Abstract
Background
Inflammatory bowel disease (IBD) is an important cause of chronic disability in humans.
Methods
We characterized a model of chronic IBD in young male Wistar rats by administering dextran sodium sulfate (DSS: 0%, 0.25%, 0.5%, or 1% in drinking water) for six weeks, with 0.5% DSS for twelve weeks, following DSS cessation or together with treatment with sulfasalazine for the last 6 weeks. We measured gastrointestinal characteristics including stool consistency, blood in stools, small intestine and colon length, intestinal transit and permeability, and gut microbiota, as well as extra-intestinal parameters including oral glucose tolerance, systolic blood pressure, fat and lean mass, and left ventricular stiffness.
Results
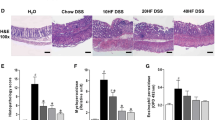
At 6 weeks, 0.25–1% DSS produced gastrointestinal changes as diarrhea and blood in stools. At 12 weeks, 0.5% DSS produced chronic and sustained gastrointestinal changes, with marked infiltration of inflammatory cells throughout the gastrointestinal tract and crypt distortion. Firmicutes increased and Bacteroidetes and Actinobacteria decreased in DSS-treated rats. Changes were reversed by DSS cessation or sulfasalazine treatment. Gastrointestinal permeability and extra-intestinal parameters did not change, so DSS changes were limited to the gastrointestinal tract.
Conclusion
Chronic 0.5% DSS produces selective and reversible gastrointestinal changes, providing an improved chronic model in rats that mimics human IBD for testing new interventions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Mowat C, Cole A, Windsor A, Ahmad T, Arnott I, Driscoll R, et al. Guidelines for the management of inflammatory bowel disease in adults. Gut 2011;60:571–607.
Kiesler P, Fuss IJ, Strober W. Experimental models of inflammatory bowel diseases. Cell Mol Gastroenterol Hepatol 2015;1:154–70.
Eichele DD, Kharbanda KK. Dextran sodium sulfate colitis murine model: an indispensable tool for advancing our understanding of inflammatory bowel diseases pathogenesis. World J Gastroenterol 2017;23:6016–29.
Perse M, Cerar A. Dextran sodium sulphate colitis mouse model: traps and tricks. J Biomed Biotechnol 2012(2012) 718617.
Chassaing B, Aitken JD, Malleshappa M, Vijay-Kumar M. Dextran sulfate sodium (DSS)-induced colitis in mice. Curr Protoc Immunol 2014;104:25 Unit 15.
Verstockt B, Ferrante M, Vermeire S, Van Assche G. New treatment options for inflammatory bowel diseases. J Gastroenterol 2018;53:585–90.
Ghattamaneni NKR, Panchal SK, Brown L. Nutraceuticals in rodent models as potential treatments for human inflammatory bowel disease. Pharmacol Res 2018;132:99–107.
Vasina V, Broccoli M, Ursino MG, Canistro D, Valgimigli L, Soleti A, et al. Non-peptidyl low molecular weight radical scavenger IAC attenuates DSS-induced colitis in rats. World J Gastroenterol 2010;16:3642–50.
Panchal SK, Poudyal H, Iyer A, Nazer R, Alam MA, Diwan V, et al. High-carbohydrate, high-fat diet-induced metabolic syndrome and cardiovascular remodeling in rats. J Cardiovasc Pharmacol 2011;57:611–24.
Capasso R, Orlando P, Pagano E, Aveta T, Buono L, Borrelli F, et al. Palmitoylethanolamide normalizes intestinal motility in a model of post-inflammatory accelerated transit: involvement of CB1 receptors and TRPV1 channels. Br J Pharmacol 2014;171:4026–37.
Wanyonyi S, du Preez R, Brown L, Paul NA, Panchal SK. Kappaphycus alvarezii as a food supplement prevents diet-induced metabolic syndrome in rats. Nutrients 2017;9:1261.
Farhadi A, Keshavarzian A, Holmes EW, Fields J, Zhang L, Banan A. Gas chromatographic method for detection of urinary sucralose: application to the assessment of intestinal permeability. J Chromatogr B Analyt Technol Biomed Life Sci 2003;784:145–54.
Rtibi K, Grami D, Wannes D, Selmi S, Amri M, Sebai H, et al. Ficus carica aqueous extract alleviates delayed gastric emptying and recovers ulcerative colitis-enhanced acute functional gastrointestinal disorders in rats. J Ethnopharmacol 2018;224:242–9.
Llewellyn SR, Britton GJ, Contijoch EJ, Vennaro OH, Mortha A, Colombel JF, et al. Interactions between diet and the intestinal microbiota alter intestinal permeability and colitis severity in mice. Gastroenterology 2018;154:1037–46 e2.
Marin M, Maria Giner R, Rios JL, Recio MC. Intestinal anti-inflammatory activity of ellagic acid in the acute and chronic dextrane sulfate sodium models of mice colitis. J Ethnopharmacol 2013;150:925–34.
Randhawa PK, Singh K, Singh N, Jaggi AS. A review on chemical-induced inflammatory bowel disease models in rodents. Korean J Physiol Pharmacol 2014;18:279–88.
Gaudio E, Taddei G, Vetuschi A, Sferra R, Frieri G, Ricciardi G, et al. Dextran sulfate sodium (DSS) colitis in rats: clinical, structural, and ultrastructural aspects. Dig Dis Sci 1999;44:1458–75.
Dong Y, Yang C, Wang Z, Qin Z, Cao J, Chen Y. The injury of serotonin on intestinal epithelium cell renewal of weaned diarrhoea mice. Eur J Histochem 2016;60:2689.
DeVoss J, Diehl L. Murine models of inflammatory bowel disease (IBD): challenges of modeling human disease. Toxicol Pathol 2014;42:99–110.
Funderburg NT, Stubblefield Park SR, Sung HC, Hardy G, Clagett B, Ignatz-Hoover J, et al. Circulating CD4+ and CD8+ T cells are activated in inflammatory bowel disease and are associated with plasma markers of inflammation. Immunology 2013;140:87–97.
Yuan B, Zhou S, Lu Y, Liu J, Jin X, Wan H, et al. Changes in the expression and distribution of claudins, increased epithelial apoptosis, and a mannan-binding lectin-associated immune response lead to barrier dysfunction in dextran sodium sulfate-induced rat colitis. Gut Liver 2015;9:734–40.
Kim TI. The role of barrier dysfunction and change of claudin expression in inflammatory bowel disease. Gut Liver 2015;9:699–700.
Kodani M, Fukui H, Tomita T, Oshima T, Watari J, Miwa H. Association between gastrointestinal motility and macrophage/mast cell distribution in mice during the healing stage after DSS-induced colitis. Mol Med Rep 2018;17:8167–72.
Wadie W, Abdel-Aziz H, Zaki HF, Kelber O, Weiser D, Khayyal MT. STW 5 is effective in dextran sulfate sodium-induced colitis in rats. Int J Colorectal Dis 2012;27:1445–53.
Aviello G, Scalisi C, Fileccia R, Capasso R, Romano B, Izzo AA, et al. Inhibitory effect of caffeic acid phenethyl ester, a plant-derived polyphenolic compound, on rat intestinal contractility. Eur J Pharmacol 2010;640:163–7.
Hobson CH, Broom MF, Ferry D, Grindley R, Chadwick VS. Involvement of M1 cholinergic receptors and enteric nerves in the spasmogenic activity of bacterial N-formyl oligopeptides on guinea-pig ileum. Aliment Pharmacol Ther 1988;2:311–6.
Becker C, Neurath MF, Wirtz S. The intestinal microbiota in inflammatory bowel disease. ILAR J 2015;56:192–204.
Gagnon M, Vimont A, Darveau A, Fliss I, Jean J. Study of the ability of bifidobacteria of human origin to prevent and treat rotavirus infection using colonic cell and mouse models. PLoS One 2016;11:e0164512.
Vasquez N, Suau A, Magne F, Pochart P, Pelissier MA. Differential effects of Bifidobacterium pseudolongum strain Patronus and metronidazole in the rat gut. Appl Environ Microbiol 2009;75:381–6.
Sasajima N, Ogasawara T, Takemura N, Fujiwara R, Watanabe J, Sonoyama K. Role of intestinal Bifidobacterium pseudolongum in dietary fructo-oligosaccharide inhibition of 2,4-dinitrofluorobenzene-induced contact hypersensitivity in mice. Br J Nutr 2010;103:539–48.
Osaka T, Moriyama E, Arai S, Date Y, Yagi J, Kikuchi J, et al. Meta-analysis of fecal microbiota and metabolites in experimental colitic mice during the inflammatory and healing phases. Nutrients 2017;9:1329.
Yu Q, Yuan L, Deng J, Yang Q. Lactobacillus protects the integrity of intestinal epithelial barrier damaged by pathogenic bacteria. Front Cell Infect Microbiol 2015;5:26.
Heidarian F, Noormohammadi Z, Asadzadeh Aghdaei H, Alebouyeh M. Relative abundance of Streptococcus spp. and its association with disease activity in inflammatory bowel disease patients compared with controls. Arch Clin Infect Dis 2017;12:e57291.
Png CW, Linden SK, Gilshenan KS, Zoetendal EG, McSweeney CS, Sly LI, et al. Mucolytic bacteria with increased prevalence in IBD mucosa augment in vitro utilization of mucin by other bacteria. Am J Gastroenterol 2010;105:2420–8.
Zheng H, Chen M, Li Y, Wang Y, Wei L, Liao Z, et al. Modulation of gut microbiome composition and function in experimental colitis treated with sulfasalazine. Front Microbiol 2017;8:1703.
Paik J, Fierce Y, Treuting PM, Brabb T, Maggio-Price L. High-fat diet-induced obesity exacerbates inflammatory bowel disease in genetically susceptible Mdr1a−/− male mice. J Nutr 2013;143:1240–7.
Zulian A, Cancello R, Ruocco C, Gentilini D, Di Blasio AM, Danelli P, et al. Differences in visceral fat and fat bacterial colonization between ulcerative colitis and Crohn’s disease. An in vivo and in vitro study. PLoS One 2013;8:e78495.
Pac-Kozuchowska E, Krawiec P, Mroczkowska-Juchkiewicz A, Pawlowska-Kamieniak A, Kominek K. Inflammatory and lipid-associated markers of cardiovascular diseases in children with first exacerbation of inflammatory bowel disease. Med Sci Monit 2016;22:1534–9.
Ott C, Scholmerich J. Extraintestinal manifestations and complications in IBD. Nat Rev Gastroenterol Hepatol 2013;10:585–95.
Filimon AM, Negreanu L, Doca M, Ciobanu A, Preda CM, Vinereanu D. Cardiovascular involvement in inflammatory bowel disease: dangerous liaisons. World J Gastroenterol 2015;21:9688–92.
Fan F, Galvin A, Fang L, White DA, Moore XL, Sparrow M, et al. Comparison of inflammation, arterial stiffness and traditional cardiovascular risk factors between rheumatoid arthritis and inflammatory bowel disease. J Inflamm (Lond) 2014;11:29.
Cappello M, Randazzo C, Bravata I, Licata A, Peralta S, Craxi A, et al. Liver function test abnormalities in patients with inflammatory bowel diseases: a hospital-based survey. Clin Med Insights Gastroenterol 2014;7:25–31.
Author information
Authors and Affiliations
Corresponding author
Supplementary data
Rights and permissions
About this article
Cite this article
Ghattamaneni, N.K.R., Panchal, S.K. & Brown, L. An improved rat model for chronic inflammatory bowel disease. Pharmacol. Rep 71, 149–155 (2019). https://doi.org/10.1016/j.pharep.2018.10.006
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2018.10.006