Abstract
Background
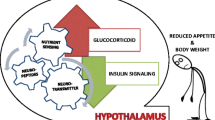
In depression, excessive glucocorticoid action may cause maladaptive brain changes, including in the pathways controlling energy metabolism. Insulin and glucagon-like peptide-1 (GLP-1), besides regulation of glucose homeostasis, also possess neurotrophic properties. Current study was aimed at investigating the influence of prenatal stress (PS) on insulin, GLP-1 and their receptor (IR and GLP-1R) levels in the hypothalamus. GLP-1 and GLP-1R were assayed also in the hippocampus and frontal cortex — brain regions mainly affected in depression. The second objective was to determine the influence of exendin-4 and insulin on CRH promoter gene activity in in vitro conditions.
Methods
Adult male PS rats were subjected to acute stress and/or received orally glucose. Levels of hormones and their receptors were assayed with ELISA method. In vitro studies were performed on mHypoA-2/12 hypothalamic cell line, stably transfected with CRH promoter coupled with luciferase.
Results
PS has reduced GLP-1 and GLP-1R levels, attenuated glucose-induced increase in insulin concentration and increased the amount of phosphorylated IR in the hypothalamus of animals subjected to additional stress stimuli, and also decreased the GLP-1R level in the hippocampus. In vitro studies demonstrated that insulin is capable of increasing CRH promoter activity in the condition of stimulation of the cAMP/PKA pathway in the applied cellular model.
Conclusion
Prenatal stress may act as a preconditioning factor, affecting the concentrations of hormones such as insulin and GLP-1 in the hypothalamus in response to adverse stimuli. The decreased GLP-1R level in the hippocampus could be linked with the disturbances in neuronal plasticity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- cAMP:
-
cyclic adenosine monophosphate
- CREB:
-
cAMP response elementbinding protein
- CRH:
-
Corticotropinreleasing hormone
- GLP-1glucagon:
-
like peptide-1
- GLP-1R:
-
glucagonlike peptide-1 receptor
- HPA:
-
hypothalamic — pituitary — adrenal axis
- IR:
-
insulin receptor
- PI3K:
-
phosphatidylinositol3-kinase
- PKA:
-
protein kinase A
References
Mainardi M, Fusco S, Grassi C. Modulation of hippocampal neural plasticity by glucose-related signaling. Neural Plast 2015;2015:, doi:https://doi.org/10.1155/2015/657928.
McEwen BS, Seeman T. Protective and damaging effects of mediators of stress: elaborating and testing the concepts of allostasis and allostatic load. Ann N Y Acad Sci 1999;896:30–47, doi:https://doi.org/10.1111/j.1749-6632.1999.tb08103.x.
Magariños AM, McEwen BS. Experimental diabetes in rats causes hippocampal dendritic and synaptic reorganization and increased glucocorticoid reactivity to stress. Proc Natl Acad Sci U S A 2000;97:11056–61, doi:https://doi.org/10.1073/pnas.97.20.11056.
Nyirenda MJ, Welberg LA, Seckl JR. Programming hyperglycaemia in the rat through prenatal exposure to glucocorticoids-fetal effect or maternal influence? J Endocrinol 2001;170:653–60.
Pan Y, Hong Y, Zhang QY, Kong LD. Impaired hypothalamic insulin signaling in CUMS rats: restored by icariin and fluoxetine through inhibiting CRF system. Psychoneuroendocrinology 2013;38:122–34, doi:https://doi.org/10.1016/j.psyneuen.2012.05.007.
Detka J, Kurek A, Basta-Kaim A, Kubera M, Lasoń W, Budziszewska B. Elevated brain glucose and glycogen concentrations in an animal model of depression. Neuroendocrinology 2014;100:178–90, doi:https://doi.org/10.1159/000368607.
Detka J, Kurek A, Kucharczyk M, Głombik K, Basta-Kaim A, Kubera M, et al. Brain glucose metabolism in an animal model of depression. Neuroscience 2015;295:198–208, doi:https://doi.org/10.1016/j.neuroscience.2015.03.046.
Sharma AN, Ligade SS, Sharma JN, Shukla P, Elased KM, Lucot JB. GLP-1 receptor agonist liraglutide reverses long-term atypical antipsychotic treatment associated behavioral depression and metabolic abnormalities in rats. Metab Brain Dis 2015;30:519–27, doi:https://doi.org/10.1007/s11011-014-9591-7.
Anderberg RH, Richard JE, Hansson C, Nissbrandt H, Bergquist F, Skibicka KP. GLP-1 is both anxiogenic and antidepressant; Divergent effects of acute and chronic GLP-1 on emotionality. Psychoneuroendocrinology 2016;65:54–66, doi:https://doi.org/10.1016/j.psyneuen.2015.11.021.
Gil-Lozano M, Romani-’Pérez M, Outeiriño-Iglesias V, Vigo E, González-Matías LC, Brubaker PL, et al. Corticotropin-releasing hormone and the sympathoadrenal system are major mediators in the effects of peripherally administered exendin-4 on the hypothalamic-pituitary-adrenal axis of male rats. Endocrinology 2014;155:2511–23, doi:https://doi.org/10.1210/en.2013-1718.
Bohringer A, Schwabe L, Richter S, Schachinger H. Intranasal insulin attenuates the hypothalamic-pituitary-adrenal axis response to psychosocial stress. Psychoneuroendocrinology 2008;33:1394–400, doi:https://doi.org/10.1016/j.psyneuen.2008.08.002.
Budziszewska B, Jaworska-Feil L, Tetich M, Basta-Kaim A, Kubera M, Leśkiewicz M, et al. Effect of antidepressant drugs on the human corticotropin-releasing-hormone gene promoter activity in neuro-2A cells. Pharmacol Rep 2002;54:711–6.
Seasholtz A, Thompson R, Douglas J. Identification of a cyclic adenosine monophosphate-responsive element in the rat corticotropin-releasing hormone gene. Mol Endocrinol 1988;12:1311–9, doi:https://doi.org/10.1210/mend-2-12-1311.
Morley-Fletcher S, Darnaudery M, Koehl M, Casolini P, Van Reeth O. Prenatal stress in rats predicts immobility behavior in the forced swim test effects of a chronic treatment with tianeptine. Brain Res 2003;989:246–51.
Detke MJ, Rickels M, Lucki I. Active behaviors in the rat forced swimming test differentially produced by serotonergic and noradrenergic antidepressants. Psychopharmacology (Berl) 1995;121:66–72.
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD, et al. Measurement of protein using bicinchoninic acid. Anal Chem 1985;150:76–85.
Hampf M, Gossen M. A protocol for combined photinus and renilla luciferase quantification compatible with protein assays. Anal Biochem 2006;356:94–9, doi:https://doi.org/10.1016/j.ab.2006.04.046.
Budziszewska B, Jaworska-Feil L, Basta-Kaim A, Kubera M, Leśkiewicz M, Regulska M, et al. The effect of antidepressant drugs on the HPA axis activity, glucocorticoid receptor level and FKBP51 concentration in prenatally stressed rats. Psychoneuroendocrinology 2009;34:822–32, doi:https://doi.org/10.1016/j.psyneuen.2008.12.012.
Zhang R, Packard BA, Tauchi M, D’Alessio DA, Herman JP. Glucocorticoid regulation of preproglucagon transcription and RNA stability during stress. Proc Natl Acad Sci U S A 2009;106:13636, doi:https://doi.org/10.1073/pnas.0907839106.
Mansur RB, Ahmed J, Cha DS, Woldeyohannes HO, Subramaniapillai M, Lovshin J, et al. Liraglutide promotes improvements in objective measures of cognitive dysfunction in individuals with mood disorders: A pilot, open-label study. J Affect Disord 2017;207:114–20, doi:https://doi.org/10.1016/j.jad.2016.09.056.
McClean PL, Gault VA, Harriott P, Hölscher C. Glucagon-like peptide-1 analogues enhance synaptic plasticity in the brain: a link between diabetes and Alzheimer’s disease. Eur J Pharmacol 2010;630:158–62, doi:https://doi.org/10.1016/j.ejphar.2009.12.023.
Rebosio C, Balbi M, Passalacqua M, Ricciarelli R, Fedele E. Presynaptic GLP-1 receptors enhance the depolarization-evoked release of glutamate and GABA in the mouse cortex and hippocampus. Biofactors 2018;44:148–57, doi:https://doi.org/10.1002/biof.1406.
Gumuslu E, Mutlu O, Celikyurt IK, Ulak G. Exenatide enhances cognitive performance and upregulates neurotrophic factor gene expression levels in diabetic mice. Fundam Clin Pharmacol 2016;30:376–84, doi:https://doi.org/10.1111/fcp.12192.
Kucharczyk M, Kurek A, Pomierny B, Detka J, Papp M, Tota K. The reduced level of growth factors in an animal model of depression is accompanied by regulated necrosis in the frontal cortex but not in the hippocampus. Psychoneuroendocrinology 2018;94:121–33, doi:https://doi.org/10.1016/j.psyneuen.2018.05.008.
Obici S, Zhang BB, Karkanias G, Rossetti L. Hypothalamic insulin signaling is required for inhibition of glucose production. Nat Med 2002;8:1376–82, doi: https://doi.org/10.1038/nm798.
Guardiola-Diaz HM, Kolinske JS, Gates LH, Seasholtz AF. Negative glucorticoid regulation of cyclic adenosine 3′, 5′-monophosphate-stimulated corticotropin-releasing hormone-reporter expression in AtT-20 cells. Mol Endocrinol 1996;10:317–29, doi:https://doi.org/10.1210/mend.10.3.8833660.
Kageyama K, Yamagata S, Akimoto K, Sugiyama A, Murasawa S, Suda T. Action of glucagon-like peptide 1 and glucose levels on corticotropin-releasing factor and vasopressin gene expression in rat hypothalamic 4B cells. Mol Cell Endocrinol 2012;362:221–6, doi:https://doi.org/10.1016/j.mce.2012.06.023.
Ahmad F, Lindh R, Tang Y, Weston M, Degerman E, Manganiello VC. Insulin-induced formation of macromolecular complexes involved in activation of cyclic nucleotide phosphodiesterase 3B (PDE3B) and its interaction with PKB. Biochem J 2007;404:257–68, doi:https://doi.org/10.1042/BJ20060960.
Fruehwald-Schultes B, Kern W, Bong W, Wellhoener P, Kerner W, Born J, et al. Supraphysiological hyperinsulinemia acutely increases hypothalamic-pituitary-adrenal secretory activity in humans. J Clin Endocrinol Metab 1999;84:3041–6, doi:https://doi.org/10.1210/jcem.84.9.5953.
Fruehwald-Schultes B, Kern W, Born J, Fehm HL, Peters A. Hyperinsulinemia causes activation of the hypothalamus-pituitary-adrenal axis in humans. Int J Obes Relat Metab Disord 2001;25(Suppl. 1):S38–40, doi:https://doi.org/10.1038/sj.ijo.0801695.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Detka, J., Ślusarczyk, J., Kurek, A. et al. Hypothalamic insulin and glucagon-like peptide-1 levels in an animal model of depression and their effect on corticotropin-releasing hormone promoter gene activity in a hypothalamic cell line. Pharmacol. Rep 71, 338–346 (2019). https://doi.org/10.1016/j.pharep.2018.11.001
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2018.11.001