Abstract
Background
Increased mortality due to type 2 diabetes mellitus (T2DM) has been associated with renal and/or cardiovascular dysfunction. Dipeptidyl dipeptidase-4 inhibitors (iDPP-4s) may exert cardioprotective effects through their pleiotropic actions via glucagon-like peptide 1-dependent mechanisms. In this study, the pharmacological profile of a new iDPP-4 (LASSBio-2124) was investigated in rats with cardiac and renal dysfunction induced by T2DM.
Methods
T2DM was induced in rats by 2 weeks of a high-fat diet followed by intravenous injection of streptozotocin. Metabolic disturbance and cardiac, vascular, and renal dysfunction were analyzed in the experimental groups.
Results
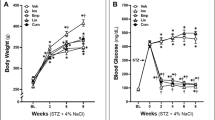
Sitagliptin and LASSBio-2124 administration after T2DM induction reduced elevated glucose levels to 319.8 ±13.2 and 279.7 ± 17.8 mg/dL, respectively (p<0.05). LASSBio-2124 also lowered the cholesterol and triglyceride levels from 76.8 ±8.0 to 42.7 ± 3.2 mg/dL and from 229.7 ±25.4 to 100.7 ± 17.1 mg/dL, in diabetic rats. Sitagliptin and LASSBio-2124 reversed the reduction of the plasma insulin level. LASSBio-2124 recovered the increased urinary flow in diabetic animals and reduced 24-h proteinuria from 23.7 ±1.5 to 13.3 ±2.8 mg (p<0.05). It also reduced systolic and diastolic left-ventricular dysfunction in hearts from diabetic rats.
Conclusion
The effects of LASSBio-2124 were superior to those of sitagliptin in the cardiovascular systems of T2DM rats. This new prototype showed promise for the avoidance of comorbidities in a T2DM experimental model, and thus may constitute an innovative therapeutic agent for the treatment of these conditions in the clinical field in future.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Jia G, Whaley-Connell A, Sowers JR. Diabetic cardiomyopathy: a hyperglycaemia- and insulin-resistance-induced heart disease. Diabetologia 2018;61:21–8.
Jia G, DeMarco VG, Sowers JR. Insulin resistance and hyperinsulinaemia in diabetic cardiomyopathy. Nat Rev Endocrinol 2016;12:144–53.
Jia G, Habibi J, DeMarco VG, Martinez-Lemus LA, Ma L, Whaley-Connell AT, et al. Endothelial mineralocorticoid receptor deletion prevents diet-induced cardiac diastolic dysfunction in females. Hypertension 2015;66:1159–67.
Orchard TJ, Secrest AM, Miller RG, Costacou T. In the absence of renal disease, 20 year mortality risk in type 1 diabetes is comparable to that of the general population: a report from the Pittsburgh Epidemiology of Diabetes Complications Study. Diabetologia 2010;53:2312–9.
Wen HL, Liang ZS, Zhang R, Yang K. Anti-inflammatory effects of triptolide improve left ventricular function in a rat model of diabetic cardiomyopathy. Cardiovasc Diabetol 2013;12:50.
Duran-Salgado MB, Rubio-Guerra AF. Diabetic nephropathy and inflammation. World J Diabetes 2014;5:393–8.
Kirino Y, Sato Y, Kamimoto T, Kawazoe K, Minakuchi K, Nakahori Y. Interrelationship of dipeptidyl peptidase IV (DPP4) with the development of diabetes, dyslipidaemia and nephropathy: a streptozotocin-induced model using wild-type and DPP4-deficient rats. J Endocrinol 2009;200:53–61.
Sato Y, Koshioka S, Kirino Y, Kamimoto T, Kawazoe K, Abe S, et al. Role of dipeptidyl peptidase IV (DPP4) in the development of dyslipidemia: DPP4 contributes to the steroid metabolism pathway. Life Sci 2011;88:43–9.
Inzucchi SE, McGuire DK. New drugs for the treatment of diabetes: part II: incretin-based therapy and beyond. Circulation 2008;117:574–84.
Ussher JR, Drucker DJ. Cardiovascular actions of incretin-based therapies. Circ Res 2014;114:1788–803.
Avogaro A, de Kreutzenberg S, Fadini G. Dipeptidyl-peptidase 4 inhibition: linking metabolic control to cardiovascular protection. Curr Pharm Des 2014;20:2387–94.
dos Santos L, Salles TA, Arruda-Junior DF, Campos LC, Pereira AC, Barreto AL, et al. Circulating dipeptidyl peptidase IV activity correlates with cardiac dysfunction in human and experimental heart failure. Circ Heart Fail 2013;6:1029–38.
Bittle PA. The use of dipeptidyl peptidase-4 inhibitors in patients with type 2 diabetes & chronic kidney disease. Nurse Pract 2017;42:31–8.
Scott LJ. Sitagliptin: a review in type 2 diabetes. Drugs 2017;77:209–24.
Zapata-Sudo G, da Costa Nunes IK, Araujo JS, da Silva JS, Trachez MM, da Silva TF, et al. Synthesis, solubility, plasma stability, and pharmacological evaluation of novel sulfonylhydrazones designed as anti-diabetic agents. Drug Des Devei Ther 2016;10:2869–79.
L.M. Lima, E.J. Barreiro, R.T. Sudo, G. Zapata-Sudo, B.E.O. Alves, E. Reina, Compostos β-amino-N-acilidrazônicos e/ou β-amino-N-acilidrazidas, seus sais farmacêuticamente aceitáveis e derivados, inibidores de enzima dipeptidil peptidasa-4 (DPP4) com atividade anti-inflamatoria e/ou anti-citocina, composições farmacêuticas contendo os mesmos, e processo para sua produção. Patent Number BR 10 2018 070913 5. Insituto Nacional de Propiedade Industrial (INPI). Brazil 10-10-2018.
Srinivasan K, Viswanad B, Asrat L, Kaul CL, Ramarao P. Combination of high-fat diet-fed and low-dose streptozotocin-treated rat: a model for type 2 diabetes and pharmacological screening. Pharmacol Res 2005;52:313–20.
Mansor LS, Gonzalez ER, Cole MA, Tyler DJ, Beeson JH, Clarke K, et al. Cardiac metabolism in a new rat model of type 2 diabetes using high-fat diet with low dose streptozotocin. Cardiovasc Diabetol 2013;12:136.
da Reimer R, Grover GJ, Koetzner L, Gahler RJ, Juneja P, Lyon MR, et al. Sitagliptin reduces hyperglycemia and increases satiety hormone secretion more effectively when used with a novel polysaccharide in obese Zucker rats. J Nutr 2012;142:1812–20, doi:https://doi.org/10.3945/jn.112.163204.
Ali SM, Khalifa H, Mostafa DK, El Sharkawy A. Suppression of connective tissue growth factor mediates the renoprotective effect of Sitagliptin rather than Pioglitazone in type 2 diabetes mellitus. Life Sci 2016;153:180–7, doi:https://doi.org/10.1016/j.lfs.2016.03.043.
Saad MI, Kamel MA, Hanafi MY. Modulation of adipocytokines production and serum NEFA level by metformin, glimepiride, and sitagliptin in HFD/STZ diabetic rats. Biochem Res Int 2015;2015:, doi:https://doi.org/10.1155/2015/138134.
Liu L, Liu J, Wong WT, Tian XY, Lau CW, Wang YX, et al. Dipeptidyl peptidase 4 inhibitor sitagliptin protects endothelial function in hypertension through a glucagon-like peptide 1-dependent mechanism. Hypertension 2012;60:833–41, doi:https://doi.org/10.1161/HYPERTENSIONAHA.112.195115.
Zapata-Sudo G, Pereira SL, Beirai HJ, Kummerle AE, Raimundo JM, Antunes F, et al. Pharmacological characterization of (3-thienylidene)-3,4- methylenedioxybenzoylhydrazide: a novel muscarinic agonist with antihypertensive profile. Am J Hypertens 2010;23:135–41.
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, et al. Fiji: an open-source platform for biological-image analysis. Nat Methods 2012;9:676–82.
Gomes CL, Leao CL, Venturotti C, Barreira AL, Guimaraes G, Fonseca RJ, et al. The protective role of fucosylated chondroitin sulfate, a distinct glycosaminoglycan, in a murine model of streptozotocin-induced diabetic nephropathy. PLoS One 2014;9:e106929.
Landgraf SS, Silva LS, Peruchetti DB, Sirtoli GM, Moraes-Santos F, Portella VG, et al. 5-Lypoxygenase products are involved in renal tubulointerstitial injury induced by albumin overload in proximal tubules in mice. PLoS One 2014;9:e107549.
Grgic I, Campanholle G, Bijol V, Wang C, Sabbisetti VS, Ichimura T, et al. Targeted proximal tubule injury triggers interstitial fibrosis and glomerulosclerosis. Kidney Int 2012;82:172–83.
Portella VG, Silva-Filho JL, Landgraf SS, de Rico TB, Vieira MA, Takiya CM, et al. Sepsis-surviving mice are more susceptible to a secondary kidney insult. Crit Care Med 2013;41:1056–68.
Frazier KS, Seely JC, Hard GC, Betton G, Burnett R, Nakatsuji S, et al. Proliferative and nonproliferative lesions of the rat and mouse urinary system. Toxicol Pathol 2012;40:14S–86S.
Radi ZA, Khan NK. Comparative expression and distribution of c-fos, estrogen receptoralpha (eralpha), and p38alpha in the uterus of rats, monkeys, and humans. Toxicol Pathol 2006;34:327–35.
Alencar AK, Pereira SL, da Silva FE, Mendes LV, Cunha Vdo M, Lima LM, et al. N- acylhydrazone derivative ameliorates monocrotaline-induced pulmonary hypertension through the modulation of adenosine AA2R activity. Int J Cardiol 2014;173:154–62.
Ma H, Li SY, Xu P, Babcock SA, Dolence EK, Brownlee M, et al. Advanced glycation endproduct (AGE) accumulation and AGE receptor (RAGE) up- regulation contribute to the onset of diabetic cardiomyopathy. J Cell Mol Med 2009;13:1751–64.
Pappachan JM, Varughese GI, Sriraman R, Arunagirinathan G. Diabetic cardiomyopathy: pathophysiology, diagnostic evaluation and management. World J Diabetes 2013;4:177–89.
Doshi SM, Friedman AN. Diagnosis and management of type 2 diabetic kidney disease. Clin J Am Soc Nephrol 2017;12:1366–73.
Laakso M. Cardiovascular disease in type 2 diabetes from population to man to mechanisms: the Kelly West Award Lecture 2008. Diabetes Care 2010;33:442–9.
Stern MP, Haffner SM. Dyslipidemia in type II diabetes. Implications for therapeutic intervention. Diabetes Care 1991;14:1144–59.
Ammon HP. Hyper- and hypoinsulinemia in type-2 diabetes: what may be wrong in the secretory mechanism of the B-cell. Exp Clin Endocrinol Diabetes 1997;105(Suppl. 2):43–7.
Glaser B. Type 2 diabetes: hypoinsulinism, hyperinsulinism, or both? PLoS Med 2007;4:e148.
Hakim ZS, Patel BK, Goyal RK. Effects of chronic ramipril treatment in streptozotocin-induced diabetic rats. Indian J Physiol Pharmacol 1997;41:353–60.
Dominguez JH, Tang N, Xu W, Evan AP, Siakotos AN, Agarwal R, et al. Studies of renal injury III: lipid-induced nephropathy in type II diabetes. Kidney Int 2000;57:92–104.
Kasiske BL, O’Donnell MP, Schmitz PG, Kim Y, Keane WF. Renal injury of diet-induced hypercholesterolemia in rats. Kidney Int 1990;37:880–91.
Visconti L, Benvenga S, Lacquaniti A, Cernaro V, Bruzzese A, Conti G, et al. Lipid disorders in patients with renal failure: role in cardiovascular events and progression of chronic kidney disease. J Clin Transl Endocrinol 2016;6:8–14.
Dhananjayan R, Koundinya KS, Malati T, Kutala V. Endothelial dysfunction in type 2 diabetes mellitus. Indian J Clin Biochem 2016;31:372–9.
Aijada A, Dandona P. Effect of insulin on human aortic endothelial nitric oxide synthase. Metabolism 2000;49:147–50.
Chakravarthy U, Hayes RG, Stitt AW, McAuley E, Archer DB. Constitutive nitric oxide synthase expression in retinal vascular endothelial cells is suppressed by high glucose and advanced glycation end products. Diabetes 1998;47:945–52.
Ding Y, Vaziri ND, Coulson R, Kamanna VS, Roh DD. Effects of simulated hyperglycemia, insulin, and glucagon on endothelial nitric oxide synthase expression. Am J Physiol Endocrinol Metab 2000;279:E11–7.
Jeremy JY, Mikhailidis DP, Dandona P. Simulating the diabetic environment modifies in vitro prostacyclin synthesis. Diabetes 1983;32:217–21.
Forster W, Beitz J, Hoffmann P. Stimulation and inhibition of PGI2 synthetase activity by phospholipids (PL), cholesterol esters (CE), unesterified fatty acids (UFA) and lipoproteins (LDL and HDL). Artery 1980;8:494–500.
Mikhailidis DP, Mikhailidis AM, Barradas MA, Dandona P. Effect of nonesterified fatty acids on the stability of prostacyclin activity. Metabolism 1983;32:717–21.
Dandona P, Chaudhuri A, Aijada A. Endothelial dysfunction and hypertension in diabetes mellitus. Med Clin North Am 2004;88:911–31 x-xi.
Arai M. Advanced glycation endproducts and their receptor: do they play a role in diabetic cardiomyopathy? J Mol Cell Cardiol 2002;34:1305–8.
Al-Rasheed NM, Al-Rasheed NM, Hasan IH, Al-Amin MA, Al-Ajmi HN, Mohamad RA, et al. Simvastatin ameliorates diabetic cardiomyopathy by attenuating oxidative stress and inflammation in rats. Oxid Med Cell Longev 2017;2017:1092015.
Haudek SB, Taffet GE, Schneider MD, Mann DL. TNF provokes cardiomyocyte apoptosis and cardiac remodeling through activation of multiple cell death pathways. J Clin Invest 2007;117:2692–701.
Baker RG, Hayden MS, Ghosh S. NF-kappaB, inflammation, and metabolic disease. Cell Metab 2011;13:11–22.
Satoh-Asahara N, Sasaki Y, Wada H, Tochiya M, Iguchi A, Nakagawachi R, et al. A dipeptidyl peptidase-4 inhibitor, sitagliptin, exerts anti-inflammatory effects in type 2 diabetic patients. Metabolism 2013;62:347–51.
Xu K, Liu XF, Ke ZQ, Yao Q, Guo S, Liu C. Resveratrol modulates apoptosis and autophagy induced by high glucose and palmitate in cardiac cells. Cell Physiol Biochem 2018;46:2031–40.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alves, B.E.O., de Alencar, A.K.N., Gamba, L.E.R. et al. Reduction of cardiac and renal dysfunction by new inhibitor of DPP4 in diabetic rats. Pharmacol. Rep 71, 1190–1200 (2019). https://doi.org/10.1016/j.pharep.2019.07.005
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2019.07.005