Abstract
Melanoma treatment is being revolutionized by the development of effective immunotherapeutic approaches1,2. These strategies include blockade of immune-inhibitory receptors on activated T cells; for example, using monoclonal antibodies against CTLA-4, PD-1, and PD-L1 (refs 3, 4, 5). However, only a subset of patients responds to these treatments, and data suggest that therapeutic benefit is preferentially achieved in patients with a pre-existing T-cell response against their tumour, as evidenced by a baseline CD8+ T-cell infiltration within the tumour microenvironment6,7. Understanding the molecular mechanisms that underlie the presence or absence of a spontaneous anti-tumour T-cell response in subsets of cases, therefore, should enable the development of therapeutic solutions for patients lacking a T-cell infiltrate. Here we identify a melanoma-cell-intrinsic oncogenic pathway that contributes to a lack of T-cell infiltration in melanoma. Molecular analysis of human metastatic melanoma samples revealed a correlation between activation of the WNT/β-catenin signalling pathway and absence of a T-cell gene expression signature. Using autochthonous mouse melanoma models8,9 we identified the mechanism by which tumour-intrinsic active β-catenin signalling results in T-cell exclusion and resistance to anti-PD-L1/anti-CTLA-4 monoclonal antibody therapy. Specific oncogenic signals, therefore, can mediate cancer immune evasion and resistance to immunotherapies, pointing to new candidate targets for immune potentiation.
Similar content being viewed by others
Main
To identify oncogenic pathways inversely associated with T-cell infiltration, we categorized 266 metastatic human cutaneous melanoma samples into those with low (non-T-cell-inflamed) and high (T-cell-inflamed) expression of T-cell signature genes6,10 (Fig. 1a). Comparative gene expression profiling revealed 1,755 genes that were preferentially expressed in the non-inflamed patient cohort (q < 0.01) (Supplementary Table 1). Pathway analysis, comparing 91 non-T-cell-inflamed to 106 T-cell-inflamed patients, indicated active β-catenin signalling (APC2, SOX2, SOX11 and WNT7B; P = 0.00116) as well as dermatan-sulfate biosynthesis (HS6ST2 and NDST3; P = 0.00196) in the non-T-cell-inflamed cohort. Previous reports suggested that active β-catenin signalling in melanoma was associated with more aggressive disease9. To determine if activation of the β-catenin pathway might be modified by specific mutations, we analysed exome-sequencing data for all 197 patients. Indeed, seven tumour samples (7.7%) with the non-T-cell-inflamed phenotype showed gain-of-function mutations in β-catenin (CTNNB1), versus one case in the T-cell-infiltrated cohort. Additionally, loss-of-function mutations in negative regulators of the pathway (APC, AXIN1, TCF1) were identified in ten non-T-cell-inflamed tumours (11%) (Supplementary Table 3). To identify the total percentage of tumours with an active β-catenin pathway, we assessed expression of six well-characterized β-catenin target genes11. Forty-eight per cent (44 patients) in the non-T-cell-inflamed subset showed expression of at least five of the six β-catenin target genes versus 3.8% (4 patients) of the T-cell-inflamed tumours (Fig. 1b). While several cases were associated with defined mutations (CTNNB1, 14%; APC, AXIN1 or TCF1, 23%) the majority (61%) of the remaining cases showed increased expression of either WNT7B (WNT7B, 29.5%; 13 patients), FZD3 (FZD3, 20.5%; 9 patients), or β-catenin itself (11%; 5 patients; Supplementary Table 3). In sum, an increased CTNNB1 score was predictive for the lack of T cells, with an odds ratio of 4.9 (Extended Data Fig. 1a). Additional analysis revealed a negative correlation between individual β-catenin target genes and CD8A transcripts, which was opposite to the pattern of PD-L1 expression (Fig. 1c and Supplementary Table 2)12. Immunohistochemical analysis of an independent sample cohort also revealed an inverse association between stabilized β-catenin and CD8+ T cells (Fig. 1d and Extended Data Fig. 1b).
a, b, Heat maps of 266 metastatic melanomas clustered in low versus high T-cell signature gene groups (a), and β-catenin target genes within the T-cell-signature high and low cohorts (b). c, Pearson correlation of CD8A expression with c-MYC, TCF1 and WNT7B (red indicates T-cell-signature high, blue indicates T-cell-signature low). d, Correlation between β-catenin and CD8 in melanoma biopsies. Fisher’s exact test with n = 49. e, Tumour incidence rates of GEMs (median time to tumour event): BrafV600E/Pten−/−: 100%, 21 days (n = 14); BrafV600E/CAT-STA: 85%, 55.5 days (n = 8); BrafV600E/Pten−/−/CAT-STA: 100%, 26 days (n = 14). f, CD3+ T cells depicted as percentage living cells and absolute numbers per gram tumour. n = 20, mean ± standard error of the mean (s.e.m.), Mann–Whitney U test. g, Representative example out of five for fluorescent immunohistochemistry staining against CD3+ T cells. Scale bars, 100 μm. See Extended Data Fig. 3 for overview. ***P ≤ 0.001, ****P ≤ 0.0001, *****P ≤ 0.00001.
We investigated directly whether active β-catenin signalling within tumour cells could adversely affect anti-tumour T-cell responses using inducible autochthonous mouse models (genetically engineered mice (GEM)) driven by conditional active Braf with or without conditional PTEN deletion and expression of active β-catenin. These GEMs developed tumours with similar latency, as reported previously (Fig. 1e and Extended Data Fig. 2a–c)8,9. We focused on BrafV600E/Pten−/− and BrafV600E/Pten−/−/CAT-STA mice due to the similar rate of onset of tumour development in these strains (Extended Data Fig. 2b, c). Using gene array analysis and histological examination we confirmed that the developing tumours were indeed melanomas (Extended Data Fig. 2d, e)8,9, albeit with less pigmentation in BrafV600E/Pten−/− tumours (Extended Data Figs 2e, f and 3a, b)9. Analysis of immune infiltrates revealed that BrafV600E/Pten−/− tumours indeed contained CD3+ T cells. However, tumours with active β-catenin showed almost a complete absence of T cells (Fig. 1f). Fluorescent immunohistology confirmed the absence of intra-tumoural CD3+ T cells in BrafV600E/Pten−/−/CAT-STA tumours (Fig. 1g and Extended Data Fig. 3a–c) with only rare T cells observed in the epidermis. These results indicate that tumour-intrinsic β-catenin activation dominantly excludes T-cell infiltration into the melanoma tumour microenvironment.
The T-cell infiltrate in BrafV600E/Pten−/− tumours consisted of both CD4+ and CD8+ T cells, with the majority of them expressing the αβ-T-cell antigen receptor (TCR) (Extended Data Fig. 4a, b). The majority were CD44hi/CD62Llo/CD45RAlo, suggesting an activated phenotype (Extended Data Fig. 4c), and 6% FoxP3+ regulatory T cells were detected (Extended Data Fig. 4d). Additionally, CD8+ T cells from BrafV600E/Pten−/− tumours showed expression of PD-1 and Lag3 (Extended Data Fig. 4e, f), markers of T-cell dysfunction in the tumour context13. Consistent with this phenotype, sorted CD3+ T cells from BrafV600E/Pten−/− tumours showed defective interleukin (IL)-2 production but were capable of producing interferon (IFN)-γ (Extended Data Fig. 4g, h). Comparable studies on the few T cells from BrafV600E/Pten−/−/CAT-STA tumours showed predominantly a naive phenotype (Extended Data Fig. 4a–e). Correspondingly, increased PD-L1 expression in BrafV600E/Pten−/− tumours was observed, consistent with previous work linking PD-L1 expression with the presence of CD8+ T cells (Extended Data Fig. 4i, j)12. We did not detect significant differences in CD11b+Gr1+ myeloid-derived suppressor cells (BrafV600E/Pten−/−, 1,047 ± 418, to BrafV600E/Pten−/−/CAT-STA, 739 ± 185 cells per gram tumour; P = 0.7429) (Extended Data Fig. 4k)14.
Although the models used in this study recapitulate defined carcinogenic processes, one drawback is the potentially low number of generated neo-antigens, which may lead to reduced immunogenicity15. To circumvent this we crossed both GEMs to a mouse strain allowing Cre-dependent expressing of the model antigen SIYRYYGL (SIY)16. We investigated whether lack of T-cell infiltration into the BrafV600E/Pten−/−/CAT-STA tumours was secondary to a lack of initial T-cell priming by adoptive transfer of carboxyfluorescein succinimidyl ester (CFSE)-labelled SIY-specific TCR-transgenic 2C T cells. While SIY-negative mice failed to accumulate 2C T cells within the tumour-draining lymph nodes (TdLNs) or the tumour site, SIY-positive mice had detectable 2C T cells in the TdLNs in both GEMs. However, no proliferation of 2C T cells was identified within the TdLNs in the BrafV600E/Pten−/−/CAT-STA/SIY+model, whereas activation of T cells within the TdLNs of BrafV600E/Pten−/− mice was brisk (Fig. 2a, b). Accordingly, the presence of proliferated 2C T cells was observed at the tumour site exclusively in BrafV600E/Pten−/− mice (Fig. 2a, b). These data indicate that tumour-intrinsic β-catenin signalling prevents the early steps of T-cell priming against tumour-associated antigens.
a, Abundance and proliferation of TCR-transgenic 2C T cells. Depicted are representative examples pre-gated on live, CD45+CD3+CD8+ cells. b, Statistical analysis of a (n = 8). c, Percentages of dendritic cell subsets within BrafV600E/Pten−/− and BrafV600E/Pten−/−/CAT-STA tumours (n = 8). d, Representative example of CD103/CD8α staining (gated CD45+MHCIIhiCD11c+). e, Quantification of CD103+ dendritic cells (n = 12). f, Amount of CD3+ T cell and CD103+ dendritic cell (DC) infiltration in BrafV600E/Pten−/− tumours reconstituted with control or Batf3−/− bone marrow (n = 4 and n = 11, respectively). g, Intra-tumoural injection of Flt3 ligand-derived dendritic cells into BrafV600E/Pten−/−/CAT-STA tumours (n = 6 control mice and 8 mice, PBS control). All data are mean ± s.e.m., Mann–Whitney U test. *P ≤ 0.05, ***P ≤ 0.001, ****P ≤ 0.0001; NS, not significant.
The absence of early T-cell priming in BrafV600E/Pten−/−/CAT-STA tumour-bearing mice suggested a defect in the antigen-presenting-cell compartment. Work using transplantable tumour models has indicated that Batf3-lineage dendritic cells are crucial for cross-presentation of tumour antigens to CD8+ T cells17,18,19. Dendritic cell subsets (CD45+MHCII+CD11c+) were analysed phenotypically within the tumour microenvironment with minimal differences observed in the number of conventional dendritic cells (B220−), plasmacytoid dendritic cells (B220+), monocytes (B220−Ly6C+), or Langerhans dendritic cells (B220−CD207+). Strikingly, the CD8α+ and CD103+ dendritic cell populations were nearly completely absent from BrafV600E/Pten−/−/CAT-STA tumours (Fig. 2c–e). CD103+ dendritic cells were also reduced in the TdLNs, while being preserved in the spleen (Fig. 2d, e; data not shown). Sorted tumour-infiltrating CD45+CD11c+ dendritic cells from BrafV600E/Pten−/−/CAT-STA tumours also showed reduced expression of the CD103+ dendritic cell transcripts Batf3, Irf8 and Itgae (Extended Data Fig. 5a, b), and dendritic cells showed reduced expression of the key innate cytokine IFN-β (Extended Data Fig. 5a). Together, these results suggest that the failed T-cell priming against tumour-associated antigen in BrafV600E/Pten−/−/CAT-STA tumours is secondary to defective recruitment and activation of Batf3-lineage dendritic cells.
To determine whether T-cell infiltration into BrafV600E/Pten−/− tumours was dependent on CD103+ dendritic cells, Batf3−/− bone marrow chimaeras were generated. Indeed, tumours from BrafV600E/Pten−/−/Batf3−/− bone marrow chimaeras failed to develop T-cell infiltration (Fig. 2f). To assess whether poor dendritic cell recruitment was indeed the major functional barrier, we generated Flt3 ligand-derived bone-marrow dendritic cells activated with polyinosinic:polycytidylic acid (poly(I:C))20 for intra-tumoural injection, which were found to restore T-cell infiltration in BrafV600E/Pten−/−/CAT-STA tumours (Fig. 2h) and led to a modest reduction in tumour weight (Extended Data Fig. 5c). Using dendritic cells generated from actin–green fluorescent protein (GFP) transgenic mice, injected dendritic cells were retained within the tumour microenvironment during this experimental timeframe (Extended Data Fig. 5d). Together, these results suggest that the major immunological defect in the context of melanomas expressing tumour-intrinsic β-catenin signalling is defective recruitment of CD103+ dendritic cells.
To pursue mechanisms explaining failed CD103+ dermal dendritic cell recruitment, gene expression profiling was performed from tumours of the two genotypes, focusing on chemokines (Supplementary Table 4). Five chemokines were differentially expressed, with four of these (CCL3, CXCL1, CXCL2 and CCL4) being expressed at lower levels in BrafV600E/Pten−/−/CAT-STA tumours (Fig. 3a, b and Supplementary Table 4). For evaluation of tumour-cell-intrinsic chemokine production in vivo, we crossed BrafV600E/Pten−/− mice to yellow fluorescent protein (YFP)-reporter mice, which allowed identification of transformed YFP+ cells. Ccl4 transcripts were detected exclusively in the YPF+ cell population from BrafV600E/Pten−/− mice, while control sorted YFP+ cells from BrafWT/Pten−/− mice or YFP− cells showed no detectable Ccl4 (Fig. 3c). A similar expression pattern was observed for CXCL1, whereas CCL3 and CXCL2 were expressed by normal melanocytes and stromal cells, respectively (Fig. 3c). CD45+CD3+ and CD45+CD3− cells sorted as controls, showing the expected patterns of Ifng and Ifnb expression (Extended Data Fig. 6c, d). Expression analysis of the corresponding chemokine receptor, Ccr5, revealed a lack of CCR5 expression by the dendritic cells isolated from BrafV600E/Pten−/−/CAT-STA tumours (Fig. 3d). CCR5 has previously been linked with the migratory capacity of CD8α+ dendritic cells21. To confirm this observation, we generated tumour cell lines from both GEMs and found increased production of CCL4 by BP (BrafV600E/Pten−/−-derived) tumour cells compared to BPC (BrafV600E/Pten−/−/CAT-STA-derived) tumour cells (Extended Data Fig. 6a, b). To strengthen a functional role for CCL4, we used an in vitro migration assay in response to recombinant murine CCL4 as well as tumour cell line supernatants (Fig. 3e). Indeed, skin-derived CD11c+CD103+ dendritic cells and lymph-node-derived dendritic cells (CD11c+CD8α+) migrated in response to CCL4 and BP supernatants but not to BPC supernatants. Together, these results indicate that failed recruitment of CD103+ dendritic cells into the tumour microenvironment of BrafV600E/Pten−/−/CAT-STA tumours was, at least in part, due to defective production of the chemokine CCL4.
a, Chemokine expression in GEM tumours assessed via gene array analysis (n = 4). b, Confirmatory quantitative polymerase chain reaction with reverse transcription (qRT–PCR) (n = 8) with fold change (FC) indicated at the top. c, Transcript levels of Ccl3, Ccl4, Cxcl1 and Cxcl2 assessed from YFP+ and CD45− YFP− cells from BrafV600E/Pten−/−/YFP+ tumours (n = 5), sorted on day 7 after tamoxifen administration. ND, not detected. d, Expression level of CCR5 in sorted CD45+CD11c+ dendritic cells (n = 8). e, Migration assay of dendritic cell subsets towards recombinant mouse CCL4 or conditioned medium (SF) (two independent experiments, duplicates per experiment). f, Atf3 transcripts in tumour tissues (n = 8). g, ATF3-specific ChIP assay in BP and BPC cell lines (two independent experiments, duplicates per experiment). h, Amount of secreted CCL4 in 48-h-conditioned siRNA-treated tumour-cell BP and BPC supernatants, assessed by enzyme-linked immunosorbent assay (ELISA) and Atf3 expression at the endpoint detected by qRT–PCR (two independent experiments, duplicates per experiment). All data are mean ± s.e.m., Mann–Whitney U test. *P ≤ 0.05, **P ≤ 0.01, ****P ≤ 0.0001; NS, not significant.
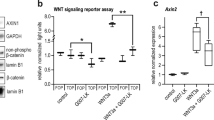
We then pursued a mechanism by which β-catenin activation might prevent Ccl4 gene expression, since CCL4 has also been associated with a T-cell infiltrate in human melanoma tumours6,22. Previous reports had suggested that Wnt/β-catenin signalling induces expression of the transcriptional repressor ATF3 (ref. 23), and that ATF3 suppresses Ccl4 (ref. 24). Indeed, Atf3 was expressed at higher levels in primary tumours as well as in BPC tumour cell lines from BrafV600E/Pten−/−/CAT-STA mice (Fig. 3f). A chromatin immunoprecipitation (ChIP) assay revealed binding of ATF3 to the Ccl4 promoter region in BrafV600E/Pten−/−/CAT-STA cells while no binding was observed for Ccl2, a chemokine lacking an ATF3-binding site (Fig. 3g). Short interfering RNA (siRNA)-mediated knockdown of Atf3 or Ctnnb1 in BPC tumour cells restored CCL4 production (Fig. 3h). To examine this relationship in human melanoma, we analysed two melanoma cell lines, mel537 and mel888, which show low or high β-catenin expression, respectively (Extended Data Fig. 7a, c). Consistent with the murine cell lines, increased ATF3 and decreased CCL4 production were observed in the β-catenin-positive mel888 cells (Extended Data Fig. 7b, e), and increased binding of ATF3 to the CCL4 promoter was also detected (Extended Data Fig. 7d). siRNA-mediated knockdown of ATF3 or β-catenin in mel888 cells restored CCL4 production (Extended Data Fig. 7e).
We additionally investigated whether decreased presence of BATF3-lineage dendritic cells was associated with active β-catenin signalling in human melanoma metastases. A Pearson correlation analysis for expression of THBD (CD141, marker for human BATF3-lineage dendritic cells25; P < 0.0001), BATF3 (P = 0.0336) and IRF8 (P < 0.0001) revealed a negative association with the CTNNB1 score (Extended Data Fig. 8 and data not shown). Furthermore, CCL4 had already been observed to correlate positively with T-cell transcripts (Fig. 1a). We conclude that β-catenin activation within melanoma cells results in decreased CCL4 gene expression, which is at least partly mediated through ATF3-dependent transcriptional repression (Extended Data Fig. 9).
To explore the therapeutic relevance of the lack of T-cell infiltration, both GEMs were treated with a combination of anti-CTLA-4 and anti-PD-L1 monoclonal antibodies3,26. While treatment of BrafV600E/Pten−/− mice resulted in a significant delay in tumour outgrowth, no therapeutic effect was detected in BrafV600E/Pten−/−/CAT-STA mice (Fig. 4a, b). To evaluate whether restoration of intra-tumoural dendritic cells could restore immunotherapy responsiveness, Flt3 ligand-induced bone-marrow dendritic cells were injected intra-tumourally into BrafV600E/Pten−/−/CAT-STA tumours. Indeed, introduction of dendritic cells had a partial therapeutic effect, which was improved significantly with anti-CTLA-4 and anti-PD-L1 monoclonal antibodies (Fig. 4c).
a, b, Tumour growth in BrafV600E/Pten−/− (a) and BrafV600E/Pten−/−/CAT-STA ( b) mice untreated or treated with anti-CTLA-4 and anti-PD-L1 therapy (n = 10). c, Tumour growth of BrafV600E/Pten−/−/CAT-STA tumour-bearing mice that were untreated, treated with anti-CTLA-4 and anti-PD-L1 therapy, intra-tumoural Flt3 ligand (Flt3-L) dendritic cell injections, or combination therapy (n = 5). BM-DC, bone-marrow dendritic cell; mAb, monoclonal antibody. All data are mean ± s.e.m., two-way analysis of variance (ANOVA) test. **P ≤ 0.01, ****P ≤ 0.0001; NS, not significant.
We conclude that melanoma-cell-intrinsic activation of an oncogenic pathway can result in exclusion of the host immune response, including the absence of a T-cell infiltrate within the tumour microenvironment. Although 48% of non-T-cell-infiltrated melanomas show active β-catenin signalling, it is conceivable that additional oncogenic signalling pathways might mediate immune exclusion in other cases. The WNT/β-catenin pathway may contribute to immune evasion in other tumour entities beyond melanoma, which would be consistent with previous in vitro work27. Within T cells, β-catenin appears to inhibit T-cell activation, suggesting that a general immune-potentiating effect may result from therapeutic targeting28. The T-cell-inflamed tumour microenvironment phenotype appears to be predictive of clinical response to immune-based therapies7,10,29. Immune escape among this subset appears to be a consequence of dominant effects of negative regulatory pathways such as PD-1, arguing that the clinical activity of anti-PD-1 is tipping the balance in favour of an ongoing immune response12. By inference, tumour-intrinsic β-catenin activation may represent one mechanism of primary resistance to these therapies.
Methods
Analysis of TCGA data set
Level 4 gene expression data and level 2 somatic mutation data were downloaded for skin cutaneous melanoma (SKCM) from TCGA, which were processed by Broad Institute’s TCGA workgroup (release date 10 October 2013). The RNA-seq level 4 gene expression data contain upper-quartile-normalized and log2-transformed RNA-seq by expectation maximization (RSEM) values summarized at gene level30. The whole-exome sequencing (WXS) level 2 mutation data contains somatic mutation calls for each subject. A total of 266 metastatic SKCM samples were analysed. For clustering of cold and hot tumours, genes expressed in less than 80% of the samples were removed. A total of 15,974 genes were kept for further analysis. Unsupervised hierarchical clustering of the genes was performed in primary tumours and metastasis samples separately using K-mean equal to 12 and Euclidean distance metrics. Clusters containing the 13 known T-cell-signature transcripts (CD8A, CCL2, CCL3, CCL4, CXCL9, CXCL10, ICOS, GZMK, IRF1, HLA-DMA, HLA-DMB, HLA-DOA, HLA-DOB) were selected for resampling-based hierarchical clustering of the samples using ConsensusClusterPlus v.1.16.0 (ref. 31). This procedure was performed with 2,000 random selections of 80% of the samples and Euclidean distance metrics. Genes differentially expressed between cold and hot tumour groups were detected using ANOVA and filtered by false discovery rate (FDR) q value < 0.01 and fold change > 2.0. Canonical pathways significantly enriched in the genes of interest were identified by Ingenuity Pathways Analysis (IPA) (Ingenuity Systems; http://www.ingenuity.com) based on experimental evidence from the Ingenuity Knowledge Base (release date 23 March 2014). The somatic variants were converted to VCF format and annotated using ANNOVAR (release date 23 August 2013)32. Each variant was annotated with known genes, exonic functions, predicted amino acid changes and minor allele frequencies derived from the 1000 Genomes Project (phase 1, release v.3, 23 November 2010) and the NHLBI Exome Sequencing Project (ESP6500SI-V2-SSA137) (EVS)33. Synonymous single-nucleotide variants (SNVs) were excluded from further analysis. The variants were then summarized at gene level and patient level for comparison of mutation profiles between the cold and hot tumour groups. Interactions between proteins encoded by genes of interest were retrieved from the STRING database based on high-confidence evidence collected from co-expression data, experiments and databases34. SNVs located in selected genes were analysed using the Variant Effect Prediction (http://www.ensembl.org/info/docs/tools/vep/index.html) software in combination with the UniProt database (http://www.uniprot.org). Calls of loss-of-function and gain-of-function were based on existing experimental data obtained from the UniProt data base, while harmful or tolerated effects on the protein structure were predicted using the SIFT prediction algorithm imbedded in the Variant Effect Predictions analysis. A continuous numerical score was generated using the six β-catenin target genes (EFNB3, APC2, TCF1, c-MYC, TCF12, VEGFA) reads. The resulting score was used to align patients based on activity of the β-catenin pathway.
Mice, tumour induction and generation of tumour cell lines
The following mouse strains were gifts from collaborators and were used to generate the mouse models used in this study: Tyr:Cre-ER (gifted by L. Chin), LSL-BrafV600E (provided by M. MacMahon), Ptenfl/fl(provided by T. Mak), LSL-CAT-STA (provided by F. Gounari), Rosa26-LSL-SIY and Rosa26-LSL-YFP (Jackson Laboratories, strain 006148) reporter16,35,36,37,38. As an initial cross, the Tyr:Cre-ER mice were crossed onto LSL-BrafV600E and subsequently crossed with the loxP-Pten mouse strain. Those mice were maintained as Tyr:Cre-ER+, LSL-BrafV600E+/−, Ptenfl/fl and will be referred to as BrafV600E/Pten−/−. Additionally Tyr:Cre-ER, LSL-BrafV600E mice were crossed to the LSL-CAT-STA mouse strain with subsequent crossing to the Ptenfl/flstrain. Those mouse strains were maintained as Tyr:Cre-ER+, LSL-BrafV600E+/−, LSL-CAT-STA+/+ and Tyr:Cre-ER+, LSL-BrafV600E+/−, Ptenfl/fl, LSL-CAT-STA+/+ and will be referred to as BrafV600E/CAT-STA or BrafV600E/Pten−/−/CAT-STA, respectively. Additionally, the BrafV600E/Pten−/− and BrafV600E/Pten−/−/CAT-STA mice were crossed to the Rosa26-LSL-SIY mouse and mice were maintained heterozygote for the Rosa26 locus. Similarly, BrafV600E/Pten−/− mice were bred onto the Rosa26-LSL-YFP reporter strain, which were also maintained with heterozygous breeders for this locus. Genotyping was performed as described previously16,35,36,37,38,39 (for primer sequences, see Supplementary Table 5). For tumour induction, 6–10-week-old mice were shaved on the back and 5 μl of 4-OH-tamoxifen (Sigma) at a concentration of 10 mg ml−1 (dissolved in acetone) were applied. Subsequently, mice were screened weekly for tumour induction and growth with endpoint criteria of 4,000 mm3. For tumour cell line generation, a single-cell suspension of the tumour tissue was generated as described later and as its entirety used for subcutaneous injections into Rag-knockout mice (RAGN12-F; Taconic). After tumour outgrowth, the tumour tissue was harvested and re-injected into Rag-knockout mice, C57BL/6 mice (Taconic), and adapted to cell culture using DMEM (Gibco) with 10% FCS (Atlanta Biologics), 1× NEAA (Gibco) and 1× MOPS (Sigma). In this work we used one cell line derived from each genotype, BrafV600E/Pten−/− and BrafV600E/Pten−/−/CAT-STA. Additionally, TCR-transgenic 2C T cells were maintained as T-cell donors40, actin–GFP mice were obtain from Jackson (strain identifier 003291), Batf3−/− mice were maintained as bone marrow donors and were originally obtained from K. Murphy18. All animal procedures were approved by the Institutional Animal Care and Use Committee of the University of Chicago. Human tumour cell lines were obtained from National Cancer Institute and maintained in RPMI medium supplemented with 10% FCS and 1× NEAA.
Tumour growth, tissue harvest and single-cell suspensions
For tumour outgrowth experiments, mice were treated at the lower back with 4-OH-tamoxifen at day 0. After day 21, tumour masses were measured by assessing length, width and height of major tumour mass using a digital calliper. Measuring the height was a critical parameter to assess tumour growth, since width and length were mainly influenced given by the spread of the TAM solution. Tumour volume TV was calculated: TV = TL × TW × TH, where TL is tumour length, TH is tumour height and TW is tumour width, since the tumour shape was rectangular and flat rather than spherical. The maximum tumour size was reached when the tumour mass reached approximately 10% of the body weight. At the indicated experimental endpoint, tumour tissue was harvested, cleared from remaining skin and minced using razor blades. Subsequently, tumour pieces were digested using the human tumour digestion kit (Miltenyi) in combination with the tissue dissociater (Miltenyi). For flow cytometric analysis and cell sorting, living cells were separated using a ficoll (GE) centrifugation step with subsequent washing of the obtained cells. For generation of tumour cell lines, the cell suspension was used directly after digestion and two washing steps.
Immunohistochemistry and fluorescent immunohistology
The immunohistology staining on human samples was performed by the Human Tissue Resource Center of the University of Chicago using biopsies from malignant melanoma patients. Staining was performed using a CD8-specific monoclonal antibody (CD8 clone C8/144B, NeoMarkers), β-catenin (clone CAT-5H1, Life Technologies) in combination with a secondary goat anti-mouse immunoglobulin G (IgG) conjugated to an alkaline phosphatase (Biocare Medical) was applied. Slides were scanned using a CRi Panoramic Scan Whole Slide Scanner. Positivity for β-catenin staining was obtained first and grading was based on the staining intensity. Subsequently, the number of CD8-positive T cells within one needle biopsy (2.5 mm diameter) was counted using ImageJ cell counter and calculated as number of CD8+ T cells per mm2. Samples with fewer than 50 CD8+ T cells per mm2 were considered T-cell-infiltrate low whereas counts >50 per mm2 were considered as T-cell high, similar to as described previously41. For mouse fluorescent immunohistology staining, formalin/paraffin-fixed tissues were used to obtain 5 μm sections for subsequent staining. Staining was performed using the following primary antibodies: anti-CD3 (clone SP7, 1:500, Abcam) and anti-Trp1 (clone EPR13063, 1:500, Abcam) in combination with goat anti-rabbit 594 (JacksonImmuno) and Hoechst counterstain. Slides were imaged using a Zeiss Axiovert 200 with a Hammatsu Orca ER firewire digital monochrome camera.
Flow cytometry and cell sorting
For flow cytometric analysis, washed cells were resuspended in staining buffer (PBS with 10% FCS and 0.5 M EDTA (Ambion)). Cells were incubated with live/dead staining dye (Invitrogen, wavelength 450 nm) and Fc Block (clone 93; Biolegend) for 20 min on ice. Subsequently, specific antibodies were added (Supplementary Table 5) and staining was continued for 40 min on ice. After a washing step, cells were either analysed directly or fixed with 4% PFA (BD) solution for 30 min and stored in a 1% PFA solution until analysis. For staining of TCR-transgenic 2C T cells a TCR specific-biotinylated monoclonal antibody (1B2 clone) was obtained from the University of Chicago Monoclonal Core Facility. Subsequent to live/dead staining, TCR-specific monoclonal antibody was added for 15 min on ice at a 1:100 dilution alone with surface antibodies targeting other antigens added in for an additional 25 min thereafter. After a washing step, a 1:500 dilution of Streptavidin APC was added and incubated on ice for 20 min before cells were fixed in 4% PFA and stored in 1% PFA solution. Flow cytometry sample acquisition was performed on a LSR2B (BD), and analysis was performed using FlowJo software (TreeStar). For cell sorting, staining protocols were carried out similarly under sterile conditions. Cell sorting was performed using an ARIAIIIu (BD) and cells were collected in 100% FCS if further used for in vitro analysis or in TriZol Reagent (Invitrogen) if used for RNA isolation. Percentage of T cells was calculated as follows ((100/number of total living cells acquired) × number of CD3+ T cells); number per gram tumour was calculated as follows (number of acquired CD3+ T cells/tumour weight).
T-cell stimulation
2.5 × 104 sorted T cells from spleen and/or tumour were either stimulated on plates coated with 1 μg ml−1 anti-CD3 antibody (145-2C11 clone; Biolegend) and 2 mg ml−1 anti-CD28 antibody (37.51 clone BD) in T-cell medium (DMEM, 10% FCS, 1× NEAA, 1× MOPS, 500 μM β-mercapthoethanol (Sigma)) or plated on tissue-culture-treated uncoated plates for 8 h. Following incubation, cells were harvested and resuspended in TriZol Reagent (Invitrogen) for subsequent RNA isolation.
RNA isolation and qRT–PCR
RNA isolation using TriZol was performed according to the manufacturer’s instruction. In the case of RNA isolation from whole tumour tissue, a piece of tumour was snap frozen in TriZol at the time of tumour harvest. Before RNA isolation the tissue was thawed at room temperature and homogenization was achieved using a tissue homogenizer (GE) with homogenizer tips (USA Scientific). Subsequent RNA isolation was performed according to the manufacturer’s instructions. Reverse transcriptase reaction was performed using High Capacity cDNA RTPCR Kit (Life Technologies) according to instructions and 1 μl of the resulting copy DNA was used for qPCR. qPCR reactions were carried out using Sybr Green or TaqMan master mix (Life Technologies) and defined primer sets or primer/probe sets (probes were obtained from Roche), respectively (Supplementary Table 5). Reactions were run on a 7300 RT PCR system machine (Applied Biosystems) and expression level and fold change were calculated as follows: ΔCT = CTgene of interest − CT18S; expression level = 2−ΔCT; fold change (ref. 42).
Adoptive T-cell transfer
For adoptive transfer experiments, tumour development was induced and transfer of 1 × 106 T cells was performed when tumour reached near endpoint sizes (approximately 3–4 weeks after induction). Transferred T cells were isolated from gender-matched 2C donor mice using the Miltenyi CD8+ enrichment Kit II for untouched CD8+ T-cell isolation. After isolation, cells were stained with 1 μM CFSE solution (eBioscience) for 8 min at 37 °C before intravenous injection. Tumour tissue, tumour-draining lymph nodes and spleen were harvested 5 days after adoptive transfer of T cells and used for flow cytometric analysis. This short timeframe was chosen to avoid the reported leakiness of the SIY transgene that has been associated with partial T-cell activation within the spleen16. For tumour tissues, the entirety of each sample was acquired and the total number of CD3+CD8+ T cells and transferred 2C cells was assessed. The percentage 2C cells was calculated as ((100/CD3+/CD8+ T cells) × 2C) and also the number of 2C cells per gram tumour.
Generation of bone marrow chimaeras
To condition host mice to generate bone marrow chimaeras, indicated mouse strains were irradiated twice with a 3 h interval and a first irradiation dose of 500 rad followed by 550 rad. Twenty-four hours after the second irradiation dose, bone marrow from gender-matched donor mice was isolated from femur and tibia of both legs, washed, and erythrocytes were lysed. 3 × 106 bone marrow cells were injected intravenously to reconstitute the mice. Two-to-three months after bone marrow transfer, tumour development was induced as described previously.
Generation and administration of bone-marrow-derived dendritic cells
For administration of bone-marrow-derived dendritic cells, bone marrow from C57BL/6 mice or GFP–actin mice was collected from the femurs and tibias of both legs. After washing and lysis of erythrocytes, bone marrow cells were cultured in RPMI (Gibco) complete medium (10% FCS, 1× NEAA, 500 μM β-ME) supplemented with 300 ng ml−1 Flt3 ligand (eBioscience) for 7 days at a concentration of 2.5 × 106 cells ml−1. Dendritic cells were then activated for 24 h with poly(I:C) (InvivoGen) at a final concentration of 5 μg ml−1 (pre-heated for 5 min at 95 °C). Activated Flt3 ligand dendritic cells were frozen in aliquots of 5 × 106 cells in 90% FCS with 10% dimethylsulfoxide (DMSO; Sigma) until use for in vivo administration. For each dendritic cell preparation, activation marker expression was analysed using flow cytometry with the majority of cells being CD11c+, CD11b+, predominantly CD8α+ and after activation high expression of CD80, CD86, MHCII and CD40 was observed. Injection of dendritic cells was initiated when the first signs of tumour lesions were identified on mice (2–3 weeks after induction) and were given intra-dermally/intra-tumourally using a 27G (Braintree) needle twice per week at a dose of 1 × 106 dendritic cells per injection.
Gene array analysis of mouse tumour tissue
For gene array analysis, RNA from whole tumour tissue was isolated. Subsequent experimental procedures were performed by the University of Chicago Genomics Core facility using the Illumina MouseWG-6 gene array chip (Illumina) according to the manufacturer’s instructions. Subsequent gene lists were analysed from differentially expressed genes with a cut off for at least twofold change between the two analysed cohorts. Significance was determined using a two-way ANOVA test.
Trans-well migration assay
Dendritic cell populations were isolated from lymph nodes and skin of naive 6-week-old C57BL/6 mice. For this purpose, skin tissue was digested in a similar way as tumour tissue and cells from skin and lymph node were stained using the previously described protocol for cell sorting. Subsequently, living, CD45+, CD11c+, CD8α− or CD8α+ cells were isolated from the lymph node sample as well as living, CD45+, CD11c+ or CD103+ cells from skin samples. Migration assays were performed as described previously with minor adaptations using 5 × 105 cells per well and pre-treatment of dendritic cells with pertussis toxin (Sigma) at a final concentration of 20 ng ml−1 for 1.5 h as indicated43. As a migration stimulus, CCL4 (R&D) was added to RPMI complete medium at 500 ng ml−1 or 48 h conditioned media from BrafV600E/Pten−/− or BrafV600E/Pten−/−/CAT-STA cell lines were used. At the endpoint, cells from the lower compartment as well as the trans-well were harvested and counted using a standard Neubauer counting chamber. Percentage of migrated cells was calculated as follows: count lower well/(count upper well + count lower well) × 100; with the sum of trans-well and lower well being >90% of the input cell count.
ELISA
ELISA assays against murine and human CCL4 were performed using CCL4-specific ELISA kits (R&D) according to the manufacturer’s instructions.
siRNA knockdown
Target gene-specific and control siRNAs were obtained from Ambion and can be found in Supplementary Table 5. For knockdown, 3 × 104 tumour cells are plated in 96-well plates at a concentration of 3 × 105 per ml. Opti-MEM (Gibco) was mixed with 1.2 pmol siRNA and 1.5% RNAiMAX reagent (Invitrogen) and added to the culture at a ratio of 1:5. Cells were incubated for 48 h before supernatant was harvested for ELISA assays and cells were collected for RNA or protein extraction.
Western blotting
Cell lysates were generated using RIPA buffer in combination with protein inhibitor (Invitrogen) and protein concentration was determined using Bradford protein assay (Biorad). Denaturated lysates were applied to a 10% SDS–PAGE and blotted using standard procedures. For protein detection, blots were incubated with primary antibodies (β-catenin clone D10A8; β-actin clone 13E5; Cell Signaling) overnight and with secondary antibodies (donkey anti-rabbit-HRP; GE Healthcare) for 2 h. Chemiluminescence was used to visualize the protein bands (GE Healthcare).
ChIP assay
For ChIP assays, the two cell lines, BP and BPC, were grown to 80% confluence in a 10 cm Petri dish. Cells were fixed with 1% formaldehyde solution for 30 min at 37 °C. Subsequent steps were performed using the EpiTect ChIP kit (Qiagen) according to the manufacturer’s instructions with some minor adaptations. In brief, the formaldehyde was removed and cells were washed before harvesting using RIPA buffer. Sonication was performed using a water bath sonicator (GE) with the following cycle of 30 s on/15 s off at maximum voltage for 15 min, and this cycle was repeated three times at 4 °C. Chromatin-containing supernatants were incubated with an ATF3-specific antibody (mouse: polyclonal rabbit IgG; human: clone 44C3a, mouse IgG; Abcam) or rabbit/mouse IgG1 isotype (Cell Signaling) for 3 h or overnight at a 1:50 dilution. Pulled-down DNA was used as template for qPCR using Sybr Green master mix and primers (Supplementary Table 5). Results were calculated as followed: ΔCT = ΔCTIP− (ΔCTIP− log2100), fold enrichment .
Monoclonal antibody therapy
Therapy using monoclonal antibodies was initiated either when the tumour was first palpable or 7 days after dendritic cell injection was initiated. Mice were assigned to groups in a randomized fashion based on their ear tag number. Antibodies (CTLA-4 clone 9H10, PD-L1 clone 10F.9G2; BioXcell) were administered every other day throughout the experiment at a dose of 100 μg per mouse per treatment and treatment was initiated 3 weeks after tamoxifen application26.
Statistical analysis
All statistical analyses were performed using GraphPad Prism (GraphPad) with the exception of analyses of the TCGA data set. Unless otherwise noted, all data are shown as mean ± s.e.m. combined with a two-tailed Mann–Whitney U test. Significance was assumed with P ≤ 0.05. For correlation studies, a Gaussian fit was performed to assure normal distribution. All experiments shown were repeated at least in two independent experiments.
References
Kaufman, H. L. et al. The Society for Immunotherapy of Cancer consensus statement on tumour immunotherapy for the treatment of cutaneous melanoma. Nature Rev. Clin. Oncol. 10, 588–598 (2013)
Mellman, I., Coukos, G. & Dranoff, G. Cancer immunotherapy comes of age. Nature 480, 480–489 (2011)
Wolchok, J. D. et al. Nivolumab plus ipilimumab in advanced melanoma. N. Engl. J. Med. 369, 122–133 (2013)
Topalian, S. L. et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J. Clin. Oncol. 32, 1020–1030 (2014)
Hodi, F. S. et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 363, 711–723 (2010)
Harlin, H. et al. Chemokine expression in melanoma metastases associated with CD8+ T-cell recruitment. Cancer Res. 69, 3077–3085 (2009)
Ji, R. R. et al. An immune-active tumor microenvironment favors clinical response to ipilimumab. Cancer Immunol. Immunother. 61, 1019–1031 (2012)
Dankort, D. et al. Braf(V600E) cooperates with Pten loss to induce metastatic melanoma. Nature Genet. 41, 544–552 (2009)
Damsky, W. E. et al. β-Catenin signaling controls metastasis in Braf-activated Pten-deficient melanomas. Cancer Cell 20, 741–754 (2011)
Galon, J. et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313, 1960–1964 (2006)
Rimm, D. L., Caca, K., Hu, G., Harrison, F. B. & Fearon, E. R. Frequent nuclear/cytoplasmic localization of β-catenin without exon 3 mutations in malignant melanoma. Am. J. Pathol. 154, 325–329 (1999)
Spranger, S. et al. Up-regulation of PD-L1, IDO, and Tregs in the melanoma tumor microenvironment is driven by CD8+ T cells. Sci. Transl. Med. 5, 200ra116 (2013)
Woo, S. R. et al. Immune inhibitory molecules LAG-3 and PD-1 synergistically regulate T-cell function to promote tumoral immune escape. Cancer Res. 72, 917–927 (2012)
Landsberg, J. et al. Melanomas resist T-cell therapy through inflammation-induced reversible dedifferentiation. Nature 490, 412–416 (2012)
Matsushita, H. et al. Cancer exome analysis reveals a T-cell-dependent mechanism of cancer immunoediting. Nature 482, 400–404 (2012)
Cheung, A. F., Dupage, M. J., Dong, H. K., Chen, J. & Jacks, T. Regulated expression of a tumor-associated antigen reveals multiple levels of T-cell tolerance in a mouse model of lung cancer. Cancer Res. 68, 9459–9468 (2008)
Fuertes, M. B. et al. Host type I IFN signals are required for antitumor CD8+ T cell responses through CD8α+ dendritic cells. J. Exp. Med. 208, 2005–2016 (2011)
Hildner, K. et al. Batf3 deficiency reveals a critical role for CD8α+ dendritic cells in cytotoxic T cell immunity. Science 322, 1097–1100 (2008)
Bedoui, S. et al. Cross-presentation of viral and self antigens by skin-derived CD103+ dendritic cells. Nature Immunol. 10, 488–495 (2009)
Mollah, S. A. et al. Flt3L dependence helps define an uncharacterized subset of murine cutaneous dendritic cells. J. Invest. Dermatol. 134, 1265–1275 (2014)
Aliberti, J. et al. CCR5 provides a signal for microbial induced production of IL-12 by CD8α+ dendritic cells. Nature Immunol. 1, 83–87 (2000)
Peng, W. et al. PD-1 blockade enhances T-cell migration to tumors by elevating IFN-γ inducible chemokines. Cancer Res. 72, 5209–5218 (2012)
Li, Y. et al. N-myc downstream-regulated gene 2, a novel estrogen-targeted gene, is involved in the regulation of Na+/K+-ATPase. J. Biol. Chem. 286, 32289–32299 (2011)
Khuu, C. H., Barrozo, R. M., Hai, T. & Weinstein, S. L. Activating transcription factor 3 (ATF3) represses the expression of CCL4 in murine macrophages. Mol. Immunol. 44, 1598–1605 (2007)
Jongbloed, S. L. et al. Human CD141+ (BDCA-3)+ dendritic cells (DCs) represent a unique myeloid DC subset that cross-presents necrotic cell antigens. J. Exp. Med. 207, 1247–1260 (2010)
Spranger, S. et al. Mechanism of tumor rejection with doublets of CTLA-4, PD-1/PD-L1, or IDO blockade involves restored IL-2 production and proliferation of CD8+ T cells directly within the tumor microenvironment. J. Immunother. Cancer (2014)
Yaguchi, T. et al. Immune suppression and resistance mediated by constitutive activation of Wnt/β-catenin signaling in human melanoma cells. J. Immunol. 189, 2110–2117 (2012)
Driessens, G. et al. β-Catenin inhibits T cell activation by selective interference with linker for activation of T cells–phospholipase C-γ1 phosphorylation. J. Immunol. 186, 784–790 (2011)
Cipponi, A., Wieers, G., van Baren, N. & Coulie, P. G. Tumor-infiltrating lymphocytes: apparently good for melanoma patients. But why? Cancer Immunol. Immunother. 60, 1153–1160 (2011)
Li, B. & Dewey, C. N. RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinformatics 12, 323 (2011)
Wilkerson, M. D. & Hayes, D. N. ConsensusClusterPlus: a class discovery tool with confidence assessments and item tracking. Bioinformatics 26, 1572–1573 (2010)
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 38, e164 (2010)
The 1000 Genomes Project Consortium. An integrated map of genetic variation from 1,092 human genomes. Nature 491, 56–65 (2012)
Jensen, L. J. et al. STRING 8—a global view on proteins and their functional interactions in 630 organisms. Nucleic Acids Res. 37, D412–D416 (2009)
Suzuki, A. et al. High cancer susceptibility and embryonic lethality associated with mutation of the PTEN tumor suppressor gene in mice. Curr. Biol. 8, 1169–1178 (1998)
Gounari, F. et al. Stabilization of β-catenin induces lesions reminiscent of prostatic intraepithelial neoplasia, but terminal squamous transdifferentiation of other secretory epithelia. Oncogene 21, 4099–4107 (2002)
Dankort, D. et al. A new mouse model to explore the initiation, progression, and therapy of BRAFV600E-induced lung tumors. Genes Dev. 21, 379–384 (2007)
Bosenberg, M. et al. Characterization of melanocyte-specific inducible Cre recombinase transgenic mice. Genesis 44, 262–267 (2006)
Jeong, J., Mao, J., Tenzen, T., Kottmann, A. H. & McMahon, A. P. Hedgehog signaling in the neural crest cells regulates the patterning and growth of facial primordia. Genes Dev. 18, 937–951 (2004)
Manning, T. C. et al. Antigen recognition and allogeneic tumor rejection in CD8+ TCR transgenic/RAG−/− mice. J. Immunol. 159, 4665–4675 (1997)
Erdag, G. et al. Immunotype and immunohistologic characteristics of tumor-infiltrating immune cells are associated with clinical outcome in metastatic melanoma. Cancer Res. 72, 1070–1080 (2012)
Schmittgen, T. D. & Livak, K. J. Analyzing real-time PCR data by the comparative CT method. Nature Protocols 3, 1101–1108 (2008)
Spranger, S. et al. Generation of Th1-polarizing dendritic cells using the TLR7/8 agonist CL075. J. Immunol. 185, 738–747 (2010)
Acknowledgements
The authors would like to thank A. Sailer and J. Turner for their assistance on mouse tissue immunofluorescent staining, M. Leung and Y. Zha for technical support, and the Special Services Animal Resources Center for assistance with mouse husbandry. We also acknowledge the Fitch Monoclonal Antibody Facility, the Human Tissue Research Core and the Integrated Microscopy core of The University of Chicago Comprehensive Cancer Center. We would like to thank A. O. Emmanuel and F. Gounari for assistance with the ChIP assay as well as for conditional β-catenin knock-in mice; C. Slingluff, D. Deacon, J. Schaefer, G. Erdag and the University of Virginia Biorepository and Tissue Research Facility for melanoma biopsy specimens, and P. Savage for critical comments. Funding for this study was provided by a Team Science Award from the Melanoma Research Alliance and a Translational Research Grant from the Cancer Research Institute. S.S. was supported by the German Research Foundation and is currently a fellow of the Cancer Research Institute.
Author information
Authors and Affiliations
Contributions
S.S. contributed to the overall project design, planned and performed experiments, and performed data analysis. R.B. performed analysis of the TCGA data set. T.F.G. designed the overall project. S.S. and T.F.G. wrote the manuscript.
Corresponding author
Extended data figures and tables
Extended Data Figure 1 Correlation between active β-catenin and CD8 T-cell infiltrate in human patients.
a, A continuous numerical score was generated using six β-catenin target genes (CTNNB1 score). Using this score, patients from the TCGA data set were grouped in high or low CTNNB1 score (centred on the average score) (low, 91 patients; high, 108 patients). Subsequent correlation analysis was performed using a Fisher’s exact test. b, Representative examples for CD8 and β-catenin staining in human needle biopsies used for analysis shown in Fig. 1d.
Extended Data Figure 2 Tumour growth of genetically engineered mice.
a, Overall survival of all three models: BrafV600E/Pten−/− with 100% lethality and mean time to death of 31 days (n = 14), BrafV600E/CAT-STA with 85% lethality and mean time to tumour event of 93 days (n = 8), and BrafV600E/Pten−/−/CAT-STA with 100% lethality and mean time to tumour event of 36 days (n = 14). b, Tumour outgrowth of BrafV600E/Pten−/− (red) and BrafV600E/Pten−/−/CAT-STA (blue) tumours shown as mm3 at days after tamoxifen application (n = 10). c, Representative macroscopic pictures for tumour growth over time when tamoxifen was applied on the lower back of the mouse (see illustration). d, Gene array analysis of tumours isolated from GEMs (n = 4, Mann–Whitney U test). e, Histology slides showing representative examples for haematoxylin and eosin stain in all three mouse models (left, ×20, scale bars indicate 100 μm; right, ×100, scale bars indicate 20 μm). *P ≤ 0.05; NS, not significant.
Extended Data Figure 3 T-cell infiltration of genetically engineered mice.
a, Representative images of immmunofluorescent staining against CD3 (red, left panel) and TRP1 (green, right panel) in all three tumour tissues (scale bar, 100 μm; ×4, ×10, ×20 with ×4 differential interference contrast (DIC) on top; nuclei Hoechst ×20 CD3 stain as shown in Fig. 1). b, Representative immmunofluorescent staining against CD3 (red, left panel) and TRP1 (green, right panel) in a highly pigmented area of BrafV600E/Pten−/− tumour tissues (scale bar, 100 μm; ×10, ×20 with ×10 DIC left) excluding that the lack of T cells is associated with increased pigmentation (nuclei Hoechst). c, Numbers of CD3+ T cells were counted within 13 different fields (0.5 mm × 1 mm) from two tumour samples. Mean of 12 T cells or 3.2 T cells per 0.5 mm2 in BrafV600E/CAT-STA or BrafV600E/Pten−/−/CAT-STA tumours, respectively, versus 100 T cells per 0.5 mm2 in BrafV600E/Pten−/− tumours. Data are given as mean with minimum and maximum, as well as individual values. Statistical analysis was performed using Mann–Whitney U test. ****P ≤ 0.0001.
Extended Data Figure 4 Characterization of the T-cell infiltrate in BrafV600E/Pten−/−/CAT-STA mice.
a, Distribution of T-cell subsets in BrafV600E/Pten−/− and BrafV600E/Pten−/−/CAT-STA tumours (n = 6). b, c, Representative flow cytometry plots to discriminate αβ-TCR T cells and γδ-TCR T cells (b), naive (CD62L+CD44−) and effector (CD62L−CD44+) T cells (pre-gated on CD3+CD8+ T cells), and one representative example of CD44/CD45RA staining (c). Quantification of naive (CD62L+CD44−CD45RA+), effector (CD62L−CD44+CD45RA−) and memory (CD62L+CD44+CD45RA−) T cells is indicated on the right (n = 6). d, Representative flow cytometry plots of FoxP3+ T regulatory cells (n = 6). e, Quantification and comparison of PD-1/Lag3 double-positive T cells in BrafV600E/Pten−/− and BrafV600E/Pten−/−/CAT-STA tumours (n = 12). f, Representative flow cytometry of PD-1- and Lag3-positive T cells (pre-gated on CD3+CD8+ T cells) in BrafV600E/Pten−/− tumours. g, Il2 transcripts present in sorted CD3+ T cells from BrafV600E/Pten−/− tumours and spleen (n = 10). h, Ifng transcripts present in sorted CD3+ T cells from BrafV600E/Pten−/− and BrafV600E/Pten−/−/CAT-STA mice (n = 10). i, Expression level of PD-L1 in whole tumour tissue from both mouse models assessed by qRT–PCR (n = 8). j, Flow cytometric analysis of PD-L1 expression of non-haematopoietic tumour cells (CD45−), CD45+CD11c+ dendritic cells (DC) and CD45+CD3+ T cells. Shown is a representative example as histogram (grey isotype, red BrafV600E/Pten−/−; blue, BrafV600E/Pten−/−/CAT-STA) with mean fluorescent intensity of n = 3 given each histogram (red, BrafV600E/Pten−/−; blue, BrafV600E/Pten−/−/CAT-STA). k, Percentage of Gr1+ cells within the CD11b+ fraction of the tumour immune cell infiltrate (n = 8; absolute numbers BrafV600E/Pten−/−: 1,047 ± 418 cells per gram tumour to BrafV600E/Pten−/−/CAT-STA: 739 ± 185 cells per gram tumour; P = 0.7429). All data are mean ± s.e.m., Mann–Whitney U test.*P ≤ 0.05, **P ≤ 0.01, ****P ≤ 0.0001; NS, not significant.
Extended Data Figure 5 Injection of Flt3 ligand-derived dendritic cells into tumours of BrafV600E/Pten−/−/CAT-STA mice is sufficient to overcome the lack of CD103+ dermal dendritic cells.
a, Expression level of Ifnb in CD45+CD11c+ sorted dendritic cells from tumours from BrafV600E/Pten−/− (open bars) and BrafV600E/Pten−/−/CAT-STA (filled bars) mice. FC, fold change. b, Expression level of Batf3, Irf8 and Itgae in sorted dendritic cells. Fold change is indicated in each graph (n = 8). c, Mean (± s.e.m.) tumour weight of BrafV600E/Pten−/−/CAT-STA assessed at the endpoint of the experiment depicted in Fig. 3e, after intra-tumoural injection of dendritic cells. d, Per cent of GFP+CD11c+ dendritic cells (DC) present at the tumour site after injections of Flt3 ligand-derived dendritic cells from actin–GFP mice. Depicted are the percentages detected in the tumour of both genotypes injected with either wild-type or actin–GFP dendritic cells as well as in the TdLNs for the actin–GFP injected mice (n = 4). All data are mean ± s.e.m., Mann–Whitney U test. *P ≤ 0.05.
Extended Data Figure 6 Chemokine expression patterns indicate that CCL4 expression from tumour cells is directly inhibited by active β-catenin-signalling.
a, Expression of Ccl4 mRNA in established tumour cell lines BP and BPC (8 independent experiments). b, Amount of secreted CCL4 in 48 h conditioned BP and BPC tumour cell supernatants, assessed by ELISA (4 independent experiments). c, d, Control qRT–PCR for the experiment shown in Fig. 4e with Ifnb expression (c) and Ifng expression (d) (n = 6). ND, not detected. All data are mean ± s.e.m., Mann–Whitney U test. *P ≤ 0.05.
Extended Data Figure 7 Active β-catenin signalling blocks CCL4 production in human melanoma cell lines.
a, Western blot on mel537 and mel888 showing stabilized β-catenin expression. b, Expression level of human ATF3 and human CCL4 in mel537 and mel888 (three independent experiments, duplicates per experiment). c, Expression level of β-catenin target genes in mel537 and mel888. d, ATF3-specific ChIP assay in mel537 and mel888 cell lines for the CCL4 gene locus (two independent experiments, duplicates per experiment). e, CCL4 secretion (left) and ATF3 transcription levels (right) after siRNA-mediated knockdown of CTNNB1 and ATF3 in mel537 and mel888 assessed by ELISA or qRT–PCR, respectively (two independent experiments, duplicates per experiment). All data are mean ± s.e.m., Mann–Whitney U test. *P ≤ 0.05.
Extended Data Figure 8 β-Catenin target gene expression correlates inversely with markers for human BATF3-lineage dendritic cells and T cells.
Pearson correlation of CTNNB1 score with CD8Α (R2 = 0.214), THBD (R2 = 0.109) and IRF8 (R2 = 0.2374) (red indicates T-cell-signature high, blue indicates T-cell-signature low).
Extended Data Figure 9 Graphical summary.
Left, tumour without active β-catenin signalling in which ATF3 transcription is not induced and thus CCL4 (red circles) is transcribed and secreted. Downstream CD103+ dendritic cells (DC) (blue) are attracted and subsequent activation of CD8+ T cells (green) is enabled. Right, tumour with active β-catenin signalling (green), which leads to induction of ATF3 transcription (red), which in turn leads, among others effects, to suppression of CCL4 transcription. This leads to an active escape from the anti-tumour immune response since dendritic cell recruitment is insufficient.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-5 comprising:1 (a) Expression of T cell genes in segregated groups (b) Gene clusters after supervised hierarchical clustering (c) List of differentially expressed genes; 2 - Pearson correlation of β-catenin target genes and CD8a transcripts; 3 - Mutation analysis summary with (a) T cell signature low patients and (b) T cell signature high patients (c) Table summarizing potential pathway activators in patients with an active β-catenin signature; 4 - Mouse gene array data of differentially expressed genes (a) and a summary table focusing on chemokine expression (b); 5 - Detailed primer and antibody information (a) Genotyping primers (b) antibodies (c) qPCR primer/ probes (d) ChIP assay primer and (e) siRNA oligos. (PDF 3428 kb)
Source data
Rights and permissions
About this article
Cite this article
Spranger, S., Bao, R. & Gajewski, T. Melanoma-intrinsic β-catenin signalling prevents anti-tumour immunity. Nature 523, 231–235 (2015). https://doi.org/10.1038/nature14404
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14404
- Springer Nature Limited
This article is cited by
-
Targeting focal adhesion kinase boosts immune response in KRAS/LKB1 co-mutated lung adenocarcinoma via remodeling the tumor microenvironment
Experimental Hematology & Oncology (2024)
-
How chemokines organize the tumour microenvironment
Nature Reviews Cancer (2024)
-
Immune escape of head and neck cancer mediated by the impaired MHC-I antigen presentation pathway
Oncogene (2024)
-
PD-L1-positive circulating endothelial progenitor cells associated with immune response to PD-1 blockade in patients with head and neck squamous cell carcinoma
Cancer Immunology, Immunotherapy (2024)
-
Investigating the effects of PTEN mutations on cGAS-STING pathway in glioblastoma tumours
Journal of Neuro-Oncology (2024)