Abstract
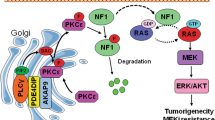
Sur8, a scaffold protein of the Ras pathway, interacts with Ras and Raf and modulates the Ras-extracellular signal-regulated kinase (ERK) pathway. Here we show that Sur8 is overexpressed in established human colorectal cancer (CRC) cell lines and CRC patient tissues. Moreover, Sur8 expression is increased during liver metastasis in CRC patients. Sur8 knockdown decreases ERK and Akt activities in CRC cell lines, regardless of their K-Ras, B-Raf or PI3K mutation status. Overexpression or knockdown of Sur8 increases or decreases, respectively, the proliferation or transformation of CRC cell lines. Sur8 knockdown attenuates the migration and invasion of HCT116 CRC cells. Subcutaneous or orthotopic injection of HCT116 cells harboring a doxycycline (Dox)-mediated Sur8 knockdown system in nude mice resulted in decreased tumorigenic potential and inhibited the liver metastatic potential of HCT116 cells. Taken together, our data support the role of Sur8 as a promoter of tumorigenesis and liver metastasis in CRC through its modulation of the Ras-ERK and PI3K-Akt signaling pathways.
Similar content being viewed by others
Introduction
Colorectal cancer (CRC) is the third most common cancer worldwide and the second leading cause of cancer-related deaths in developed countries.1 Oncogenic activation of the epidermal growth factor receptor-Ras pathway has a major role in the progression and metastasis of CRC.2, 3 The activating mutations that lock K-Ras into a GTP-bound active form occur at early and intermediate stages of human colorectal tumorigenesis at a frequency of 32–57%.4, 5 In addition to K-Ras mutations, activating mutations in B-Raf and PI3K (the gene encoding the p110α subunit of phosphoinositide 3-kinase (PI3K)) are found in 10–15% and 10–20% of colorectal tumors, respectively.3, 6
Approximately 40–50% of CRC patients develop liver metastases, and 80–90% of these metastases are unresectable, which contributes to a 5-year patient survival rate of less than 10%.7, 8 Common sites of metastasis in patients with CRC include the liver, peritoneum and lungs. Thus, identification of new targets involved in colorectal tumorigenesis and metastasis is a critical step toward the development of novel therapeutics that can significantly increase the survival rate of CRC patients.
Oncogenic Ras is involved in tumorigenesis and metastasis via the activation of both the extracellular signal-regulated kinase (ERK) and PI3K signaling pathways.9, 10, 11, 12 Regulation of the pathways downstream of Ras is coordinated by scaffold proteins such as KSR and Sur8 (also known as Shoc2).13 Sur8 was initially identified in a screening test for molecules that regulate Ras-mediated multivulva production in C. elegans as a Ras-binding protein with leucine rich repeats (LRR).14 The human homolog of Sur8 was subsequently characterized and implicated in fibroblast growth factor receptor signaling.15 Previous studies showed that Sur8 positively regulates Ras-ERK signaling either by forming a complex with Ras and Raf16 or by dephosphorylating the Ser-259 inhibitory site of Raf when Sur8 is in a complex with the catalytic subunit of protein phosphatase 1 (PP1c) and M-Ras.17, 18 To date, investigations of Sur8 have mainly focused on the pathogenesis of Noonan-like syndrome,19, 20, 21 and its role in cancer is poorly understood despite its identification as a Ras/Raf scaffold protein. In a recent study, we found that Sur8 interacts with the p110α subunit of PI3K as well as with Ras and Raf and that it positively regulates not only the Ras-ERK signaling pathway but also the PI3K-Akt signaling pathway, suggesting a role for Sur8 in cell motility and tumor metastasis.22
In this study, we show, for the first time, that the Sur8 protein is significantly overexpressed in tissues obtained from human CRC patients as well as in established CRC cell lines. We also investigated the involvement of Sur8 in colorectal tumorigenesis and metastasis. Using doxycycline (Dox)-mediated Sur8 knockdown and overexpression lentiviral systems, we found that Sur8 modulates both of the ERK and PI3K-Akt signaling pathways, promoting growth, transformation, migration, and invasion of CRC cells. Using Dox-mediated Sur8 knockdown, we further showed the involvement of Sur8 in colorectal tumorigenesis and metastasis in mouse models. Overall, our study has identified a novel role for the scaffold protein Sur8 in regulating colorectal tumor growth and liver metastasis and has identified Sur8 as a new therapeutic target in CRC treatment.
Materials and methods
Cell culture and transfection
Human embryonic kidney 293 T (HEK293T) cells, human normal colon cells (CCD18Co), and human CRC cells (Colo205, WiDr, HT29, RKO, Caco2, LoVo, SW480, DLD-1, HCT15 and HCT116) were obtained from the American Type Culture Collection (ATCC; Manassas, VA). DLD-1, Colo205, HT29, RKO, Caco2, HCT15 and SW480 cells were maintained in RPMI 1640 medium (Gibco BRL, Carlsbad, CA, USA). HEK293T, LoVo, and WiDr cells were maintained in Dulbecco’s modified Eagle’s medium (Gibco BRL). HCT116 cells were maintained in McCoy's 5A medium (Gibco BRL). CCD18Co cells were grown in Dulbecco’s modified Eagle’s medium supplemented with 20% fetal bovine serum (Gibco BRL) and 1 × non-essential amino acids (Sigma-Aldrich, St Louis, MO, USA). All other media used were supplemented with 10% fetal bovine serum, 100 units per ml penicillin, and 100 μg ml−1 streptomycin (Gibco BRL). Cells were transfected using Lipofectamine transfection reagent (Invitrogen, Carlsbad, CA, USA) following the manufacturer’s instructions.
Patient tissues and tissue microarray array
Human normal and tumor colon samples used for immunoblotting were obtained from the National Cancer Hospital, Seoul, Korea. The use of human samples was approved by the Institutional Review Board of the National Cancer Hospital. Tissue microarray array (Catalog #BC05002) consisting of human normal colon and tumor samples was purchased from US Biomax. Paraffin-embedded CRC tissues from different tumor stages were obtained from the Severance Hospital, Yonsei University (Seoul, Korea) as previously described.12
Plasmids, lentivirus production and establishment of Sur8 knockdown or overexpression cell lines
The GST-PAK-CD (GST-PAK-CRIB-Domain) plasmid was provided by Dr John G. Collard, The Netherlands Cancer Institute, The Netherlands.23 For the Dox-mediated knockdown of Sur8, the lentiviral shRNA construct inpLKO.1 (Catalog #21915, Addgene, Cambridge, MA, USA) was used, and the following plasmids with respective target sequences were constructed: InshCon (5′-CCTAAGGTTAAGTCGCCCTCGCTCGAGCGAGGGCGACTTAACCTTAGG-3′), InshSur8#1 (5′-CCGGGCTGCGGATGCTTGATTTACTCGAGTAAATCAAGCATCCGCAGCTTTTTG-3′) and InshSur8#2 (5′-CCGGAACCTTGACTTGCAGCACAATCTCGAGATTGTGCTGCAAGTCAAGGTTTTTTTG-3′). For the production of the Dox-mediated Con (InConOE) or Sur8 (InSur8OE)-overexpressing lentiviral plasmids, human Sur8 complementary DNA (cDNA) was amplified by reverse transcription (RT)-PCR using F-primer 5′-GCGCCGGCCGGATCCATGAGTAGTTTAGGAAAA-3′ and R-primer 5′-ACCACACTGGGATCCTCAGACCATGGCACGATATGG-3′ and was introduced into the pLVX-Tight-Puro vector (Catalog #632162, Clontech, Palo Alto, CA, USA). For virus production, HEK293T cells were transfected with lentiviral DNA constructs combined with pMD2G and pAX2G at a ratio of 2:1:1, respectively. Immunoblotting analysis of HCT116 cells infected with either InshSur8 #1 or #2 lentivirus showed that both constructs effectively knocked down endogenous Sur8 (Supplementary Figure S1). InshSur8 #2 viral supernatant was used for Sur8 knockdown in all subsequent experiments. To construct the shRNA-resistant form of Sur8 (shR-Sur8), the following silent mutations shown in bold were introduced: AACCTTGATCTACAGCACAAT.
Immunoblotting
Immunoblotting was performed as previously described.24 The following antibodies were used: anti-phosphorylated (p)-ERK, anti-p-Akt, and anti-Ki67 (Cell Signaling, Beverly, MA, USA); anti-cyclin D1, anti-ERK, anti-β-actin, anti-proliferating cell nuclear antigen, and anti-c-myc (Santa Cruz Biotechnology, Santa Cruz, CA, USA); anti-Rac1 (Upstate Biotechnology, Lake Placid, NY, USA); anti-α-tubulin (Oncogene Research Products, Cambridge, MA, USA); anti-GTP-Rac (NewEast Biosciences, Malvern, PA, USA); and anti-Sur8 produced and purified as previously described.25 Horseradish peroxidase (HRP)-conjugated anti-mouse (Cell Signaling) and anti-rabbit (Bio-Rad Laboratories, Hercules, CA, USA) secondary antibodies were used.
RNA isolation and RT-PCR
cDNA was used for PCR analysis using the following primers: glyceraldehyde-3-phosphate dehydrogenase F-primer 5′-AAGGTCGGAGTCAACGGATTTGGT-3′ and R-primer 5′-AGTGATGGCATGGACTGTGGTCAT-3′; Sur8 F-primer 5′-TCCCTGAGGATGTGTCTGGTCTCG-3′ and R-primer 5′-CCATGGCACGATATGGACCCTGC-3′; matrix metalloproteinase (MMP)-2F-primer 5′-GAGATCTGCAAACAGGACAT-3′ and R-primer 5′-GGTTCTCCAGCTTCAGGTAA-3′; and MMP-9F-primer 5′-CGACGAGTTGTGGTCGCTGG-3′ and R-primer 5′-GCACGCTGGAATGATCTGAG-3′.
Cell proliferation assay
Cells were seeded in triplicate at a density of 1 × 104 cells per well in 12-well plates and treated with Dox for 5 days. Cells were fixed with 4% paraformaldehyde and stained with crystal violet. Cells were then lysed with 10% acetic acid, and the absorbance at 590 nm was measured using a FLUOstar Optima plate reader (BMG Lab Technologies, Offenburg, Germany).
Anchorage-independent soft agar and foci formation assays
The anchorage-independent soft agar assay was performed as previously described.26 Briefly, cells were mixed in growth media containing 0.3% agar (USB Corporation Cleveland, OH, USA) and plated on a culture dish containing a 0.6% agar base. After 3 weeks, images of colonies were captured using an ECLIPSE TE2000-U microscope (Nikon, Melville, NY, USA). For the foci formation assay, cells were plated at a density of 500 cells per well in six-well plates. After 14 days, cells were fixed with 4% paraformaldehyde and stained with crystal violet.
Rac activation assay
Rac activity assays were performed as previously described23 by incubating the cell lysates with recombinant GST-PAK-CD fusion protein bound to glutathione-coupled agarose beads. Protein complexes were analyzed by immunoblotting with a Rac1 antibody.
Wound-healing assay
Cells were grown until they were confluent, and a scratch was introduced. After 36 h, cells were fixed with 4% paraformaldehyde and stained with crystal violet. The total distance travelled by the cells was quantified using NIS-Elements AR 3.1 software (Nikon).
Invasion assay
InshCon or InshSur8 HCT116 cells were treated with Dox for 72 h and seeded at a density of 5 × 104 cells on Matrigel-coated chambers (BD Bioscience, Bedford, MA, USA). After 24 h, cells on the outer surface were fixed with 4% paraformaldehyde and stained with crystal violet.
Animals, xenografts and metastasis assays
All animal experiments were performed in accordance with the Korean Food and Drug Administration guidelines. Animal studies were approved by the Institutional Review Board of Yonsei University. Approximately 4- to 6-week-old male athymic Crl: NU-Foxn1nu nude mice were purchased from Orient Biotech (Seoul, Korea). Stable InshCon or InshSur8 HCT116 cells (1 × 106 cells per site) were mixed with 40% Matrigel and were subcutaneously injected into both flanks of athymic nude mice for the xenograft assay. For the metastasis assay, stable InshCon or InshSur8 HCT116 cells (5 × 105 cells) were orthotopically implanted into the spleens of nude mice.
Immunohistochemistry
The sectioned tissues were autoclaved in 10 mM sodium citrate buffer (pH 6.0) for antigen retrieval. Sections were blocked in 5% bovine serum albumin and 1% goat serum in PBS and incubated with primary antibodies overnight at 4 °C. For diaminobenzidine (DAB) staining, immunohistochemistry (IHC) was performed with the UltraTek HRP kit (ScyTek Laboratories, Logan, UT, USA). For fluorescent IHC, the sections were incubated with the anti-rabbit Alexa Fluor 555 (Invitrogen) secondary antibody and counterstained with 4′,6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich). For quantitative analysis, the intensity of staining was determined using NIS-Elements AR 3.1 software (Nikon) or HistoQuest software (TissueGnostics, Vienna, Austria).
Statistical analysis
The data are presented as the mean±s.d. Student’s t-test was used for statistical analysis using GraphPad Prism (La Jolla, CA, USA) and the results are indicated in the figures as follows: *P<0.05, **P<0.005 and ***P<0.0005. P-values <0.05 were considered statistically significant.
Results
Sur8 is overexpressed in the established CRC cell lines and human CRC patient tissues
To assess the involvement of Sur8 in human CRC, we analyzed Sur8 expression levels in 10 different human CRC cell lines and an immortalized normal colon cell line, CCD18Co. Compared to the levels in CCD18Co, Sur8 was highly overexpressed in 9 CRC cell lines, with the exception of SW480 cells (Figure 1a). Sur8 was also highly increased in various human CRC tissues compared to the paired normal colon tissues (Figure 1b). Elevated Sur8 expression levels, compared with those in the normal colon tissues, were also detected in the colorectal tumor tissues as assessed by IHC using CRC tissue microarray array (Figures 1c and d). In human CRC tissues, Sur8 expression was significantly increased in liver metastases, compared with that in non-metastatic tumor tissues (Figures 1e and f).
The expression of Sur8 in established CRC cell lines and patients tissues. (a, b) WCLs of cell lines (a) and human CRC tissues (tumor and paired normal) (b) were analyzed by immunoblotting using anti-Sur8, -α-tubulin or -ERK antibodies. (c) TMA containing normal colon (n=5) and tumor tissues (n=7) was subjected to IHC analysis using an anti-Sur8 antibody, and cell nuclei were visualized by counterstaining with DAPI. Scale bar=250 μm. (d) The mean intensities of Sur8 expression in c were quantified using the NIS-Element AR image program. ***P<0.0005. (e) Liver metastatic (n=10) or non-metastatic (n=39) tumor tissues of CRC were stained with DAB using an anti-Sur8 antibody. Scale bar=250 μm. (f) The mean intensities of Sur8 expression in e were quantified using HistoQuest software. *P<0.05. CRC, colorectal cancer; ERK, extracellular signal-regulated kinase; IHC, immunohistochemistry; TMA, tissue microarray array; WCLs, whole-cell lysates.
Sur8 has a role in the activation of ERK and Akt and in the proliferation of CRC cells
When Sur8 was knocked down in various CRC cell lines, we observed that both ERK and Akt activities were decreased, irrespective of the mutational status of K-Ras, B-Raf or PI3K (Figure 2a). These decrements were rescued by expressing a shRNA-resistant form of Sur8 (Supplementary Figures S2a and S2b). As Sur8 was overexpressed not only in CRC cell lines but also in malignant CRC tissues, we examined the role of Sur8 in the proliferation of CRC cells. When Sur8 was knocked down in HCT116 and LoVo CRC cells, cell proliferation was reduced by 50% and 46%, respectively, after 5 days of cell culture (Figure 2b). Consistent with these results, immunoblotting analysis confirmed the reduction of proliferation-related markers such as proliferating cell nuclear antigen, c-myc, and cyclin D1 upon knockdown of Sur8 in HCT116 and LoVo CRC cells (Supplementary Figures S3a and S3b). In contrast, proliferation of the CCD18Co normal colon and SW480 CRC cells, which expressed low levels of endogenous Sur8 (Figure 1a), was increased by 54% and 50%, respectively, when Sur8 was overexpressed (Figure 2c and Supplementary Figure S4a). The Dox-induced overexpression of Sur8 in SW480 cells activated both ERK and Akt, as revealed by the increase in phosphorylation of both proteins (Supplementary Figure S4b).
Effect of Sur8 modulation on ERK and Akt activities, and proliferation of CRC cells. (a) Different stable CRC cells infected with either InshCon or InshSur8 lentivirus were treated with Dox for 72 h. WCLs were immunoblotted with the indicated antibodies. (b, c) The indicated stable CRC cells infected with either InshCon or InshSur8 (b), and with InConOE or InSur8OE lentivirus (c), were seeded in 12-well plates. Cells were treated with fresh Dox every 24 h to induce Sur8 knockdown (b) or overexpression (c) and cell growth was measured at different time points as described in the Materials and Methods section. The data shown are the mean±s.d. of three independent experiments. CRC, colorectal cancer; Dox, doxycycline; ERK, extracellular signal-regulated kinase; IHC, immunohistochemistry; WCLs, whole-cell lysates.
Sur8 plays a role in the transformation of CRC cells
When Sur8 was knocked down in HCT116 and LoVo CRC cells, their transforming abilities were significantly reduced (Figures 3a and b). However, when Sur8 was overexpressed in normal colon (CCD18Co) and SW480 CRC cells, which showed low levels of endogenous Sur8 (Figure 1a), anchorage-independent growth was increased in both cell lines (Figures 3c and d).
Effect of Sur8 knockdown or overexpression on the transforming potential of CRC cells. (a, b) Stable HCT116 or LoVo cells infected with either InshCon or InshSur8 lentivirus were seeded in six-well plates and treated with fresh Dox every 24 h for 14 days to induce Sur8 knockdown. Foci were stained with crystal violet, and images were captured. (c, d) Stable CCD18Co and SW480 cells infected with either InConOE or InSur8OE lentivirus were seeded in 24-well plates for performing the anchorage-independent soft agar assay. Cells were treated with fresh Dox every 24 h to induce Sur8 overexpression, and images of the colonies were captured after 3 weeks. The data shown are the mean±s.d. of three independent experiments. ***P<0.0005. Dox, doxycycline.
Sur8 knockdown suppresses the growth of xenograft tumors derived from HCT116 CRC cells
To explore the role of Sur8 in CRC tumorigenesis in vivo, we used the mouse xenograft model in which HCT116 cells harboring K-Ras and PI3K mutations were implanted into nude mice. We subcutaneously injected stable InshCon or InshSur8 HCT116 cells harboring the Dox-mediated Sur8 knockdown lentiviral system. When Dox was added to the feeding water to induce acute shutdown of endogenous Sur8 in the tumor cells implanted in vivo, the xenograft tumors from InshSur8-injected HCT116 cells were significantly decreased within 11 days of Dox treatment, compared to those from the InshCon-injected cells (Figures 4a and b). The anti-tumor effect of Sur8 knockdown on the xenograft tumors appeared to be caused by the inhibition of both the Ras-ERK and PI3K-Akt pathways, as revealed by the decrease in the positive signals of both p-ERK and p-Akt in the IHC analysis (Figure 4c). GTP-Rac was also found to be decreased on the xenograft tumors upon knockdown of Sur8. Both the staining intensity and the nuclear localization of the proliferation marker Ki67 were also decreased upon Sur8 knockdown. Interestingly, the liver and peritoneal metastases were observed only in the mice injected with InshCon HCT116 cells (Figure 4d), suggesting that Sur8 may have a role in driving liver/peritoneal metastases in CRC.
Effect of Sur8 knockdown on the growth of xenograft tumors derived from HCT116 cells. (a–d) HCT116 cells (1 × 106) infected with either InshCon or InshSur8 lentivirus were subcutaneously injected into both flanks of nude mice (n=5), and the mice were fed with Dox-water (2 mg ml−1 Dox) for 11 days after the tumor volume reached 200 mm3. (a) The gross images of isolated xenograft tumors. Scale bar=30 mm. (b) Quantitative analysis of tumor volumes on the indicated days. (c) The paraffin-embedded tumor tissues generated in the xenograft model (a) were subjected to H&E staining and DAB analyses using the indicated antibodies. Scale bar=25 μm. (d) Representative images of liver and peritoneal metastases observed in the nude mice injected with InshCon lentivirus-infected HCT116 cells. White arrows represent metastatic tumors. Two-sided Student’s t-test was used to determine the statistical significance. **P<0.005. Dox, doxycycline.
Sur8 knockdown reduces the migration and invasion potential of HCT116 CRC cells
Consistent with the changes in the xenograft tumor model in vivo, Rac activity was reduced by Sur8 knockdown in HCT116 cells in vitro (Figure 5a). In addition, expression of both MMP-2 and MMP-9 were decreased upon Sur8 knockdown (Figure 5b). As Sur8 knockdown decreased Rac and MMP activation, two key factors involved in Sur8-mediated cell migration and invasion, we next examined the involvement of Sur8 in the migration and invasion of HCT116 cells. We found that the migration ability and invasive properties of HCT116 cells were decreased when Sur8 was knocked down (Figures 5c–f).
Effect of Sur8 knockdown on HCT116 cell migration and invasion. (a–f) Stable HCT116 cells infected with either InshCon or InshSur8 lentivirus were treated with Dox for 72 h before performing the experiments. (a) WCLs were incubated with GST-PAK-CD, and GTP-bound active Rac was detected by immunoblotting with an anti-Rac1 antibody. For all other measurements, WCLs were immunoblotted with the indicated antibodies. (b) RT-PCR analyses for Sur8, MMP-2, MMP-9 or GAPDH were performed. (c, d) Cells were grown until confluent, and scratches were made using a 200-μl tip. Cells were collected at 36 h and stained with crystal violet, images were captured and the total distance travelled by the cells was quantified. Scale bar=200 μm. (e, f) Cells were seeded on the Matrigel-coated chamber. Cells that invaded through the Matrigel were stained with crystal violet, representative images were captured, and the cells were counted. Scale bar=200 μm. ***P<0.0005. Dox, doxycycline; GADPH, glyceraldehyde-3-phosphate dehydrogenase; MMP, matrix metalloproteinase; WCLs, whole-cell lysates.
The liver metastatic potential of HCT116 CRC cells is reduced by Sur8 knockdown
To investigate the role of Sur8 in mediating CRC metastasis, stable InshCon or InshSur8 HCT116 cells harboring the Dox-mediated Sur8 knockdown system were orthotopically implanted into the spleens of nude mice. One day after the splenic injection of cells, mice were fed Dox-water for 3 weeks (Figure 6a). The acute shutdown of endogenous Sur8 significantly decreased the liver metastatic capacities of HCT116 cells from the spleen (Figures 6b and c). In the metastasized liver tumors, Sur8 expression was significantly decreased, concomitant with decreased ERK, Akt, and Rac activities, when Sur8 was knocked down (Figure 6d). These data support a role for Sur8 in driving liver metastasis in CRC.
Effect of Sur8 knockdown on the ability of HCT116 cells to form liver metastases. (a–d) HCT116 cells (5 × 105) infected with either InshCon or InshSur8 lentivirus were injected into the spleens of nude mice (n=5). Dox-water (2 mg ml−1 Dox) was fed to the mice on the following day after the injection of cells, and liver metastases were monitored after 3 weeks. (a) Schematic representation of the experimental design. The representative gross images (b) and quantification of the liver metastases (c). Black arrows indicate metastasized liver tumors. Scale bar=7 mm. (d) Paraffin-embedded metastasized liver tissue sections were subjected to DAB analysis using the indicated antibodies. Scale bar=25 μm. Student’s t-test was used to determine statistical significance. **P<0.005. Dox, doxycycline.
Discussion
In this study, we provide evidence for the involvement of Sur8, initially identified as a scaffold protein of the Ras-ERK pathway, in regulating tumorigenesis and liver metastasis of CRC. The role of Sur8 in colorectal tumorigenesis is indicated by overexpression of Sur8 in both established CRC cell lines and in CRC patient-derived tissues. The elevated expression of Sur8 in metastatic tumor tissues compared to that in non-metastatic tissues suggests that Sur8 may have a role in metastasis of CRC. ERK and Akt activities were attenuated upon Sur8 knockdown, indicating that Sur8 mediates tumorigenesis of CRC via activation of both the Ras-ERK and PI3K pathways. The inactivation of Akt and ERK by Sur8 knockdown correlates with our recent study showing that Sur8 interacts with PI3K and Raf and has a role in tumor malignancy via regulation of both the PI3K-Akt and Ras-Raf-ERK pathways.22
Our results from the Dox-mediated Sur8 knockdown/overexpression CRC cellular model also showed the involvement of Sur8 in regulating proliferation and transformation of CRC cells in vitro. Consistent with these observations, the tumor size was critically reduced when Sur8 was knocked down in the xenograft tumors of HCT116 cells in vivo. Sur8 knockdown decreased the activation of Rac and the expression of MMPs, thereby reducing the migration and invasion potential of HCT116 cells. In addition, shutdown of endogenous Sur8 in HCT116 cells injected into the spleens of nude mice decreased the liver metastatic abilities of these cells. These results indicate that Sur8 may have a critical role in colorectal tumorigenesis and metastasis, and especially in promoting liver metastases.
CRC is one of the most common cancer types harboring mutations in K-Ras, B-Raf or PI3K. Although chemotherapeutic agents have been used for the treatment of CRC, various adverse effects have been observed in CRC patients. Epidermal growth factor receptor-targeted monoclonal antibodies, which were considered the most promising new therapeutic option for CRC, are of limited utility, especially due to restricted responses in patients, especially in patients carrying K-Ras, B-Raf or PI3K mutations.2, 6, 27, 28 As Sur8 modulates both the Ras-ERK and PI3K-Akt signaling pathways in CRC cells, despite the mutational status of K-Ras, B-Raf or PI3K, we suggest that the scaffold protein Sur8 may be a potential therapeutic target for colorectal tumorigenesis and metastasis.
References
Hawk ET, Levin B . Colorectal cancer prevention. J Clin Oncol 2005; 23: 378–391.
Lievre A, Blons H, Laurent-Puig P . Oncogenic mutations as predictive factors in colorectal cancer. Oncogene 2010; 29: 3033–3043.
Yokota T . Are KRAS/BRAF mutations potent prognostic and/or predictive biomarkers in colorectal cancers? Anticancer Agents Med Chem 2012; 12: 163–171.
Bos JL, Fearon ER, Hamilton SR, Verlaan-de Vries M, van Boom JH, van der Eb AJ et al. Prevalence of ras gene mutations in human colorectal cancers. Nature 1987; 327: 293–297.
Brink M, de Goeij AF, Weijenberg MP, Roemen GM, Lentjes MH, Pachen MM et al. K-ras oncogene mutations in sporadic colorectal cancer in The Netherlands Cohort Study. Carcinogenesis 2003; 24: 703–710.
Cathomas G . PIK3CA in Colorectal Cancer. Front Oncol 2014; 4: 35.
Field K, Lipton L . Metastatic colorectal cancer-past, progress and future. World J Gastroenterol 2007; 13: 3806–3815.
Silvestri A, Pin E, Huijbers A, Pellicani R, Parasido EM, Pierobon M et al. Individualized therapy for metastatic colorectal cancer. J Intern Med 2013; 274: 1–24.
Giehl K . Oncogenic Ras in tumour progression and metastasis. Biol Chem 2005; 386: 193–205.
Pylayeva-Gupta Y, Grabocka E, Bar-Sagi D . RAS oncogenes: weaving a tumorigenic web. Nat Rev Cancer 2011; 11: 761–774.
Steelman LS, Chappell WH, Abrams SL, Kempf RC, Long J, Laidler P et al. Roles of the Raf/MEK/ERK and PI3K/PTEN/Akt/mTOR pathways in controlling growth and sensitivity to therapy-implications for cancer and aging. Aging 2011; 3: 192–222.
Moon BS, Jeong WJ, Park J, Kim TI, Min do S, Choi KY . Role of oncogenic K-Ras in cancer stem cell activation by aberrant Wnt/beta-catenin signaling. J Natl Cancer Inst 2014; 106: djt373.
Morrison DK, Davis RJ . Regulation of MAP kinase signaling modules by scaffold proteins in mammals. Annu Rev Cell Dev Biol 2003; 19: 91–118.
Sieburth DS, Sun Q, Han M . SUR-8, a conserved Ras-binding protein with leucine-rich repeats, positively regulates Ras-mediated signaling in C. elegans. Cell 1998; 94: 119–130.
Selfors LM, Schutzman JL, Borland CZ, Stern MJ . soc-2 encodes a leucine-rich repeat protein implicated in fibroblast growth factor receptor signaling. Proc Natl Acad Sci USA 1998; 95: 6903–6908.
Li W, Han M, Guan KL . The leucine-rich repeat protein SUR-8 enhances MAP kinase activation and forms a complex with Ras and Raf. Genes Dev 2000; 14: 895–900.
Rodriguez-Viciana P, Oses-Prieto J, Burlingame A, Fried M, McCormick F . A phosphatase holoenzyme comprised of Shoc2/Sur8 and the catalytic subunit of PP1 functions as an M-Ras effector to modulate Raf activity. Mol Cell 2006; 22: 217–230.
Young LC, Hartig N, Munoz-Alegre M, Oses-Prieto JA, Durdu S, Bender S et al. An MRAS, SHOC2, and SCRIB complex coordinates ERK pathway activation with polarity and tumorigenic growth. Mol Cell 2013; 52: 679–692.
Cordeddu V, Di Schiavi E, Pennacchio LA, Ma'ayan A, Sarkozy A, Fodale V et al. Mutation of SHOC2 promotes aberrant protein N-myristoylation and causes Noonan-like syndrome with loose anagen hair. Nat Genet 2009; 41: 1022–1026.
Hoban R, Roberts AE, Demmer L, Jethva R, Shephard B . Noonan syndrome due to a SHOC2 mutation presenting with fetal distress and fatal hypertrophic cardiomyopathy in a premature infant. Am J Med Genet A 2012; 158A: 1411–1413.
Komatsuzaki S, Aoki Y, Niihori T, Okamoto N, Hennekam RC, Hopman S et al. Mutation analysis of the SHOC2 gene in Noonan-like syndrome and in hematologic malignancies. J Hum Genet 2010; 55: 801–809.
Kaduwal S, Jeong WJ, Park JC, Lee KH, Lee YM, Jeon SH et al. Sur8/Shoc2 promotes cell motility and metastasis through activation of Ras-PI3K signaling. Oncotarget 2015; 6: 33091–33105.
Sander EE, ten Klooster JP, van Delft S, van der Kammen RA, Collard JG . Rac downregulates Rho activity: reciprocal balance between both GTPases determines cellular morphology and migratory behavior. J Cell Biol 1999; 147: 1009–1022.
Jeong WJ, Yoon J, Park JC, Lee SH, Lee SH, Kaduwal S et al. Ras stabilization through aberrant activation of Wnt/beta-catenin signaling promotes intestinal tumorigenesis. Sci Signal 2012; 5: ra30.
Moon BS, Kim HY, Kim MY, Yang DH, Lee JM, Cho KW et al. Sur8/Shoc2 involves both inhibition of differentiation and maintenance of self-renewal of neural progenitor cells via modulation of extracellular signal-regulated kinase signaling. Stem Cells 2011; 29: 320–331.
Zhao JJ, Gjoerup OV, Subramanian RR, Cheng Y, Chen W, Roberts TM et al. Human mammary epithelial cell transformation through the activation of phosphatidylinositol 3-kinase. Cancer Cell 2003; 3: 483–495.
Lievre A, Bachet JB, Le Corre D, Boige V, Landi B, Emile JF et al. KRAS mutation status is predictive of response to cetuximab therapy in colorectal cancer. Cancer Res 2006; 66: 3992–3995.
Love RR, Leventhal H, Easterling DV, Nerenz DR . Side effects and emotional distress during cancer chemotherapy. Cancer 1989; 63: 604–612.
Acknowledgements
We thank Hye-Young Kim for providing the human normal and tumor colon samples. This work was supported by the National Research Foundation of Korea (NRF) grants funded by the Korean government (MSIP) (2009–0083522 and 2015R1A2A1A05001873). SK, KHL and JCP were supported by a Brain Korea 21 (BK21) studentship from the NRF.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Experimental & Molecular Medicine website
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Lee, YM., Kaduwal, S., Lee, K. et al. Sur8 mediates tumorigenesis and metastasis in colorectal cancer. Exp Mol Med 48, e249 (2016). https://doi.org/10.1038/emm.2016.58
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/emm.2016.58
- Springer Nature Limited
This article is cited by
-
Accessory proteins of the RAS-MAPK pathway: moving from the side line to the front line
Communications Biology (2021)