Abstract
Background
Tauopathies are a class of neurodegenerative illnesses associated with the aberrant accumulation of the tau protein in the brain. The best known out of these diseases is Alzheimer’s disease, a disorder where the microtubule associated tau protein becomes hyperphosphorylated (which lowers its binding affinity to microtubules) and accumulates inside neurons in the form of tangles. In this study, we attempt to find out whether brain ischemia may play an important role in tau protein gene alterations.
Methods
We have investigated the relationship between hippocampal ischemia and Alzheimer’s disease by means of a transient 10-min global brain ischemia in rats and determining the effect on Alzheimer’s disease tau protein gene expression during 2, 7 and 30 days post injury.
Results
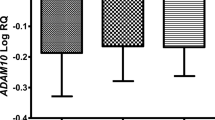
We found the significant overexpression of tau protein gene on the 2nd day, but on day’s 7 and 30 post-ischemia there a significant opposite tendency was observed.
Conclusion
The obtained results offer a novel insight into tau protein gene in regulating delayed neuronal death in the ischemic hippocampus. Finally, these findings further elucidate the long-term impact of brain ischemia on Alzheimer’s disease development.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Desmond D.W., Moroney JT, Sano M, Stern Y. Incidence of dementia after ischemic stroke: results of a longitudinal study. Stroke 2002;33:2254–60.
Leys D, Henon H, Mackowiak-Cordoliani MA, Pasquier F. Poststroke dementia. Lancet Neurol 2005;4:752–9.
Barra de la Tremblaye P, Plamondon H. Impaired conditioned emotional response and object recognition are concomitant to neuronal damage in the amygdale and perirhinal cortex in middle-aged ischemic rats. Behav Brain Res 2011;219:227–33.
Kiryk A, Pluta R, Figiel I, Mikosz M, Ułamek M, Niewiadomska G, et al. Transient brain ischemia due to cardiac arrest causes irreversible long-lasting cognitive injury. Behav Brain Res 2011;219:1–7.
Li J, Wang YJ, Zhang M, Fang CQ, Zhou HD. Cerebral ischemia aggravates cognitive impairment in a rat model of Alzheimer’s disease. Life Sci 2011;89:86–92.
Gemmell E, Bosomworth H, Allan L, Hall R, Khundakar A, Oakley AE, et al. Hippocampal neuronal atrophy and cognitive function in delayed poststroke and aging-related dementias. Stroke 2012;43:808–14.
Cohan CH, Neumann JT, Dave KR, Alekseyenko A, Binkert M, Stransky K, et al. Effect of cardiac arrest on cognitive impairment and hippocampal plasticity in middle-aged rats. PLoS One 2015;10:e0124918.
Portegies ML, Wolters FJ, Hofman A, Ikram MK, Koudstaal PJ, Ikram MA. Prestroke vascular pathology and the risk of recurrent stroke and poststroke dementia. Stroke 2016;47:2119–22.
Kudo T, Imaizumi K, Tanimukai H, Katayama T, Sato N, Nakamura Y, et al. Are cerebrovascular factors involved in Alzheimer’s disease? Neurobiol Aging 2000;21:215–24.
Delacourte A, Sergeant N, Wattez A, Maurage CA, Lebert F, Pasquier F, et al. Tau aggregation in the hippocampal formation: an ageing or a pathological process. Exp Gerontol 2002;37:1291–6.
German DC, Eisch AJ. Mouse models of Alzheimer’s disease: insight into treatment. Rev Neurosci 2004;15:353–69.
Pluta R. Resuscitation of the rabbit brain after acute complete ischemia lasting up to 1 h. Pathophysiological and pathomorphological observations. Resuscitation 1987;15:267–87.
Pluta R, Ułamek M, Jabłoński M. Alzheimer’s mechanisms in ischemic brain degeneration. Anat Rec 2009;292:1863–81.
Gendron TF, Petrucelli L. The role of tau in neurodegeneration. Mol Neurodegener 2009;4:13.
Dani M, Brooks DJ, Edison P. Tau imaging in neurodegenerative diseases. Eur J Nucl Med Mol Imaging 2016;43:1139–50.
Bekris LM, Yu CE, Bird TD, Tsuang DW. Genetics of Alzheimer disease. J Geriatr Psychiatr Neurol 2010;23:213–27.
Alzheimer’s Association. Alzheimer’s disease facts and figures. Alzheimers Dement 2015;2015(11):332–84.
Pooler AM, Usardi A, Evans CJ, Philpott KL, Noble W, Hanger DP. Dynamic association of tau with neuronal membranes is regulated by phosphorylation. Neurobiol Aging 2012;33:27–38.
Wen Y, Yang SH, Liu R, Perez EJ, Brun-Zinkernagel AM, Koulen P, et al. Cdk5 is involved in NFT-like tauopathy induced by transient cerebral ischemia in female rats. Biochim Biophys Acta 2007;1772:473–83.
Zhao JK, Guan FL, Duan SR, Zhao JW, Sun RH, Zhang LM, et al. Effect of focal mild hypothermia on expression of MMP-9, TIMP-1, Tau-1 and beta-APP in rats with cerebral ischaemia/reperfusion injury. Brain Inj 2013;27:1190–8.
Hanger DP, Wray S. Tau cleavage and tau aggregation in neurodegenerative disease. Biochem Soc Trans 2010;38:1016–20.
Marchesi VT. Alzheimer’s dementia begins as a disease of small blood vessels, damaged by oxidative-induced inflammation and dysregulated amyloid metabolism: implications for early detection and therapy. FASEB J 2011;25:5–13.
Henriksen K, Byrjalsen I, Christiansen C, Karsdal MA. Relationship between serum levels of tau fragments and clinical progression of Alzheimer’s disease. J Alzheimers Dis 2015;43:1331–41.
Mörtberg E, Zetterberg H, Nordmark J, Blennow K, Catry C, Decraemer H, et al. Plasma tau protein in comatose patients after cardiac arrest treated with therapeutic hypothermia. Acta Anaesthesiol Scand 2011;55:1132–8.
Randall J, Mörtberg E, Provuncher GK, Fournier DR, Duffy DC, Rubertsson S, et al. Tau proteins in serum predict neurological outcome after hypoxic brain injury from cardiac arrest: results of a pilot study. Resuscitation 2013;84:351–6.
Magnoni S, Esparza TJ, Conte V, Carbonara M, Carrabba G, Holtzman DM, et al. Tau elevations in the brain extracellular space correlate with reduced amyloid-β levels and predict adverse clinical outcomes after severe traumatic brain injury. Brain 2012;135:1268–80.
Ułamek-Kozioł M, Pluta R, Bogucka-Kocka A, Januszewski S, Kocki J, Czuczwar SJ. Brain ischemia with Alzheimer phenotype dysregulates Alzheimer’s disease-related proteins. Pharmacol Rep 2016;68:582–91.
Ułamek-Kozioł M, Pluta R, Januszewski S, Kocki J, Bogucka-Kocka A, Czuczwar SJ. Expression of Alzheimer’s disease risk genes in ischemic brain degeneration. Pharmacol Rep 2016;68:1345–9.
Yang SH, Simpkins JW. Ischemia-reperfusion promotes tau and beta-amyloid pathology and a progressive cognitive impairment. In: Pluta R, editor. Ischemia-reperfusion pathways in Alzheimer’s disease. New York: Nova Science Publishers; 2007 p. 113.
Pluta R, Misicka A, Barcikowska M, Spisacka S, Lipkowski AW, Januszewski S. Possible reverse transport of β-amyloid peptide across the blood-brain barrier. Acta Neurochir (Suppl) 2000;76:73–7.
Pluta R. Blood-brain barrier dysfunction and amyloid precursor protein accumulation in microvascular compartment following ischemia-reperfusion brain injury with 1-year survival. Acta Neurochir Suppl 2003;86:117–22.
Pluta R, Ułamek M, Januszewski S. Micro-blood-brain barrier openings and cytotoxic fragments of amyloid precursor protein accumulation in white matter after ischemic brain injury in long-lived rats. Acta Neurochir Suppl 2006;96:267–71.
Ułamek-Kozioł M, Kocki J, Bogucka-Kocka A, Petniak A, Gil-Kulik P, Januszewski S, et al. Dysregulation of autophagy, mitophagy, and apoptotic genes in the medial temporal lobe cortex in an ischemic model of Alzheimer’s disease. J Alzheimers Dis 2016;54:113–21.
Wiśniewski HM, Pluta R, Lossinsky AS, Mossakowski MJ. Ultrastructural studies of cerebral vascular spasm after cardiac arrest-related global cerebral ischemia in rats. Acta Neuropathol 1995;90:432–40.
Pluta R. The role of apolipoprotein E in the deposition of β-amyloid peptide during ischemia-reperfusion brain injury. A model of early Alzheimer’s disease. Ann NY Acad Sci 2000;903:324–34.
Pluta R, Jabłonski M, Ułamek-Kozioł M, Kocki J, Brzozowska J, Januszewski S, et al. Sporadic Alzheimer’s disease begins as episodes of brain ischemia and ischemically dysregulated Alzheimer’s disease genes. Mol Neurobiol 2013;48:500–15.
Pluta R, Lossinsky AS, Mossakowski MJ, Faso L, Wisniewski HM. Reassessment of a new model of complete cerebral ischemia in rats Method of induction of clinical death, pathophysiology and cerebrovascular pathology. Acta Neuropathol 1991;83:1–11.
Kocki J, Ułamek-Kozioł M, Bogucka-Kocka A, Januszewski S, Jabłonski M, Gil-Kulik P, et al. Dysregulation of amyloid-β protein precursor, β-secretase, presenilin 1 and 2 genes in the rat selectively vulnerable CA1 subfield of hippocampus following transient global brain ischemia. J Alzheimers Dis 2015;47:1047–56.
Pluta R, Kocki J, Ułamek-Kozioł M, Bogucka-Kocka A, Gil-Kulik P, Januszewski S, et al. Alzheimer-associated presenilin 2 gene is dysregulated in rat medial temporal lobe cortex after complete brain ischemia due to cardiac arrest. Pharmacol Rep 2016;68:155–61.
Pluta R, Kocki J, Ułamek-Kozioł M, Petniak A, Gil-Kulik P, Januszewski S, et al. Discrepancy in expression of β-secretase and amyloid-β protein precursor in Alzheimer-related genes in the rat medial temporal lobe cortex following transient global brain ischemia. J Alzheimers Dis 2016;51:1023–31.
Chomczynski P, Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 1987;162:156–9.
Yao L, Chen X, Tian Y, Lu X, Zhang P, Shi Q, et al. Selection of housekeeping genes for normalization of RT-PCR in hypoxic neural stem cells of rat in vitro. Mol Biol Rep 2012;39:569–76.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2T−ΔΔC method. Methods 2001;25:402–8.
Ułamek-Kozioł M, Kocki J, Bogucka-Kocka A, Januszewski S, Bogucki J, Czuczwar SJ, et al. Autophagy, mitophagy and apoptotic gene changes in the hippocampal CA1 area in a rat ischemic model of Alzheimer’s disease. Pharmacol Rep 2017;69:1289–94.
Quintanilla RA, Dolan PJ, Jin YN, Johnson GV. Truncated tau and A (cooperatively impair mitochondria in primary neurons. Neurobiol Aging 2012;33(619):e25–35.
Jarero-Basulto JJ, Luna-Munoz J, Mena R, Kristofikova Z, Ripova D, Perry G, et al. Proteolytic cleavage of polymeric tau protein by caspase-3: implications for Alzheimer’s disease. J Neuropathol Exp Neurol 2013;72:1145–61.
McIlwain DR, Berger T, Mak TW. Caspase functions in cell death and disease. Cold Spring Harb Perspect Biol 2013;5:a008656.
Means JC, Venkatesan A, Gerdes B, Fan JY, Bjes ES, Price JL. Drosophila spaghetti and double time link the circadian clock and light to caspases, apoptosis and tauopathy. PLoS Genet 2015;7:e1005171.
De Calignon A, Fox LM, Pitstick R, Carlson GA, Bacskai BJ, Spires-Jones TL, et al. Caspase activation precedes and leads to tangles. Nature 2010;464:1201–4.
Shackelford DA, Yeh RY. Dephosphorylation of tau during transient forebrain ischemia in the rat. Mol Chem Neuropathol 1998;34:103–20.
Mailliot C, Podevin-Dimster V, Rosenthal RE, Sergeant N, Delacourte A, Fiskum G, et al. Rapid tau protein dephosphorylation and differential rephosphorylation during cardiac arrest-induced cerebral ischemia and reperfusion. J Cereb Blood Flow Metab 2000;20:543–9.
Morioka M, Kawano T, Yano S, Kai Y, Tsuiki H, Yoshinaga Y, et al. Hyperphosphorylation at serine 199/202 of tau factor in the gerbil hippocampus after transient forebrain ischemia. Biochem Biophys Res Commun 2006;347:273–8.
Igbal K, Liu F, Gong CX, Grundke-Iqbal I. Tau in Alzheimer disease and related tauopathies. Curr Alzheimer Res 2010;7:656–64.
Pluta R, Ułamek-Kozioł M, Januszewski S, Czuczwar SJ. Dysregulation of Alzheimer’s disease-related genes and proteins following cardiac arrest. Folia Neuropathol 2017;55:283–8.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Joint first authors.
These authors are co-senior authors.
Rights and permissions
About this article
Cite this article
Pluta, R., Bogucka-Kocka, A., Ułamek-Kozioł, M. et al. Ischemic tau protein gene induction as an additional key factor driving development of Alzheimer’s phenotype changes in CA1 area of hippocampus in an ischemic model of Alzheimer’s disease. Pharmacol. Rep 70, 881–884 (2018). https://doi.org/10.1016/j.pharep.2018.03.004
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2018.03.004