Abstract
We aimed to evaluate the hypotensive effect and the time to attain the maximal antihypertensive effect (stabilization time) of 8 mg candesartan/6.25 mg hydrochlorothiazide (HCTZ) combination therapy (combination regimen) and therapy with an increased candesartan dose (12 mg; maximum dose regimen) using home blood pressure (BP) measurements. A prospective, multicenter, open-label, randomized, comparative trial was conducted. Essential hypertensive patients who failed to achieve adequate BP control (systolic BP (SBP) ⩽135 mm Hg) on 8 mg candesartan alone were randomized to two groups: the combination regimen (n=103) and the maximum dose regimen (n=103). Home morning SBP reduction at 8 weeks after randomization was 11.4±1.3 mm Hg in the combination regimen and 7.8±1.2 mm Hg in the maximum dose regimen. The combination regimen provided additional reduction of 4.0 mm Hg (95% confidence interval (CI): 0.8–7.2 mm Hg, P=0.01) in home morning SBP over the maximum dose regimen at 8 weeks after randomization. The maximal antihypertensive effect and stabilization time for home SBP were 9.4 mm Hg and 37.1 days (P<0.0001), respectively, with the combination regimen. The maximum dose regimen decreased home SBP with a very gentle slope, and estimated maximal effect and estimated stabilization time were not significant (P>0.2). The rate of achieving target BP (home morning SBP <135 mm Hg) was significantly higher with the combination regimen than with the maximum dose regimen (52.4 vs. 30.1%, P=0.002). In conclusion, changing from 8 mg candesartan to combination therapy was more effective in reducing home SBP and achieving goal BP more rapidly than increasing the candesartan dose.
Similar content being viewed by others
Introduction
Use of an angiotensin II receptor blocker (ARB) is recommended as first-line therapy in Japan because of its high efficacy and tolerability. When blood pressure (BP) control by an ARB is insufficient, combination therapy with low-dose diuretics is also recommended.1 The combination tablet of candesartan/hydrochlorothiazide (HCTZ; ECARD® LD/HD), which is marketed in Japan, is one of the recommended combinations. The renin–angiotensin–aldosterone system is stimulated by HCTZ; HCTZ stimulates renin release and increases plasma renin activity, which attenuates the hypotensive effect of HCTZ, and an ARB potentiates the antihypertensive effect of HCTZ.2 Furthermore, the presence of candesartan can abolish the transient fall in glomerular filtration rate induced by HCTZ.3 In addition, a fixed-dose combination can reduce the risk of non-compliance.4 However, the antihypertensive effects of the candesartan/thiazide diuretic combination and an incremental dose of candesartan alone have not been compared using home BP measurements.
Home BP measurement has been recognized as a useful tool for accurate diagnosis and appropriate treatment of hypertension and is the best method for evaluating the BP-lowering effects of antihypertensive drugs in clinical trials.5, 6 Mori et al. examined using home BP to compare antihypertensive effect between morning administration and evening administration.7 We previously reported that nocturnal BP could predict the risk of cardiovascular diseases better than daytime BP, especially in treated hypertensive patients.8 It has been reported that adding a very low dose of HCTZ (6.25 mg) to an ARB was very effective in lowering nocturnal BP evaluated by ambulatory BP monitoring.9 To evaluate nocturnal BP among antihypertensive regimen, ambulatory BP monitoring is often used.9, 10 Recently, home BP measuring devices that can monitor nocturnal BP during sleep have been developed.11, 12 However, there is no information about the effect of a low-dose HCTZ on nocturnal BP evaluated by home BP measurements.
Home BP measurements can provide daily serial measurements. Mashima et al. first reported using exponential decay function analysis to determine the velocity of the hypotensive effect of a drug using home BP measurements.13 In modern antihypertensive treatment, prompt achievement of goal BP is expected.14 On the other hand, it is assumed that excessively rapid BP lowering induced by antihypertensive drug therapy is not desirable because of adverse effects,15 especially in the elderly.16 Therefore, in the evaluation of the effect of antihypertensive drugs, the velocity, as well as the amplitude, of the antihypertensive effects must be taken into account. The aims of this study were (1) to evaluate the antihypertensive effects of two regimens using home BP measurements in the morning, as well as at night during sleep, and (2) to investigate the time to achieve the maximal antihypertensive effects of the two regimens.
Methods
Patients
Newly diagnosed, untreated hypertensive patients or those who were on monotherapy with any antihypertensive drug alone, ranging in age from 20 to 80 years, were screened. Patients with secondary hypertension, cardiac failure, past history of major cardiovascular events or intervention within 6 months before the screening, severe hepatic or renal diseases, uncontrolled arrhythmia, intolerance to ARBs or thiazide diuretics, pregnant or breastfeeding women or those expecting to become pregnant, or patients with other reasons rendering them ineligible were not included.
BP measurement
Clinic BP and pulse rate (PR) were measured twice on each outpatient clinic visit (−4 and 0 weeks of the run-in period and 4 and 8 weeks of the randomized period) using a mercury sphygmomanometer or calibrated automatic device based on the cuff-oscillometric principle.
Home BP was measured using a HEM-7080IC automatic device (Omron Health Care, Kyoto, Japan) based on the cuff-oscillometric principle, which measures systolic BP (SBP), diastolic BP (DBP) and PR, and stores the date and time of measurement, as well as the SBP, DBP and PR, in the integrated-circuit memory. All devices used in this study were validated11 and satisfied the criteria of the Association for the Advancement of Medical Instrumentation.17
Physicians and nurses taught subjects how to measure their morning, evening and nocturnal BP during sleep. The subjects were asked to measure their home BP once every morning within 1 h after awaking, after micturition, before breakfast, before taking their morning dose, in the sitting position and after more than 1 min of rest. Evening home BP was obtained once every evening just before going to bed. These measurement conditions are based on the Japanese Society of Hypertension Guideline for Self-monitoring of BP at home.6 To measure nocturnal BP during sleep, the subjects were asked to remove tight or restrictive clothing, apply the cuff of the HEM-7080IC device to the upper arm at bedside, and keep their arm covered by the cuff during sleep. The device then automatically measured BP twice at the programmed clock time (0200 h) and 4 h after going to bed and stored the reading in the memory chip in the device. The subjects were also asked to record the quality of sleep on a diary card after waking. On the diary card, subjects were asked to select one of the following options on the quality of sleep during each nocturnal BP measurement: ‘no sleep disturbance and no awareness of measurement’ or ‘mild to serious sleep disturbance with an awareness of measurement.’ The former was defined as a ‘sleeping’ state and the latter as a ‘waking’ state. The subjects were asked to take their own nocturnal BP for 3 days before the next clinic visit. All values from each nocturnal BP measurement were used in the analysis. It was further defined that all of the states that were ‘sleeping’ were the sleeping state, and any one of the states that was ‘waking’ was the waking state.
Randomization
Patients were seen every 4 weeks for 8 weeks, and home BP data from the outpatient clinic microcomputer were transmitted to the host computer every 4 weeks.
The Japan home vs. office BP measurement evaluation with candesartan and restricted dose of diuretic (J-HOME-CARD) study is a prospective, multicenter, open-label, randomized, comparative trial. When home SBP averaged for 5 days in the morning just before randomization was greater than 135 mm Hg, the host computer randomized to the two drugs, and the decisions were transmitted to the outpatient clinic microcomputer over the Internet.
Intervention
Four weeks after treatment with 8 mg candesartan monotherapy (run-in period), those whose home SBP in the morning was greater than 135 mm Hg were randomized to receive a once-daily dose of 8 mg candesartan/6.25 mg HCTZ single pill combination (the combination regimen) or 12 mg candesartan, which is the maximum dose clinically permitted in Japan (the maximum dose regimen) for 8 weeks.
Outcomes
The primary outcome of this study was to determine the difference in home morning SBP reduction between the two groups during 8-week treatment. Time-dependent changes in the antihypertensive effects and nocturnal BP reduction were compared between the two groups during 8-week treatment as secondary outcomes.
Sample size
Based on a previous report, the reduction of SBP with the medium-dose ARB and HCTZ combination was assumed to be 12.8 mm Hg. The average SBP reduction with maximal-dose of 5 ARB clinically permitted in Japan was assumed to be 7.6±1.2 mm Hg on the basis of each ARB’s medium-dose monotherapy.18 Therefore, a treatment difference of at least a 5.2 mm Hg mean SBP reduction between combination and maximal-dose ARB treatment was expected. The target sample size was 168 patients (84 patients per treatment group) with equal randomization between the two regimens. A dropout rate of 10% was assumed, so that a total of approximately 184 patients were randomized. This sample size provided 80% power to detect a significant difference at a two-sided significance level of 0.05 in the setting described.
Data collection and statistical analysis
Patient information was collected using a questionnaire administered by the attending physicians. The estimated glomerular filtration rate using a modified three-variable equation based on creatinine clearance for Japanese individuals as follows: estimated glomerular filtration rate (ml min−1 per 1.73 m2)=194 × (serum creatinine in enzymatic method)−1.094 × age−0.287 ( × 0.739, if female).19 All analyses were conducted according to the intention-to-treat principle. The last observation carried forward method was implemented to account for missing data. Changes from baseline values (average for 5 days) to 4-week (average for 5 days) and 8-week (average for 5 days) values were analyzed by a two-way analysis of covariance model with treatment and region as factors and baseline home BP as a covariate. The variables for the baseline characteristics of the study patients and laboratory data were compared using Fisher’s exact test, the χ2 test, the t-test or the Wilcoxon rank-sum test.
Daily serial home BP changes were also evaluated by a linear mixed model. For these analyses, a two-tailed P value less than 0.0009 (0.05/56) was considered significant after the Bonferroni correction was applied, as 56 home BPs were tested.
As the screened patients were heterogeneous from the perspective of preceding antihypertensive treatment, the data of the subsequent 4-week run-in period (treatment with 8 mg candesartan alone) were not analyzed, but the morning home BP averaged for the last 5 days of the 4-week run-in period was used as a baseline value.
Using an exponential decay function analysis, the maximal antihypertensive effect (A) and the stabilization time (the time when the maximal effect was observed) were evaluated using a nonlinear mixed model. The exponential function is shown as follows (Figure 1):
Fitting to the exponential decay function curve. The exponential function is expressed as Y=Ae−kt+C. Y is home systolic blood pressure (BP), A is the extent of the home systolic BP-lowering effect, k is the time constant for home systolic BP decay (per day), t is the time (days) and C is the final stabilized systolic BP. Stabilization time (3.0/k) is the time to achieve 95% of the maximal antihypertensive effect observed.

where Y is the home SBP value, A is the extent of the home SBP lowering effect, k is the time constant for home SBP decay (day−1), t is the time (days) and C is the final stabilized home SBP.
As an index of the velocity of home SBP reduction, stabilization time, which demonstrates the time to achieve a stable treatment effect, was calculated. Stabilization time was defined as the time to achieve 95% of the maximal effect (A). The value A+C−95A/100 was substituted for Y, and the exponential decay function equation was solved as follows:







resulting in a stabilization time of 3.0/k.
The data were analyzed using the SAS package (version 9.3 SAS Institute, Cary, NC, USA).
This study was approved by the Institutional Review Board of Kojinkai Central Hospital and was registered with the UMIN Clinical Trial Registry (number UMIN 000003356). All participants gave their written, informed consent.
Results
Patient selection
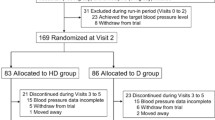
From March 2010 to April 2012, 258 eligible patients were recruited. Of those, 52 were not included in the randomization because: home SBP in the morning was less than 135 mm Hg with 8 mg candesartan alone (n=27); of not visiting hospital after registration (n=6); of changing hospital after registration (n=2); of violation of study protocol (n=9); of no hypotensive effect (n=2); of dropout for unknown reasons (n=4) or the patient chose to withdraw from the study (n=2; Figure 2).
The remaining 206 patients met the study eligibility criteria and were randomized to the two regimens. Of these, 103 patients were allocated to the maximum dose regimen and 103 patients were allocated to the combination regimen. Of the 103 patients allocated to the maximum dose regimen and the 103 patients allocated to the combination regimen, two (one due to withdrawing from the trial, one due to an unknown reason) and five (one due to withdrawing from the trial, two due to adverse events (one for elevated serum uric acid level and one for vertigo and heaviness of the head) and three due to unknown reasons) patients, respectively, discontinued study treatment during the trial (Figure 2).
Characteristics and BP
The baseline characteristics of the patients were similar between the regimens except for home evening BP (Table 1). Home BP reduction in the morning at 4 weeks after randomization was 8.9±1.1 mm Hg for SBP and 4.0±0.7 mm Hg for DBP in the combination group and 5.1±1.2 mm Hg for SBP and 2.1±0.7 mm Hg for DBP in the maximum dose group. The home BP reduction at 8 weeks after randomization was 11.4±1.3 mm Hg for SBP and 5.3±0.7 mm Hg for DBP in the combination group and 7.8±1.2 mm Hg for SBP and 3.6±0.6 mm Hg for DBP in the maximum dose group. Analyses using analysis of covariance adjusted by baseline home BP values as covariates showed that the combination regimen provided additional reductions of 4.0 mm Hg (95% CI: 0.8–7.2 mm Hg) for SBP and 1.8 mm Hg (95% CI: −0.02 to 3.7 mm Hg) for DBP over the maximum dose regimen at 4 weeks after randomization, whereas at 8 weeks after randomization, these reductions were 4.0 mm Hg (95% CI: 0.9–7.2 mm Hg) and 1.7 mm Hg (95% CI: −0.05 to 3.4 mm Hg), respectively (Figure 3).
Home morning SBP was well controlled (<135 mm Hg) in 52.4% of the combination group and 30.1% of the maximum dose group at 8 weeks after randomization (P=0.002).
For clinic and home evening BPs, both regimens caused a significant reduction in SBP and DBP at 8 weeks after randomization, and significant additional reduction at 8 weeks after randomization was observed in the combination regimen compared with the maximum dose regimen, except for home evening DBP (see Supplementary Figures 1 and 2, which illustrate the hypotensive effects on home evening and conventional BP). These two regimens did not affect either clinic or home PR at 8 weeks after randomization (data not shown).
For home nocturnal BP, the analysis included the 53 patients who measured nocturnal BP at baseline, 4 and 8 weeks after randomization. At 4 and 8 weeks, both regimens significantly decreased nocturnal SBP and DBP. There were no significant additional reductions in nocturnal SBP and DBP at 8 weeks regardless of regimens (Figure 4). There were no significant differences in the rate of the sleeping state between the two regimens. The reduction in home nocturnal BP at 8 weeks after randomization was not different between the two regimen groups (see Supplementary Table 1, which demonstrates the nocturnal BP reduction and the status during nocturnal BP measurement).
Laboratory data
Laboratory data at baseline and at 8 weeks after randomization are shown in Supplementary Table 2. After treatment for 8 weeks, red blood cell and hemoglobin levels were significantly decreased in both regimen groups. Only in the combination regimen group, uric acid, potassium and chloride levels at 8 weeks after randomization were significantly lower than at baseline. Red blood cell, hemoglobin, uric acid, potassium and chloride levels were within normal limits after treatment.
Stabilization time
Using a nonlinear mixed model, in the combination group, the maximal effect (A) and stabilization time of home morning SBP were 9.4±5.3 mm Hg and 37.1±62.8 days, respectively (all P<0.0001). As shown in Figure 5, the maximum dose regimen decreased home SBP with a very gentle slope. With the maximum dose regimen, the estimated maximal effect was 15.6±94.3 mm Hg, and the stabilization time was 279.9±2317.5 days, but these values were not significant (all P>0.2), which suggested that the data were not well matched to the model. The maximum effect and stabilization time after 8 weeks of treatment in the maximum dose regimen are estimated values and do not depend on the actual measurement value of home BP. Thus, the daily SBP changes in the morning after randomization were also calculated using a linear mixed model, and the results are also shown in Figure 5. At 8 weeks after randomization, the difference in the depressor effect between the two regimens for home morning SBP was 4.4 mm Hg (P=0.098). The differences in the depressor effect for home morning SBP calculated using a linear mixed model were as great as the additional reduction between the two regimens, as shown in Figure 3 (4.0 mm Hg).
The maximum depressor effect and stabilization time of the two regimens on home blood pressure in the morning; the combination regimen where hydrochlorothiazide (6.25 mg) was added to 8 mg candesartan (n=103, open circles) and the maximum dose regimen where candesartan increased from 8 to 12 mg (n=103, closed circles).
Discussion
The present study clearly demonstrated that 8 mg candesartan/6.25 mg HCTZ combination therapy had a more potent and faster antihypertensive effect, a shorter duration to achieve a stable treatment effect and a higher rate of achieving target BP than 12 mg candesartan therapy for patients who did not attain target BP value with 8 mg candesartan therapy based on home BP measurements. In our present study, all of the patients were on 8 mg of candesartan monotherapy for 4 weeks before randomization. Thus, we could reveal the pure effect of 8 mg candesartan/6.25 mg HCTZ combination and 12 mg candesartan therapies on the basis of 8 mg candesartan monotherapy.
It has been reported that HCTZ combined with candesartan was effective in hypertensive patients,9 but this study was based on clinic and ambulatory BP measurements, and there was no comparison to the effect of candesartan alone. In the present study, morning home BP at 8 weeks after randomization was significantly lower than baseline in both regimens. The BP reduction was larger in the combination group than in the maximum dose group. The present results suggest that, in patients whose morning home BP is not well controlled on 8 mg of candesartan alone, the combination therapy is more effective for lowering home BP than increasing the candesartan dose.
For nocturnal home BP at 8 weeks after randomization, a significant difference in BP reduction from baseline was observed with both regimens. However, only 25% of participants measured nocturnal BP, and there were no significant differences in the hypotensive effects for nocturnal BP at 8 weeks between the two groups. It has been reported that adding a very low dose of HCTZ (6.25 mg) to an ARB was very effective in lowering ABP, particularly nocturnal BP.9 The nocturnal BP fall was significantly enhanced by diuretic therapy.20 However, in the present study, superiority of the combination of ARB with low-dose HCTZ on the effect on nocturnal BP was not observed. It is possible that an insufficient and unequal number of subjects in each regimen may have affected the results. The completed participants who were available to measure nocturnal BP were only about 25% of total subjects. This small number of subjects may limit the statistical power of the analysis. Although, in this study, we asked participants to measure nocturnal BP, we did not force them to measure it. The participants might be reluctant to measure nocturnal BP, because they might worry about disrupting their sleep by measuring it. There were no significant differences in the baseline characteristics and morning home BP between the participants who had full data (0, 4 and 8 weeks) of nocturnal BP were available and who were excluded from the analysis about nocturnal BP (data not shown). Additional studies are needed to further evaluate the effect of HCTZ on nocturnal home BP.
We previously reported the stabilization time of other ARB monotherapy (12–15 days)21, 22, 23, 24 and 50 mg losartan/12.5 mg HCTZ combination therapy (7.3 days).18 Although there are differences in patients’ characteristics, the stabilization time of combination therapy in the present study (8 mg candesartan/6.25 mg HCTZ, 38 days) was relatively long compared with the previous report.18 The half dose of HCTZ compared with the previous study might have contributed to the present results. These results indicate the stabilization time would differ according to the ARB subtypes or the dose of additional HCTZ. With incremental therapy of candesartan from 8 to 12 mg, the stabilization time (280 days) seems to be very long compared with a previous report.18 With incremental therapy of losartan from 50 to 100 mg, the maximal effect and stabilization time of home morning SBP were 7.6 mm Hg and 68.7 days, respectively.18 The stabilization time was calculated using a nonlinear mixed model, and these values would be estimated values. Thus, the stabilization time does not necessarily reveal the real time to attain the maximum effect, but it may merely indicate a slow attainment of the maximum effect of the ARB when the dose was increased.
In the present study, stabilization time was shorter in the combination group than in the maximum dose group. It has been reported that combination therapy using different classes of drugs is approximately five times more effective in lowering BP than increasing the dose of one drug.25 Gene polymorphisms that are responsible for salt-sensitive hypertension are significantly more frequent in Japanese than in Caucasians,26 and it is well known that Japanese hypertensive patients consume higher amounts of salt. In salt-sensitive hypertension, diuretic therapy is effective for lowering BP. ARBs have been shown to increase salt sensitivity. Therefore, a very low-dose HCTZ added to an ARB might be more effective for lowering BP than increasing the ARB dose, especially in Japanese hypertensive patients.
This study had several limitations. First, the antihypertensive effects of both regimens, 12 mg candesartan and 8 mg candesartan/6.25 mg HCTZ, were compared following treatment with 8 mg candesartan alone. In the present study, the dose of the ARB was increased or ARB monotherapy was changed to combination therapy. If untreated patients were treated from the beginning with the regimen used in the present study, the antihypertensive effect might have been different. Further study is needed to characterize the maximal effect and the velocity of the antihypertensive effect of 12 mg candesartan and 8 mg candesartan/6.25 mg HCTZ monotherapy. Second, the effects of the two regimens were examined for only 8 weeks after randomization. The estimated stabilization time of the maximum dose regimen was very long, whereas the stabilization time in the maximum dose regimen group was not significant. Further studies with a longer treatment period are needed to obtain the maximal effect and stabilization time for the maximum dose regimen.
In conclusion, in hypertensive patients whose home BP was not controlled by 8 mg of candesartan, combination therapy with 8 mg candesartan/6.25 mg HCTZ lowered home BP more effectively and achieved goal BP more rapidly than the maximum dose regimen in which candesartan was increased from 8 to 12 mg.
References
Ogihara T, Kikuchi K, Matsuoka H, Fujita T, Higaki J, Horiuchi M, Imai Y, Imaizumi T, Ito S, Iwao H, Kario K, Kawano Y, Kim-Mitsuyama S, Kimura G, Matsubara H, Matsuura H, Naruse M, Saito I, Shimada K, Shimamoto K, Suzuki H, Takishita S, Tanahashi N, Tsuchihashi T, Uchiyama M, Ueda S, Ueshima H, Umemura S, Ishimitsu T, Rakugi H, on behalf of The Japanese Society of Hypertension Committee. The Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2009). Hypertens Res 2009; 32: 3–107.
Lijnen P, Fagard R, Staessen J, Amery A . Effect of chronic diuretic treatment on the plasma renin-angiotensin-aldosterone system in essential hypertension. Br J Clin Pharmacol 1981; 12: 387–392.
Knauf H, Bailey MA, Hasenfuss G, Mutschler E . The influence of cardiovascular and antiinflammatory drugs on thiazide-induced hemodynamic and saluretic effects. Eur J Clin Pharmacol 2006; 62: 885–892.
Bangalore S, Kamalakkannan G, Parkar S, Messerli FH . Fixed-dose combinations improve medication compliance: a meta-analysis. Am J Med 2007; 120: 713–719.
Imai Y, Obara T, Asamaya K, Ohkubo T . The reason why home blood pressure measurements are preferred over clinic or ambulatory blood pressure in Japan. Hypertens Res 2013; 36: 661–672.
Imai Y, Otsuka K, Kawano Y, Shimada K, Hayashi H, Tochikubo O, Miyakawa M, Fukiyama K, Japanese Society of H. Japanese society of hypertension (JSH) guidelines for self-monitoring of blood pressure at home. Hypertens Res 2003; 26: 771–782.
Mori H, Yamamoto H, Ukai H, Yuasa S, Nakajima K, Mikawa T, Niizuma M, Hirao K, Umemura S, COMPATIBLE Study Group. Comparison of effects of angiotensin II receptor blocker on morning home blood pressure and cardiorenal protection between morning administration and evening administration in hypertensive patients: the COMPATIBLE study. Hypertens Res 2013; 36: 202–207.
Boggia J, Li Y, Thijs L, Hansen TW, Kikuya M, Bjorklund-Bodegard K, Richart T, Ohkubo T, Kuznetsova T, Torp-Pedersen C, Lind L, Ibsen H, Imai Y, Wang J, Sandoya E, O'Brien E, Staessen JA, International Database on Ambulatory blood pressure monitoring in relation to Cardiovascular Outcomes investigators (IDACO). Prognostic accuracy of day versus night ambulatory blood pressure: a cohort study. Lancet 2007; 370: 1219–1229.
Eguchi K, Hoshide S, Kabutoya T, Shimada K, Kario K . Is very low dose hydrochlorothiazide combined with candesartan effective in uncontrolled hypertensive patients? Blood Press Monit 2010; 15: 308–311.
Parati G, Bilo G, Redon J., Committee SS. The effects of telmisartan alone or with hydrochlorothiazide on morning and 24- h ambulatory BP control: results from a practice-based study (SURGE 2). Hypertens Res 2013; 36: 322–327.
Chonan K, Kikuya M, Araki T, Fujiwara T, Suzuki M, Michimata M, Hashimoto J, Ohkubo T, Hozawa A, Yamamoto N, Miyawaki Y, Matsubara M, Imai Y . Device for the self-measurement of blood pressure that can monitor blood pressure during sleep. Blood Press Monit 2001; 6: 203–205.
Hosohata K, Kikuya M, Ohkubo T, Metoki H, Asayama K, Inoue R, Obara T, Hashimoto J, Totsune K, Hoshi H, Satoh H, Imai Y . Reproducibility of nocturnal blood pressure assessed by self-measurement of blood pressure at home. Hypertens Res 2007; 30: 707–712.
Mashima K, Nakatsu T, Murakami T, Kusachi S, Tominaga Y, Yamane S, Uesugi T, Mayumi E, Mitsuda T, Tsuji T . Exponential-exponential cosine fitting of blood pressure decay induced by a long-acting calcium blocker, amlodipine, using home blood pressure measurement. Clin Exp Hypertens 2003; 25: 145–154.
Julius S, Kjeldsen SE, Weber M, Brunner HR, Ekman S, Hansson L, Hua T, Laragh J, McInnes GT, Mitchell L, Plat F, Schork A, Smith B, Zanchetti A, VALUE trial group. Outcomes in hypertensive patients at high cardiovascular risk treated with regimens based on valsartan or amlodipine: the VALUE randomised trial. Lancet 2004; 363: 2022–2031.
Furberg CD, Psaty BM, Meyer JV . Nifedipine. Dose-related increase in mortality in patients with coronary heart disease. Circulation 1995; 92: 1326–1331.
Jackson G, Pierscianowski TA, Mahon W, Condon J . Inappropriate antihypertensive therapy in the elderly. Lancet 1976; 2: 1317–1318.
Association for the Advancement of Medical Instrumentation: American National Standards for Electronic or Automated Sphygmomanometers (ANSI/AAMI SP 10–1987). Washington DC, Association for the Advancement of Medical Instrumentation, 1987.
Metoki H, Ohkubo T, Kikuya M, Asayama K, Inoue R, Obara T, Hirose T, Sato M, Hashimoto T, Imai Y, group JH-AS. The velocity of antihypertensive effect of losartan/hydrochlorothiazide and angiotensin II receptor blocker. J Hypertens 2012; 30: 1478–1486.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, Yamagata K, Tomino Y, Yokoyama H, Hishida A . Collaborators developing the Japanese equation for estimated GFR. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis 2009; 53: 982–992.
Uzu T, Harada T, Namba T, Yamamoto R, Takahara K, Yamauchi A, Kimura G . Thiazide diuretics enhance nocturnal blood pressure fall and reduce proteinuria in immunoglobulin A nephropathy treated with angiotensin II modulators. J Hypertens 2005; 23: 861–865.
Obara T, Yasu M, Hanazawa T, Ogasawara K, Katada S, Metoki H, Asayama K, Inoue R, Kikuya M, Ohkubo T, Mano N, Imai Y . Rapidity of home blood pressure reduction by angiotensin II receptor blocker candesartan. Diagnosis Treatment 2010; 98: 1039–1045.
Obara T, Yasu M, Hanazawa T, Ogasawara K, Katada S, Metoki H, Asayama K, Inoue R, Kikuya M, Ohkubo T, Mano N, Imai Y . Rapidity of home blood pressure reduction by angiotensin II receptor blocker olmesartan. Ther Res 2010; 17: 569–575.
Yasu M, Obara T, Hanazawa T, Ogasawara K, Katada S, Metoki H, Asayama K, Inoue R, Kikuya M, Ohkubo T, Imai Y . Rapidity of home blood pressure reduction by angiotensin II receptor blocker telmisartan. J Blood Press 2010; 17: 445–449.
Yasu M, Obara T, Hanazawa T, Ogasawara K, Katada S, Metoki H, Asayama K, Inoue R, Kikuya M, Ohkubo T, Mano N, Imai Y . Rapidity of home blood pressure reduction by angiotensin II receptor blocker losartan. J Blood Press 2010; 17: 145–150.
Wald DS, Law M, Morris JK, Bestwick JP, Wald NJ . Combination therapy versus monotherapy in reducing blood pressure: meta-analysis on 11,000 participants from 42 trials. Am J Med 2009; 122: 290–300.
Katsuya T, Ishikawa K, Sugimoto K, Rakugi H, Ogihara T . Salt sensitivity of Japanese from the viewpoint of gene polymorphism. Hypertens Res 2003; 26: 521–525.
Acknowledgements
The study was designed, conducted and interpreted by the investigators, independently of the sponsors. This study was conducted by the J-HOME-CARD study group. This work was supported by Nouvelle Place, Japan, with an unrestricted grant from the Miyagi Kidney Foundation, which is partly funded by Takeda Pharmaceutical; Grants for Scientific Research (23249036, 23390171, 24390084, 24591060, 24790654, 25253059, 25461205, 25461083, 25860156 and 26860093) from the Ministry of Education, Culture, Sports, Science and Technology, Japan; Grant-in-Aid for Japan Society for the Promotion of Science (JSPS) fellows (25.9328 and 25.7756); a Health Labor Sciences Research Grant (H23-Junkankitou [Seishuu]-Ippan-005) from the Ministry of Health, Labor and Welfare, Japan.
The J-HOME-CARD Study Group
Principal investigator: Yutaka Imai.
Coordinating and data management members: Hirohito Metoki, Takayoshi Ohkubo, Ryusuke Inoue, Taku Obara, Kei Asayama, Masahiro Kikuya, Kazuhito Totsune.
Participating practitioners: Makoto Sekino (Sendai), Hirohito Metoki (Iwanuma), Yasuhiko Kawamorita (Shiwa), Youkou Emura (Morioka), Kazuteru Ohira (Hanamaki), Takako Sugawara (Sakata), Atsuhiro Kanno (Iwanuma), Nanako Hiwatari (Tagajo), Shuichi Sasaki (Tomiya), Tetsuko Watanabe (Tomiya), Shiro Kyogoku (Sendai), Daisuke Nakayama (Matsushima), Kiyoshi Morita (Kesennuma), Koutarou Nakamura (Fukushima), Naoto Yagi (Toda), Takao Noshiro (Sano), Masaaki Miyakawa (Yokohama), Hisao Mori (Yokohama), Hareaki Yamamoto (Sagamihara), Soichiro Sato (Sagamihara), Keiichi Chin (Sagamihara), Hiroyuki Takase (Hamamatsu), Katsumi Yoshida (Niihama), Yuji Takemoto (Izumiotsu), Tomohiro Katsuya (Amagasaki), Taisuke Iwaoka (Kumamoto).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Hypertension Research website
Rights and permissions
About this article
Cite this article
Hosaka, M., Metoki, H., Satoh, M. et al. Randomized trial comparing the velocities of the antihypertensive effects on home blood pressure of candesartan and candesartan with hydrochlorothiazide. Hypertens Res 38, 701–707 (2015). https://doi.org/10.1038/hr.2015.64
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2015.64
- Springer Nature Singapore Pte Ltd.
Keywords
This article is cited by
-
The impact of clinical inertia on uncontrolled blood pressure in treated hypertension: real-world, longitudinal data from Japan
Hypertension Research (2023)
-
Antihypertensive medication and risk of kidney stones: a Canadian wake-up call
Hypertension Research (2017)
-
The effects of increasing calcium channel blocker dose vs. adding a diuretic to treatment regimens for patients with uncontrolled hypertension
Hypertension Research (2017)