Abstract
Background/objective:
The prevalence of childhood obesity continues to rise in most countries, but the exposures responsible remain unclear. The shape of the body mass index (BMI) distribution curve defines how a population responds, and can be described by its three parameters—skew (L), median (M) and variance (S). We used LMS analysis to explore differences in the BMI trajectories of contemporary UK children with those of 25 years ago, and to draw inferences on the exposures responsible.
Subjects/methods:
We applied Cole’s LMS method to compare the BMI trajectories of 307 UK children (EarlyBird cohort) measured annually from 5–16 years (2000–2012) with those of the BMI data set used to construct the UK 1990 growth centiles, and used group-based trajectory modelling (GBTM) to establish whether categorical trajectories emerged.
Results:
Gender-specific birth weights were normally distributed and similar between both data sets. The skew and variance established by 5 years in the 1990 children remained stable during the remainder of their childhood, but the pattern was different for children 25 years on. The skew at 5 years among the EarlyBird children was greatly exaggerated, and involved selectively the offspring of obese parents, but returned to 1990 levels by puberty. As the skew diminished, so the variance in BMI rose sharply. The median BMI of the EarlyBird children differed little from that of 1990 before puberty, but diverged from it as the variance rose. GBTM uncovered four groups with distinct trajectories, which were related to parental obesity.
Conclusions:
There appear to be two distinct environmental interactions with body mass among contemporary children, the one operating selectively according to parental BMI during early childhood, the second more generally in puberty.
Similar content being viewed by others
Introduction
Childhood obesity has become a global epidemic.1 The UK recorded only subtle changes in its prevalence during the decade 1974–1984, but a substantial rise from 5.0 to 9.0% during the next 10 years (1984–1994),2 and to 16% by 2010.3 Most, though not all,4 industrialised and industrialising countries have experienced something similar. Obesity is important because of its co-morbidities later in life,5 and a knowledge of its timing is important for a better understanding of the factors responsible. Gestation and early infancy are increasingly recognised as key periods in the development of obesity,6 but less is known about childhood for lack of published data from truly longitudinal studies. Multiple cross-sectional data, such as might be available year-on-year from national screening programmes, cannot be used to construct true trends.
Obesity rates are usually reported as a percentage or as a mean, and the popular perception of weight gain in children tends to be one of whole population shift, sometimes referred to as a ‘drifting iceberg’.7 However, neither the percentage nor the mean defines the distribution, which is key in understanding environmental impact. Thus, the mean can rise without change in the median (skew or ‘landslip’), and the variance can rise without change in the mean (although the mean usually rises, along with the median, because changes in variance are rarely symmetrical). Obesity ‘rates’ increase with both skew and variance, as more children in both instances cross the obesity threshold—but for different reasons. The development of a skew implies that a sector only of the population is exposed (or susceptible) to an environmental risk, while increasing variance suggests a more generalised exposure or susceptibility.
Height is normally distributed, and so are weight and body mass index (BMI) in pre-industrialised children.8 Skew in these variables is a feature of industrialisation.9 Cole proposed that the three parameters, ‘skew’ (λ/L), ‘median’ (μ/M) and ‘variance’ (σ/S) be used to construct growth curves in children.10 Together, they define the shape of a distribution and individually reflect the type of interaction between environmental risk and genetic susceptibility. In this report, we have compared the LMS parameters from a contemporary cohort with those derived from the children used to populate the 1990 UK Growth Standards. Parental BMI is generally considered the single most important influence on the BMI of their offspring, and assortative mating might be expected to strengthen it further, but the correlation in BMI between contemporary parents is now reportedly low (r=0.12, P<0.08).11 Our aim was to establish whether environmental impacts have changed over a period that was arguably too short for genetic change (period effect—between-group differences over a generation), and how such changes might have influenced the pattern of obesity (age effect—within-group differences over childhood).
Materials and methods
Data were sourced from the EarlyBird study, which monitored the height and weight (BMI) of 307 contemporary children (170 boys) recruited at the age of 5 years in the city of Plymouth, UK. Details of the study design are published elsewhere.12 In brief, the children were selected randomly from the 53 of 72 primary schools that agreed to take part. The great majority (98%) were white Caucasian, with a wide socio-economic mix representative of the UK as a whole (mean Index of Multiple Deprivation 21.7, range 6.5–73.0; UK mean 26.3). Measures were made annually from age 5 to 16 years in blind duplicate by trained research nurses in a hospital environment. At least 80% of the measures were available at each visit for the boys, and 75% for the girls. Birth weight was collected from the Plymouth Child Information Register, based on hospital records. The height and weight of both mother and father were measured directly and in duplicate by the same trained nurses at baseline. Demographic information was collected in person from the mother by questionnaire.
Stata 2011 (Stata Statistical Software: Release 12. College Station, TX, USA) was used for the analysis. One sample two-tail t-tests were carried out to compare mean weight standard deviation score (SDS) and BMI SDS of the EarlyBird children in relation to those of 1990. One sample two-tail proportion test was used to test the hypothesis that the proportion of obese children in the EarlyBird cohort was different from the 2% (>98th centile) obese in the reference population. Statistical significance was tested at P<0.05.
LMS
LMS parameters for EarlyBird data were fitted by using LMS chartmaker by observing the changes in penalised deviance measures in sequential steps.13 Best model was chosen when deviance value was least and provided a smooth fit of the curves. The individual trends in age and sex-specific L, M and S were compared graphically with the corresponding LMS values from the 1990 data (provided with the LMS Microsoft Excel add-in).14 The 1990 UK growth standards were sourced originally from 12 surveys of the 1980s, together involving ~8000 children.11
Group-base trajectory modelling
The trend in BMI was not uniform across the distribution, so that modelling of average BMI would fail to resolve latent growth trajectories. Accordingly, we assumed that the EarlyBird cohort was composed of distinct groups defined by their growth pattern, within which trajectories were similar and between which they were different, and we carried out group-based trajectory modelling (GBTM) to capture the differences.15 As pointed out by Nagin and Odgers,16 there is no single definition for a ‘best-fit’ model, so we fitted several models in order to compare the Bayesian Information Criteria17—the Akaike Information Criterion18 and Lo-Mendell-Rubin likelihood ratio test statistic,19 the average posterior probability for group membership in each model, and significant (P<0.05) linear/higher order polynomials fitted within each group. GBTM was carried out using the Stata plug-in ‘traj’,20 and in order to generate meaningful trajectories given the size of the cohort, we modelled both genders together. Gender was incorporated as a time constant variable, in order to adjust for any gender-related effect that could influence the group trajectories and thus free the extracted group trajectories from gender bias.
GBTM estimations are unbiased where data are missing at random.16 Some 95% of our BMI data were available at multiple measurement points and, as the missing patterns were not attributable to any particular characteristic, imputations were made to minimise loss of information using ICE (Imputation by Chained Equation), a user-written programme in Stata designed for imputing longitudinal data.21 Age, gender, height, weight and BMI were included in the imputation model, and group-based trajectory modelling was performed on the data set whose BMI distribution approximated most closely to the original. Finally, we developed a multinomial logistic regression model to explore the role early life factors that may have been associated with the increased risk of being in the vulnerable groups identified by the GBTM.
Results
Mean birth weight (kg) for boys in the EarlyBird cohort was 3.45 kg confidence interval (CI) 3.37–3.53, and for girls 3.34 kg CI 3.25–3.45. The characteristics of the EarlyBird cohort are summarised in Table 1, which tests for differences from the 1990 UK Growth Standards in age-standardised weight SDS, BMI SDS and proportion of obese children in each gender at age 5, 10 and 16 years (age-specific full table, Supplementary Appendix: Supplementary Table 1).
Weight and BMI SDS
The birth weight of the EarlyBird cohort was no different from that of the preceding generation. By 5 years, however, EarlyBird boys and girls were heavier, and their BMI correspondingly greater. Both genders gained excess weight year-by-year throughout childhood so that, by 16 years, the mean BMI of the boys had risen to 0.43 SDS and that of the girls to 0.85 SDS.
Proportion obese
Although they were no different in weight at birth from the 1990 children, 4% (CI 1.0–7.0, P<0.05) of the EarlyBird boys and 5% (CI 1.0–9.0, P<0.01) of the girls were already obese by 5 years, compared with 2% (by definition) of the 1990 children. The proportion increased with age so that, by 16 years, 11% of the EarlyBird boys and 16% of the girls were obese.
L, M and S
Birth
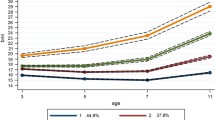
Birth weight distribution was close to Normal (Figure 1a) (L for boys: EarlyBird −0.33, 1990 +0.77; L for girls: EarlyBird +1.93, 1990 +0.66).
(a–c) Comparison of the EarlyBird cohort, by gender, with the UK Growth Standards of 1990 in relation to skew in BMI (a), median (b) and variance (c). The marker between birth and 5 years indicates non-availability of data from 1–4 years, and the estimate for ‘L’ and ‘S’ at birth relates to weight, not to BMI.
0–5 Years
The EarlyBird cohort gained substantial excess weight from 0–5 years, but it was largely related to skew (‘landslip’ rather than a ‘drifting iceberg’—Figure 1a), with little change in the median BMI in either gender (Figure 1b), and none in its variance (Figure 1c). Thus, while a rightwards (negative) skew in BMI was already established by 5 years in 1990, it was considerably greater in 5-year-old EarlyBird children 25 years later, and particularly so in the boys (L values for boys: EarlyBird –3.12, 1990 –1.48; girls: EarlyBird: –2.23, 1990 –1.22).
5–9 Years
The greater skew in the BMI of EarlyBird children at 5 years returned to 1990 values by the onset of puberty, and rather earlier in the girls than the boys (Figure 1a). At the same time, the variance in BMI among both began to rise steeply, and to diverge sharply from the trend of 1990, more so in the girls (Figure 1c). As the variance in BMI of the EarlyBird children moved away from the trajectory of 1990, so did the median BMI (Figure 1b).
9–16 Years
The median BMI of the EarlyBird children continued to diverge from the 1990 values as puberty progressed (Figure 1b). The difference in variance, on the other hand, peaked at around 13 years in both genders (Figure 1c). The skew in BMI established by 5 years remained strikingly stable throughout childhood in the 1990 data, whereas that of the EarlyBird children returned to the values of 1990 by early puberty, before rising again in later adolescence (Figure 1a).
To summarise, the mean BMI of the EarlyBird children was higher than that of the 1990 standards throughout the course of childhood (Table 1), but for reasons that changed as the children grew. Exaggerated skew accounted for the difference early on (the median changed little), while widening variance accounted for it later (the median rose). In both instances, the mean rose progressively, and the prevalence of obesity increased. The sequence of events points to trends in the distribution of BMI over the course of contemporary childhood that are different from a generation ago, when skew and variance were relatively stable.
Group-based trajectory modelling (GBTM)
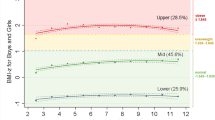
We carried out GBTM to establish whether there were groups of EarlyBird children who behaved sufficiently differently from each other over childhood to account for the changes in distribution of BMI that we observed over a generation (Figure 2).
The effect of multiple imputations on the GBTM dataset was minimal. In response to imputation, the mean BMI for girls changed from 19.39 to 19.44, Δ+0.05 (boys 18.07 to 18.30, Δ +0.23) and the s.d. from 4.14 to 4.08, Δ −0.06 (boys 3.43 to 3.57, Δ+0.14). Several models were fitted and Bayesian Information Criteria/Akaike Information Criterion/L statistics were compared in the iterative process of choosing the final model for meaningful group trajectories. Supplementary Table 2 (Supplementary Appendix) shows the stepwise development of the final model. Five-group solutions were sought initially, with higher order polynomials as a function of age. Bayesian Information Criteria/Akaike Information Criterion/L values were closer towards zero in the five-group solutions (model-1, model-2) but they resulted in a group with small numbers (1.7%, not reported), which made the model difficult to interpret. A four-group solution was therefore retained (model-5), whose model statistics were slightly lower (Bayesian Information Criteria: −8004.5, Akaike Information Criterion: −7976.55, L: −7961.55) in comparison with the five-group solutions, but whose average posterior probability for group membership was similar (0.97), and whose fitted lines were significant (P<0.01) with small standard errors.
The four distinct groups identified in the final model are arbitrarily labelled Group-1 to Group-4, according to gradient of BMI. As expected (Figure 2a), the majority (76%) of the children remained in Group-1 or Group-2 throughout (G1 26.1%, G2 49.5%), while their mean BMI rose from 15 to 22, well within the reference range for normal. In contrast, the mean BMI of Group-4 children (5.8%) crossed 25 (overweight) at age 9 years and 30 (obese) by 13 years, to reach 32.3 by 16 years. The mean BMI of Group-3 (18.5%) departed from the 1990 trajectory later than that of Group-4 moving more slowly towards obesity (Figure 2b). The mean BMI of Group-3 was 17.5 at 5 years and 26.5 at 16 years. Groups 3 and 4 together comprised 24.3% of the sample and, as expected, the mean BMI of Group-4 children was consistently and substantially higher in both genders than that of the 1990 children 98th centile.
Early life factors are important to subsequent growth, so we used multinomial logistic regression analysis, with the groups as the outcome variable, to establish whether such factors were associated with GBTM group trajectory. Parental BMI, birth weight SDS, mothers’ smoking habit during pregnancy, gestational period, mothers’ age at child birth, child birth order, socio-economic deprivation index and duration of breastfeeding were all considered in the model. Comparative risk ratios are presented in Table 2 with reference to Group G-1, whose BMI trajectory was normal throughout.
Only birth weight SDS and mothers’ BMI had a statistically significant impact on group allocation, though not universally. Thus, a birth weight greater by 1 SDS was associated with twice the risk of allocation to Group G-3 (P<0.01) compared with Group G-1. Again, every unit difference in the mothers’ BMI was associated with a 1.18-fold (P<0.01) greater risk of allocation to Group G-4. Interestingly, although the mean BMI of the parents was high (mothers 30.6 CI 26–34.3; fathers 30.2 CI 25.6–34), only children from Group G-4 were associated with parental BMI, and then only the mothers’. Fathers’ BMI did not have a significant impact on group allocation.
Discussion and limitation
We compared the BMI LMS parameters of contemporary children with those of children who lived a generation ago to examine the differences, and modelled group-based trajectories within the contemporary group to establish whether such differences could be explained by the behaviour of discrete groups of children. The results are straightforward but novel, and suggest that the obesogenic pressures to which modern children are exposed differ not only from those of a generation ago, but according to the stage of childhood as well. The mother’s BMI was particularly associated with an early negative skew in offspring weight, while birth weight appeared to influence a later increase in its variance. Both effects registered as an increase in overweight and obesity, but for very different reasons.
Weight at birth was distributed symmetrically about the mean, but somewhere between birth and 5 years a skew developed in the BMI with little change in the median. The lack of shift in median implies that only a sector of the childhood population, not the entire population, is susceptible to, or selectively exposed to, some early environmental pressure. One possibility, given that the skew emerged after birth and that these children were in large part the offspring of overweight or obese parents, is behavioural. It seems possible, as we have suggested previously,11 that the (inappropriate) dietary habits of today’s obese parents, and particularly the mother, are revisited on their children from a young age.22 A second contribution to the rising prevalence of obesity among today’s children, this time unrelated to parental weight and not connected to skew, appears to be a widening variance from early puberty. Variance implies a more general environmental exposure, and involves a broader group of children. Importantly, group-based trajectory modelling suggested that the children subject to the widening variance in early puberty were different children from those responsible for the exaggerated skew some years earlier. The age-related differences in skew and variation of BMI appear to have emerged since 1990, and their implications may be important, as the effective prevention of childhood obesity depends crucially on understanding its cause. Thus two quite distinct strategies may be needed to reduce obesity in today’s children—the one aimed at parental conduct during the early years (or even pre-conception if epigenetic change proves to be important), the other at the wider environment later on.
Group-based trajectory modelling is an objective probabilistic approach to capturing latent growth trajectories, which are assumed to exist in the population. It was based here on independent longitudinal analysis, and used to complement the LMS curves, not to be compared with them. Each of the four groups deemed by the model to provide optimal resolution incorporated contemporary children who behaved in like fashion as a group over the course of childhood, but differently from the other groups. The trajectory solutions that we found are similar to those of larger studies,23 which severally report 72.8 and 83%, in comparison to our 76%, as the proportion of children in a contemporary population that retains normal weight throughout childhood. Those children who were obese at 5 years (5.8%) tended to remain obese throughout, and accounted for much of today’s obesity in pre-pubertal children. The rising prevalence of obesity in adolescence was attributable to a separate group of children whose BMI first rose rapidly in puberty. The transition was associated with, and we believe accounted for, by the increase in variance with rise in median BMI that characterises puberty. One such second pressure might be the increasing freedom (both over the course of time, and with age) for growing children to consume the processed meals, sugary drinks and calorie-dense ‘snacks’ that many consider underpin the current levels of childhood obesity.
The replacement of skew by widening variance as the basis for rising obesity in a genetically stable cohort suggests that one set of environmental factors, which involves a select few when they are very young, gives place to different set of factors with a more general impact when they are older. Although largely limited to the children of obese parents, the contribution of early weight gain to childhood obesity is substantial—over 90% of the excess weight gained before puberty (9 years) in girls, and over 70% in boys, is gained before the age of 5 years.24
The study has strengths and limitations. EarlyBird is small by epidemiological standards, and there is at least one larger longitudinal dataset spanning contemporary childhood (ALSPAC) in which our conclusions could be tested. EarlyBird draws power from the serial and precise measurement of the same cohort over a prolonged period of time. Nearly 80% of the cohort was retained over the 12 years, and all auxology was carried out in blind duplicate by a team of three research nurses in a hospital setting. The cohort, however was 98% white Caucasian, which favours homogeneity of the analysis but limits generalisability of the results.
Conclusion
We interpret the current data to mean that that the weight gain in contemporary childhood, at least in the UK, may have two different components. The early skew in BMI suggests an environmental pressure to which very young children are either selectively susceptible or selectively exposed. The later increase in variance suggests a pressure to which the population is either randomly susceptible or randomly exposed. Two, largely independent, causes of obesity operating at different ages may signal the need for two fundamentally different strategies in its prevention.
References
Wang Y, Lim H . The global childhood obesity epidemic and the association between socio-economic status and childhood obesity. Int Rev Psychiatry 2012; 24: 176–188.
Chinn S, Rona RJ . Prevalence and trends in overweight and obesity in three cross sectional studies of British Children, 1974-94. BMJ 2001; 322: 24–26.
http://www.noo.org.uk/uploads/doc/vid_16006_ChildWeightFactsheet-July2012.pdf. NOO data fact sheet, 2012.
Rokholm B, Baker JL, Sorensen TI . The levelling off of the obesity epidemic since the year 1999–a review of evidence and perspectives. Obesity Rev 2010; 11: 835–846.
Must A, Strauss RS . Risks and consequences of childhood and adolescent obesity. Int J Obesity Relat Metab Disord 1999; 23: S2–S11.
Dietz WH . Critical periods in childhood for the development of obesity. Am J Clin Nutr 1994; 59: 955–959.
Penman AD, Johnson WD . The changing shape of the body mass index distribution curve in the population: implications for public health policy to reduce the prevalence of adult obesity. Prev Chronic Dis 2006; 3: A74.
Lakshmi S, Metcalf B, Joglekar C, Yajnik CS, Fall CH, Wilkin TJ . Differences in body composition and metabolic status between white U.K. and Asian Indian children (EarlyBird 24 and the Pune Maternal Nutrition Study). Pediatr Obes 2012; 7: 347–354.
Popkin BM, Adair LS, Ng SW . Global nutrition transition and the pandemic of obesity in developing countries. Nutr Rev 2012; 70: 3–21.
Cole TJ . The LMS method for constructing normalized growth standards. Eur J Clin Nutr 1990; 44: 45–60.
Cole TJ, Green PJ . Smoothing reference centile curves: the LMS method and penalized likelihood. Stat Med 1992; 11: 1305–1319.
Cole TJ . Fitting smoothed centile curves to reference data. J R Stat Soc 1988; 151: 33.
Perez-Pastor EM, Metcalf BS, Hosking J, Jeffery AN, Voss LD, Wilkin TJ . Assortative weight gain in mother-daughter and father-son pairs: an emerging source of childhood obesity. Longitudinal study of trios (EarlyBird 43). Int J Obes (Lond) 2009; 33: 727–735.
Voss LD, Kirkby J, Metcalf BS, Jeffery AN, O'Riordan C, Murphy MJ et al. Preventable factors in childhood that lead to insulin resistance, diabetes mellitus and the metabolic syndrome: the EarlyBird diabetes study 1. J Pediatr Endocrinol Metab 2003; 16: 1211–1224.
Pan H, Cole T . LMSchartmaker, a program to construct growth references using the LMS method. Version 2011; 2: 54.
LMSgrowth, a Microsoft Excel add-in to access growth references based on the LMS method. [program]. Version 2.77. version, 2012.
Nagin DS . Group-Based Modeling of Development. Harvard University Press: Cambridge, MA, USA, 2005.
Nagin DS, Odgers CL . Group-based trajectory modeling in clinical research. Annu Rev Clin Psychol 2010; 6: 109–138.
Raftery A . Bayesian Model Selection in Social Research. Cambridge: MA: Blackwell Science, 1995.
Akaike H . A new look at the statistical model identification. IEEE Trans Autom Control 1974; 19: 7.
Lo Y, Mendell N, Rubin D . Testing the number of components in a normal mixture. Biometrika 2001; 88: 767–778.
Jones BL, Nagin DS A Stata Plugin for Estimating Group-Based Trajectory Models, 2012.
Royston P . Multiple imputation of missing values. Stata J 2004; 4: 227–241.
Lean ME . Childhood obesity: time to shrink a parent. Int J Obes (Lond) 2010; 34: 1–3.
Acknowledgements
We thank Karen Brookes, Val Morgan, the EarlyBird children, their parents and our volunteers for their continued support. EarlyBird is currently funded by the Bright Future Trust, BUPA Foundation, Peninsula Foundation, Kirby Laing Trust and the EarlyBird Diabetes Trust.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Mostazir, M., Jeffery, A., Voss, L. et al. Childhood obesity: evidence for distinct early and late environmental determinants a 12-year longitudinal cohort study (EarlyBird 62). Int J Obes 39, 1057–1062 (2015). https://doi.org/10.1038/ijo.2015.68
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2015.68
- Springer Nature Limited
This article is cited by
-
Evidence for energy conservation during pubertal growth. A 10-year longitudinal study (EarlyBird 71)
International Journal of Obesity (2016)