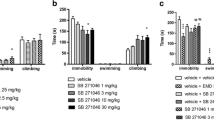
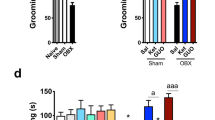
Abstract
Background
The removal of the olfactory bulbs has been attributed to behavioral changes and neuroplasticity manifesting themselves among others like increases in brain neurotrophin expression and neurogenesis. Earlier data presented that EMD386088, a 5-HT6 receptor partial agonist, exerts antidepressant-like properties after chronic administration in olfactory bulbectomy (OB) model as was it compared with amitriptyline (AMI). The aim of this study was to compare acute and chronic biochemical effects of EMD386088, administered in its antidepressant active (2.5 mg/kg) and non-active (1.25 mg/kg) doses, found in the open field test in OB rats, with those of AMI (10 mg/kg). The levels of 5-HT6 receptor protein and selected neurotrophins in prefrontal cortex (PFC) and hippocampus (Hp) of rats have been examined.
Methods
5-HT6 receptor protein and selected neurotrophins: brain-derived neurotrophic factor (BDNF), cAMP-response element binding protein (CREB), the product of the immediate early gene c-fos (cFos) protein levels were assessed using a Western blot analysis in PFC and Hp of bulbectomized rats after acute or chronic (14-day) EMD386088 or AMI intraperitoneal (ip) treatment.
Results
The acute treatment with EMD386088 caused significant increases in CREB and BDNF protein levels in PFC, and an increase in BDNF in Hp of OB rats, while AMI injection decreased CREB and did not change BDNF levels. After the chronic administration of EMD386088, the increasing levels of BDNF and CREB were still observed in PFC and Hp.
Conclusions
The antidepressant-like effect of EMD386088 may be associated with the neuroplasticity activation in PFC and Hp in rats.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Kohen R., Metcalf MA, Khan N, Druck T, Huebner K, Lachowicz JE, et al. Cloning, characterization, and chromosomal localization of a human 5-HT6 serotonin receptor. J Neurochem 1996;66:47–56.
Ruat M, Traiffort E, Arrang JM, Tardivel-Lacombe J, Diaz J, Leurs R, et al. A novel rat serotonin (5-HT6) receptor: molecular cloning, localization and stimulation of cAMP accumulation. Biochem Biophys Res Commun 1993;193:268–76.
Monsma Jr. FJ, Shen Y, Ward RP, Hamblin MW, Sibley DR. Cloning and expression of a novel serotonin receptor with high affinity for tricyclic psychotropic drugs. Mol Pharmacol 1993;43:320–7.
Hyung-Mun Y, Sunoh K, Hyun-Ji K, Evi K, Jae IK, Jae YS, et al. The novel cellular mechanism of human 5-HT6 receptor through an interaction with fyn. J Biol Chem 2007;282:5496–505, doi:https://doi.org/10.1074/jbc.M606215200.
Zhang YM, Zhang L, Wang Y, Sun YN, Guo Y, Du CX, et al. Activation and blockade of prelimbic 5-HT6receptors produce different effects on depressive-like behaviors in unilateral 6-hydroxydopamine-induced Parkinson’s rats. Neuropharmacology 2016;110:25–36, doi:https://doi.org/10.1016/j.neuropharm.2016.07.014.
Wang HY, Lu CW, Lin TY, Kuo JR, Wang SJ. WAY208466 inhibits glutamate release at hippocampal nerve terminals. Eur J Pharmacol 2016;781:117–27, doi:https://doi.org/10.1016/j.ejphar.2016.04.010.
Gérard C, Martres MP, Lefèvre K, Miquel MC, Vergé D, Lanfumey L, et al. Immune-localization of serotonin 5-HT6 receptor-like material in the rat central nervous system. Brain Res 1997;746:207–19, doi:https://doi.org/10.1016/S0006-8993(96)01224-3.
Ward RP, Hamblin MW, Lachowicz JE, Hoffman BJ, Sibley DR, Dorsa DM. Localization of serotonin subtype 6 receptor messenger RNA in the rat brain by in situ hybridization histochemistry. Neuroscience 1995;64:1105–11, doi:https://doi.org/10.1016/0306-4522(94)00439-C.
Yoshioka M, Matsumoto M, Togashi H, Mori K, Saito H. Central distribution and function of 5-HT6 receptor subtype in the rat brain. Life Sci 1998;62:1473–7, doi:https://doi.org/10.1016/S0024-3205(98)00092-7.
Takamori K, Yoshida S, Okuyama S. Repeated treatment with imipramine, fluvoxamine and tranylcypromine decreases the number of escape failures by activating dopaminergic systems in a rat learned helplessness test. Life Sci 2001;69:1919–26.
Boess FG, Monsma FJ, Carolo C, Meyer V, Rudler a, Zwingelstein C, et al. Functional and radioligand binding characterization of rat 5-HT6 receptors stably expressed in HEK293 cells. Neuropharmacology 1997;36:713–20, doi:https://doi.org/10.1016/S0028-3908(97)00019-1.
Woolley ML, Marsden CA, Fone KCF. 5-ht6 receptors. Curr Drug Targets CNS Neurol Disord 2004;3:59–79.
Svenningsson P, Tzavara ET, Qi H, Carruthers R, Witkin JM, Nomikos GG, et al. Biochemical and behavioral evidence for antidepressant-like effects of 5-HT6 receptor stimulation. J Neurosci 2007;27:4201–9, doi:https://doi.org/10.1523/JNEUROSCI.3110-06.2007.
Wesołowska A, Nikiforuk A, Stachowicz K. Anxiolytic-like and antidepressant-like effects produced by the selective 5-HT6 receptor antagonist SB-258585 after intrahippocampal administration to rats. Behav Pharmacol 2007;18:439–46, doi:https://doi.org/10.1097/FBP.0b013e3282d28f9c.
Hirano K, Piers TM, Searle KL, Miller ND, Rutter AR, Chapman PF. Procognitive 5-HT6 antagonists in the rat forced swimming test: potential therapeutic utility in mood disorders associated with Alzheimer’s disease. Life Sci 2009;84:558–62, doi:https://doi.org/10.1016/j.lfs.2009.01.019.
Carr GV, Schechter LE, Lucki I. Antidepressant and anxiolytic effects of selective 5-HT6 receptor agonists in rats. Psychopharmacology (Berl) 2011;213:499–507, doi:https://doi.org/10.1007/s00213-010-1798-7.
Nikiforuk A, Kos T, Wesołowska A. The 5-HT6 receptor agonist EMD 386088 produces antidepressant and anxiolytic effects in rats after intrahippocampal administration. Psychopharmacology (Berl) 2011;217:411–8, doi:https://doi.org/10.1007/s00213-011-2297-1.
Jastrzębska-Więsek M, Siwek A, Partyka A, Szewczyk B, Sowa-Kućma M, Wasik A, et al. Antidepressant-like activity of EMD 386088, a 5-HT6 receptor partial agonist, following systemic acute and chronic administration to rats. Naunyn Schmiedebergs Arch Pharmacol 2015;388:1079–88, doi:https://doi.org/10.1007/s00210-015-1141-2.
Jastrzębska-Więsek M, Siwek A, Partyka A, Antkiewicz-Michaluk L, Michaluk J, Romańska I, et al. Study of a mechanism responsible for potential antidepressant activity of EMD 386088, a 5-HT6 partial agonist in rats. Naunyn Schmiedebergs Arch Pharmacol 2016;389:839–49, doi:https://doi.org/10.1007/s00210-016-1245-3.
Jastrzębska-Więsek M, Siwek A, Kazek G, Nawieśniak B, Partyka A, Marcinkowska M, et al. Partial agonist efficacy of EMD386088, a 5-HT6 receptor ligand, in functional in vitro assays. Pharmacol Rep 2013;65:998–1005.
Mattsson C, Sonesson C, Sandahl A, Greiner HE, Gassen M, Plaschke J, et al. 2-Alkyl-3-(1,2,3,6-tetrahydropyridin-4-yl)-1H-indoles as novel 5-HT6 receptor agonists. Bioorg Med Chem Lett 2005;15:4230–4, doi:https://doi.org/10.1016/j.bmcl.2005.06.067.
Jastrzębska-Więsek M, Siwek A, Partyka A, Kołaczkowski M, Walczak M, Smolik M, et al. Study on the effect of EMD386088, a 5-HT6 receptor partial agonist, in enhancing the anti-immobility action of some antidepressants in rats. Naunyn Schmiedebergs Arch Pharmacol 2017, doi:https://doi.org/10.1007/s00210-017-1431-y.
Pittenger C, Duman RS. Stress, depression, and neuroplasticity: a convergence of mechanisms. Neuropsychopharmacology 2008;33:88–109, doi:https://doi.org/10.1038/sj.npp.1301574.
Krishnan V, Nestler EJ. The molecular neurobiology of depression. Nature 2008;455:894–902, doi:https://doi.org/10.1038/nature07455.
de Foubert G, O’Neill MJ, Zetterström TSC. Acute onset by 5-HT6-receptor activation on rat brain brain-derived neurotrophic factor and activity-regulated cytoskeletal-associated protein mRNA expression. Neuroscience 2007;147:778–85, doi:https://doi.org/10.1016/j.neuroscience.2007.04.045.
Duman RS, Monteggia LM. A neurotrophic model for stress-related mood disorders. Biol Psychiatry 2006;59:1116–27, doi:https://doi.org/10.1016/j.biopsych.2006.02.013.
Nibuya M, Nestler EJ, Duman RS. Chronic antidepressant administration increases the expression of cAMP response element binding protein (CREB) in rat hippocampus. J Neurosci 1996;16:2365–72, doi:https://doi.org/10.1016/S0006-3223(03)00117-3.
Nibuya M, Morinobu S, Duman RS. Regulation of BDNF and trkB mRNA in rat brain by chronic electroconvulsive seizure and antidepressant drug treatments. J Neurosci 1995;15:7539–47.
Tamburella A, Micale V, Navarria A, Drago F. Antidepressant properties of the 5-HT4receptor partial agonist, SL65.0155: Behavioral and neurochemical studies in rats. Prog Neuro-Psychopharmacol Biol Psychiatry 2009;33:1205–10, doi:https://doi.org/10.1016/j.pnpbp.2009.07.001.
Tamburella A, Micale V, Leggio GM, Drago F. The β3adrenoceptor agonist, amibegron (SR58611A) counteracts stress-induced behavioral and neurochemical changes. Eur Neuropsychopharmacol 2010;20:704–13, doi:https://doi.org/10.1016/j.euroneuro.2010.04.006.
Tamburella A, Leggio GM, Micale V, Navarria A, Bucolo C, Cicirata V, et al. Behavioural and neurochemical changes induced by stress-related conditions are counteracted by the neurokinin-2 receptor antagonist saredutant. Int J Neuropsychopharmacol 2013;16:813–23, doi:https://doi.org/10.1017/S1461145712000612.
Boess FG, Monsma FJ, Carolo C, Meyer V, Rudler A, Zwingelstein C, et al. Functional and radioligand binding characterization of rat 5-HT6 receptors stably expressed in HEK293 cells. Neuropharmacology 1997;36:713–20.
Gdula-Argasińska J, Czepiel J, Totoń-Żurańska J, Wołkow P, Librowski T, Czapkiewicz A, et al. n-3 Fatty acids regulate the inflammatory-state related genes in the lung epithelial cells exposed to polycyclic aromatic hydrocarbons. Pharmacol Rep 2016;68:319–28, doi:https://doi.org/10.1016/j.pharep.2015.09.001.
Song C, Leonard BE. The olfactory bulbectomised rat as a model of depression. Neurosci Biobehav Rev 2005;29:627–47, doi:https://doi.org/10.1016/j.neubiorev.2005.03.010.
Czéh B, Fuchs E, Wiborg O, Simon M. Animal models of major depression and their clinical implications. Prog Neuro-Psychopharmacol Biol Psychiatry 2016;64:293–310, doi:https://doi.org/10.1016/j.pnpbp.2015.04.004.
Jiménez-Sánchez L, Linge R, Campa L, Valdizán EM, Pazos A, Díaz Á, et al. Behavioral, neurochemical and molecular changes after acute deep brain stimulation of the infralimbic prefrontal cortex. Neuropharmacology 2016;108:91–102, doi:https://doi.org/10.1016/j.neuropharm.2016.04.020.
Yurttas C, Schmitz C, Turgut M, Strekalova T, Steinbusch HWM. The olfactory bulbectomized rat model is not an appropriate model for studying depression based on morphological/stereological studies of the hippocampus. Brain Res Bull 2017;134:128–35, doi:https://doi.org/10.1016/j.brainresbull.2017.07.010.
Ménard C, Hodes GE, Russo SJ. Pathogenesis of depresson: insights from human and rodent studies. Neuroscience 2016;321:138–62, doi:https://doi.org/10.1016/j.neuroscience.2015.05.053.Pathogenesis.
Harkin A, Kelly JP, Leonard BE. A review of the relevance and validity of olfactory bulbectomy as a model of depression. Clin Neurosci Res 2003;3:253–62, doi:https://doi.org/10.1016/S1566-2772(03)00087-2.
Roche M, Harkin A, Kelly JP. Chronic fluoxetine treatment attenuates stressor-induced changes in temperature, heart rate, and neuronal activation in the olfactory bulbectomized rat. Neuropsychopharmacology 2007;32:1312–20, doi:https://doi.org/10.1038/sj.npp.1301253.
Grecksch G, Zhou D, Franke C, Schröder U, Sabel B, Becker a, et al. Influence of olfactory bulbectomy and subsequent imipramine treatment on 5-hydroxytryptaminergic presynapses in the rat frontal cortex: behavioural correlates. Br J Pharmacol 1997;122:1725–31, doi:https://doi.org/10.1038/sj.bjp.0701530.
de Foubert G, Khundakar AA, Zetterström TS. Effects of repeated 5-HT6 receptor stimulation on BDNF gene expression and cell survival. Neurosci Lett 2013;553:211–5, doi:https://doi.org/10.1016/j.neulet.2013.08.029.
Takahashi K, Nakagawasai O, Nemoto W, Odaira T, Arai Y, Hisamitsu T, et al. Time-dependent role of prefrontal cortex and hippocampus on cognitive improvement by aripiprazole in olfactory bulbectomized mice. Eur Neuropsychopharmacol 2017;27:1000–10, doi:https://doi.org/10.1016/j.euroneuro.2017.08.071.
de Foubert G, O’Neill MJ, Zetterström TSC. Acute onset by 5-HT6-receptor activation on rat brain brain-derived neurotrophic factor and activity-regulated cytoskeletal-associated protein mRNA expression. Neuroscience 2007;147:778–85, doi:https://doi.org/10.1016/j.neuroscience.2007.04.045.
Jindal A, Mahesh R, Bhatt S. Type 4 phosphodiesterase enzyme inhibitor, rolipram rescues behavioral deficits in olfactory bulbectomy models of depression: involvement of hypothalamic-pituitary-adrenal axis, cAMP signaling aspects and antioxidant defense system. Pharmacol Biochem Behav 2015;132:20–32, doi:https://doi.org/10.1016/j.pbb.2015.02.017.
Grychowska K, Satała G, Kos T, Partyka A, Colacino E, Chaumont-Dubel S, et al. Novel 1H — pyrrolo[3,2-c]quinoline based 5-HT6 receptor antagonists with potential application for the treatment of cognitive disorders associated with Alzheimer’s disease. ACS Chem Neurosci 2016;7:972–83, doi:https://doi.org/10.1021/acschemneuro.6b00090.
Strekalova T, Bahzenova N, Trofimov A, Schmitt-Böhrer AG, Markova N, Grigoriev V, et al. Pro-neurogenic memory-enhancing and anti-stress effects of DF302, a novel fluorine gamma-carboline derivative with multi-target mechanism of action. Mol Neurobiol 2016;2017:1–15, doi:https://doi.org/10.1007/s12035-017-0745-6.
Wesołowska A, Nikiforuk A, Stachowicz K. Anxiolytic-like and antidepressant-like effects produced by the selective 5-HT6 receptor antagonist SB-258585 after intrahippocampal administration to rats. Behav Pharmacol 2007;18:439–46, doi:https://doi.org/10.1097/FBP.0b013e3282d28f9c.
Wesołowska A. Study into a possible mechanism responsible for the antidepressant-like activity of the selective 5-HT6 receptor antagonist SB-399885 in rats. Pharmacol Rep 2007;59:664–71.
Wesołowska A. The anxiolytic-like effect of the selective 5-HT6 receptor antagonist SB-399885: the impact of benzodiazepine receptors. Eur J Pharmacol 2008;580:355–60, doi:https://doi.org/10.1016/j.ejphar.2007.11.022.
Carr GV, Lucki I. The role of serotonin receptor subtypes in treating depression: a review of animal studies. Psychopharmacology (Berl) 2011;213:265–87, doi:https://doi.org/10.1007/s00213-010-2097-z.
De Foubert G, Carney SL, Robinson CS, Destexhe EJ, Tomlinson R, Hicks CA, et al. Fluoxetine-induced change in rat brain expression of brain-derived neurotrophic factor varies depending on length of treatment. Neuroscience 2004;128:597–604, doi:https://doi.org/10.1016/j.neuroscience.2004.06.054.
Mackowiak M, O’Neill MJ, Hicks CA, Bleakman D, Skolnick P. An AMPA receptor potentiator modulates hippocampal expression of BDNF: an in vivo study. Neuropharmacology 2002;43:1–10.
Hamon M, Doucet E, Lefevre K, Miquel MC, Lanfumey L, Insausti R, et al. Antibodies and antisense oligonucleotide for probing the distribution and putative functions of central 5-HT6receptors. Neuropsychopharmacology 1999;21:68S–76S, doi:https://doi.org/10.1016/S0893-133X(99)00044-5.
Schechter LE, Lin Q, Smith DL, Zhang G, Shan Q, Platt B, et al. Neuropharmacological profile of novel and selective 5-HT6 receptor agonists: WAY-181187 and WAY-208466. Neuropsychopharmacology 2008;33:1323–35, doi:https://doi.org/10.1038/sj.npp.1301503.
Neto FL, Borges G, Torres-Sanchez SA, Mico J, Berrocoso E. Neurotrophins role in depression neurobiology: a review of basic and clinical evidence. Curr Neuropharmacol 2011;9:530–52, doi:https://doi.org/10.2174/157015911798376262.
Dale E, Pehrson AL, Jeyarajah T, Li Y, Leiser SC, Smagin G, et al. Effects of serotonin in the hippocampus: how SSRIs and multimodal antidepressants might regulate pyramidal cell function. CNS Spectr 2016;21:143–61, doi:https://doi.org/10.1017/S1092852915000425.
Chang CH, Chen MC, Lu J. Effect of antidepressant drugs on the vmPFC-limbic circuitry. Neuropharmacology 2015;92:116–24, doi:https://doi.org/10.1016/j.neuropharm.2015.01.010.
Castrén E, Võikar V, Rantamäki T. Role of neurotrophic factors in depression. Curr Opin Pharmacol 2007;7:18–21, doi:https://doi.org/10.1016/j.coph.2006.08.009.
Sato A, Nakagawasai O, Tan-No K, Onogi H, Niijima F, Tadano T. Effect of non-selective dopaminergic receptor agonist on disrupted maternal behavior in olfactory bulbectomized mice. Behav Brain Res 2010;210:251–6, doi:https://doi.org/10.1016/j.bbr.2010.02.042.
Miyata S, Hamamura T, Lee Y, Miki M, Habara T, Oka T, et al. Contrasting Fos expression induced by acute reboxetine and fluoxetine in the rat forebrain: neuroanatomical substrates for the antidepressant effect. Psychopharmacology (Berl) 2005;177:289–95, doi:https://doi.org/10.1007/s00213-004-2072-7.
Kovács KJ. c-Fos as a transcription factor: a stressful (re)view from a functional map. Neurochem Int 1998;33:287–97, doi:https://doi.org/10.1016/S0197-0186(98)00023-0.
Santarelli L, Saxe M, Gross CT, Surget A, Battaglia F, Dulawa S, et al. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science (80-) 2003;301:805–9, doi:https://doi.org/10.1126/science.1083328.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jastrzębska-Więsek, M., Gdula-Argasińska, J., Siwek, A. et al. Chronic antidepressant-like effect of EMD386088, a partial 5-HT6 receptor agonist, in olfactory bulbectomy model may be connected with BDNF and/or CREB signalling pathway. Pharmacol. Rep 70, 1047–1056 (2018). https://doi.org/10.1016/j.pharep.2018.05.009
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2018.05.009