Abstract
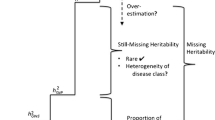
Genome-wide association studies have detected many loci underlying susceptibility to disease, but most of the genetic factors that contribute to disease susceptibility remain unknown. Here we provide evidence that part of the 'missing heritability' can be explained by an overestimation of heritability. We estimated the heritability of 12 complex human diseases using family history of disease in 1,555,906 individuals of white ancestry from the UK Biobank. Estimates using simple family-based statistical models were inflated on average by ∼47% when compared with those from structural equation modeling (SEM), which specifically accounted for shared familial environmental factors. In addition, heritabilities estimated using SNP data explained an average of 44.2% of the simple family-based estimates across diseases and an average of 57.3% of the SEM-estimated heritabilities, accounting for almost all of the SEM heritability for hypertension. Our results show that both genetics and familial environment make substantial contributions to familial clustering of disease.

Similar content being viewed by others
References
Falconer, D.S. Inheritance of liability to certain diseases estimated from incidence among relatives. Annal. Hum. Genet. 29, 51–76 (1965).
Manolio, T.A. et al. Finding the missing heritability of complex diseases. Nature 461, 747–753 (2009).
Tenesa, A. & Haley, C.S. The heritability of human disease: estimation, uses and abuses. Nat. Rev. Genet. 14, 139–149 (2013).
Allen, N. et al. UK Biobank: current status and what it means for epidemiology. Health Policy Technol 1, 123–126 (2012).
UK Biobank Coordinating Centre. UK Biobank: protocol for a large-scale prospective epidemiological resource (Protocol number UKBB-PROT-09-06 (Main Phase)) (UK Biobank, 2007).
Kaprio, J. et al. Concordance for type 1 (insulin-dependent) and type 2 (non-insulin-dependent) diabetes mellitus in a population-based cohort of twins in Finland. Diabetologia 35, 1060–1067 (1992).
Lichtenstein, P. et al. Environmental and heritable factors in the causation of cancer—analyses of cohorts of twins from Sweden, Denmark, and Finland. N. Engl. J. Med. 343, 78–85 (2000).
Iliadou, A. et al. Repeated blood pressure measurements in a sample of Swedish twins: heritabilities and associations with polymorphisms in the renin–angiotensin–aldosterone system. J. Hypertens. 20, 1543–1550 (2002).
Gatz, M. et al. Role of genes and environments for explaining Alzheimer disease. Arch. Gen. Psychiatry 63, 168–174 (2006).
Zdravkovic, S., Wienke, A., Pedersen, N.L. & de Faire, U. Genetic influences on angina pectoris and its impact on coronary heart disease. Eur. J. Hum. Genet. 15, 872–877 (2007).
Hallberg, J. et al. Interaction between smoking and genetic factors in the development of chronic bronchitis. Am. J. Respir. Crit. Care Med. 177, 486–490 (2008).
Korja, M. et al. Genetic epidemiology of spontaneous subarachnoid hemorrhage: Nordic Twin Study. Stroke 41, 2458–2462 (2010).
Polderman, T.J.C. et al. Meta-analysis of the heritability of human traits based on fifty years of twin studies. Nat. Genet. 47, 702–709 (2015).
Annest, J.L., Sing, C.F., Biron, P. & Mongeau, J.G. Familial aggregation of blood pressure and weight in adoptive families. II. Estimation of the relative contributions of genetic and common environmental factors to blood pressure correlations between family members. Am. J. Epidemiol. 110, 492–503 (1979).
Mancuso, N. et al. The contribution of rare variation to prostate cancer heritability. Nat. Genet. 48, 30–35 (2016).
Yang, J. et al. Genetic variance estimation with imputed variants finds negligible missing heritability for human height and body mass index. Nat. Genet. 47, 1114–1120 (2015).
Zaitlen, N. et al. Using extended genealogy to estimate components of heritability for 23 quantitative and dichotomous traits. PLoS Genet. 9, e1003520 (2013).
Liu, C. et al. Revisiting heritability accounting for shared environmental effects and maternal inheritance. Hum. Genet. 134, 169–179 (2015).
Aschard, H., Vilhjálmsson, B.J., Joshi, A.D., Price, A.L. & Kraft, P. Adjusting for heritable covariates can bias effect estimates in genome-wide association studies. Am. J. Hum. Genet. 96, 329–339 (2015).
Tada, H. et al. Risk prediction by genetic risk scores for coronary heart disease is independent of self-reported family history. Eur. Heart J. 37, 561–567 (2016).
Risch, N. Linkage strategies for genetically complex traits. I. Multilocus models. Am. J. Hum. Genet. 46, 222–228 (1990).
Falconer, D.S. & Mackay, T.F. Introduction to Quantitative Genetics (Longman, 1996).
Dempster, E.R. & Lerner, I.M. Heritability of threshold characters. Genetics 35, 212–236 (1950).
Boker, S. et al. OpenMx: an open source extended structural equation modeling framework. Psychometrika 76, 306–317 (2011).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Canela-Xandri, O., Law, A., Gray, A., Woolliams, J.A. & Tenesa, A. A new tool called DISSECT for analysing large genomic data sets using a Big Data approach. Nat. Commun. 6, 10162 (2015).
Stuart, A. & Ord, J.K. Kendall's Advanced Theory of Statistics (Hoder Arnold, 1994).
Acknowledgements
We thank I. White for his helpful comments. This research has been conducted using the UK Biobank resource and funded by the Roslin Institute Strategic Programme Grant from the BBSRC (BB/J004235/1). C.S.H. and A.T. also acknowledge funding from the MRC. SNP heritability estimates were calculated using the ARCHER UK National Supercomputing Service.
Author information
Authors and Affiliations
Contributions
A.T. and C.S.H. conceived and designed the study. M.M. and A.T. performed the statistical analysis. O.C.-X. and K.R. carried out the SNP filtering and quality control. M.M., C.S.H. and A.T. wrote the manuscript. R.P.-W. performed the simulations and contributed ideas and quantitative genetics expertise. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1 and 2, Supplementary Tables 1–16 and Supplementary Note. (PDF 665 kb)
Rights and permissions
About this article
Cite this article
Muñoz, M., Pong-Wong, R., Canela-Xandri, O. et al. Evaluating the contribution of genetics and familial shared environment to common disease using the UK Biobank. Nat Genet 48, 980–983 (2016). https://doi.org/10.1038/ng.3618
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.3618
- Springer Nature America, Inc.
This article is cited by
-
Increasing number of long-lived ancestors marks a decade of healthspan extension and healthier metabolomics profiles
Nature Communications (2023)
-
Genetic correlations between traits associated with hyperuricemia, gout, and comorbidities
European Journal of Human Genetics (2021)
-
Interaction between lifestyle behaviors and genetic polymorphism in SCAP gene on blood pressure among Chinese children
Pediatric Research (2019)
-
Indirect assortative mating for human disease and longevity
Heredity (2019)
-
Relatedness disequilibrium regression estimates heritability without environmental bias
Nature Genetics (2018)