Abstract
Proliferation of vascular smooth muscle cells (VSMCs) is thought to have a key role in the development of atherosclerotic lesions. Neuropeptide Y (NPY), norepinephrine and dopamine are sympathetic neurotransmitters. NPY has been particularly shown to stimulate proliferation of VSMCs. NPY, norepinephrine and dopamine are all sympathetic transmitters. In our previous study, we found that in the presence of the dopamine receptor, the α1-adrenergic receptor-mediated VSMC proliferation is reduced. We hypothesize that the activation of the D1-like receptor might inhibit the NPY-mediated VSMC proliferation. In our present study, we found that NPY, mainly via the Y1 receptor, increased VSMC proliferation. This was determined by [3H]-thymidine incorporation, in a concentration (10−11 to 10−8 m)-dependent manner. In the presence of the D1-like receptor agonist, fenoldopam (10−12 to 10−5m), the stimulatory effect of NPY on VSMC proliferation was reduced. The involvement of the D1-like receptor was confirmed when the inhibitory effect of fenoldopam was reversed in the presence of the D1-like receptor antagonist SCH-23390 (10−8m). Moreover, the inhibitory effect of fenoldopam on NPY-mediated VSMC proliferation was also blocked in the presence of the PKA inhibitor 14–22 (10−6 m). Protein kinase A activator 8-(4-chlorophenylthio) adenosine-3,5-cyclic monophosphorothioate, Sp-isomer sodium salt (10−6 m) could simulate the stimulatory effect of fenoldopam. It indicated that the inhibitory effect of D1-like receptors on NPY-mediated VSMC proliferation may have an important role in the regulation of blood pressure or prevention of atherosclerosis.
Similar content being viewed by others
Introduction
Understanding the regulatory mechanism1, 2, 3, 4 of vascular smooth muscle cell (VSMC) proliferation is important in elucidating the pathogenesis of atherosclerosis as well as in the design of therapeutic drugs for the prevention of atherogenic disorders.5, 6
Neuropeptide Y (NPY) is a 36-amino acid neurotransmitter that belongs to a family of peptides containing peptide YY and a pancreatic polypeptide. It was isolated originally from porcine brain and has been shown to be the most abundant neuropeptide in the mammalian central and peripheral nervous systems.7, 8 NPY, together with norepinephrine, ATP and dopamine, is released from the sympathetic nerve terminals to act as vaso-neurotransmitters. According to the duration of the vasoconstrictive activity, it is thought that ATP, norepinephrine and NPY mediate the immediate,9 intermediate and long-lasting components of sympathetic nerve stimulation-induced vasoconstriction, respectively. Hypertension and atherosclerosis are chronic progressive diseases, therefore, the long-lasting effect, as mediated by NPY, seems to be much more important.
Dopamine has recently been characterized as an important modulator of blood pressure,10, 11 sodium balance and renal and adrenal functions, and it is relevant to the pathogenesis and/or maintenance of hypertension.12, 13 Dopamine receptors are classified into two families: the D1-like receptor subfamily includes the D1 and the D5 receptors, while the D2, D3 and D4 receptors belong to the D2-like subfamily. Whereas the D1-like receptors couple to the stimulatory G protein, GαS, D2-like receptors couple to the inhibitory G protein, Gαi/GαO.13 Our previous study showed that in the presence of the dopamine receptor, the α1-adrenergic receptor-mediated VSMC proliferation is reduced.4 As a major mitogenic factor, NPY has been shown to stimulate the proliferation of VSMCs.3 We hypothesize that the activation of the D1-like receptor might inhibit the NPY-mediated VSMC proliferation. Therefore, our present study was designed to investigate the possible role of D1-like receptors on NPY-mediated VSMC proliferation and examine its potential mechanism(s).
Methods
SD rat aortic smooth muscle cell culture
VSMCs were isolated from the aortic strips of male Sprague–Dawley (SD) rats (200–250 g) with an explant technique previously described.1, 2 Briefly, the arteries were dissected free of fat and excess adventitial tissue and then opened along its longitudinal axis, with the endothelial lining removed by vigorously scraping of the lumen with a scalpel blade. Small fragments (~1 mm3) were transferred to a flask and cultured in Dulbecco’s modified Eagle’s medium containing 20% fetal bovine serum supplemented with 1% penicillin/streptomycin/bFGF/insulin/EGF (all from Sigma, St Louis, MO) in a humidified atmosphere (95% air/5% CO2) at 37 °C. The purity and identity of VSMCs were verified by their typical morphological pattern (multilayer sheets and ‘hills and valleys’) and by immunohistochemistry staining using specific mouse monoclonal antibody against smooth muscle cell α-actin (Sigma). Cells from passages 3 through 5 were used. The cells were then placed in serum-free media for 24 h to render them quiescent.14, 15 The animal experiments were approved by the Animal Use and Care Committee of Daping Hospital.
Measurement of [3H]-thymidine incorporation
Cell proliferation was determined by measuring the incorporation of [3H]-thymidine (Atomic Energy Research Establishment of China, Beijing, China) into the DNA of cells cultured in 96-well plates. After the induction of quiescence, VSMCs were re-stimulated with or without 10−8mol l−1 NPY (Sigma) for 24 h. [3H]-thymidine (1 μCi ml−1) was added to each well 6 h before the measurements. At the end of the incubation period, the medium was removed and the cells were treated with 0.25 ml of 0.05% trypsin–0.53 mm EDTA for 5 min and diluted to 10 ml with ice-cold 1 × phosphate-buffered saline. The cells were incubated in ice-cold 10% trichloroacetic acid for 30 min. After being washed twice with phosphate-buffered saline, the cells were lysed with 0.2 m NaOH, neutralized with 0.2 m HCl and subjected to liquid scintillation counting (Beckman LS6500, Beckman, Fullerton, CA, USA). Three independent samples were measured in each experiment and the experiment was repeated at least three times.14, 16
The D1-like receptor antagonists SCH-23390 (Sigma) was used to block the effect of the D1-like agonist, fenoldopam (Sigma). Owing to the fact that no ligand can distinguish between D1 and D5 receptors, the receptors involved in this experiment are likely to be D1-like receptors (D1 and D5), which are expressed in VSMCs of rats. To determine whether the proliferative effect of NPY is exerted at the NPY Y1 receptor, the NPY Y1 receptor antagonist BIBP3226 (Sigma) was also used. A D3 receptor agonist PD128907 (10−8 m) and a D3 receptor antagonist GR105691 (10−8 m) were used for testing the role of D3 receptor in NPY-induced VSMC proliferation.
MTT assay
The viability of cells was determined on the basis of the activity of mitochondrial dehydrogenase to reduce 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT; Sigma) to formazan. Briefly, cells were plated in 96-well plates at a density of 1 × 103 cells per well and treated with the indicated drugs for 24 h. MTT was then added to each well and incubation continued for 4 h in culture. Thereafter, 150 μl of dimethyl sulfoxide was added to each well and an absorbance at 490 nm was read on a microplate reader (Model 680, Bio-Rad, Hercules, CA, USA).17, 18
Estimation of cell number
VSMCs were seeded in six-well plastic culture dishes at a density of 1 × 104 cells per well in Dulbecco’s modified Eagle’s medium containing 10% fetal bovine serum and cultured for 24 h. After induction of quiescence, the culture media was replaced with serum-free media with the indicated drug and incubated for another 24 h. Cell viability was determined by the uptake of 0.4% Trypan blue after 5 min of mixing (Invitrogen Life Technologies, Carlsbad, CA), and were counted using a hemocytometer (Trypan blue uptake was observed in <10% of the cells). Counting was performed in triplicate.19
Rat carotid artery balloon injury model
Carotid artery balloon injury was induced in male SD rats (230–300 g) as described in a previous study.20 Briefly, rats were anesthetized using a mixture of ketamine (80 mg kg−1) and xylazine (12 mg kg−1), and then the left common and external carotid arteries were exposed through a midline cervical incision. A 2F Fogarty catheter (Edwards Life Sciences, Irvine, CA) was introduced into the common carotid artery through an arteriotomy in the external carotid artery and then the catheter was advanced to the proximal edge of the omohyoid muscle. To produce carotid artery injury, we inflated the balloon with saline and withdrew it three times from just under the proximal edge of the omohyoid muscle to the carotid bifurcation. After injury, the external carotid artery was ligated and the blood flow was restored.
After the balloon injury, rats were intraperitoneally injected with NPY (5 mg kg−1 per day, i.p.) or similar volume of distilled water (2 ml kg−1) daily for 14 days. D1-like receptor activator fenoldopam (6 mg kg−1 per day, i.p.) or/and D1-like receptor antagonist SCH-23390 (6 mg kg−1 per day, i.p.) were administrated in different experimental groups. Then, both carotid arteries were dissected and the sections were stained with hematoxylin and eosin and observed under a light microscope (Olympus, Tokyo, Japan). Media and neointima areas of injured arteries were quantified by planimetry (μm2) and intima-to-media ratios were calculated as reported.21
Immunohistochemistry staining
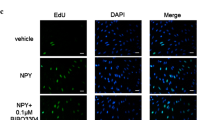
Briefly, paraformaldehyde-fixed vessel samples were conducted to paraffin-embedded sections (5 μm) and mounted on slides. The slides were incubated at 4 °C overnight, with or without primary anti Ki67 or a smooth muscle actin (α-SMA) antibodies at 1:1000 dilution. The slides were then incubated with secondary antibodies at 1:1000 at room temperature for 2 h. Pictures were taken under fluorescence microscopy. The antibodies were purchased from Invitrogen Life Technologies.
Statistical analysis
The data are expressed as mean±s.e.m. For analysis of differences among experimental groups, analysis of variance with the Bonferroni post hoc test was performed to determine statistical significance. A P-value of 0.05 was considered significant. In this study, n is the number of repeated sets of experiments for in vitro studies or the number of animals for in vivo studies.
Results
NPY, via the NPY Y1 receptor, increases proliferation of VSMCs from rat aorta of SD rats
Treatment of VSMCs with varying concentrations of NPY (10−11 to 10−6 m) for 24 h increased [3H]-thymidine incorporation in aortic VSMCs from SD rats in a concentration-dependent manner (Figure 1a). As major receptor subtypes of NPY receptors, we determined the role of Y1 in the VSMC proliferation. Results showed that the increase in [3H]-thymidine incorporation induced by NPY was blocked by the NPY Y1 receptor antagonist BIBP3226 (Figure 1b). This indicates that the NPY-mediated proliferation was via the Y1 receptor.
Effect of D1-like receptor on NPY-mediated proliferation of aortic vascular smooth muscle cells (VSMCs) from Sprague–Dawley (SD) rats. (a) Effect of NPY on VSMC proliferation. The primary cultured VSMCs were incubated with different concentration of NPY (10−11 to 10−8 m), the VSMC proliferation was determined by [3H]-thymidine incorporation. Results were expressed as fold increase of control. *P<0.05 vs. control (n=5). (b) Role of NPY Y1 in the NPY-mediated VSMC proliferation. VSMCs were incubated with NPY (10−8 m) in the presence of Y1 receptor antagonist BIBP3226 (10−7 m). The VSMC proliferation was determined by [3H]-thymidine incorporation. Results were expressed as fold increase of control. *P<0.05 vs. control, #P<0.05 vs. NPY (n=6). Analysis of variance with the Bonferroni post hoc test was performed to determine statistical significance. NPY, neuropeptide Y; BIBP, NPY Y1 receptor antagonist BIBP3226.
Effect of D1-like receptor on NPY-mediated proliferation of VSMCs
To investigate the interaction between NPY Y1 and D1-like receptors, VSMCs were incubated with NPY and with the D1-like receptor agonist fenoldopam for 24 h. The results showed that fenoldopam reduced the stimulatory effect of NPY in a concentration-dependent manner (10−11 to 10−7 m; Figure 2a). The effect of fenoldopam was via the D1-like receptor, because the inhibitory effect of fenoldopam on NPY-mediated proliferation was blocked by the D1-like receptor antagonist SCH-23390 (Figure 2b).
Effect of D1-like receptors on neuropeptide Y (NPY)-mediated proliferation of vascular smooth muscle cells (VSMCs). (a) The dose-dependent effect of D1-like receptor agonist fenoldopam on NPY-mediated VSMC proliferation. The VSMC proliferation was determined by [3H]-thymidine incorporation. Results were expressed as fold increase of control (n=6). (b) Effect of a D1-like receptor antagonist SCH-23390 on NPY-mediated VSMC proliferation (n=7). (c and d) The VSMC proliferation was determined by cell number count (c) and MTT analysis (d) (n=5–6). *P<0.05 vs. control, #P<0.05 vs. NPY. Analysis of variance with the Bonferroni post hoc test was performed to determine statistical significance. Fen, fenoldopam; SCH, D1-like receptor antagonist SCH-23390 (10−8 m).
To confirm the results from [3H]-thymidine incorporation study, we also determined the effect of fenoldopam on NPY-mediated VSMC proliferation, as indicated by cell number count and the MTT analysis. The data showed that NPY increased the VSMC number and MTT viability, which were significantly reduced in the presence of the D1-like receptor agonist fenoldopam (10−7 m; Figures 2c and d).
Role of PKA in the inhibitory effect by fenoldopam on VSMC proliferation
To explore the potential mechanisms for the effect of dopamine receptors on NPY-induced VSMC proliferation, we determined the roles of PKA and PKC signalings. Results showed that the PKA inhibitor (PKAI) 14–22 amide (10−6 m), by itself, had no effect on VSMC proliferation, but negated the inhibitory effect of fenoldopam, indicating that PKA was engaged in the inhibitory action of fenoldopam. To further confirm the role of PKA, we used the PKA agonist (PKAa), Sp-cAMP[S] (10−6 m) in this study. It showed that in the presence of Sp-cAMP[S], the stimulatory effect of NPY was reduced, which was blocked by PKA inhibitor 14–22 amide (10−6 m). PKA inhibitor itself did not influence VSMC proliferation (Figure 3). We also detected the role of PKC signaling and Ca2+ channel in the antiproliferative effect of fenoldopam. The results showed that neither PKC inhibitor nor Ca2+ channel blocker influenced the effect of fenoldopam on NPY-induced VSMC proliferation (Figure 3).
Role of PKA and PKC signalings and Ca2+ channel in the inhibitory effect of D1-like receptor on neuropeptide Y (NPY)-mediated vascular smooth muscle cell (VSMC) proliferation. VSMC proliferation was determined by [3H]-thymidine incorporation. Results were expressed as fold increase of control. *P<0.05 vs. control, #P<0.05 vs. NPY (n=6). Analysis of variance with the Bonferroni post hoc test was performed to determine statistical significance. PKAa, PKA agonist Sp-cAMP[S] (10−6 m); PKAI, PKA inhibitor 14–22 amide (10−6 m).
We have previously shown that PKA activation is the mechanism underlying the antiproliferative effects of D3 receptor activation on norepinephrine-stimulated VSMCs.10 We further determined the effects of D3 receptor activation on NPY-stimulated VSMCs. As shown in Supplementary Figure 1, D3 receptor activation with D3 receptor agonist PD128907 blocked NPY-induced VSMC proliferation. The effect of D3 receptor activation was blocked by a PKA inhibitor. These data indicated that the function of D3 receptor activation is additive to the effect of D1-like receptor activation, which is associated with PKA signaling.
Effect of D1-like receptor activation on angioplasty-induced neointima
To further identify the beneficial effect of D1-like receptor activation in vivo, we examined the role of fenoldopam in a rat model of atherosclerosis and neointimal formation according to the description in the previous study.20 As shown in Figure 4, NPY significantly promoted angioplasty-induced neointima, which was blocked by pretreatment with dopamine D1-like receptor activator fenoldopam. D1-like receptor antagonist SCH-23390 abolished the beneficial effect of fenoldopam. With Ki67 staining analysis, our data showed that NPY significantly promoted VSMC proliferation, which was blocked by fenoldopam. SCH-23390 abolished the antiproliferative effect of fenoldopam (Supplementary Figure 2). Thus, these data provided further evidence of the beneficial effect of D1-like receptor activation on atherosclerosis and neointimal formation.
Effect of fenoldopam on angioplasty-induced neointima with neuropeptide Y (NPY) stimulation in a rat model. (a1–a5) Representative images of hematoxylin and eosin staining of rat carotid arteries from different treatment groups. (b) The bar graphs show quantification of the area of the intima-to-media ratio (I/M ratio ). n=5, *P<0.05 vs. injury+vehicle; #P<0.05 vs. injury+NPY; &P<0.05 vs. injury+NPY+Fen. Analysis of variance with the Bonferroni post hoc test was performed to determine statistical significance. Fen, fenoldopam. A full color version of this figure is available at Hypertension Research online.
Discussion
NPY has an important role in the regulation of blood pressure. It is reported that NPY accounts for about 30% of sympathetic nerve-mediated vasoconstriction in resistance vessels.22 NPY potentiates other vasoconstrictor agents, such as norepinephrine, epinephrine and endothelin, to further increase blood pressure. Mice lacking the Y1 receptor have a basal blood pressure, but lack the ability to potentiate norepinephrine-induced vasoconstriction. In addition, NPY has a hypertrophic effect on vascular smooth muscles,23, 24 as confirmed that NPY exerts direct stimulation to mRNA synthesis in VSMCs.2, 25 Furthermore, an NPY locus on chromosome 4 has been found to co-segregate with blood pressure in spontaneously hypertensive rats (SHRs). We also found that compared with Wistar–Kyoto rats, SHRs have higher plasma NPY concentration and higher NPY receptor-binding sites/affinity in VSMCs.25 These evidences suggest that NPY is important in the development and maintenance of hypertension.
Previous studies have shown that dopamine receptors have an important role in the regulation of blood pressure.12, 13 Dopamine D1-like receptor-deficient mouse shows a higher blood pressure as compared with its control counterpart.25 Stimulation of the D1-like receptor induces diuresis and natriuresis,12, 26 leading to vasodilation in arteries preconstricted by other constrictive factors such as norepinephrine. VSMC proliferation has an important role in hypertension pathogenesis27 and vascular remodeling in atherosclerosis.28, 29 In the hypertensive state, the D1-like receptor-mediated natriureis is impaired due to receptor hyperphosphorylation,30 which leads to its uncoupling from the G protein. As a major mitogenic factor, NPY has been shown to stimulate the proliferation of VSMCs.3
Our previous study found that in the presence of the dopamine receptor, the α1-adrenergic receptor-mediated VSMC proliferation is reduced. The present study also found D1-like receptor agonist reduced the NPY-mediated VSMC proliferation, indicating that the dopamine receptor inhibits the VSMC proliferation induced by NPY.
Dopamine D1-like receptor agonists increased PKA activity whereby reducing the MAPK activity in VSMCs, and it blocked the hypertrophic effect mediated by platelet-derived growth factor-BB in VSMCs.31 In our present study, to determine the signal pathway involved in the inhibitory effect of fenoldopam, we observed the effect of different agonists/antagonists, including PKC agonist/antagonist, PKA agonist/antagonist and Ca2+ channel blockers. The results showed that the inhibitory effect of fenoldopam is negated by PKA inhibition. Moreover, the PKA agonist could imitate the effect of fenoldopam, that is, PKA agonist blocks the effect of proliferation mediate by NPY in VSMCs.
The VSMC proliferation is a key process in the pathogenesis of atherosclerosis. VSMC proliferation is also associated with hypertension-related vascular dysfunction, restenosis and pulmonary hypertension.32 NPY, released from the sympathetic nerve terminals as a vaso-neurotransmitter, acts via G protein-coupled receptors to stimulate constriction and VSMC proliferation through interactions with its Y1 receptors.33 NPY release is long-lastingly activated in these pathological states, thereby inducing VSMC proliferation that contributes to the pathogenesis of these diseases. Our findings showed that the activation of the D1-like receptor inhibit the NPY-mediated VSMC proliferation. This may lead to developing new therapeutic strategies for the treatment of cardiovascular diseases related to VSMC proliferation.
In summary, we have demonstrated that the activation of D1-like receptor, via PKA signaling pathway, inhibits the proliferative effect of NPY in VSMCs. This indicates that the D1-like dopamine receptor may be a novel treating target for limiting the pathological progress of cardiovascular diseases such as atherosclerosis.
References
Nishimura J, Bi D, Kanaide H . Dependence of proliferating dedifferentiated vascular smooth muscle contraction on Rho-Rho kinase system. Trends Cardiovasc Med 2006; 16: 124–128.
Nilsson T, Edvinsson L . Neuropeptide Y stimulates DNA synthesis in human vascular smooth muscle cells through neuropeptide Y Y1 receptors. Can J Physiol Pharmacol 2000; 78: 256–259.
Pons J, Kitlinska J, Jacques D, Perreault C, Nader M, Everhart L, Zhang Y, Zukowska Z . Interactions of multiple signaling pathways in neuropeptide Y-mediated bimodal vascular smooth muscle cell growth. Can J Physiol Pharmacol 2008; 86: 438–448.
Li Z, Yu C, Han Y, Ren H, Shi W, Fu C, He D, Huang L, Yang C, Wang X, Zhou L, Asico LD, Zeng C, Jose PA . Inhibitory effect of D1-like and D3 dopamine receptors on norepinephrine-induced proliferation in vascular smooth muscle cells. Am J Physiol Heart Circ Physiol 2008; 294: H2761–H2768.
Rudijanto A . The role of vascular smooth muscle cells on the pathogenesis of atherosclerosis. Acta Med Indones 2007; 39: 86–93.
Selzman CH, Miller SA, Harken AH . Therapeutic implications of inflammation in atherosclerotic cardiovascular disease. Ann Thorac Surg 2001; 71: 2066–2074.
McDermott BJ, Millar BC, Piper HM . Cardiovascular effects of neuropeptide Y: receptor interactions and cellular mechanisms. Cardiovasc Res 1993; 27: 893–905.
Dumont Y, Quirion R . An overview of neuropeptide Y: pharmacology to molecular biology and receptor localization. EXS 2006; 95: 7–33.
Coelho EF, Ferrari MF, Maximino JR, Fior-Chadi DR . Change in the expression of NPY receptor subtypes Y1 and Y2 in central and peripheral neurons related to the control of blood pressure in rats following experimental hypertension. Neuropeptides 2004; 38: 77–82.
Zeng C, Felder RA, Jose PA . A new approach for treatment of hypertension: modifying D1 dopamine receptor function. Cardiovasc Hematol Agents Med Chem 2006; 4: 369–377.
Tayebati SK, Lokhandwala MF, Amenta F . Dopamine and vascular dynamics control: present status and future perspectives. Curr Neurovasc Res 2011; 8: 246–257.
Zeng C, Eisner GM, Felder RA, Jose PA . Dopamine receptor and hypertension. Curr Med Chem Cardiovasc Hematol Agents 2005; 3: 69.
Zeng C, Jose PA . Dopamine receptors: important antihypertensive counterbalance against hypertensive factors. Hypertension 2011; 57: 11–17.
Lu SY, Zhu MZ, Wang DS, Chen SY, Zhang WD, Dong H, Yu J, Guo HT . Inhibition of the proliferation of smooth muscle cells from human coronary bypass vessels by vasonatrin peptide. Physiol Res 2004; 53: 387–393.
Lu SY, Wang DS, Zhu MZ, Zhang QH, Hu YZ, Pei JM . Inhibition of hypoxia-induced proliferation and collagen synthesis by vasonatrin peptide in cultured rat pulmonary artery smooth muscle cells. Life Sci 2005; 77: 28–38.
Nagata D, Takeda R, Sata M, Satonaka H, Suzuki E, Nagano T, Hirata Y . AMP-activated protein kinase inhibits angiotensin II-stimulated vascular smooth muscle cell proliferation. Circulation 2004; 110: 444–451.
Zhang H, Han Y, Tao J, Liu S, Yan C, Li S . Cellular repressor of E1A-stimulated genes regulates vascular endothelial cell migration by the ILK/AKT/mTOR/VEGF (165) signaling pathway. Exp Cell Res 2011; 317: 2904–2913.
Nabzdyk CS, Lancero H, Nguyen KP, Salek S, Conte MS . RNA interference-mediated survivin gene knockdown induces growth arrest and reduced migration of vascular smooth muscle cells. Am J Physiol Heart Circ Physiol 2011; 301: H1841–H1849.
Yu ML, Wang JF, Wang GK, You XH, Zhao XX, Jing Q, Qin YW . Vascular smooth muscle cell proliferation is influenced by let-7d microRNA and its interaction with KRAS. Circ J 2011; 75: 703–709.
Li L, Jönsson-Rylander AC, Abe K, Zukowska Z . Chronic stress induces rapid occlusion of angioplasty-injured rat carotid artery by activating neuropeptide Y and its Y1 receptors. Arterioscler Thromb Vasc Biol 2005; 25: 2075–2080.
Lu J, Ji J, Meng H, Wang D, Jiang B, Liu L, Randell E, Adeli K, Meng QH . The protective effect and underlying mechanism of metformin on neointima formation in fructose-induced insulin resistant rats. Cardiovasc Diabetol 2013; 12: 58.
Han S, Yang CL, Chen X, Naes L, Cox BF, Westfall T . Direct evidence for the role of neuropeptide Y in sympathetic nerve stimulation-induced vasoconstriction. Am J Physiol 1998; 274: H290–H294.
Goldberg Y, Taimor G, Piper HM, Schlüter KD . Intracellular signaling leads to the hypertrophic effect of neuropeptide Y. Am J Physiol 1998; 275: C1207–C1215.
Movafagh S, Hobson JP, Spiegel S, Kleinman HK, Zukowska Z . Neuropeptide Y induces migration, proliferation, and tube formation of endothelial cells bimodally via Y1, Y2, and Y5 receptors. FASEB J 2006; 20: 1924–1926.
Zeng C, Wang X, Liu G, Yang C . Effects of ACE inhibitor and beta-adrenergic blocker on plasma NPY and NPY receptors in aortic vascular smooth muscle cells from SHR and WKY rats. Neuropeptides 2002; 36: 353–361.
Salomone LJ, Howell NL, McGrath HE, Kemp BA, Keller SR, Gildea JJ, Felder RA, Carey RM . Intrarenal dopamine D1-like receptor stimulation induces natriuresis via an angiotensin type-2 receptor mechanism. Hypertension 2007; 49: 155–161.
Du CQ, Yang L, Yang J, Han J, Hu XS, Wu T, Hu SJ . Inhibition of farnesyl pyrophosphate synthase prevents norepinephrine-induced fibrotic responses in vascular smooth muscle cells from spontaneously hypertensive rats. Hypertens Res 2014; 37: 26–34.
Ozasa Y, Akazawa H, Qin Y, Tateno K, Ito K, Kudo-Sakamoto Y, Yano M, Yabumoto C, Naito AT, Oka T, Lee JK, Minamino T, Nagai T, Kobayashi Y, Komuro I . Notch activation mediates angiotensin II-induced vascular remodeling by promoting the proliferation and migration of vascular smooth muscle cells. Hypertens Res 2013; 36: 859–865.
Liu G, Hitomi H, Rahman A, Nakano D, Mori H, Masaki T, Ma H, Iwamoto T, Kobori H, Nishiyama A . High sodium augments angiotensin II-induced vascular smooth muscle cell proliferation through the ERK 1/2-dependent pathway. Hypertens Res 2014; 37: 13–18.
Jose PA, Eisner GM, Felder RA . Dopamine receptor-coupling defect in hypertension. Curr Hypertens Rep 2002; 4: 237–244.
Yasunari K, Kohno M, Hasuma T, Horio T, Kano H, Yokokawa K, Minami M, Yoshikawa J . Dopamine as a novel antimigration and antiproliferative factor of vascular smooth muscle cells through dopamine D1-like receptors. Arterioscler Thromb Vasc Biol 1997; 17: 3164–3173.
Crnkovic S, Egemnazarov B, Jain P, Seay U, Gattinger N, Marsh LM, Bálint Z, Kovacs G, Ghanim B, Klepetko W, Schermuly RT, Weissmann N, Olschewski A, Kwapiszewska G . NPY/Y1 receptor-mediated vasoconstrictory and proliferative effects in pulmonary hypertension. Br J Pharmacol 2014; 171: 3895–3907.
Lagraauw HM, Westra MM, Bot M, Wezel A, van Santbrink PJ, Pasterkamp G, Biessen EA, Kuiper J, Bot I . Vascular neuropeptide Y contributes to atherosclerotic plaque progression and perivascular mast cell activation. Atherosclerosis 2014; 235: 196–203.
Acknowledgements
These studies were supported in part by grants from the National Natural Science Foundation of China (31430043, 31130029) and grants from the National Basic Research Program of China (2012CB517801).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Hypertension Research website
Rights and permissions
About this article
Cite this article
Zhou, Y., Shi, W., Luo, H. et al. Inhibitory effect of D1-like dopamine receptors on neuropeptide Y-induced proliferation in vascular smooth muscle cells. Hypertens Res 38, 807–812 (2015). https://doi.org/10.1038/hr.2015.84
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2015.84
- Springer Nature Singapore Pte Ltd.
Keywords
This article is cited by
-
Different effects of neuropeptide Y on proliferation of vascular smooth muscle cells via regulation of Geminin
Molecular and Cellular Biochemistry (2017)