Abstract
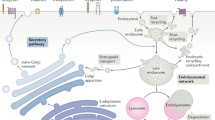
Following endocytosis into the endosomal network, integral membrane proteins undergo sorting for lysosomal degradation or are retrieved and recycled back to the cell surface. Here we describe the discovery of an ancient and conserved multiprotein complex that orchestrates cargo retrieval and recycling and, importantly, is biochemically and functionally distinct from the established retromer pathway. We have called this complex ‘retriever’; it is a heterotrimer composed of DSCR3, C16orf62 and VPS29, and bears striking similarity to retromer. We establish that retriever associates with the cargo adaptor sorting nexin 17 (SNX17) and couples to CCC (CCDC93, CCDC22, COMMD) and WASH complexes to prevent lysosomal degradation and promote cell surface recycling of α5β1 integrin. Through quantitative proteomic analysis, we identify over 120 cell surface proteins, including numerous integrins, signalling receptors and solute transporters, that require SNX17–retriever to maintain their surface levels. Our identification of retriever establishes a major endosomal retrieval and recycling pathway.







Similar content being viewed by others
References
Huotari, J. & Helenius, A. Endosome maturation. EMBO J. 30, 3481–3500 (2011).
Gallon, M. & Cullen, P. J. Retromer and sorting nexins in endosomal sorting. Biochem. Soc. Trans. 43, 33–47 (2015).
Schreij, A. M., Fon, E. A. & McPherson, P. S. Endocytic membrane trafficking and neurodegenerative disease. Cell. Mol. Life Sci. 73, 1529–1545 (2016).
Henne, W. M., Buchkovich, N. J. & Emr, S. D. The ESCRT pathway. Dev. Cell 21, 77–91 (2011).
Burd, C. & Cullen, P. J. Retromer: a master conductor of endosome sorting. Cold Spring Harb. Perspect. Biol. 6, a016774 (2014).
Seaman, M. N., Marcusson, E. G., Cereghino, J. L. & Emr, S. D. Endosome to Golgi retrieval of the vacuolar protein sorting receptor, Vps10p, requires the function of the VPS29, VPS30, and VPS35 gene products. J. Cell Biol. 137, 79–92 (1997).
Seaman, M. N., McCaffery, J. M. & Emr, S. D. A membrane coat complex essential for endosome-to-Golgi retrograde transport in yeast. J. Cell Biol. 142, 665–681 (1998).
Arighi, C. N., Hartnell, L. M., Aguilar, R. C., Haft, C. R. & Bonifacino, J. S. Role of the mammalian retromer in sorting of the cation-independent mannose 6-phosphate receptor. J. Cell Biol. 165, 123–133 (2004).
Seaman, M. N. Cargo-selective endosomal sorting for retrieval to the Golgi requires retromer. J. Cell Biol. 165, 111–122 (2004).
Seaman, M. N. Identification of a novel conserved sorting motif required for retromer-mediated endosome-to-TGN retrieval. J. Cell Sci. 120, 2378–2389 (2007).
Canuel, M., Lefrancois, S., Zeng, J. & Morales, C. R. AP-1 and retromer play opposite roles in the trafficking of sortilin between the Golgi apparatus and the lysosomes. Biochem. Biophys. Res. Commun. 366, 724–730 (2008).
Harterink, M. et al. A SNX3-dependent retromer pathway mediates retrograde transport of the Wnt sorting receptor Wntless and is required for Wnt secretion. Nat. Cell Biol. 13, 914–923 (2011).
Zhang, P., Wu, Y., Belenkaya, T. Y. & Lin, X. SNX3 controls Wingless/Wnt secretion through regulating retromer-dependent recycling of Wntless. Cell Res. 21, 1677–1690 (2011).
Lucas, M. et al. Structural mechanism for cargo recognition by the retromer complex. Cell 167, 1623–1635 (2016).
Lauffer, B. E. et al. SNX27 mediates PDZ-directed sorting from endosomes to the plasma membrane. J. Cell Biol. 190, 565–574 (2010).
Temkin, P. et al. SNX27 mediates retromer tubule entry and endosome-to-plasma membrane trafficking of signalling receptors. Nat. Cell Biol. 13, 715–721 (2011).
Gallon, M. et al. A unique PDZ domain and arrestin-like fold interaction reveals mechanistic details of endocytic recycling by SNX27-retromer. Proc. Natl Acad. Sci. USA 111, E3604–E3613 (2014).
Steinberg, F. et al. A global analysis of SNX27-retromer assembly and cargo specificity reveals a function in glucose and metal ion transport. Nat. Cell Biol. 15, 461–471 (2013).
Clairfeuille, T. et al. A molecular code for endosomal recycling of phosphorylated cargos by the SNX27-retromer complex. Nat. Struct. Mol. Biol. 23, 921–932 (2016).
Harbour, M. E., Breusegem, S. Y. & Seaman, M. N. Recruitment of the endosomal WASH complex is mediated by the extended ‘tail’ of Fam21 binding to the retromer protein Vps35. Biochem. J. 442, 209–220 (2012).
Jia, D., Gomez, T. S., Billadeau, D. D. & Rosen, M. K. Multiple repeat elements within the FAM21 tail link the WASH actin regulatory complex to the retromer. Mol. Biol. Cell 23, 2352–2361 (2012).
Gomez, T. S. & Billadeau, D. D. A FAM21-containing WASH complex regulates retromer-dependent sorting. Dev. Cell 17, 699–711 (2009).
Derivery, E. et al. The Arp2/3 activator WASH controls the fission of endosomes through a large multiprotein complex. Dev. Cell 17, 712–723 (2009).
Harbour, M. E. et al. The cargo-selective retromer complex is a recruiting hub for protein complexes that regulate endosomal tubule dynamics. J. Cell Sci. 123, 3703–3717 (2010).
Puthenveedu, M. A. et al. Sequence-dependent sorting of recycling proteins by actin-stabilized endosomal microdomains. Cell 143, 761–773 (2010).
Steinberg, F., Heesom, K. J., Bass, M. D. & Cullen, P. J. SNX17 protects integrins from degradation by sorting between lysosomal and recycling pathways. J. Cell Biol. 197, 219–230 (2012).
McMillan, K. J. et al. Atypical parkinsonism-associated retromer mutant alters endosomal sorting of specific cargo proteins. J. Cell Biol. 214, 389–399 (2016).
McGough, I. J. et al. Retromer binding to FAM21 and the WASH complex is perturbed by the Parkinson disease-linked VPS35(D620N) mutation. Curr. Biol. 24, 1670–1676 (2014).
Zavodszky, E. et al. Mutation in VPS35 associated with Parkinson’s disease impairs WASH complex association and inhibits autophagy. Nat. Commun. 5, 3828 (2014).
Vilarino-Guell, C. et al. VPS35 mutations in Parkinson disease. Am. J. Hum. Genet. 89, 162–167 (2011).
Tsika, E. et al. Parkinson’s disease-linked mutations in VPS35 induce dopaminergic neurodegeneration. Hum. Mol. Genet. 23, 4621–4638 (2014).
Böttcher, R. T. et al. Sorting nexin 17 prevents lysosomal degradation of β1 integrins by binding to the β1-integrin tail. Nat. Cell Biol. 14, 584–592 (2012).
Stockinger, W. et al. The PX-domain protein SNX17 interacts with members of the LDL receptor family and modulates endocytosis of the LDL receptor. EMBO J. 21, 4259–4267 (2002).
Burden, J. J., Sun, X. M., Garcia, A. B. & Soutar, A. K. Sorting motifs in the intracellular domain of the low density lipoprotein receptor interact with a novel domain of sorting nexin-17. J. Biol. Chem. 279, 16237–16245 (2004).
van Kerkhof, P. et al. Sorting nexin 17 facilitates LRP recycling in the early endosome. EMBO J. 24, 2851–2861 (2005).
Farfan, P. et al. A sorting nexin 17-binding domain within the LRP1 cytoplasmic tail mediates receptor recycling through the basolateral sorting endosome. Traffic 14, 823–838 (2013).
Ghai, R. et al. Structural basis for endosomal trafficking of diverse transmembrane cargos by PX-FERM proteins. Proc. Natl Acad. Sci. USA 110, E643–E652 (2013).
McGough, I. J. et al. Identification of molecular heterogeneity in SNX27-retromer-mediated endosome-to-plasma-membrane recycling. J. Cell Sci. 127, 4940–4953 (2014).
Phillips-Krawczak, C. A. et al. COMMD1 is linked to the WASH complex and regulates endosomal trafficking of the copper transporter ATP7A. Mol. Biol. Cell 26, 91–103 (2015).
Li, H. et al. Endosomal sorting of Notch receptors through COMMD9-dependent pathways modulates Notch signaling. J. Cell Biol. 211, 605–617 (2015).
Hierro, A. et al. Functional architecture of the retromer cargo-recognition complex. Nature 449, 1063–1067 (2007).
Koumandou, V. L. et al. Evolutionary reconstruction of the retromer complex and its function in Trypanosoma brucei. J. Cell Sci. 124, 1496–1509 (2011).
Fitzgerald, D. J. et al. Protein complex expression by using multigene baculoviral vectors. Nat. Methods 3, 1021–1032 (2006).
Stenmark, H. et al. Inhibition of rab5 GTPase activity stimulates membrane fusion in endocytosis. EMBO J. 13, 1287–1296 (1994).
Seaman, M. N., Harbour, M. E., Tattersall, D., Read, E. & Bright, N. Membrane recruitment of the cargo-selective retromer subcomplex is catalysed by the small GTPase Rab7 and inhibited by the Rab-GAP TBC1D5. J. Cell Sci. 122, 2371–2382 (2009).
Rojas, R. et al. Regulation of retromer recruitment to endosomes by sequential action of Rab5 and Rab7. J. Cell Biol. 183, 513–526 (2008).
Li, Y., Calvo, S. E., Gutman, R., Liu, J. S. & Mootha, V. K. Expansion of biological pathways based on evolutionary inference. Cell 158, 213–225 (2014).
Tseng, H. Y. et al. Sorting nexin 31 binds multiple β integrin cytoplasmic domains and regulates β1 integrin surface levels and stability. J. Mol. Biol. 426, 3180–3194 (2014).
Lee, J. et al. Adaptor protein sorting nexin 17 regulates amyloid precursor protein trafficking and processing in the early endosomes. J. Biol. Chem. 283, 11501–11508 (2008).
Yin, W. et al. SNX17 regulates Notch pathway and pancreas development through the retromer-dependent recycling of Jag1. Cell Regen. 1, 4 (2012).
Bergant Marušič, M., Ozbun, M. A., Campos, S. K., Myers, M. P. & Banks, L. Human papillomavirus L2 facilitates viral escape from late endosomes via sorting nexin 17. Traffic 13, 455–467 (2012).
Buckley, C. M. et al. WASH drives early recycling from macropinosomes and phagosomes to maintain surface phagocytic receptors. Proc. Natl Acad. Sci. USA 113, E5906–E5915 (2016).
Zech, T. et al. The Arp2/3 activator WASH regulates α5β1-integrin-mediated invasive migration. J. Cell Sci. 124, 3753–3759 (2011).
Nagel, B. M., Bechtold, M., Rodriguez, L. G. & Bogdan, S. Drosophila WASH is required for integrin-mediated cell adhesion, cell motility and lysosomal neutralization. J. Cell Sci. 130, 344–359 (2017).
van Weering, J. R. et al. Molecular basis for SNX-BAR-mediated assembly of distinct endosomal sorting tubules. EMBO J. 31, 4466–4480 (2012).
Mi, H., Poudel, S., Muruganujan, A., Casagrande, J. T. & Thomas, P. D. PANTHER version 10: expanded protein families and functions, and analysis tools. Nucleic Acids Res. 44, D336–D342 (2016).
Trowitzsch, S., Bieniossek, C., Nie, Y., Garzoni, F. & Berger, I. New baculovirus expression tools for recombinant protein complex production. J. Struct. Biol. 172, 45–54 (2010).
Buck, C. B., Pastrana, D. V., Lowy, D. R. & Schiller, J. T. Generation of HPV pseudovirions using transfection and their use in neutralization assays. Methods Mol. Med. 119, 445–462 (2005).
Acknowledgements
We thank the Wolfson Bioimaging Facility at the University of Bristol and also the Life Imaging Center (LIC) of the University of Freiburg for their support. P.J.C. is supported by the Medical Research Council (MR/K018299/1 and MR/P018807/1) and the Wellcome Trust (089928 and 104568). Funding to B.M.C. from the Australian Research Council (ARC) (DP160101743) and National Health and Medical Research Council (NHMRC) (APP1058734) is acknowledged. The NIH (R01DK073639, R01DK107733) supported E.B. and (R01AI65474) D.D.B. L.B. gratefully acknowledges research support from the Associazione Italiana per la Ricerca sul Cancro. L.B. acknowledges research support from the Associazione Italiana per la Ricerca sul Cancro grant no. 18578. Wellcome Trust PhD Studentships from the Dynamic Cell Biology programme (083474) supported K.E.M. and M.G. An NHMRC Dementia Fellowship (APP1097185) supported R.G., and B.M.C. is supported by an NHMRC Career Development Fellowship (APP1061574). F.S. is funded by the German Research Council (DFG) Emmy Noether grant (STE2310/1-1).
Author information
Authors and Affiliations
Contributions
Initial conceptualization, K.E.M., F.S. and P.J.C. Evolution of conceptualization, K.E.M., F.S., I.B., D.D.B., E.B. and P.J.C. Formal analysis, R.S. Investigation, K.E.M., F.S., R.F., M.G., K.J.H., D.P. and R.G. Writing—original draft, K.E.M. and P.J.C. Writing—review and editing, all authors. Funding acquisition, F.S., L.B., B.M.C., D.D.B., E.B. and P.J.C. Resources, P.L., N.P., C.M.D., L.L.M., B.L.O., A.S. and H.N. Supervision, F.S., L.B., B.M.C., D.D.B., E.B. and P.J.C.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 SNX17 and VPS29 rescue experiments.
(A) Lower magnification images of Figure 1D. Scale bars = 20 μm. Representative images from 3 independent experiments. (B) Parental, VPS35 KO, VPS29 KO and VPS29 KO HeLa cells where VPS29 has been re-expressed (VPS29 KO rescue), were treated with 10 μg/ml cycloheximide for 0, 4 or 8 hours before lysis. Lysates were then analysed by Western blot for the indicated proteins. Representative blot from 3 independent experiments. (C,D) Quantification of B. Band intensities were measured using the Odyssey software. Band intensities of integrin α5 or integrin β1 are represented as a % of the band intensity at 0 hours cycloheximide treatment for each of the four conditions. % of integrin in KO cells were compared to parental HeLa at each time point using two-tailed t-test. Points represent the mean of n = 3 independent experiments. Error bars represent s.d. ∗∗ p < 0.01, ∗ p < 0.05. Unprocessed original scans of immunoblots are shown in Supplementary figure 8. See statistics source data for precise p values.
Supplementary Figure 2 Production of recombinant human retriever complex using MultiBac44.
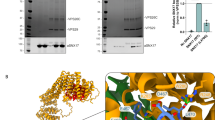
(A) Strep-DSCR3, untagged C16orf62 and VPS29-6xHis were cloned into MultiBac plasmids pACEBac1, pIDC and pIDK, respectively. The resulting expression constructs were conjoined by Cre/loxP reaction44, giving rise to a co-expression construct combining all three genes. This construct was inserted into the EMBacY baculoviral genome and virus prepared as described44. Expression in Sf21 insect cells was followed by protein purification using immobilized metal affinity chromatography IMAC) and size exclusion chromatography (SEC), resulting in highly purified retriever complex. (B) SEC profile of the recombinant retriever complex. Profile from one size exclusion chromatography experiment. (C) Analysis of the second peak in SEC by Coomassie-stained SDS-PAGE evidences excess VPS29-6xHis. Unprocessed original scans of the Coomassie gel are shown in Supplementary figure 8.
Supplementary Figure 3 DSCR3, C16orf62 and VPS29 localise on retromer positive endosomes.
(A) GFP-SNX17 transduced RPE-1 cells were fixed and stained for endogenous VPS35 and either C16orf62 or FAM21. Representative field of view from 3 independent fixations and stainings. Scale bars represent 20 μm. (B) RPE-1 cells were transduced with GFP-VPS29. Antibodies against endogenous SNX17 or C16orf62 stained endosomes which were positive for GFP-VPS29 and VPS35. Images are a representative field from 3 independent experiments. Scale bars represent 20 μm. (C) RPE-1 cells were transduced to express low levels of GFP-DSCR3. GFP-DSCR3 labelled endosomes stained positive for endogenous C16orf62 and VPS35. Representative field of view from 3 independent experiments. Scale bars represent 20 μm.
Supplementary Figure 4 The WASH complex co-evolved with retriever and is required for its endosomal localisation.
(A) Lower magnification images of figure 4C. Scale bars = 20 μm. (B) Validation of FAM21 knock-out in HeLa cells by immunofluorescence. Scale bars = 10 μm. Representative field of view from 2 independent experiments. (C) Quantification of Figure 4F. Pearson’s and colocalisation coefficients were calculated from 9 individual fields of view in each of the n = 2 independent experiments (18 fields of view in total). Bars = mean. (D) The evolutionary conservation of C16orf62 was mapped using the CLIME algorithm (Li et al., 2014). The pattern of C16orf62 conservation created an evolutionary conserved module. All genes in the genome were then compared to this evolutionary conserved module, searching for genes which share similar patterns of conservation and are therefore in inferred to have co-evolved with C16orf62. Proteins which are inferred to have co-evolved with C16orf62 have a LLR higher than 0, with a higher score indicating increased likeliness of co-evolution. PG = paralogue group, LLR = log-likelihood ratio scale.
Supplementary Figure 5 Characterisation of DSCR3 knock-out HeLas.
(A) Endogenous SNX17 IPs were performed in parental or CCDC22 knock-out HeLa cells. Representative blot from 2 independent experiments. (B) Lysates from DSCR3 knock-out HeLas were analysed by Western blotting for knock-out and effects on the levels of other proteins. Representative blot from 4 independent experiments. (C) Endogenous CCDC22 immunoprecipitations in parental or DSCR3 KO HeLa cells. This experiment was performed once. (D) Immunofluorescence of indicated endogenous proteins in parental HeLa cells. Representative field of view from 3 independent experiments. Scale bars = 10 μm for zoomed out image, 5 μm for zoomed in images. (E) Same as D but staining performed in DSCR3 knock-out HeLas. Scale bars = 20 μm for a field of cells, 5 μm for zoomed in images. (F) Parental HeLas and knock-out DSCR3 HeLa cells were analysed by immunofluorescence for their distribution of integrin α5. Bottom panels represent zoomed images of the white boxes. Scale bars = 10 μm for a field of cells, 5 μm for zoomed in images. (G) Parental or knock-out DSCR3 HeLa cells were either untransfected or transfected with Bafilomycin A to prevent lysosomal acidification. Lysates were then analysed by Western blotting against the indicated proteins. Representative blot from 3 independent cell lysis events. Unprocessed original scans of immunoblots are shown in Supplementary figure 8.
Supplementary Figure 6 The C terminal leucine of SNX17 and SNX31 is essential in engaging retriever and the CCC complex.
(A) GFP traps of HEK 293 cells expressing SNX17 mutants indicated in figure 4B. Representative Western blot from three GFP trap experiments. (B) GFP-SNX17 or the indicated mutants were transiently transfected into RPE-1 cells. Cells were fixed and stained for EEA1 and DAPI. Representative field of cells from three independent experiments. Scale bars = 20 μm. (C) GFP traps and Western analysis of GFP-SNX17 or GFP-SNX31 and their equivalent loss of function mutations (L470G and L490G). Representative blot from three independent experiments. Unprocessed original scans of immunoblots are shown in Supplementary figure 8.
Supplementary Figure 7 Characterisation of SNX17 knock-out HeLa cells, SNX17 knock-downs for cell surface proteomics and pseudo-virion infection.
(A) Parental HeLa cells or populations of SNX17 cells which have been targeted by CRISPR-Cas9 against either exon 1 or exon 2. Cells were fixed and stained for integrin α5 and the lysosomal marker LAMP1. Scale bars = 10 μm. Representative field of view from one experiment. (B) Cells in (A) were lysed and analysed by Western blot for knock-out of SNX17 and total levels of integrin α5. This experiment was performed once. (C) Knockdowns of SNX17 in SILAC labelled HeLa cells used for cell surface proteomics. 3 independent knock-down experiments used for SILAC proteomics analysed on one blot. (D) Steady-state cell surface analysis of indicated cargo proteins and their dependency on SNX17, retriever and retromer. HeLa cells were knocked-out for the indicated proteins using CRISPR/Cas9. Cell surface proteins were randomly biotinylated with a membrane-impermeable biotin conjugate. Cells were lysed and biotinylated proteins isolated with streptavidin sepharose followed by Western blotting. This experiment was performed once. (E) siRNA knockdowns of cells used in pseudovirion infection assay. Representative blot of 3 independent knock-down experiments. Unprocessed original scans of immunoblots are shown in Supplementary figure 8.
Supplementary information
Supplementary Information
Supplementary Information (PDF 19206 kb)
Supplementary Table 1
Supplementary Information (XLSX 563 kb)
Supplementary Table 2
Supplementary Information (XLSX 2391 kb)
Supplementary Table 3
Supplementary Information (XLSX 34 kb)
Supplementary Table 4
Supplementary Information (XLSX 12 kb)
Rights and permissions
About this article
Cite this article
McNally, K., Faulkner, R., Steinberg, F. et al. Retriever is a multiprotein complex for retromer-independent endosomal cargo recycling. Nat Cell Biol 19, 1214–1225 (2017). https://doi.org/10.1038/ncb3610
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb3610
- Springer Nature Limited
This article is cited by
-
Endosomal protein DENND10/FAM45A integrates extracellular vesicle release with cancer cell migration
BMC Biology (2024)
-
The commander complex is the Swiss Army knife of endosomal trafficking
Nature Structural & Molecular Biology (2024)
-
Structure and interactions of the endogenous human Commander complex
Nature Structural & Molecular Biology (2024)
-
Assembly and fission of tubular carriers mediating protein sorting in endosomes
Nature Reviews Molecular Cell Biology (2024)
-
Profiling the proximal proteome of the activated μ-opioid receptor
Nature Chemical Biology (2024)