Abstract
Chimeric antigen receptor (CAR) T-cell immunotherapies have shown unprecedented success in treating leukemia but limited clinical efficacy in solid tumors. Here, we generated 1928zT2 and m28zT2, targeting CD19 and mesothelin, respectively, by introducing the Toll/interleukin-1 receptor domain of Toll-like receptor 2 (TLR2) to 1928z and m28z. T cells expressing 1928zT2 or m28zT2 showed improved expansion, persistency and effector function against CD19+ leukemia or mesothelin+ solid tumors respectively in vitro and in vivo. In a patient with relapsed B-cell acute lymphoblastic leukemia, a single dose of 5 × 104/kg 1928zT2 T cells resulted in robust expansion and leukemia eradication and led to complete remission. Hence, our results demonstrate that TLR2 signaling can contribute to the efficacy of CAR T cells. Further clinical trials are warranted to establish the safety and efficacy of this approach.
Similar content being viewed by others
Introduction
The success of the adoptive transfer of CD19-specific chimeric antigen receptor (CAR) T cells represents a major step in cancer immunotherapy. Both CD3ζ and costimulatory domains1 are needed to generate effective CARs that provide the signals necessary for full activation, expansion and survival of CAR T cells. The incorporation of costimulatory molecules CD28,2 4-1BB,3, 4 OX-40,5 CD276 and ICOS7 significantly improve the expansion, cytokine production, in vivo persistence and antitumor efficacy of CAR T cells. Moreover, the efficacy of CAR T cells for solid tumors is substantially poorer than that for leukemia.8, 9 Given the few costimulatory molecules that have been used in the design of CARs, novel intracellular signals are needed to improve their efficacy so as to effectively treat not only leukemia but also solid tumors.
The intracellular signaling domains of CARs mainly derive from the costimulatory receptors expressed on T cells that are critical for their adaptive immune responses. Nevertheless, the innate immune receptor Toll-like receptor 2 (TLR2) is highly expressed on activated and memory T cells, and the TLR2 signaling increases T-cell expansion and cytokine production,10, 11, 12, 13 lowers the activation threshold for costimulatory signals delivered by antigen-presenting cells, facilitates the generation of memory CD8 T cells,14, 15, 16 and directly triggers Th1 effector functions.17 In addition, TLR2-mediated signaling can abolish the suppressive capacities of regulatory T cells.18 Furthermore, T-cell hyporesponsiveness with impaired inerferon (IFN)-γ and interkeukin (IL)-2 production can partially be accounted for by diminished expression of TLR2 on T cells.19 Strikingly, TLR2 ligands induce the regression of established leukemia and solid tumors by compromising the suppressive function of Foxp3+ regulatory T cells and enhancing the cytotoxicity of tumor-specific T cells.20, 21 TLR2 can form heterodimers with TLR1 or TLR6 to initiate a proinflammatory response.22, 23 However, TLR2 may also form weak homodimers, which allow it to directly bind to MyD88.24 Therefore, we proposed that TLR2 signaling could potentiate the antitumor activity of CAR T cells.
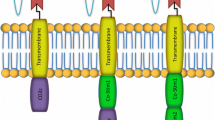
To test our hypothesis, we generated CARs targeting CD19 (1928z) and mesothelin (m28z) by linking scFv with CD28 and CD3ζ signaling domains and then generated 1928zT2 or m28zT2 by adding the Toll/interleukin-1 receptor domain of TLR2 to the 3’ end of CD3ζ. We compared the functions of CAR T cells with or without the TLR2 signaling domain and found that the addition of a TLR2 domain led to enhanced antitumor responses for both leukemia and solid tumors. To test the efficacy and safety of TLR2 construct in humans, we conducted a pilot trial in a patient with relapsed B-cell acute lymphoblastic leukemia (B-ALL) with 1928zT2 T cells and found that a single dose (5 × 104/kg) of 1928zT2 T cells led to complete remission. Our data revealed that the innate immune receptor TLR2, in addition to canonical costimulatory receptors, can improve the antitumor efficacy of CARs for both leukemia and solid tumors.
Materials and methods
The details are provided in Supplementary material.
Results
TLR2 enhanced the effector functions of CD19-specific CAR T cells in vitro
The CD19-specific second-generation CARs have generated exciting results in clinical trials.25, 26, 27, 28, 29, 30 We used 1928z, a CAR-linking FMC63-scFv, CD28 transmembrane and endodomain, and the CD3ζ signaling domain as benchmarks to evaluate TLR2 function in CARs. The Toll/interleukin-1 receptor domain (amino acid 639–784) of TLR2 was appended to the CD3ζ signaling domain. Lentiviral vectors that contained GFP (negative control), 1928z and 1928zT2 (Supplementary Figure 1a) were transfected into T cells to generate GFP T, 1928z T and 1928zT2 T cells, respectively. The transduction efficiencies were measured by the percentages of GFP+ cells (Supplementary Figure 1b). To confirm the role of GFP in representing CAR expression, we detected the coexpression of GFP and anti-CD19 CAR (Supplementary Figure 1c). K562, K562-CD19, NALM6 and REH cells were tagged with both GFP and luciferase (GL). K562-CD19-GL, NALM6-GL, and REH-GL cells, but not K562-GL cells, were specifically killed by 1928z T and 1928zT2 T cells, and notably, the killing percentages of 1928zT2 T cells were higher than those of 1928z T cells at low effector to target (E/T) ratios (Figure 1a). Expression of GZMB, PRF1 and IFNG were higher in 1928zT2 T cells compared with 1928z T cells upon stimulation by NALM6 cells (Supplementary Figure 2). Consistently, 1928zT2 T cells secreted significantly higher amounts of IL-2, IFN-γ and GM-CSF (granulocyte-macrophage colony-stimulating factor), but a lower level of tumor necrosis factor-alpha (TNF-α) (Figure 1b). Interestingly, suppression of TNF-α secretion from macrophages by TLR2 agonist has been reported.31 In addition, the capacities of 1928zT2 T cells to expand upon serial stimulations and to continuously eliminate target cells were higher than 1928z T cells (Figure 1c). Moreover, the killing capacity of 1928zT2 T cells was enhanced compared with 1928BBz T cells (Supplementary Figure 3). Taken together, the incorporation of the TLR2 signaling domain into 1928z led to enhanced antitumor effector functions in vitro.
1928zT2 T cells showed enhanced effector functions compared with 1928z T cells in vitro. (a) Killing percentages of K562-GL, K562-CD19-GL, NALM6-GL and REH-GL cells by GFP T, 1928z T and 1928zT2 T cells after 18 h coculture in vitro. E:T ratios mean the ratios of absolute number of CAR T cells to target cells. The GFP percentages of CAR T cells were equalized by non-transduced T cells from the same donor. Results are representative of at least three independent experiments with T cells from different healthy donors. (b) IL-2, IFN-γ, GM-CSF and TNFα concentration in the supernatant of 24 h coculture of the indicated T cells with K562-GL or K562-CD19-GL cells (E:T=1, 2 × 105 cells each). (c) Enhanced expansion and serial killing potency of 1928zT2 T cells compared with 1928z T cells. 1928z T and 1928zT2 T cells were serially co-cultured with NALM6 cells at 1:4 E/T ratio, the numbers of 1928z T cells and 1928zT2 T cells were counted (left), and percentages of CD19+ NALM6 cells (right) were detected by flow cytometry, every 24 h for three times. Error bars denote s.e.m and results were compared through two-sided unpaired t-test. *P<0.05, **P<0.01, ***P<0.001.
1928zT2 T cells exhibited enhanced antitumor efficacy in vivo
Next, the killing capacities of 1928z T and 1928zT2 T cells were compared in cell line-derived xenografts. Immunodeficient NSI mice32 were intravenously injected with 1 × 106 NALM6-GL cells followed by two infusions of 5 × 106 GFP T, 1928z T or 1928zT2 T cells. Bioluminescence imaging (BLI) showed that a reduced tumor burden was present in mice infused with 1928zT2 T cells compared with 1928z T cells (Figure 2a). Consistently, the percentages of NALM6-GL cells in the peripheral blood mononuclear cells (PBMC) were significantly lower in mice treated with 1928zT2 T cells compared with 1928z T cells (Figure 2b). In addition, the mice treated with 1928zT2 T cells exhibited delayed weight loss (Supplementary Figure S4a). Furthermore, mice treated with 1928zT2 T cells showed enhanced secretion of IL-2, IFN-γ and GM-CSF, but not TNF-α, in serum (Supplementary Figure 4b). To confirm the improved efficacy of the TLR2 construct in terms of survival, 2 × 105 NALM6 cells, and 2 days later, 2 × 106 GFP T, 1928z T, 1928zT2 T or no T cells, were intravenously injected into each NSI mice (n=10). 1928zT2 T cells improved the survival of NALM6 burdened mice better than 1928z T cells (Figure 2c). The percentages of NALM6 cells and T cells in BM when mice died (Supplementary Figure 4c) indicated that the major cause of death was high NALM6 burden for mice treated with no T, GFP T and 1928z T cells, but was graft-versus-host disease for mice treated with 1928zT2 T cells as severe symptoms such as loss of hair and weight were observed. Thus, 1928zT2 T cells were more potent than 1928z T cells at killing NALM6-GL cells in vivo.
1928zT2 T cells showed superior anti-leukemia efficacy in vivo. (a) Bioluminescence imaging of NALM6-GL-burdened mice treated with GFP T, 1928z T or 1928zT2 T cells. In total, 1 × 106 NALM6-GL cells were injected intravenously into NSI mice, and on day 2 and 7, 5 × 106 GFP T, 1928z T or 1928zT2 T cells were injected intravenously into each NALM6-GL-NSI mice (n=5). On day 8, 14 and 19, bioluminescence imaging was conducted. (b) The percentages of CD19+ (NALM6-GL) cells in the PBMC of mice on day 14. (c) Survival analysis of mice treated with GFP T, 1928z T or 1928zT2 T cells (n=10). A total of 2 × 105 NALM6 cells were intravenously injected into NSI mice, and 2 days later, 2 × 106 T cells were injected (GFP% normalized to 10%), and survival of mice was observed. Error bars denote s.e.m and groups were compared through two-sided unpaired t-test. *P<0.05, **P<0.01, ***P<0.001.
Furthermore, the efficacies of 1928z T and 1928zT2 T cells were compared in patient-derived xenograft models. B-ALL cells were eliminated by 1928zT2 T cells, but not by 1928z T cells, and the percentages of total human T cells and CAR T cells were higher in 1928zT2 group compared with 1928z group (Supplementary Figure 5a). Due to the less aggressiveness of primary B-ALL cells, all the B-ALL burdened mice including the GFP T group survived until when the mice were culled for analysis on day 78, and splenomegaly was found only in the GFP and 1928z groups, but not in the 1928zT2 group (Supplementary Figure 5b). Collectively, these results demonstrated that 1928zT2 T cells had significantly higher anti-leukemia efficacy than 1928z T cells in vivo.
TLR2 costimulation upregulated genes associated with cell adhesion, synaptic transmission, T-cell migration and mTOR signaling
To decipher the molecular mechanisms of TLR2 costimulation, we performed RNA sequencing to compare the transcriptomes of 1928z T and 1928zT2 T cells with or without stimulation by NALM6 cells. Approximately 11 013 of the 33 382 RefSeq genes were detectably expressed in all four populations. We identified the genes specifically upregulated in stimulated 1928zT2 T cells and found that most of these genes were related to cell adhesion and synaptic transmission (Figure 3a). Quantitative real-time PCR confirmed the upregulation of genes associated with cell adhesion and synaptic transmission, along with two genes IRF3 and IRF7 downstream of the TLRs pathway (Figure 3b). TLR2 signaling has been reported to enhance mTOR signaling and expression of T-bet.33 Here, we also detected elevated transcription of T-bet, mTOR, 4E-BP1 and P70S6K (Figure 3b). MMPs (matrix metallopeptidases) have been shown to play critical roles in T-cell activation34 and migration into tissues.35 TLR2 incorporation also induced the upregulation of MMP2 and MMP24 expression in 1928zT2 T cells (Figures 3a and b). Therefore, the migration capacities of GFP T, 1928z T and 1928zT2 T cells were compared and results indicated that 1928zT2 T cells transmigrated the matrigel more efficiently than 1928z T cells (Figure 3c). These results suggest that TLR2 incorporation enhances the antitumor efficacy of CAR T cells partly through improved adhesion, synaptic transmission and migration capacities, and enhanced T-bet expression through mTOR pathway.
TLR2 costimulation upregulated genes associated with cell adhesion, synaptic transmission, infiltration and mTOR pathway. (a) Heatmap of RNA sequencing results showing genes specifically upregulated in 1928zT2 T cells after stimulation (+sti) (left). These genes were associated with cell adhesion, synapse components and transmission and cell invasion of T cells (right). (b) Relative expression of genes associated with cell adhesion (CD24, CDH2, TYRO3, CXADR), T-cell infiltration (MMP2), synaptic transmission (CADPS2), TLRs pathway (IRF3 and IRF7) and mTOR pathway (T-bet, mTOR, 4E-BP1 and p70S6K). (c) Transwell cell migration assay indicated an improved capacity of 1928zT2 T cells to degrade and transmigrate Matrigel. T-cell culture medium was used as a chemoattractant in the lower chamber. T cells were cultured in the insert with or without coated Matrigel for 24 h, and the cells that transmigrated to the lower chamber were counted. The percentage of invasion was calculated as follows: (mean of cells migrating through the Matrigel chamber membrane/mean of cells migrating through the control insert membrane) × 100. Error bars denote s.e.m and groups were compared through two-sided unpaired t-test. *P<0.05, **P<0.01, ***P<0.001.
TLR2 costimulation enhanced antitumor efficacy of mesothelin-specific CAR T cells
Mesothelin is highly expressed in various solid tumors including lung adenocarcinoma cells,36 and clinical trials of mesothelin-targeting CAR T cells are ongoing (such as NCT02414269 and NCT02930993). Thus, to assess the effect of TLR2 costimulation on CAR T cells targeting solid tumors, we incorporated TLR2 into a CAR specific to mesothelin and generated GFP T, m28z T and m28zT2 T cells through lentiviral transfection (Supplementary Figure 6a and 6b). m28zT2 T cells showed enhanced killing capacity against mesothelin+ A549-GL cells compared with m28z T cells (Figure 4a). m28zT2 T cells secreted more IL-2, IFN-γ and GM-CSF, but reduced TNF-α compared with m28z T cells upon stimulation by A549 cells (Figure 4b). The expansion of m28zT2 T cells during repetitive stimulation was improved compared with m28z T cells, and the potency of m28zT2 T cells at killing A549 cells were significantly higher than m28z T cells (Supplementary Figure 7a). Meanwhile the exhaustion markers on T cells after repetitive coculture were compared, and the expression of TIM-3, but not PD-1 or LAG-3, was lower on m28zT2 T cells relative to m28z T cells (Supplementary Figure 7b). These results indicated that TLR2 incorporation in mesothelin-specific CAR also improved its effector functions in vitro.
m28zT2 T cells showed enhanced antitumor efficacy compared with m28z T cells. (a) Killing percentages of mesothelin+ A549-GL and mesothelin- H23-GL cells after 18 h coculture with GFP T, m28z T or m28zT2 T cells at the indicated ratios. (b) IL-2, IFN-γ, GM-CSF and TNF-α concentration in the supernatant of 24 h coculture of GFP T, m28z T or m28zT2 T cells with A549-GL cells. E: T=1:1, 2 × 105 cells each. (c) Weight of subcutaneous A549-GL tumors from NSI mice treated with no T cells, GFP T, m28z T or m28zT2 T cells. On day 0, 2 × 105 A549-GL cells were injected subcutaneously into NSI mice, and on day 7 and 9, no T, 5 × 106 GFP T, m28z T or m28zT2 T cells were injected through the tail vein into each mouse (n=6). On day 43, mice were killed and tumors were analyzed. Error bars denote s.e.m and groups were compared through two-sided unpaired t-test. *P<0.05, **P<0.01, ***P<0.001.
We further compared the antitumor efficacy of m28zT2 T, m28z T and GFP T cells in subcutaneous A549-GL xenografted mice. Tumors from m28zT2 group were smaller than those from the blank, GFP and m28z groups (Figure 4c). Moreover, mice treated with m28zT2 T cells produced more cytokines IL-2, IFN-γ and GM-CSF, but not TNF-α, than those treated with m28z T cells (Supplementary Figure 8). As metastasis is a major hurdle in the treatment of solid tumors, we compared the efficacies of m28z T and m28zT2 T cells against metastasized tumors through the blood in intravenous A549-GL xenografted mice. The bioluminescence imaging results showed that m28zT2 T cells efficiently inhibited the growth of A549-GL cells in NSI mice; however, surprisingly, the mice grew a greater burden of tumors when treated with m28z T cells than the mice that were injected with GFP T cells (Supplementary Figure 9a). The histologic analysis confirmed that the tumor cells in the uterus of the mice from the m28z group were HLA+ A549-GL cells (Supplementary Figure 9b). These results indicated that TLR2 incorporation could enhance the efficacy of mesothelin-specific CAR T cells against solid tumors.
Clinical trial of 1928zT2 T cells in a patient with relapsed and refractory B-ALL
We conducted a pilot clinical trial in a patient with relapsed and refractory B-ALL (NCT02822326). 1928zT2 T cells were produced and well characterized phenotypically and functionally (Supplementary Figures 10a–f). The patient received two days of treatment with Fludarabine (30 mg/m2/day) and cyclophosphamide (300 mg/m2/day) for lymphocyte depletion, followed by one dose of 1928zT2 T cells at 5 × 104 cells/kg (Supplementary Figure 11a). After infusion of 1928zT2 T cells, the patient experienced side effects including fever, coughing, skin rashes and hypotension, but not neurological defects. During this trial, the symptoms of the patients did not necessitate ICU stay, vasopressor support or any steroids treatment. A marked elevation of serum IL-6 was detected on day 2 and tocilizumab, an anti-IL-6R monoclonal antibody,7 was administered at 4 mg/kg, which resulted in rapid defervescence and complete reversal of symptoms over a period of 4 days; however, the patient’s concentration of C-reactive protein fluctuated in another pattern, and the highest peak on day 45 was due to urological infection (Supplementary Figure 11b). In addition, we monitored the dynamic changes of other cytokines (Supplementary Figure 11c). B-ALL cells comprised ~15% of the PBMC and >70% of the BM before treatment (Figure 5a), but decreased to an undetectable level on day 22 (Figure 5b). Complete remission (CR) was formally diagnosed on day 51 (Figure 5c). Percentage changes of 1928zT2 T cells and CD19+ B-ALL cells in PBMC (Figure 5d) and BM (Figure 5e) were depicted. On day 22, the percentages of CD4+CD8−, CD62L+CD25− and CD45RO+CD45RA− populations in 1928zT2 T cells were higher than those in wild-type T cells in both PBMC and BM (Supplementary Figure 12a). Interestingly, the elevation of CD4+ 1928zT2 T-cell percentage was concomitant with the increase of the percentage of 1928zT2 T cells in both PBMC and BM (Supplementary Figures 12b and c). In addition, the percentages of CD25+ 1928zT2 T cells were negatively correlated with the percentages of 1928zT2 T cells in PBMC (Supplementary Figure 12d), whereas CD62L expression on 1928zT2 T cells in PBMC remained high until the B-ALL cells were eradicated (Supplementary Figure 12e). 1928zT2 T cells were detected in cerebrospinal fluid, indicating the potential role of 1928zT2 T cells in eradicating leukemia in the central nervous system (Supplementary Figure 12f). The clinical results here indicated that small dosages of 1928zT2 T cells could kill leukemic cells with potent efficacy.
1928zT2 T cells showed potent efficacy in a patient with relapsed B-ALL. (a) FACS analysis of B-ALL cells in PBMC and BM from the patient on the day before infusion of 1928zT2 T cells. (b) B-ALL cells were eradicated from PBMC and BM of the patient on day 19 and 22, respectively. (c) Wright-Giemsa staining of BM biopsies of the patient on day 18 before infusion (left) and on day 51 post infusion (right) of 1928zT2 T cells. Scale bar=10 μm. (d) Dynamic changes of the percentages of CD19+ cells and 1928zT2 T cells in PBMC of the patient. (e) Dynamic changes of the percentages of CD19+ cells and 1928zT2 T cells in BM of the patient.
Discussion
Although CD19-specific CAR T-cell immunotherapy has achieved promising results in clinical trials, efficacy of other CAR T cells needs improvement, especially those for solid tumors. The development of CARs from the first generation (with only the CD3ζ signaling domain) to the second generation (with CD3ζ and a costimulatory domain) has revealed a critical role of costimulatory molecules in CARs. Incorporation of novel costimulatory molecules into CARs should be a feasible strategy to improve their efficacy.
Interestingly, the innate immune receptor TLR2 is expressed on activated T cells as a costimulatory receptor.10 Innate immunity serves as an immediate defense mechanism against pathogenic organisms. Adaptive immunity plays a delayed but more delicate role in controlling diseases and forming memories. The two types of immunity are not independent of each other and the interaction between them is critical to the outcome of overall immune response. CARs with combinatorial moieties of both adaptive and innate immunity may exert dual functions of mobilizing innate responses as well as promoting T-cell expansion, antigen-specific reaction, T-cell contraction and memory formation. We demonstrated that 1928zT2 and m28zT2 T cells were more potent at killing CD19+ leukemia cells and mesothelin+ lung cancer cells in vitro. TLR2 signaling in CD8 T cells can augment T-bet protein levels and IFN- γ, perforin and granzyme B expression through the mTOR pathway, and contribute to the efficient clearance of intracellular pathogens,33 reminiscent of the results we provide here. To decipher other mechanisms of TLR2-enhanced antitumor efficacy, we found the genes upregulated by TLR2 costimulation, most of which were associated with cell adhesion, synaptic transmission and migration. Therefore, TLR2 costimulation can serve as an innate immune moiety to enhance the effector functions and promote adhesion and immune synaptic signaling between CAR T cells and target cells.
In our pilot study, only 5 × 104/kg of 1928zT2 T cells expanded to a population that constituted ~40% of lymphocytes in patient PBMC, indicating the remarkable expansion capacity of 1928zT2 T cells. As correlation between infused CAR T-cell dose and incidence and severity of cytokine release syndrome has been suggested,26, 37, 38 it should be beneficial to reduce the CAR T-cell dose. In addition, whereas large numbers of CAR T cells can be generated, a reduction in CAR T-cell dose may reduce the time for producing enough CAR T cells for therapy and manufacturing cost, and provide capacity advantages, which will be important for widespread adoption of CAR T-cell therapies. Nevertheless, other patients in our clinical trial showed less severe side effects and none needed tocilizumab treatment (data unpublished). Therefore, our TLR2 construct should be potent but without increased incidence and severity of cytokine release syndrome. We provided an unprecedentedly detailed dynamic changes of CAR T cells and B-ALL cells in the patient, a pattern which could be reference for other patients to predict CR. However, results of our ongoing clinical trial will be needed to further determine the safety and efficacy of TLR2 costimulation in larger patient cohorts.
CAR T cells for solid tumors face five major challenges—trafficking, tumor recognition and killing, proliferation and persistence, counteract of microenvironment and control of CAR T cells,39 and the former four of which can be tackled by optimization of costimulations in CARs. In our A549-GL metastatic xenograft mouse models (Supplementary Figure 9a), the tumors in the m28z group were larger than that not only in the m28zT2 group but also in the GFP group. Conversely, we demonstrated that m28z T cells were able to kill A549-GL cells in vitro (Figure 4a) and suppress the growth of A549-GL in subcutaneous xenograft models (Figure 4c). The differences of the results from these experiments show the importance of validating the efficacies of cellular immunotherapies in multiple models. The size of tumors is determined by the combination effects of tumor growth and tumor killing in vivo. It is possible that inflammatory factors secreted by m28z T cells or tumor cells, and cell debris during killing stimulated and recruited mouse tumor-supportive cells, such as MDSCs40, 41 that easily access to and accumulate in uterus.42, 43 Then the tumor-supportive cells promoted the expansion of A549-GL cells and overcame the antitumor effects of m28z T cells.44 However, m28zT2 T cells eliminated A549-GL cells efficiently enough so that tumors hardly formed and grew in NSI mice. Of course, further studies were required to test this hypothesis in the future.
To summarize, our data demonstrate that TLR2 incorporation can generate more powerful CARs, which have superior antitumor efficacies in vitro and in vivo and can be used in patients. These findings reveal an important new strategy for CAR design which bridges the conventional costimulation of adaptive immunity with unconventional innate immune signaling.
References
Hombach A, Abken H . Costimulation tunes tumor-specific activation of redirected T cells in adoptive immunotherapy. Cancer Immunol Immunother 2007; 56: 731–737.
Kowolik CM, Topp MS, Gonzalez S, Pfeiffer T, Olivares S, Gonzalez N et al. CD28 costimulation provided through a CD19-specific chimeric antigen receptor enhances in vivo persistence and antitumor efficacy of adoptively transferred T cells. Cancer Res 2006; 66: 10995–11004.
Milone MC, Fish JD, Carpenito C, Carroll RG, Binder GK, Teachey D et al. Chimeric receptors containing CD137 signal transduction domains mediate enhanced survival of T cells and increased antileukemic efficacy in vivo. Mol Ther 2009; 17: 1453–1464.
Long AH, Haso WM, Shern JF, Wanhainen KM, Murgai M, Ingaramo M et al. 4-1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nat Med 2015; 21: 581–590.
Pule MA, Straathof KC, Dotti G, Heslop HE, Rooney CM, Brenner MK . A chimeric T cell antigen receptor that augments cytokine release and supports clonal expansion of primary human T cells. Mol Ther 2005; 12: 933–941.
Song DG, Ye Q, Poussin M, Harms GM, Figini M, Powell DJ Jr . CD27 costimulation augments the survival and antitumor activity of redirected human T cells in vivo. Blood 2012; 119: 696–706.
Shen CJ, Yang YX, Han EQ, Cao N, Wang YF, Wang Y et al. Chimeric antigen receptor containing ICOS signaling domain mediates specific and efficient antitumor effect of T cells against EGFRvIII expressing glioma. J Hematol Oncol 2013; 6: 33.
Zhang BL, Qin DY, Mo ZM, Li Y, Wei W, Wang YS et al. Hurdles of CAR-T cell-based cancer immunotherapy directed against solid tumors. Sci China Life sci 2016; 59: 340–348.
Brown CE, Alizadeh D, Starr R, Weng L, Wagner JR, Naranjo A et al. Regression of glioblastoma after chimeric antigen receptor T-cell therapy. N Engl J Med 2016; 375: 2561–2569.
Komai-Koma M, Jones L, Ogg GS, Xu DM, Liew FY . TLR2 is expressed on activated T cells as a costimulatory receptor. Proc Natl Acad Sci USA 2004; 101: 3029–3034.
Cottalorda A, Mercier BC, Mbitikon-Kobo FM, Arpin C, Teoh DY, McMichael A et al. TLR2 engagement on memory CD8(+) T cells improves their cytokine-mediated proliferation and IFN-gamma secretion in the absence of Ag. Eur J Immunol 2009; 39: 2673–2681.
Sinnott BD, Park B, Boer MC, Lewinsohn DA, Lancioni CL . Direct TLR-2 costimulation unmasks the proinflammatory potential of neonatal CD4+ T cells. J Immunol 2016; 197: 68–77.
Salerno F, Guislain A, Cansever D, Wolkers MC . TLR-mediated innate production of IFN-gamma by CD8+ T cells is independent of glycolysis. J Immunol 2016; 196: 3695–3705.
Cottalorda A, Verschelde C, Marcais A, Tomkowiak M, Musette P, Uematsu S et al. TLR2 engagement on CD8 T cells lowers the threshold for optimal antigen-induced T cell activation. Eur J Immunol 2006; 36: 1684–1693.
Mercier BC, Cottalorda A, Coupet CA, Marvel J, Bonnefoy-Berard N . TLR2 engagement on CD8 T cells enables generation of functional memory cells in response to a suboptimal TCR signal. J Immunol 2009; 182: 1860–1867.
Quigley M, Martinez J, Huang X, Yang Y . A critical role for direct TLR2-MyD88 signaling in CD8 T-cell clonal expansion and memory formation following vaccinia viral infection. Blood 2009; 113: 2256–2264.
Imanishi T, Hara H, Suzuki S, Suzuki N, Akira S, Saito T et al. TLR2 directly triggers Th1 effector functions. J Immunol 2007; 178: 6715–6719.
Oberg HH, Ly TT, Ussat S, Meyer T, Kabelitz D, Wesch D . Differential but direct abolishment of human regulatory T cell suppressive capacity by various TLR2 ligands. J Immunol 2010; 184: 4733–4740.
Babu S, Blauvelt CP, Kumaraswami V, Nutman TB . Cutting edge: diminished T cell TLR expression and function modulates the immune response in human filarial infection. J Immunol 2006; 176: 3885–3889.
Zhang Y, Luo F, Cai Y, Liu N, Wang L, Xu D et al. TLR1/TLR2 agonist induces tumor regression by reciprocal modulation of effector and regulatory T cells. J Immunol 2011; 186: 1963–1969.
Zhang Y, Luo F, Li A, Qian J, Yao Z, Feng X et al. Systemic injection of TLR1/2 agonist improves adoptive antigen-specific T cell therapy in glioma-bearing mice. Clin Immunol 2014; 154: 26–36.
Sandor F, Latz E, Re F, Mandell L, Repik G, Golenbock DT et al. Importance of extra- and intracellular domains of TLR1 and TLR2 in NFkappa B signaling. J Cell Biol 2003; 162: 1099–1110.
O'Neill LA, Bowie AG . The family of five: TIR-domain-containing adaptors in Toll-like receptor signalling. Nat Rev Immunol 2007; 7: 353–364.
Brown V, Brown RA, Ozinsky A, Hesselberth JR, Fields S . Binding specificity of Toll-like receptor cytoplasmic domains. Eur J Immunol 2006; 36: 742–753.
Brentjens RJ, Davila ML, Riviere I, Park J, Wang X, Cowell LG et al. CD19-targeted T cells rapidly induce molecular remissions in adults with chemotherapy-refractory acute lymphoblastic leukemia. Sci Transl Med 2013; 5: 177ra138.
Davila ML, Riviere I, Wang X, Bartido S, Park J, Curran K et al. Efficacy and toxicity management of 19-28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Sci Transl Med 2014; 6: 224ra225.
Grupp SA, Kalos M, Barrett D, Aplenc R, Porter DL, Rheingold SR et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N Engl J Med 2013; 368: 1509–1518.
Kochenderfer JN, Dudley ME, Carpenter RO, Kassim SH, Rose JJ, Telford WG et al. Donor-derived CD19-targeted T cells cause regression of malignancy persisting after allogeneic hematopoietic stem cell transplantation. Blood 2013; 122: 4129–4139.
Maude SL, Frey N, Shaw PA, Aplenc R, Barrett DM, Bunin NJ et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med 2014; 371: 1507–1517.
Porter DL, Levine BL, Kalos M, Bagg A, June CH . Chimeric antigen receptor-modified T cells in chronic lymphoid leukemia. N Engl J Med 2011; 365: 725–733.
Dobrovolskaia MA, Medvedev AE, Thomas KE, Cuesta N, Toshchakov V, Ren T et al. Induction of in vitro reprogramming by Toll-like receptor (TLR)2 and TLR4 agonists in murine macrophages: effects of TLR ‘homotolerance’ versus ‘heterotolerance’ on NF-kappa B signaling pathway components. J Immunol 2003; 170: 508–519.
Ye W, Jiang Z, Li GX, Xiao Y, Lin S, Lai Y et al. Quantitative evaluation of the immunodeficiency of a mouse strain by tumor engraftments. J Hematol Oncol 2015; 8: 59.
Geng D, Zheng L, Srivastava R, Asprodites N, Velasco-Gonzalez C, Davila E . When Toll-like receptor and T-cell receptor signals collide: a mechanism for enhanced CD8 T-cell effector function. Blood 2010; 116: 3494–3504.
Boussif O, Lezoualc'h F, Zanta MA, Mergny MD, Scherman D, Demeneix B et al. A versatile vector for gene and oligonucleotide transfer into cells in culture and in vivo: polyethylenimine. Proc Natl Acad Sci USA 1995; 92: 7297–7301.
Ye W, Jiang Z, Li GX, Xiao Y, Lin S, Lai Y et al. Quantitative evaluation of the immunodeficiency of a mouse strain by tumor engraftments. J Hematol Oncol 2015; 8: 59.
Ho M, Bera TK, Willingham MC, Onda M, Hassan R, FitzGerald D et al. Mesothelin expression in human lung cancer. Clin Cancer Res 2007; 13: 1571–1575.
Lee DW, Kochenderfer JN, Stetler-Stevenson M, Cui YK, Delbrook C, Feldman SA et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet 2015; 385: 517–528.
Park JH, Geyer MB, Brentjens RJ . CD19-targeted CAR T-cell therapeutics for hematologic malignancies: interpreting clinical outcomes to date. Blood 2016; 127: 3312–3320.
Lim WA, June CH . The principles of engineering immune cells to treat cancer. Cell 2017; 168: 724–740.
Sevko A, Umansky V . Myeloid-derived suppressor cells interact with tumors in terms of myelopoiesis, tumorigenesis and immunosuppression: thick as thieves. J Cancer 2013; 4: 3–11.
Ortiz ML, Lu L, Ramachandran I, Gabrilovich DI . Myeloid-derived suppressor cells in the development of lung cancer. Cancer Immunol Res 2014; 2: 50–58.
Ostrand-Rosenberg S, Sinha P, Figley C, Long R, Park D, Carter D et al. Frontline Science: myeloid-derived suppressor cells (MDSCs) facilitate maternal-fetal tolerance in mice. J Leukoc Biol 2017; 101: 1091–1101.
Zhao H, Kalish F, Schulz S, Yang Y, Wong RJ, Stevenson DK . Unique roles of infiltrating myeloid cells in the murine uterus during early to midpregnancy. J Immunol 2015; 194: 3713–3722.
Umansky V, Blattner C, Gebhardt C, Utikal J . The role of myeloid-derived suppressor cells (MDSC) in cancer progression. Vaccines 2016; 4: pii: E36.
Acknowledgements
We sincerely thank Dr Robert Weinkove at Malaghan Institute of Medical Research for his help in revising the manuscript. This study was supported by National Natural Science Foundation of China (81522002), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB19030205), the Natural Science Fund for Distinguished Young Scholars of Guangdong Province (2014A030306028), the Guangdong Provincial Applied Science and Technology Research& Development Program (2016B020237006), the Guangdong Provincial Outstanding Young Scholars Award (2014TQ01R068), the Frontier and key technology innovation special grant from the Department of Science and Technology of Guangdong province, (2015B020227003, 2014B020225005, 2016B030229006), the Guangdong Provincial Research and Commercialization Program (2014B090901044), and the Guangzhou Science Technology and Innovation Commission Project (201504010016). A patent application has been filed for the TLR2 incorporated CAR vector, and PL and YL are listed as authors on this application. The clinical trial is being sponsored by Guangdong Zhaotai Invivo Biomedicine Co. Ltd. and Hunan Zhaotai Yongren Medical Innovation Ltd.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Rights and permissions
About this article
Cite this article
Lai, Y., Weng, J., Wei, X. et al. Toll-like receptor 2 costimulation potentiates the antitumor efficacy of CAR T Cells. Leukemia 32, 801–808 (2018). https://doi.org/10.1038/leu.2017.249
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2017.249
- Springer Nature Limited
This article is cited by
-
Distinct functions of CAR-T cells possessing a dectin-1 intracellular signaling domain
Gene Therapy (2023)
-
Strategies to enhance CAR-T persistence
Biomarker Research (2022)
-
CAR-T cell potency: from structural elements to vector backbone components
Biomarker Research (2022)
-
CXCL9-modified CAR T cells improve immune cell infiltration and antitumor efficacy
Cancer Immunology, Immunotherapy (2022)
-
Human induced-T-to-natural killer cells have potent anti-tumour activities
Biomarker Research (2022)