Abstract
Objective:
Compared with pressure-controlled ventilation (PCV), volume-targeted ventilation is associated with decreased neonatal complications, including the combined outcome of death or bronchopulmonary dysplasia. However, little is known about its effect on neurodevelopmental outcome. We evaluated the hypothesis that as compared with PCV, volume-targeted ventilation reduces the risk of the combined outcome of neurodevelopmental impairment or death in very low birth weight infants.
Study Design:
We studied a cohort of extremely preterm infants managed with either volume guarantee pressure support ventilation (VGPSV; n=135) or PCV (n=135). Infants were evaluated at 18 months adjusted age with a standardized neurological examination and the Bayley Scales of Infant and Toddler Development-third edition. Logistic regression models were used to evaluate the association of ventilation mode and neurodevelopmental outcome.
Result:
Rates of pulmonary interstitial emphysema (odds ratio 0.6; 95% confidence limits: 0.4, 0.8), hypotension (odds ratio: 0.7; 95% confidence limits: 0.5, 0.9) and mortality (odds ratio 0.45; 95% confidence limits: 0.22, 0.9) were lower among infants treated with VGPSV. The infants in the VGPSV group had a significantly shorter duration on mechanical ventilation compared with infants in the PCV group (log-rank test P<0.01). Seventy percent (155/221) of survivors were evaluated at 18 months adjusted age. A trend towards benefit for the combined outcome of death or neurodevelopmental impairment was seen in the VGPSV group but did not reach statistical significance (odds ratio: 0.59; 95% confidence limits: 0.32, 1.08).
Conclusion:
VGPSV was associated with a decreased risk of short-term complications but not long-term developmental impairment in this modest-sized cohort.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
A majority of extremely preterm infants are treated with mechanical ventilation, which is associated with an increased risk of chronic lung disease, neonatal brain damage and neurodevelopmental impairments (NDIs).1, 2, 3 Interventions to decrease the risk of chronic lung disease include innovations in mechanical ventilation. Volume guarantee pressure support ventilation (VGPSV) is a hybrid mode, which delivers a set tidal volume within a set pressure range, using a computer-controlled feedback mechanism.4 At lower lung compliance, higher pressures are generated to deliver the desired tidal volume, and as compliance improves the pressure needed to achieve the set tidal volume is automatically reduced. A consistent tidal volume could be particularly beneficial to patients with rapidly changing lung compliance, such as extremely premature infants with respiratory distress syndrome who receive surfactant.5, 6, 7, 8, 9
Volume-targeted ventilation is associated with a decreased risk of at least three risk factors for NDI, that is, prolonged mechanical ventilation, severe intraventricular hemorrhage, and hypocarbia.10, 11, 12, 13, 14, 15 Published studies of neurodevelopmental outcomes after introduction of this ventilation mode are limited by small sample sizes.16, 17 In this study, we used data from a larger cohort of very low birth weight (VLBW) infants to evaluate the hypothesis that VGPSV is associated with a decreased rate of combined outcome of severe NDI or death.
Methods
Study design
This was a retrospective observational study comparing the neurodevelopmental outcomes at 18 months postmenstrual age of VLBW infants born before (1 July 2007 to 30 June 2008) or after (1 July 2008 to 31 December 2009) a change in the type of mechanical ventilator used in our neonatal intensive care unit (NICU) at Forsyth Medical Center, Winston-Salem, NC USA. The research protocol for this study was approved by the Institutional Review Boards of Wake Forest School of Medicine and Forsyth Medical Center, Winston-Salem, NC, USA.
Mechanical intervention
In the earlier 12-month epoch, pressure-limited ventilation was used, whereas in the later 18-month epoch, volume-targeted ventilation was used as the only mode of conventional ventilation. Both forms of mechanical ventilation were provided with Dräger Babylog 8000 Plus ventilators (Dräger, Lübeck, Germany). A failure of conventional mechanical ventilation in either group was an indication for high-frequency ventilation. Infants born between 1 July 2007 and 30 June 2008 constituted pressure-controlled ventilation (PCV) group, whereas infants born between 1 July 2008 and 31 December 2009 constituted the VGPSV group. The criteria used for initiating mechanical ventilation in the two epochs were similar. In general, mechanical ventilation was initiated for all infants with significant respiratory distress or apnea and was not initiated for infants who had neither respiratory distress nor apnea. The following criteria were used in the delivery room: (1) intubation with administration of surfactant then maintenance on mechanical ventilation for transport and initial care in NICU for the least mature category of infants of 22 0/7 to 24 6/7 weeks gestational age; (2) intubation with administration of surfactant then extubation to nasal continuous airway pressure with positive end expiratory pressure of +5, if FiO2 requirement was <0.4 for the category of infants born between 25 0/7 and 26 6/7 weeks gestation; (3) management on nasal continuous airway pressure with positive end expiratory pressure of +5, if FiO2 requirement was <0.4 for infants with gestational age⩾27 weeks. If FiO2 requirement exceeded 0.4, one dose of surfactant was administered; extubation was subsequently performed in the delivery room if FiO2 requirement dropped below 0.4.
Participants
We studied all VLBW infants with birth weight⩽1250 g who were intubated and treated with mechanical ventilation by 6 h of postnatal age, excluding those with lethal congenital or chromosomal anomalies, terminal conditions or treatment with high-frequency ventilation prior to treatment with conventional ventilation.
Data sources
Maternal and neonatal data were collected from review of medical records. Neonatal data during initial hospitalization included the total duration of mechanical ventilation, air leak syndrome, sepsis, patent ductus arteriosus, grade 3 and 4 intraventricular hemorrhage, periventricular leukomalacia and retinopathy of prematurity.
Data about follow-up outcomes were collected at ~18 months of adjusted age. Using a standardized neurological examination and assessment with the Gross Motor Function Classification System, we identified infants with moderate or severe cerebral palsy.18, 19 We obtained scores for the Cognitive, Motor and Language Scales using the Bayley Scales of Infant and Toddler Development-third edition.20 Examiners were not aware of the type of conventional mechanical ventilator with which infants were treated.
Data definitions
We defined chronic lung disease as use of Supplementary oxygen at 36 weeks postmenstrual age in association with characteristic radiographic changes. Infants were classified as having pulmonary interstitial emphysema, if this condition was mentioned in a radiologist’s interpretation of a chest radiograph. Pneumothorax was defined as a pneumothorax that required chest tube placement. Necrotizing enterocolitis was defined as stage I or higher as per modified Bell’s criteria.21 Sepsis was defined as a positive blood culture treated for at least 7 days with antibiotics.22 Severe intraventricular hemorrhage was defined as either intraventricular hemorrhage with ventricular dilatation or intracerebral (parenchymal) hemorrhage on cranial ultrasound.23 Periventricular leukomalacia was defined as periventricular cystic lesions. Severe retinopathy of prematurity was defined as retinopathy diagnosed by a pediatric ophthalmologist that required laser treatment.24 Bevacizumab injections were not used in our NICU during the study interval. Hypotension was defined as treatment with intravenous vasopressor agents for the purpose of increasing blood pressure.25 Infants were classified as having NDI if they had moderate-to-severe cerebral palsy (cerebral palsy with Gross Motor Function Classification System >1) or a Bayley Scales of Infant and Toddler Development-third edition Cognitive or Motor Scale <70.
Statistical analysis
Patient characteristics and outcomes were summarized as proportions and means, s.d.s and ranges. Unadjusted comparisons were performed using X2-tests or Fisher’s exact test for categorical outcomes, t-tests for continuous variables and the log-rank test for event times. We used multivariate logistic regression models to estimate odds ratios and 95% confidence limits for the association between ventilator group and outcomes, adjusted for confounders. The data analysis plan included an analysis of the association between ventilator group and the outcome of interest in the smallest category of infants with birth weight ⩽1000 g, infants at the highest risk for NDI or death. For inclusion in multivariate models, we selected variables that were associated with both the ventilator group and the outcome at a significance level of 0.2.
Results
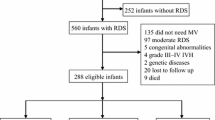
Between 1 July 2007 and 31 December 2009, 420 infants who were ⩽1250 g at birth were admitted to the intensive care unit at Forsyth Medical Center (Figure 1). Of these, 270 VLBW infants met eligibility criteria and were enrolled in our study. Among the infants enrolled, 135 infants were treated with PCV and 135 were treated with VGPSV. Seventy-eight infants in the PCV group and 77 in the VGPSV group were seen at 18 months postmenstrual age for neurodevelopmental assessment. There were no significant differences between the two study groups with regard to any of the baseline maternal demographics and characteristics or infant characteristics (Table 1).
Data for short-term morbidities are presented in Table 2. Infants treated with volume-targeted ventilation had lower rates of hypotension, pulmonary interstitial emphysema and mortality prior to discharge from the NICU. The lower rates of pneumothorax in the volume target group did not reach statistical significance. Infants in the VGPSV group had a shorter time on mechanical ventilation than infants in the PCV group (median of 26.2 versus 13.7 days in the PCV as compared with VGPSV group, respectively; log rank P<0.01). Infants treated with VGPSV had significantly higher rates of stage 1 or higher necrotizing enterocolitis (NEC) per modified Bell’s criteria as compared with infants treated with PCV.
The demographic and clinical characteristics of mothers of infants who survived but did not return for neurodevelopmental assessment were similar to those of infants who returned for follow-up at the 18 months (see Supplementary Table 1). Compared with infants not seen for follow-up evaluation, infants seen at follow-up were smaller at birth and had higher rates of periventricular leukomalacia, and patent ductus arteriosus detected with echocardiography.
Twenty-nine infants (21%) in the PCV group and 15 infants (11%) in VGPSV group died during their initial NICU hospitalization (P=0.02). After discharge from neonatal intensive care, two infants in the PCV group and three in the VGPSV group died. The overall mortality rate was not significantly different between study groups (23% in the PCV group and 13% in the VGPSV group; P=0.06).
Univariate analysis of neurodevelopment outcome data is summarized in Table 3. Two infants in the PCV group who were diagnosed with cerebral palsy were not testable with the Bayley Scales. A trend towards lower rates in the VGPSV group was found for each individual component of neurodevelopmental outcome. The composite outcome of NDI or death reached statistical difference between study groups in the smallest category of infants with birth weight ⩽1000 g. The PCV group has a greater proportion of infants below each of the three cognitive composite cut points (55, 70 and 85, respectively), and the difference is significant when the cut point is at 55 or 70 (Figure 2). Similar results were seen for motor composite and language scores. However when motor composite, cognitive composite and language scores were considered as continuous variables, there was no difference between the study groups in Bayley Cognitive, Motor or Language Scales. Similar results were obtained when we restricted analyses to infants with birth weight⩽1000 g.
Adjusting for birth weight, race and doses of antenatal steroids, there was a trend for reduced risk for cognitive composite <70 (odds ratio: 0.17; 95% CI: 0.01, 2.33; P=0.18), motor composite <70 (odds ratio: 0.41; 95% CI 0.1, 1.74; P=0.23), and composite measure of NDI or death in the VGPSV group that did not reach statistical significance. The study group difference in the NDI or death outcome in infants with birth weight ⩽1000 g maintained statistical significance after adjustment for confounders (odds ratio 0.5; 95% CI 0.25, 0.99; P=0.05 ). The individual components of neurodevelopment outcome in this subgroup of infants did not reach statistical difference.
Discussion
Our primary finding was that VGPSV was not associated with a higher rate of survival without NDI among VLBW infants (birth weight of ⩽1250 g). The lack of improvement in the combined outcome of death or NDI might be due to low statistical power. Our study agrees with a meta-analysis that found shorter duration on mechanical ventilation, and lower rates of air leak syndrome in the volume-targeted ventilation group as compared with pressure-limited ventilation group.10 The lower risk of hypotension in the VGPSV group could reflect a more stable tidal volume and systemic venous return.26 Typically, with volume-targeted ventilation, lower airway pressures are required thus minimizing the hemodynamic impact.27 Change in clinician practices over time might also have contributed to the lowered frequency of hypotension in the second epoch of our study (that is, the volume-targeted ventilation epoch). The NEC rate was significantly higher in the VGPSV group as compared with PCV group. As stages lower than 2 on the modified Bell’s criteria for NEC were collected, misclassification of some of the sepsis-like cases as NEC cases is possible. Small randomized controlled studies of VGPSV that collected data on NEC as a secondary outcome did not show an increased risk of NEC with the use of VGPSV as compared with PCV.28
Two previously published studies investigated but found no association between volume-targeted ventilation and improved neurodevelopmental outcome. Singh et al.16 found no difference in the rate of death or disability in a study that compared PCV with volume control ventilation, a type of volume-targeted ventilation that differs from the type we studied. D’Angio et al.17 compared developmental outcomes of preterm infants treated with either pressure-regulated volume control, an assist/control mode of ventilation, or synchronized intermittent mandatory ventilation mode. Neurodevelopmental outcome at 6 to 9 months’ corrected age did not differ between the two groups; however, the study limited by small sample size.
Limitations of our study include the retrospective observational design, the modest sample size and the lost to follow-up of 30 percent of survivors. To test the hypothesis that volume-targeted ventilation is associated with a decreased risk of the combined outcome of death or NDI, our sample provided 80% power to detect a decrease from 0.42 to 0.23 (41% reduction) in the combined outcome of moderate-to-severe NDI/death.
Conclusion
VGPSV was associated with lower mortality and improved short-term outcomes in VLBW infants and a statistically nonsignificant trend toward lower risk for combined death and long-term neurodevelopmental outcome as defined in our study. To provide strong evidence of clinical efficacy of VGPS on neurodevelopmental outcome, a well-powered, properly executed randomized controlled trial is warranted.
References
Stoll BJ, Hansen NI, Bell EF, Shankaran S, Laptook AR, Walsh MC et al. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics 2010; 126 (3): 443–456.
Philip AG . Bronchopulmonary dysplasia: then and now. Neonatology 2012; 102 (1): 1–8.
Skidmore MD, Rivers A, Hack M . Increased risk of cerebral palsy among very low birth weight infants with chronic lung disease. Dev Med Child Neurol 1990; 32: 325.
Goldsmith & Karoktin Assisted Ventilation of the Neonate, 5th edn, Saunders Elsevier, St Louis, Missouri, 2011.
Jobe AH, Ikegami M . Mechanisms initiating lung injury in the preterm. Early Hum Dev 1998; 53 (1): 81–94.
Dreyfuss D, Saumon G . Ventilator-induced lung injury: lessons from experimental studies. Am J Respir Crit Care Med 1998; 157: 294–323.
Clark RH, Gerstmann DR, Jobe AH, Moffitt ST, Slutsky AS, Yoder BA . Lung injury in neonates: causes, strategies for prevention, and long-term consequences. J Pediatr 2001; 139: 478–486.
Dreyfuss D, Saumon G . Role of tidal volume, FRC, and end-inspiratory volume in the development of pulmonary edema following mechanical ventilation. Am Rev Resp Dis 1993; 148: 1194–1203.
Sinha S, Donn S . Minimizing ventilator induced lung injury in preterm infants. Arch Dis Child Fetal Neonatal Ed 2006; 91: F226–F230.
Wheeler KI, Klingenberg C, Morley CJ, Davis PG . Volume-targeted versus pressure-limited ventilation for preterm infants: a systematic review and meta-analysis. Neonatology 2011; 100 (3): 219–227.
Greisen G, Munck H, Lou H . May hypocarbia cause ischaemic brain damage in the preterm infant? Lancet 1986; 2 (8504): 460.
Erickson SJ, Grauaug A, Gurrin L, Swaminathan M . Hypocarbia in the ventilated preterm infant and its effect on intraventricular haemorrhage and bronchopulmonary dysplasia. J Paediatr Child Health 2002; 38: 560–562.
Giannakopoulou C, Korakaki E, Manoura A, Bikouvarakis S, Papageorgiou M, Gourgiotis D et al. Significance of hypocarbia in the development of periventricular leukomalacia in preterm infants. Pediatr Int 2004; 46: 268–273.
Keszler M, Abubakar K . Volume guarantee: stability of tidal volume and incidence of hypocarbia. Pediatr Pulmonol 2004; 38: 240–245.
Cheema IU, Sinha AK, Kempley ST, Ahluwalia JS . Impact of volume guarantee ventilation on arterial carbon dioxide tension in newborn infants: a randomized controlled trial. Early Hum Dev 2007; 83: 183–189.
Singh J, Sinha SK, Alsop E, Gupta S, Mishra A, Donn SM . Long term follow-up of very low birthweight infants from a neonatal volume versus pressure mechanical ventilation trial. Arch Dis Child Fetal Neonatal Ed 2009; 94: F360–F362.
D’Angio CT, Chess PR, Kovacs SJ, Sinkin RA, Phelps DL, Kendig JW et al. Pressure-regulated volume control ventilation vs synchronized intermittent mandatory ventilation for very low-birth-weight infants: a randomized controlled trial. Arch Pediatr Adolesc Med 2005; 159: 868–875.
Palisano R, Rosenbaum P, Walter S, Russell D, Wood E, Galuppi B . Development and reliability of a system to classify gross motor function in children with cerebral palsy. Dev Med Child Neurol 1997; 39: 214–223.
Vohr BR, Wright LL, Dusick AM, Mele L, Verter J, Steichen JJ et al. Neurodevelopmental and functional outcomes of extremely low birth weight infants in the National Institute of Child Health and Human Development Neonatal Research Network, 1993-1994. Pediatrics 2000; 105: 1216–1226.
Bayley N . Technical Manual of the Bayley Scales of Infant and Toddler Development, 3rd edn, Harcourt Assessment Inc: San Antonio, TX, USA, 2006.
Gordon PV, Swanson JR, Attridge JT, Clark R . Emerging trends in acquired neonatal intestinal disease: is it time to abandon Bell’s criteria? J Perinatol 2007; 11: 661–671.
Klein JO . Bacterial sepsis and meningitis. In: Remington JS, Klein JO (eds). Infectious Diseases of Fetus and Newborn Infant, 5th edn, Sauders: Philadelphia, PA, USA, 2001; 943–998.
Papile LA, Burstein J, Burstein R, Koffler H . Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. J Pediatr 1978; 92: 529–534.
Committee for the Classification of Retinopathy of Prematurity. The international classification of retinopathy of prematurity revisited. Arch Ophthalmol 2005; 123: 991–999.
Engle WD . Definition of normal blood pressure range: the elusive target. In: Kleinman CS, Seri I (eds). Neonatology Questions and Controversies: Hemodynamics and Cardiology. Saunders/Elsevier Co: Philadelphia, PA, USA, 2008; 39–66.
Pryds O . Control of cerebral circulation in the high-risk neonate. Ann Neurol 1991; 30: 321–329.
Swamy R, Gupta S, Singh J, Donn SM, Sinha SK . Tidal volume delivery and peak inspiratory pressure in babies receiving volume targeted or time-cycled, pressure limited ventilation: a randomized controlled trial. J Neonatal Perinatal Med 2008; 4: 239–243.
Nafday SM, Green RS, Lin J, Brion LP, Ochshorn I, Holzman IR . Is there an advantage of using pressure support ventilation with volume guarantee in the initial management of premature infants with respiratory distress syndrome? A pilot study. J Perinatol 2005; 25: 193–197.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Journal of Perinatology website
Supplementary information
Rights and permissions
About this article
Cite this article
Stefanescu, B., Frewan, N., Slaughter, J. et al. Volume guarantee pressure support ventilation in extremely preterm infants and neurodevelopmental outcome at 18 months. J Perinatol 35, 419–423 (2015). https://doi.org/10.1038/jp.2014.228
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2014.228
- Springer Nature America, Inc.
This article is cited by
-
Volume-targeted ventilation vs pressure-controlled ventilation for very low birthweight infants: a protocol of a randomized controlled trial
Trials (2023)
-
Volume guarantee pressure support ventilation in extremely preterm infants and neurodevelopmental outcome at 18 months
Journal of Perinatology (2015)