Abstract
Background
Transient receptor potential ankyrin 1 (TRPA1) channels may have a role in migraine as some substances known to cause headache activate the channel. In the craniovascular system such activation causes a calcitonin gene-related peptide (CGRP)-dependent increase in meningeal blood flow. TRPA1 channels in the endothelium of cerebral arteries cause vasodilation when activated. The headache preventive substance feverfew inhibits activation of TRPA1 channels. In this study we aim to compare and characterize the effect of the TRPA1 agonist allyl isothiocyanate (AITC) on the diameter of rat dural and pial arteries in vivo.
Methods
The genuine closed-cranial window technique in rats was used to examine changes in dural and pial artery diameter and mean arterial blood pressure (MABP) after intracarotid infusion of AITC. Blockade experiments were performed by intravenous infusion of olcegepant, HC-030031, sumatriptan or capsazepine immediately after infusion of AITC, in four different groups of rats.
Results
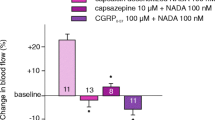
AITC caused a significant dilation of dural arteries, which was inhibited by HC-030031, olcegepant and sumatriptan, but not by capsazepine.
In pial arteries AITC caused a significant dilation, which was not inhibited by any of the pre-treatments, suggesting a poor penetration of the blood-brain barrier or autoregulation due to dimethyl sulfoxide (DMSO) mediated decrease in MABP during HC-030031 infusion.
AITC did not cause a significant change in MABP.
Conclusion
AITC causes dilation of dural arteries via a mechanism dependent on CGRP and TRPA1 that is sensitive to sumatriptan. AITC causes a small but significant dilation of pial arteries.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Clapham DE. TRP channels as cellular sensors. Nature 2003;426(6966):517–24.
Story GM, Peier AM, Reeve AJ, Eid SR, Mosbacher J, Hricik TR, et al. ANKTM1, a TRP-like channel expressed in nociceptive neurons, is activated by cold temperatures. Cell 2003;112(6):819–29.
Wang YY, Chang RB, Waters HN, McKemy DD, Liman ER. The nociceptor ion channel TRPA1 is potentiated and inactivated by permeating calcium ions. J Biol Chem 2008;283(47):32691–703.
Andersson DA, Gentry C, Moss S, Bevan S. Transient receptor potential A1 is a sensory receptor for multiple products of oxidative stress. J Neurosci 2008;28(10):2485–94.
Andre E, Campi B, Materazzi S, Trevisani M, Amadesi S, Massi D, et al. Cigarette smoke-induced neurogenic inflammation is mediated by alpha, beta-unsaturated aldehydes and the TRPA1 receptor in rodents. J Clin Invest 2008;118(7):2574–82.
Bandell M, Story GM, Hwang SW, Viswanath V, Eid SR, Petrus MJ, et al. Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin. Neuron 2004;41(6):849–57.
Bautista DM, Movahed P, Hinman A, Axelsson HE, Sterner O, Hogestatt ED, et al. Pungent products from garlic activate the sensory ion channel TRPA1. Proc Natl Acad Sci U S A 2005;102(34):12248–52.
Bautista DM, Jordt SE, Nikai T, Tsuruda PR, Read AJ, Poblete J, et al. TRPA1 mediates the inflammatory actions of environmental irritants and proalgesic agents. Cell 2006;124(6):1269–82.
Kunkler PE, Ballard CJ, Oxford GS, Hurley JH. TRPA1 receptors mediate environmental irritant-induced meningeal vasodilatation. Pain 2011;152(1):38–44.
Edelmayer RM, Le LN, Yan J, Wei X, Nassini R, Materazzi S, et al. Activation of TRPA1 on dural afferents: a potential mechanism of headache pain. Pain 2012;153(9):1949–58.
Nassini R, Materazzi S, Vriens J, Prenen J, Benemei S, De Siena G, et al. The’ headache tree’ via umbellulone and TRPA1 activates the trigeminovascular system. Brain: J Neurol 2012;135(Pt 2):376–90.
Materazzi S, Benemei S, Fusi C, Gualdani R, De Siena G, Vastani N, et al. Parthenolide inhibits nociception and neurogenic vasodilatation in the trigeminovascular system by targeting the TRPA1 channel. Pain 2013;154(12):2750–8.
Graepel R, Fernandes ES, Aubdool AA, Andersson DA, Bevan S, Brain SD. 4-oxo-2-nonenal (4-ONE): evidence of transient receptor potential ankyrin 1-dependent and -independent nociceptive and vasoactive responses in vivo. J Pharmacol Exp Ther 2011;337(1):117–24.
Bautista DM, Pellegrino M, Tsunozaki M. TRPA1: a gatekeeper for inflammation. Annu Rev Physiol 2013;75:181–200.
Lin YS, Hsu CC, Bien MY, Hsu HC, Weng HT, Kou YR. Activations of TRPA1 and P2X receptors are important in ROS-mediated stimulation of capsaicin-sensitive lung vagal afferents by cigarette smoke in rats. J Appl Physiol (Bethesda, Md: 1985) 2010;108(5):1293–303.
Julius D. TRP channels and pain. Annu Rev Cell Dev Biol 2013;29:355–84.
Raisinghani M, Zhong L, Jeffry JA, Bishnoi M, Pabbidi RM, Pimentel F, et al. Activation characteristics of transient receptor potential ankyrin 1 and its role in nociception. Am J Physiol Cell Physiol 2011;301(3):C587–600.
Kremeyer B, Lopera F, Cox JJ, Momin A, Rugiero F, Marsh S, et al. A gain-of-function mutation in TRPA1 causes familial episodic pain syndrome. Neuron 2010;66(5):671–80.
Huang D, Li S, Dhaka A, Story GM, Cao YQ. Expression of the transient receptor potential channels TRPV1, TRPA1 and TRPM8 in mouse trigeminal primary afferent neurons innervating the dura. Mol Pain 2012;8:66.
Earley S. Endothelium-dependent cerebral artery dilation mediated by transient receptor potential and Ca2+-activated K+ channels. J Cardiovasc Pharmacol 2011;57(2):148–53.
Gupta S, Bhatt DK, Boni LJ, Olesen J. Improvement of the closed cranial window model in rats by intracarotid infusion of signalling molecules implicated in migraine. Cephalalgia: Int J Headache 2010;30(1):27–36.
Dux M, Will C, Vogler B, Filipovic MR, Messlinger K. Meningeal blood flow is controlled by H2 S-NO crosstalk activating a HNO-TRPA1-CGRP signalling pathway. Br J Pharmacol 2016;173(3):431–45.
Jordt SE, Bautista DM, Chuang HH, McKemy DD, Zygmunt PM, Hogestatt ED, et al. Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature 2004;427(6971):260–5.
Everaerts W, Gees M, Alpizar YA, Farre R, Leten C, Apetrei A, et al. The capsaicin receptor TRPV1 is a crucial mediator of the noxious effects of mustard oil. Curr Biol 2011;21(4):316–21.
Sullivan MN, Gonzales AL, Pires PW, Bruhl A, Leo MD, Li W, et al. Localized TRPA1 channel Ca2+ signals stimulated by reactive oxygen species promote cerebral artery dilation. Sci Signal 2015;8(358) ra2.
Gerkens JF. Barbiturate inhibition of endothelium-dependent dilatation of blood-and Krebs-perfused rat tail arteries. Eur J Pharmacol 1987;134(3):293–301.
Beggs S, Liu XJ, Kwan C, Salter MW. Peripheral nerve injury and TRPV1-expressing primary afferent C-fibers cause opening of the blood-brain barrier. Mol Pain 2010;6(1):74.
Reid J, McCulloch J. Capsaicin and blood-brain barrier permeability. Neurosci Lett 1987;81(1–2):165–70.
Macintyre PD, Bhargava B, Hogg KJ, Gemmill J, Hillis WS. Effect of subcutaneous sumatriptan, a selective 5HT1 agonist, on the systemic, pulmonary, and coronary circulation. Circulation 1993;87(2):401–5.
Vanmolkot F, de Hoon J, Barrington P, Peck R, Dallow N, et al. Non-invasive assessment of selective 5-HT 1B/1D-receptor agonist-induced peripheral vascular effects in humans: comparison of different techniques. Eur J Clin Pharmacol 2002;58(5):303–8.
Kobari M, Fukuuchi Y, Tomita M, Tanahashi N, Konno S, Takeda H. Effects of sumatriptan on the cerebral intraparenchymal microcirculation in the cat. Br J Pharmacol 1993;110(4):1445–8.
Pagniez F, Valentin JP, Vieu S, Colpaert FC, John GW. Pharmacological analysis of the haemodynamic effects of 5-HT1B/D receptor agonists in the normotensive rat. Br J Pharmacol 1998;123(2):205–14.
Strassman AM, Levy D. Response properties of dural nociceptors in relation to headache. J Neurophysiol 2006;95(3):1298–306.
Hansen-Schwartz J, Hoel NL, Nilsson E, Tfelt-Hansen P, Edvinsson L. Endothelium-dependent relaxant responses to selective 5-HT1B/1D receptor agonists in the isolated middle cerebral artery of the rat. J Vasc Res 2003;40(6):561–6.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hansted, A.K., Bhatt, D.K., Olesen, J. et al. Effect of TRPA1 activator allyl isothiocyanate (AITC) on rat dural and pial arteries. Pharmacol. Rep 71, 565–572 (2019). https://doi.org/10.1016/j.pharep.2019.02.015
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2019.02.015