Abstract
In this retrospective analysis using the Transplant Registry Unified Management Program, we identified 145 patients with human herpesvirus (HHV)-6 encephalitis among 6593 recipients. The cumulative incidences of HHV-6 encephalitis at 100 days after transplantation in all patients, recipients of bone marrow or PBSCs and recipients of cord blood were 2.3%, 1.6% and 5.0%, respectively. Risk factors identified in multivariate analysis were male sex, type of transplanted cells (relative risk in cord blood transplantation, 11.09, P<0.001; relative risk in transplantation from HLA-mismatched unrelated donor, 9.48, P<0.001; vs transplantation from HLA-matched related donor) and GvHD prophylaxis by calcineurin inhibitor alone. At 100 days after transplantation, the overall survival rate was 58.3% and 80.5% among patients with and without HHV-6 encephalitis, respectively (P<0.001). Neuropsychological sequelae remained in 57% of 121 evaluated patients. With both foscarnet and ganciclovir, full-dose therapy (foscarnet ⩾180 mg/kg, ganciclovir ⩾10 mg/kg) was associated with better response rate (foscarnet, 93% vs 74%, P=0.044; ganciclovir, 84% vs 58%, P=0.047). HHV-6 encephalitis is not rare not only in cord blood transplant recipients but also in recipients of HLA-mismatched unrelated donors. In this study, development of HHV-6 encephalitis was associated with a poor survival rate, and neurological sequelae remained in many patients.
Similar content being viewed by others
Introduction
Reactivation of human herpesvirus (HHV)-6 is observed in 30–70% of allogeneic hematopoietic stem cell transplantation (allo-HSCT) recipients.1, 2, 3 Accumulating evidence supports a causal association between HHV-6 reactivation and encephalitis.4, 5, 6
Previous analyses have suggested that the mortality rates attributable to HHV-6 encephalitis are high,7, 8, 9, 10, 11, 12 and many survivors display cognitive sequelae.13 HHV-6 encephalitis is therefore increasingly recognized as an important complication of allo-HSCT; however, many questions remain unanswered. Reported incidence of HHV-6 encephalitis among allo-HSCT recipients ranges from 0.96 to 11.6%,5 mainly owing to small cohorts of cases. Although umbilical cord blood transplantation (CBT) represents a risk factor for HHV-6 encephalitis,5, 12, 14 other risk factors remain unconfirmed. Treatment recommendations have been made based on in vitro data,15, 16, 17 but few data are available on the clinical effectiveness. Our study aimed to define the epidemiology, clinical characteristics and outcome of HHV-6 encephalitis, and the effect of antiviral treatment on the outcome.
Patients and methods
Data source and patient selection
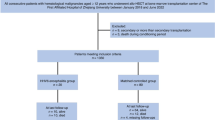
For this retrospective observational study, clinical data of patients ⩾16 years old who received allo-HSCT between 1 January 2007 and 31 December 2011 were obtained from the Japan Society of Hematopoietic Cell Transplantation (JSHCT) and the Japanese Data Center for Hematopoietic Cell Transplantation (JDCHCT), using the Transplant Registry Unified Management Program (TRUMP).18, 19 Patient data in TRUMP including survival and long-term complications are followed longitudinally using an annual follow-up form. Additional data, including detailed conditions, treatments and outcomes, were collected for this study. The study was approved by the data management committee of the JSHCT and by the ethics committee of Oita University Faculty of Medicine.
Definitions
HHV-6 encephalitis was diagnosed if the patient satisfied all of the following three criteria: (1) presence of central nervous system (CNS) dysfunction; (2) a positive PCR result for HHV-6 DNA in cerebrospinal fluid (CSF); and (3) absence of other identified causes of CNS dysfunction, including other infectious agents. CNS dysfunction was defined as the presence of disorientation as to time and place, loss of consciousness, personality change, convulsions, memory loss or dysesthesia not attributable to peripheral neuropathy.11, 20 Diagnosis and grading of acute GvHD were based on clinical criteria described elsewhere.21 Engraftment syndrome was defined according to the proposal for a uniform definition by Spitzer.22 Pre-engraftment immune reaction was defined as CBT recipients who satisfied criteria described elsewhere.23, 24 Steroid treatment was defined as treatment with ⩾0.5 mg/kg of prednisolone or ⩾0.4 mg/kg of methylprednisolone. Disease phase was evaluated against patients with hematological neoplasms. Early stage was defined as acute leukemia in a first to second remission, CML in a first chronic phase or myelodysplastic syndrome classified as refractory anemia without excess blasts; all others were considered non-early stage. We defined the following dosages as reduced-intensity conditioning regimens: TBI ⩽5 Gy in a single fraction or 5–8 Gy in multiple fractions; busulfan <9 mg/kg (IV busulfan <7.2 mg/kg); or melphalan ⩽140 mg/m2. Donor and recipient pairs were considered HLA matched when the HLA-A, HLA-B and HLA-DRB1 loci were matched in HSCT from a related donor or CBT. For unrelated HSCT other than CBT, matching at the HLA-A, HLA-B, HLA-Cw and HLA-DRB-1 loci was considered indicative of matching. HLA mismatches were defined by the presence of ⩾1 disparity among HLA loci. Regarding the effects of antiviral therapy, response was defined as disappearance or obvious improvement of CNS symptoms after starting treatment. With respect to the outcomes of HHV-6 encephalitis, ‘return to society’ meant that the patient was able to return to work, school or household activities.
Statistical considerations
The cumulative incidence of HHV-6 encephalitis was estimated by considering competing risks regression models according to the method of Fine and Gray.25 Death without HHV-6 encephalitis was treated as competing risk. Factors associated with at least borderline significance (P<0.1) in the univariate analyses were subjected to multivariate analysis. The Fine and Gray26 proportional hazards model was used for the multivariate analysis. The cumulative incidence of acute GvHD was estimated by considering competing risks regression models, and treating death without GvHD as a competing risk. The probability of overall survival was estimated using the Kaplan–Meier method. To evaluate the influence of development of HHV-6 encephalitis on survival, proportional hazard modeling was used, treating the development of HHV-6 encephalitis as a time-dependent covariate. Survival estimates were compared using the log-rank test. Factors associated with outcome of HHV-6 encephalitis were analyzed using Fisher’s exact test and Cox modeling. Proportional hazard modeling was used to evaluate the effect of antivirals on survival, treating the start of antiviral therapy as a time-dependent covariate. All statistical tests were two sided and performed using a 5% level of significance and were carried out using Stata 13 software (StataCorp LP, College Station, TX, USA).
Results
Data collection
Using the TRUMP database, we identified a total of 574 recipients, aged ⩾16 years, who received allo-HSCT between January 2007 and December 2011 and whose records showed they had viral infections owing to HHV-6. Most patients were not monitored for HHV-6 reactivation. A questionnaire was mailed to each associated transplant institution (n=128) to collect additional patient data. Questionnaires were returned from 372 recipients with HHV-6 infection (data collection rate, 65%) from 82 transplant institutions (institution participation rate, 63%). Patients comprised 239 with HHV-6-associated diseases and 133 with asymptomatic reactivation of HHV-6. Among the 239 patients with HHV-6-associated disease, 213 (89%) had been diagnosed with HHV-6 encephalitis by an attending physician (Supplementary Table S1). Among these, 145 fulfilled the criteria for HHV-6 encephalitis.
Incidence and risk factors of HHV-6 encephalitis
Cumulative incidence of HHV-6 encephalitis in the overall study population (patients at institutes from whom questionnaires were returned who were ⩾16 years old and had received allo-HSCT between January 2007 and December 2011; n=6 593) at 100 days after HSCT was 2.3% (Figure 1a). Cumulative incidence of HHV-6 encephalitis at day 100 was significantly higher in patients receiving CBT than in those receiving another stem cell source (5.0% vs 1.6%, P<0.001). By univariate analysis, patient sex, type of transplanted cells (Figure 1b) and measures of GvHD prophylaxis were associated with incidence of HHV-6 encephalitis (Table 1). GvHD prophylaxis by calcineurin inhibitor (CI) alone was associated with increased incidence of HHV-6 encephalitis among CBT recipients (Supplementary Table S2, Figure 1c) but was not a significant risk factor in non-CBT (Supplementary Table S3). Multivariate analysis (Table 2) revealed that male sex, HSCT from other than an HLA-matched related donor and GvHD prophylaxis by CI alone were significant risk factors for HHV-6 encephalitis. Among CBT recipients, GvHD prophylaxis by CI alone was the only significant factor for HHV-6 encephalitis. Regarding the associations between GvHD prophylactic methods and incidence of GvHD in CBT recipients, cumulative incidences of grade 2–4 acute GvHD at 100 days were 37.1%, 43.0% and 54.4% in patients receiving CI with methotrexate, CI with mycophenolate mofetil and CI alone, respectively (P<0.001).
Cumulative probabilities of HHV-6 encephalitis. (a) All cases. Cumulative incidence of HHV-6 encephalitis at 100 days after HSCT was 2.3%. (b) Stratified by donor source: related or unrelated bone marrow transplantation (BMT) or peripheral blood stem cell transplantation (PBSCT), HLA match or mismatch. Cumulative incidence of HHV-6 encephalitis at 100 days after HSCT was 0.36%, 1.7%, 1.3%, 3.3% and 5.0% in patients receiving related BMT or PBSCT (HLA match), related BMT or PBSCT (HLA mismatch), unrelated BMT or PBSCT (HLA match), unrelated BMT or PBSCT (HLA mismatch) and CBT, respectively. (c) Stratified by method of GvHD prophylaxis in CBT recipients: CI alone, CI with mycophenolate mofetil (MMF) or CI with methotrexate (MTX). Cumulative incidence of HHV-6 encephalitis at 100 days after CBT was 3.5%, 5.8% and 8.4% in patients who received GvHD prophylaxis using CI with MTX, CI with MMF and CI alone, respectively.
Characteristics of HHV-6 encephalitis
Characteristics of HHV-6 encephalitis are shown in Table 3. A total 125 (84.4%) of the 145 included patients developed HHV-6 encephalitis during weeks 3–5 (15–35 days) after HSCT. Among CBT recipients, criteria-defined pre-engraftment immune reaction was observed 0–8 days before development of HHV-6 encephalitis in 35 (52%) of 67 patients. Differentiation of HHV-6 was performed for 48 patients, all of whom had HHV-6B. Limbic encephalitis was demonstrated in a total of 78 (60%) of 131 patients who underwent brain magnetic resonance imaging examination.
Outcome
The probabilities of overall survival on day 100 and day 365 after HSCT were 58.3% (95% confidence interval (CI): 49.6–66.1%) and 27.5% (95% CI: 20.3–35.2%) in patients who developed HHV-6 encephalitis and 80.5% (95% CI: 79.5–81.5%) and 58.7% (95% CI: 57.5–59.9%) in those who did not develop HHV-6 encephalitis, respectively (P<0.001; Figure 2). Cumulative incidence of the mortality attributable to HHV-6 encephalitis on day 30 and day 100 after development of HHV-6 encephalitis was 2.1% and 12.9%, respectively.
Supplementary Table S4 shows proportional hazards modeling of the mortality risk after development of HHV-6 encephalitis. In the final multivariable Cox model, a positive but statistically nonsignificant relationship was confirmed between male sex and death after the development of HHV-6 encephalitis.
Figure 3 shows outcomes for those patients who developed HHV-6 encephalitis. Among patients who were evaluated for CNS dysfunction after completing antiviral therapy (n=121), a total of 69 patients (57%) were considered to have retained various neuropsychological dysfunctions. The most frequent sequela was memory disturbance.
Outcome for patients who developed HHV-6 encephalitis. For patients with HHV-6 encephalitis, median follow-up after development of HHV-6 encephalitis was 1651 days for surviving patients (range, 649–2680 days). Sequelae of HHV-6 encephalitis could not be evaluated in 24 patients owing to early death (n=20) or intubation (n=4). Information on the return to society was not obtained for 3 surviving patients and 7 patients who died.
Effect of antiviral treatment
Among 145 patients with HHV-6 encephalitis, information about response to antiviral therapy was available for 133 patients. The reasons for missing data are as follows: response was unable to be evaluated (n=9), no therapy (n=1) and treatment data not available (n=2). A total of 123 patients received foscarnet or ganciclovir monotherapy, and the other 10 patients were treated with combination foscarnet and ganciclovir therapy.
Response rates of neurological symptoms after starting antiviral therapy were 83.8% and 71.4% with foscarnet monotherapy and ganciclovir monotherapy, respectively. This difference between groups was not significant (P=0.10, Fisher’s exact test). With both foscarnet and ganciclovir, the response rate of full-dose monotherapy (foscarnet ⩾180 mg/kg, ganciclovir ⩾10 mg/kg) was significantly higher than that of non-full-dose monotherapy (foscarnet, 93% vs 74%, P=0.044; ganciclovir, 84% vs 58%, P=0.047, Fisher’s exact test; Figure 4). The response rate of patients receiving combination therapy using various doses of foscarnet and ganciclovir was 100%.
The rate of sequelae or death owing to HHV-6 encephalitis was significantly lower in patients who received full-dose therapy than in those who did not receive full-dose therapy (ganciclovir <10 mg/kg and foscarnet <180 mg/kg; Table 4). The rate of death from any cause within 30 days after the development of HHV-6 encephalitis was significantly lower in patients who received foscarnet therapy and significantly higher in patients who received ganciclovir therapy (Table 4). Using Kaplan–Meier analysis, the probabilities of overall survival on day 30 and day 100 after development of HHV-6 encephalitis were 88.3% (95% CI: 79.4–93.6%) and 60.8% (95% CI: 49.7–70.2%) in patients who were treated with foscarnet as first-line therapy, and 67.0% (95% CI: 52.3–78.0%) and 49.3% (95% CI: 35.0–62.0%) in those who were not treated with foscarnet as first-line therapy, respectively (P=0.14); these probabilities were 83.3% (95% CI: 71.2–90.7%) and 62.1% (95% CI: 48.7–72.9%) in patients who were treated with full-dose therapy, and 78.2% (95% CI: 67.4–85.9%) and 52.1% (95% CI: 40.5–62.6%) in those who were not treated with full-dose therapy, respectively (P=0.15).
Discussion
In this study, we determined the epidemiology, characteristics and outcomes of HHV-6 encephalitis by accessing a nationwide database. We found that the prevalence of HHV-6 encephalitis was unexpectedly high in patients receiving HSCT from an HLA-mismatched unrelated donor. The finding is consistent with a prior study.12 We also showed risk factors that were previously unconfirmed, including GvHD prophylaxis by CI alone in CBT. The prognosis of patients who developed HHV-6 encephalitis was poor and many surviving patients retained psychophysiological dysfunctions.
Although CBT is an established risk factor for HHV-6 encephalitis, considerable differences in incidence can be found among centers.11, 27 Methods of GvHD prophylaxis were shown to be associated with the development of HHV-6 encephalitis in CBT; previous reports have supported the clinical plausibility of such a relationship. An inflammatory milieu early after allo-HSCT, such as pre-engraftment immune reaction,11 engraftment syndrome28 and GvHD,12, 28 represents risk factors for subsequent development of HHV-6 encephalitis. In fact, more than half (52%) of CBT recipients in this study displayed pre-engraftment immune reaction before developing HHV-6 encephalitis. GvHD prophylaxis without methotrexate is known to be associated with a high incidence of post-CBT immune reaction.24, 29 In this cohort, prophylaxis using CI alone was a significant risk factor for acute GvHD. We suggest that better prevention of GvHD and related immune reactions will reduce HHV-6 encephalitis incidence.
This study clearly showed a poor prognosis for patients who developed HHV-6 encephalitis. Not only encephalitis but also various other conditions were associated with patient death, including GvHD. Reactivated HHV-6 may affect such subsequent complications.30 Even among survivors, more than half of patients retained some degree of neurological compromise; the proportion of patients who could return to society at least once was only 21%.
We showed that full-dose antiviral therapy was associated with lower incidences of sequelae and death owing to HHV-6 encephalitis, and the use of foscarnet, but not ganciclovir, was associated with a low incidence of early death. Some of these findings are biologically plausible. Previous in vitro studies have shown that foscarnet offers better in vitro activity against HHV-6 than ganciclovir.31, 32 Antiviral CSF concentrations reach the therapeutic range in patients receiving full-dose foscarnet;33 a case report suggested that this may not be the case in patients receiving full-dose ganciclovir.34 Although the reasons why ganciclovir is associated with an increased risk of early death are unknown, myelosuppression owing to ganciclovir might increase the risk of infectious diseases. Combination therapy with foscarnet and ganciclovir seemed to be most effective in our study, but its evaluation is difficult because only 10 patients received the combination therapy and the doses of foscarnet and ganciclovir varied.
Several limitations must be considered in the interpretation of our results. First, optimal evaluation of CNS dysfunction and monitoring of HHV-6 reactivation after transplantation could not be performed in this retrospective study. Although we used strict criteria for the presence of HHV-6 DNA to define HHV-6 encephalitis, many recipients who actually develop HHV-6 encephalitis may not receive CSF examination in clinical practice. A lack of opportunities for optimal evaluation of CNS dysfunction and stringent diagnostic criteria will cause underestimation of the incidence of HHV-6 encephalitis. Second, we could not rule out the possibility that positive HHV-6 DNA in the CSF was due to chromosomally integrated HHV-6 (ciHHV-6).35 Population-based studies have estimated the prevalence of ciHHV-6 to be ∼1% in the general population.36, 37 In our study population (n=6593), a total of 412 patients showed CNS dysfunction, based on the TRUMP database. Assuming that all patients with CNS dysfunction were tested for the presence of HHV-6 in the CSF and that incidence of ciHHV-6 in both donors and recipients was 1%, the positive results for HHV-6 DNA in CSF from about 8 patients with CNS dysfunction were thought to be owing to ciHHV-6. Therefore, we consider that there is some bias owing to the participation of patients with ciHHV-6, leading to misdiagnosis. Third, results of the analysis of antiviral effects require careful interpretation as data on the presence of organ dysfunction at the time of developing HHV-6 encephalitis were not collected in this study. There may be some patients who did not receive full-dose antiviral therapy owing to renal dysfunction or poor general status. Prospective studies of antiviral therapy with pre-specific dose and schedules are warranted to demonstrate the effect of antivirals for the treatment of HHV-6 encephalitis.
In summary, we found risk factors for HHV-6 encephalitis, including male sex, type of transplanted cells and GvHD prophylaxis by CI alone for CBT recipients. The incidence of HHV-6 encephalitis was unexpectedly high in patients receiving HSCT from an HLA-mismatched unrelated donor. The prognosis of HHV-6 encephalitis is poor, but we showed an association between appropriate antiviral treatment and better outcomes. The results of this large-scale study serve to improve understanding regarding the clinical impact of HHV-6 encephalitis in allo-HSCT and serve as a benchmark for future trials.
References
Yamane A, Mori T, Suzuki S, Mihara A, Yamazaki R, Aisa Y et al. Risk factors for developing human herpesvirus 6 (HHV-6) reactivation after allogeneic hematopoietic stem cell transplantation and its association with central nervous system disorders. Biol Blood Marrow Transplant 2007; 13: 100–106.
Zerr DM, Corey L, Kim HW, Huang ML, Nguy L, Boeckh M . Clinical outcomes of human herpesvirus 6 reactivation after hematopoietic stem cell transplantation. Clin Infect Dis 2005; 40: 932–940.
Ogata M, Satou T, Kadota J, Saito N, Yoshida T, Okumura H et al. Human herpesvirus 6 (HHV-6) reactivation and HHV-6 encephalitis after allogeneic hematopoietic cell transplantation: a multicenter, prospective study. Clin Infect Dis 2013; 57: 671–681.
Zerr DM, Ogata M In: Flamand L, Lautenschlager I, Krueger G, Ablashi D (eds). Human Herpesviruses HHV-6A, HHV-6B & HHV-7. Diagnosis and Clinical Management 3rd edn Elsevier: Oxford, UK, 2014 pp 217–234.
Ogata M, Fukuda T, Teshima T . Human herpesvirus-6 encephalitis after allogeneic hematopoietic cell transplantation: what we do and do not know. Bone Marrow Transplant 2015; 50: 1030–1036.
Zerr DM . Human herpesvirus 6 (HHV-6) disease in the setting of transplantation. Curr Opin Infect Dis 2012; 25: 438–444.
Zerr DM . Human herpesvirus 6 and central nervous system disease in hematopoietic cell transplantation. J Clin Virol 2006; 37: 52–56.
Fujimaki K, Mori T, Kida A, Tanaka M, Kawai N, Matsushima T et al. Human herpesvirus 6 meningoencephalitis in allogeneic hematopoietic stem cell transplant recipients. Int J Hematol 2006; 84: 432–437.
Seeley WW, Marty FM, Holmes TM, Upchurch K, Soiffer RJ, Antin JH et al. Post-transplant acute limbic encephalitis: clinical features and relationship to HHV6. Neurology 2007; 69: 156–165.
Muta T, Fukuda T, Harada M . Human herpesvirus-6 encephalitis in hematopoietic SCT recipients in Japan: a retrospective multicenter study. Bone Marrow Transplant 2009; 43: 583–585.
Mori Y, Miyamoto T, Nagafuji K, Kamezaki K, Yamamoto A, Saito N et al. High incidence of human herpes virus 6-associated encephalitis/myelitis following a second unrelated cord blood transplantation. Biol Blood Marrow Transplant 2010; 16: 1596–1602.
Hill JA, Koo S, Guzman Suarez BB, Ho VT, Cutler C, Koreth J et al. Cord-blood hematopoietic stem cell transplant confers an increased risk for human herpesvirus-6-associated acute limbic encephalitis: a cohort analysis. Biol Blood Marrow Transplant 2012; 18: 1638–1648.
Sakai R, Kanamori H, Motohashi K, Yamamoto W, Matsuura S, Fujita A et al. Long-term outcome of human herpesvirus-6 encephalitis after allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2011; 17: 1389–1394.
Scheurer ME, Pritchett JC, Amirian ES, Zemke NR, Lusso P, Ljungman P . HHV-6 encephalitis in umbilical cord blood transplantation: a systematic review and meta-analysis. Bone Marrow Transplant 2013; 48: 574–580.
Dewhurst S . Human herpesvirus type 6 and human herpesvirus type 7 infections of the central nervous system. Herpes 2004; 11: 105A–111A.
Ljungman P, de la Camara R, Cordonnier C, Einsele H, Engelhard D, Reusser P et al. Management of CMV, HHV-6, HHV-7 and Kaposi-sarcoma herpesvirus (HHV-8) infections in patients with hematological malignancies and after SCT. Bone Marrow Transplant 2008; 42: 227–240.
Tunkel AR, Glaser CA, Bloch KC, Sejvar JJ, Marra CM, Roos KL et al. The management of encephalitis: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis 2008; 47: 303–327.
Atsuta Y, Suzuki R, Yoshimi A, Gondo H, Tanaka J, Hiraoka A et al. Unification of hematopoietic stem cell transplantation registries in Japan and establishment of the TRUMP System. Int J Hematol 2007; 86: 269–274.
Atsuta Y . Introduction of Transplant Registry Unified Management Program 2 (TRUMP2): scripts for TRUMP data analyses, part I (variables other than HLA-related data). Int J Hematol 2016; 103: 3–10.
Shintaku M, Kaneda D, Tada K, Katano H, Sata T . Human herpes virus 6 encephalomyelitis after bone marrow transplantation: report of an autopsy case. Neuropathology 2010; 30: 50–55.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J et al. 1994 Consensus Conference on Acute GVHD Grading. Bone Marrow Transplant 1995; 15: 825–828.
Spitzer TR . Engraftment syndrome following hematopoietic stem cell transplantation. Bone Marrow Transplant 2001; 27: 893–898.
Kishi Y, Kami M, Miyakoshi S, Kanda Y, Murashige N, Teshima T et al. Early immune reaction after reduced-intensity cord-blood transplantation for adult patients. Transplantation 2005; 80: 34–40.
Narimatsu H, Terakura S, Matsuo K, Oba T, Uchida T, Iida H et al. Short-term methotrexate could reduce early immune reactions and improve outcomes in umbilical cord blood transplantation for adults. Bone Marrow Transplant 2007; 39: 31–39.
Gooley TA, Leisenring W, Crowley J, Storer BE . Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med 1999; 18: 695–706.
Fine JP, Gray RJ . A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc 1999; 94: 496–509.
Olson AL, Dahi PB, Zheng J, Devlin SM, Lubin M, Gonzales AM et al. Frequent human herpesvirus-6 viremia but low incidence of encephalitis in double-unit cord blood recipients transplanted without antithymocyte globulin. Biol Blood Marrow Transplant 2014; 20: 787–793.
Ogata M, Satou T, Kawano R, Takakura S, Goto K, Ikewaki J et al. Correlations of HHV-6 viral load and plasma IL-6 concentration with HHV-6 encephalitis in allogeneic stem cell transplant recipients. Bone Marrow Transplant 2010; 45: 129–136.
Uchida N, Wake A, Nakano N, Ishiwata K, Takagi S, Tsuji M et al. Mycophenolate and tacrolimus for graft-versus-host disease prophylaxis for elderly after cord blood transplantation: a matched pair comparison with tacrolimus alone. Transplantation 2011; 92: 366–371.
Zerr DM, Boeckh M, Delaney C, Martin PJ, Xie H, Adler AL et al. HHV-6 reactivation and associated sequelae after hematopoietic cell transplantation. Biol Blood Marrow Transplant 2012; 18: 1700–1708.
Manichanh C, Grenot P, Gautheret-Dejean A, Debre P, Huraux JM, Agut H . Susceptibility of human herpesvirus 6 to antiviral compounds by flow cytometry analysis. Cytometry 2000; 40: 135–140.
De Clercq E, Naesens L, De Bolle L, Schols D, Zhang Y, Neyts J . Antiviral agents active against human herpesviruses HHV-6, HHV-7 and HHV-8. Rev Med Virol 2001; 11: 381–395.
Raffi F, Taburet AM, Ghaleh B, Huart A, Singlas E . Penetration of foscarnet into cerebrospinal fluid of AIDS patients. Antimicrob Agents Chemother 1993; 37: 1777–1780.
Morita D, Hirabayashi K, Katsuyama Y, Morokawa H, Motobayashi M, Kurata T et al. Viral load and ganciclovir (GCV) concentration in cerebrospinal fluid of patients successfully treated with GCV or valGCV for human herpesvirus 6 encephalitis/myelitis following umbilical cord blood transplantation. Transpl Infect Dis 2016; 18: 773–776.
Pellett PE, Ablashi DV, Ambros PF, Agut H, Caserta MT, Descamps V et al. Chromosomally integrated human herpesvirus 6: questions and answers. Rev Med Virol 2012; 22: 144–155.
Tanaka-Taya K, Sashihara J, Kurahashi H, Amo K, Miyagawa H, Kondo K et al. Human herpesvirus 6 (HHV-6) is transmitted from parent to child in an integrated form and characterization of cases with chromosomally integrated HHV-6 DNA. J Med Virol 2004; 73: 465–473.
Leong HN, Tuke PW, Tedder RS, Khanom AB, Eglin RP, Atkinson CE et al. The prevalence of chromosomally integrated human herpesvirus 6 genomes in the blood of UK blood donors. J Med Virol 2007; 79: 45–51.
Acknowledgements
We express our appreciation for the work of all of the physicians and data managers who contributed valuable data to the JSHCT and JDCHCT. We also thank members of the Transplant Registry Unified Management committees for their data management. This work was supported in part by the Practical Research Project for Allergic Diseases and Immunology from the Japan Agency for Medical Research and Development, AMED (to YA) and Research Program on Practical Research for Innovative Cancer Control from AMED (to TF).
Author contributions
MO designed the study, analyzed the data and wrote the manuscript. KO, TI and KT designed the study, analyzed the data and revised the manuscript. HK, TK, YU, TM, HH, HO, TE, TU and TM collected data and revised the manuscript. TI revised the manuscript and was responsible for data management for the JDCHCT. YA managed the registry database and revised the manuscript. TF designed the study, revised the manuscript and was responsible for the Complication Working Group.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Bone Marrow Transplantation website
Rights and permissions
About this article
Cite this article
Ogata, M., Oshima, K., Ikebe, T. et al. Clinical characteristics and outcome of human herpesvirus-6 encephalitis after allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 52, 1563–1570 (2017). https://doi.org/10.1038/bmt.2017.175
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2017.175
- Springer Nature Limited
We’re sorry, something doesn't seem to be working properly.
Please try refreshing the page. If that doesn't work, please contact support so we can address the problem.