimental autoimmune encephalomyelitis by NK cells: alteration of primary adaptive responses. J. Immunol. 180, 4495–4506 (2008).
Liu, Q. et al. Neural stem cells sustain natural killer cells that dictate recovery from brain inflammation. Nat. Neurosci. 19, 243–252 (2016).
Bielekova, B. et al. Regulatory CD56(bright) natural killer cells mediate immunomodulatory effects of IL-2Ralpha-targeted therapy (daclizumab) in multiple sclerosis. Proc. Natl. Acad. Sci. USA 103, 5941–5946 (2006).
Hao, J. et al. Central nervous system (CNS)-resident natural killer cells suppress Th17 responses and CNS autoimmune pathology. J. Exp. Med. 207, 1907–1921 (2010).
Pikovskaya, O. et al. Cutting edge: eomesodermin is sufficient to direct type 1 innate lymphocyte development into the conventional NK lineage. J. Immunol. 196, 1449–1454 (2016).
Becher, B., Spath, S. & Goverman, J. Cytokine networks in neuroinflammation. Nat. Rev. Immunol. 17, 49–59 (2017).
Rothhammer, V. et al. Th17 lymphocytes traffic to the central nervous system independently of α4 integrin expression during EAE. J. Exp. Med. 208, 2465–2476 (2011).
Reboldi, A. et al. C-C chemokine receptor 6–regulated entry of TH-17 cells into the CNS through the choroid plexus is required for the initiation of EAE. Nat. Immunol. 10, 514–523 (2009).
Bauer, M. et al. Beta1 integrins differentially control extravasation of inflammatory cell subsets into the CNS during autoimmunity. Proc. Natl. Acad. Sci. USA 106, 1920–1925 (2009).
Dungan, L.S., McGuinness, N.C., Boon, L., Lynch, M.A. & Mills, K.H. Innate IFN-γ promotes development of experimental autoimmune encephalomyelitis: a role for NK cells and M1 macrophages. Eur. J. Immunol. 44, 2903–2917 (2014).
Kipnis, J. Multifaceted interactions between adaptive immunity and the central nervous system. Science 353, 766–771 (2016).
Song, J. et al. Focal MMP-2 and MMP-9 activity at the blood-brain barrier promotes chemokine-induced leukocyte migration. Cell Rep. 10, 1040–1054 (2015).
Chiang, E.Y. et al. Targeted depletion of lymphotoxin-α–expressing TH1 and TH17 cells inhibits autoimmune disease. Nat. Med. 15, 766–773 (2009).
Chung, Y. et al. Critical regulation of early Th17 cell differentiation by interleukin-1 signaling. Immunity 30, 576–587 (2009).
Pikor, N.B. et al. Integration of Th17- and lymphotoxin-derived signals initiates meningeal-resident stromal cell remodeling to propagate neuroinflammation. Immunity 43, 1160–1173 (2015).
Codarri, L. et al. RORγt drives production of the cytokine GM-CSF in helper T cells, which is essential for the effector phase of autoimmune neuroinflammation. Nat. Immunol. 12, 560–567 (2011).
El-Behi, M. et al. The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23-induced production of the cytokine GM-CSF. Nat. Immunol. 12, 568–575 (2011).
Jenne, C.N. et al. T-bet-dependent S1P5 expression in NK cells promotes egress from lymph nodes and bone marrow. J. Exp. Med. 206, 2469–2481 (2009).
van Helden, M.J. et al. Terminal NK cell maturation is controlled by concerted actions of T-bet and Zeb2 and is essential for melanoma rejection. J. Exp. Med. 212, 2015–2025 (2015).
Narni-Mancinelli, E. et al. Fate mapping analysis of lymphoid cells expressing the NKp46 cell surface receptor. Proc. Natl. Acad. Sci. USA 108, 18324–18329 (2011).
Intlekofer, A.M. et al. Anomalous type 17 response to viral infection by CD8+ T cells lacking T-bet and eomesodermin. Science 321, 408–411 (2008).
Gossa, S., Nayak, D., Zinselmeyer, B.H. & McGavern, D.B. Development of an immunologically tolerated combination of fluorescent proteins for in vivo two-photon imaging. Sci. Rep. 4, 6664 (2014).
Acknowledgements
We thank Y. Belkaid and A. Singer for critical review of the manuscript; S. Sharrow, L. Granger and A. Adams for flow cytometry and cell sorting; T. Loo for technical assistance; and the members of the laboratories of A. Singer and H. Park for discussions. S. Reiner (Columbia University) generously provided the Tbx21f/f mice. This study was supported by the Intramural Research Program of the US National Institutes of Health, National Cancer Institute, Center for Cancer Research (ZIA BC 011431 and ZIA BC 011432 to V.L.) and National Institute of Neurological Disorders and Stroke (1ZIA NS 003111-08 and 1ZIA NS 003112-08 to D.B.M.).
Author information
Authors and Affiliations
Contributions
B.K. conducted the experiments, analyzed the data and contributed to the first draft of the manuscript. R.R., Y.G., J.F. and Y.W. conducted experiments and analyzed data. D.B.M. contributed to experimental design and manuscript preparation. M.J.K. performed quantification of apoptotic cells in the CNS in a blinded manner. E.V. and J.Z. generated NKp46-Cre+ and T-bet ZsGreen reporter mice, respectively. V.L. designed and directed the study, conducted the experiments, analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
E.V. is the cofounder of and a shareholder in Innate Pharma.
Integrated supplementary information
Supplementary Figure 1 T-bet expression in endogenous (host-derived) T cells is not required for EAE induction by 2D2 WT TH17 cells.
(a) Flow cytometric analysis of CNS-infiltrating CD4+ T cells following adoptive transfer of 7.5 × 106 2D2 WT TH17 cells into Tbx21f/f or Tbx21f/f CD4-Cre+ recipient mice. CNS-infiltrating cells were harvested at the peak of EAE disease (day 16 post-transfer) and analyzed for the fraction of 2D2 (Vβ11+) versus endogenous (Vβ11−) cells among live CD4+ T cells. (b-c) Cytokine production of CNS-infiltrating endogenous or 2D2 CD4+ T cells at the peak of EAE disease was determined by intracellular cytokine staining following brief ex vivo PMA/ionomycin re-stimulation. (d) Mean clinical scores in Tbx21f/f or Tbx21f/f CD4-Cre+ mice receiving 2D2 WT TH17 cells (7.5 × 106). NS, P > 0.05; *, P < 0.01 (two-tailed Student’s t-test (b,c) or two-way ANOVA (d)). Data are combined from two independent experiments (a-c; n = 9, 10 mice per group) or three independent experiments (d; n = 15, 16 mice per group).
Supplementary Figure 2 T-bet expression in dendritic cells is not required for EAE induction by 2D2 WT TH17 cells.
(a) Intracellular cytokine staining of CNS-infiltrating 2D2 CD4+ T cells (CD4+Vβ11+) from Tbx21f/f or Tbx21f/f CD11c-Cre+ mice following adoptive transfer of 7.5 × 106 2D2 WT TH17 cells. CNS-infiltrating cells were harvested at the peak of EAE disease (day 15-17 post-transfer) and re-stimulated ex vivo with PMA/ionomycin. (b) Mean clinical scores in Tbx21f/f or Tbx21f/f CD11c-Cre+ mice which received 2D2 WT TH17 cells (7.5 × 106). NS, P > 0.05 (two-way ANOVA). Data are combined from two independent experiments (a; mean ± s.e.m. of n = 10, 12 mice per group) or three independent experiments (b; mean ± s.e.m. of n = 17, 19 mice per group).
Supplementary Figure 3 Efficiency of NK cell deletion in anti-NK1.1-treated mice.
Depletion of NK cells in WT (BL6) mice by anti-NK1.1 (clone PK136) treatment. 500 μg of αNK1.1 antibody per dose, or an equivalent volume of PBS, was injected intravenously via the tail vein into WT mice, beginning 5 days prior to the adoptive transfer of 2D2 WT TH17 cells and continuing until 16 days post-transfer (indicated by red arrows). On the day of the adoptive transfer of 2D2 WT TH17 cells (day 0), peripheral lymphoid and non-lymphoid tissues were harvested from representative mice, and NK cells were quantified to assess the efficacy of depletion. On day 21 post-transfer, CNS-infiltrating cells were harvested from representative mice to assess the efficacy and duration of NK cell depletion in the CNS. Data are representative of three independent experiments; numbers indicate the frequency of CD3−NKp46+ cells among total live cells.
Supplementary Figure 4 NK cell deficiency in Eomesf/f NKp46-Cre+mice.
(a-b) Frequency and absolute numbers of conventional NK cells (TCRβ−NKp46+NK1.1+IL-7Rα−) in lymphoid and non-lymphoid tissues of Eomesf/f or Eomesf/f NKp46-Cre+mice 12-14 days after active immunization with MOG35-55/CFA and pertussis toxin. Data are representative of two independent experiments (n = 3 mice per group).
Supplementary Figure 5 IFN-γ is required for the activation/maturation of meningeal APCs.
(a) Mean clinical scores of Ifng−/− and WT mice, which received 7.5 × 106 2D2 WT TH17 cells. (b) Absolute number of 2D2 CD4+ T cells (CD4+Vβ11+) in the spinal cord meninges and CNS parenchyma of WT or Ifng−/− mice as quantitated by flow cytometry. (c) Absolute number of CD206+ perivascular macrophages (PVMs), myeloid DCs (mDCs), inflammatory monocytes (iMO) and neutrophils in the meninges of WT or Ifng−/− mice as quantitated by flow cytometry (Supplementary Fig. 6 for gating strategy). (d) Flow cytometric analyses of the maturation markers on meningeal antigen presenting cells in WT or Ifng−/− mice following the adoptive transfer of 7.5 × 106 2D2 WT TH17 cells. Activation of spinal cord meningeal antigen presenting cells was determined by staining perivascular macrophages and myeloid-derived dendritic cells with antibodies to MHC ClassII (I-A/I-E) molecules, CD80 and ICAM1. *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.0001 (two-way ANOVA (a) or two-tailed Student’s t-test (b-d)). Data are representative of two independent experiments (a; mean ± s.e.m. of n = 10 mice per group) or combined from two independent experiments (b-d; mean ± SEM of n = 10 mice per group).
Supplementary Figure 6 Gating strategy for resident and infiltrating meningeal APCs.
Spinal cord meningeal cells were harvested at the peak of EAE disease (day 17 - 22 post-transfer), gated on live CD45+ cells, and further identified as follows: perivascular macrophages (Thy1.2− CD11b+ Ly6C− Ly6G−F4/80+ CD206+), myeloid-derived dendritic cells (Thy1.2−MHCII+CD11c+CD11b+), inflammatory monocytes (Thy1.2− CD11b+Ly6C+ Ly6G−) and neutrophils (Thy1.2− CD11b+Ly6C−Ly6G+).
Supplementary Figure 7 NKp46+ ILCs are dispensable for activation of meningeal APCs.
(a) Absolute number of CD206+ perivascular macrophages (PVMs), myeloid DCs (mDCs), inflammatory monocytes (iMO) and neutrophils in the meninges of Tbx21f/f or Tbx21f/f NKp46-Cre+ mice as quantitated by flow cytometry (Supplementary Fig. 6 for gating strategy). (b) Flow cytometric analyses of the activation markers on meningeal antigen presenting cells in Tbx21f/f or Tbx21f/f NKp46-Cre+ mice following the adoptive transfer of 7.5 × 106 2D2 WT TH17 cells. Activation of spinal cord meningeal antigen presenting cells was determined by staining perivascular macrophages and myeloid-derived dendritic cells with antibodies to MHC ClassII (I-A/I-E) molecules, CD80 and ICAM1. Naïve healthy WT (BL6) mice were used to assess the steady-state expression of maturation markers. Expression levels are presented as mean fluorescence intensity (MFI) of staining (b); data are combined from 3 independent experiments with 11 mice per group.
Supplementary Figure 8 T-bet-dependent NKp46+ ILCs are not required for GM-CSF production by IL-17A-producing 2D2 CD4+ T cells.
Intracellular cytokine staining of 2D2 CD4+ T cells (CD4+Vβ11+) isolated from the meninges and CNS parenchyma of Tbx21f/f or Tbx21f/f NKp46-Cre+ mice following adoptive transfer of 7.5 × 106 2D2 WT TH17 cells. CNS-infiltrating cells were harvested at the peak of EAE disease (day 17-22 post-transfer) and re-stimulated ex vivo with PMA/ionomycin prior to staining. Bar graphs present mean ± s.e.m of cytokine producing 2D2 CD4+ T cells isolated from the spinal cord meninges and parenchyma of Tbx21f/f or Tbx21f/f NKp46-Cre+ mice. NS, P > 0.05; *, P < 0.01 (two-tailed Student’s t-test). Data are combined from 2 independent experiments (n = 6, 8 mice per group).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Table 1.
Rights and permissions
About this article
Cite this article
Kwong, B., Rua, R., Gao, Y. et al. T-bet-dependent NKp46+ innate lymphoid cells regulate the onset of TH17-induced neuroinflammation. Nat Immunol 18, 1117–1127 (2017). https://doi.org/10.1038/ni.3816
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3816
- Springer Nature America, Inc.
This article is cited by
-
Brain endothelial CXCL12 attracts protective natural killer cells during ischemic stroke
Journal of Neuroinflammation (2023)
-
The effect of glatiramer acetate, IFNβ-1a, fingolimod, and dimethyl fumarate on the expression of T-bet, IFN-γ, and MEG3 in PBMC of RRMS patients
BMC Research Notes (2023)
-
The role of peripheral inflammatory insults in Alzheimer’s disease: a review and research roadmap
Molecular Neurodegeneration (2023)
-
Transcription factor EGR2 controls homing and pathogenicity of TH17 cells in the central nervous system
Nature Immunology (2023)
-
The immunology of multiple sclerosis
Nature Reviews Immunology (2022)