Abstract
Background
In this study, we aimed to investigate the role of ATP-sensitive potassium (KATP) channel, Na+/K+-ATPase activity, and intracellular calcium levels on the vasodilatory effect of N-acetylcysteine (NAC) in thoracic aorta by using electrophysiological and molecular techniques.
Methods
Rat thoracic aorta ring preparations and cultured thoracic aorta cells were divided into four groups as control, 2 mM NAC, 5 mM NAC, and 10 mM NAC. Thoracic aorta rings were isolated from rats for measurements of relaxation responses and Na+/K+-ATPase activity. In the cultured thoracic aorta cells, we measured the currents of KATP channel, the concentration of intracellular calcium and mRNA expression level of KATP channel subunits (KCNJ8, KCNJ11, ABCC8 and ABCC9).
Results
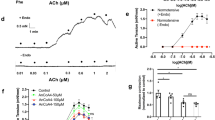
The relaxation rate significantly increased in all NAC groups compared to control. Similarly, Na+/K+-ATPase activity also significantly decreased in NAC groups. Outward KATP channel current significantly increased in all NAC groups compared to the control group. Intracellular calcium concentration decreased significantly in all groups with compared control. mRNA expression level of ABCC8 subunit significantly increased in all NAC groups compared to the control group. Pearson correlation analysis showed that relaxation rate was significantly associated with KATP current, intracellular calcium concentration, Na+/K+-ATPase activity and mRNA expression level of ABCC8 subunit.
Conclusion
Our findings suggest that NAC relaxes vascular smooth muscle cells through a direct effect on KATP channels, by increasing outward K+ flux, partly by increasing mRNA expression of KATP subunit ABCC8, by decreasing in intracellular calcium and by decreasing in Na+/K+-ATPase activity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Cotgreave IA. N-Acetylcysteine: pharmacological considerations and experimental and clinical applications. Adv Pharmacol 1997;38:205–27.
Radomska-Leśniewska DM, Skopińska-Rózewska E, Jankowska-Steifer E, Sobiecka M, Sadowska AM, Hevelke A, et al. N-Acetylcysteine inhibits IL-8 and MMP-9 release and ICAM-expression by bronchoalveolar cells from interstitial lung diseasepatients. Pharmacol Rep 2010;62(1):131–8.
Pawlas N, Malecki A. Neuroprotective effect of N-acetylcysteine in neurons exposed to arachidonic acid during simulated ischemia in vitro. Pharmacol Rep 2009;61(4):743–50.
Mondo CK, Zhang Y, de Macedo Possamai V, Miao Y, Schyvens CG, McKenzie, et al. N-acetylcysteine antagonizes the development but does not reverse ACTH-induced hypertension in the rat. Clin Exp Hypertens 2006;28(2):73–84.
Martina V, Masha A, Gigliardi VR, Brocato L, Manzato E, Berchio A, et al. Long-term N-acetylcysteine and L-arginine administration reduces endothelial activation and systolic blood pressure in hypertensive patients with type 2 diabetes. Diabetes Care 2008;31(5):940–4.
Olson LJ, Knych ET, Herzig TC, Drewett JG. Selective guanylyl cyclase inhibitor reverses nitric oxide-induced vasorelaxation. Hypertension 1997;29:254–61.
Khan SA, Higdon NR, Meisheri KD. Coronary vasorelaxation by nitroglycerin: involvement of plasmalemmal calcium-activated K+ channels and intracellular Ca++ stores. J Pharmacol Exp Ther 1998;284(3):838–46.
Ceron PI, Cremonez DC, Bendhack LM, Tedesco AC. The relaxation induced by S-nitroso-glutathione and S-nitroso-N-acetylcysteine in rat aorta is not related to nitric oxide production. J Pharmacol Exp Ther 2001;298(2):686–94.
Han WQ, Zhu DL, Wu LY, Chen QZ, Guo SJ, Gao PJ. N-acetylcysteine-induced vasodilation involves voltage-gated potassium channels in rat aorta. Life Sci 2009;84(21–22):732–7.
Cole WC, Clément-Chomienne O. ATP-sensitive K+ channels of vascular smooth muscle cells. J Cardiovasc Electrophysiol 2003; 14(1):94–103.
Bychkov R, Gollasch M, Ried C, Luft FC, Haller H. Effects of pinacidil on K+ channels in human coronary artery vascular smooth muscle cells. Am J Physiol 1997;273(1):161–71.
Gelband GH, McCullough JR. Modulation of rabbit aortic Ca(2+)-activated K+ channels by pinacidil, cromakalim, and glibenclamide. Am J Physiol 1993;264:1119–27.
Novakovic A, Pavlovic M, Stojanovic I, Milojevic P, Babic M, Ristic S, et al. Different K+ channels are involved in relaxation of arterial and venous graft induced by nicorandil. J Cardiovasc Pharmacol 2011;58(6):602–8.
Skou JC, Esmann M. The Na, K-ATPase. J Bioenerg Biomembr 1992;24(3):249–61.
McKenna MJ, Medved I, Goodman CA, Brown MJ, Bjorksten AR, Murphy KT, et al. N-Acetylcysteine attenuates the decline in muscle Na+,K+-pump activity and delays fatigue during prolonged exercise in humans. J Physiol 2006;576(Pt.1):279–88.
Girouard H, Chulak C, Wu L, Lejossec M, de Champlain J. N-acetylcysteine improves nitric oxide and alpha-adrenergic pathways in mesenteric beds of spontaneously hypertensive rats. Am J Hypertens 2003;16(7):577–84.
Mazzanti L, Rabini RA, Testa I, Bertoli E. Modifications induced by diabetes on the physicochemical and functional properties of erythrocyte plasma membrane. Eur J Clin Invest 1989; 19:84–9.
Lowry OH, Rosenbrough NJ, Farr AL. Protein measurement with the folin phenol reagent. J Biol Chem 1951;193:265–75.
Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch 1981;391(2):85–100.
Heinemann SH. Guide to data acquisition and analysis. In: Sakmann B, Neher E, editors. Single-channel recording. 2nd edition New York: Plenum; 1995. p. 53–90.
Colquhoun D, Sigworth FJ. Fitting and statistical analysis of single channel records. In: Sakmann B, Neher E, editors. Single-channel recording. 2nd edition New York: Plenum; 1995. p. 483–585.
Ashcroft FM. Ion channels and diseases. New York: Academic Press; 2000. p. 136–9.
Miyoshi H, Nakaya Y. Calcitonin gene-related peptide activates the K+ channels of vascular smooth muscle cells via adenylate cyclase. Basic Res Cardiol 1995;90:332–6.
Kleppisch T, Nelson MT. Adenosine activates ATP-sensitive potassium channels in arterial myocytes via A2 receptors and cAMP-dependent protein kinase. Proc Natl Acad Sci U S A 1995;92(26):12441–5.
Zafarullaha M, Li WQ, Sylvester J, Ahmad M. Molecular mechanisms of N-acetylcysteine actions. Cell Mol Life Sci 2003;60:6–20.
Dias FM, Ribeiro Jr. RF, Fernandes AA, Fiorim J, Travaglia TC, Vassallo DV, et al. Na+K+-ATPase activity and K+ channels differently contribute to vascular relaxation in male and female rats. PLoS One 2014;9(9):e106345.
Bełtowski J, Marciniak A, Wójcicka G, Górny D. Nitric oxide decreases renal medullary Na+, K+-ATPase activity through cyclic GMP-protein kinase G dependent mechanism. J Physiol Pharmacol 2003;54:191–210.
Muriel P, Sandoval G. Hepatic basolateral plasma high-affinity Ca2+-ATPase is inhibited by nitric oxide and peroxynitrite anion. J Appl Toxicol 2000;20:435–9.
Muriel P, Castañeda G, Ortega M, Noël F. Insights into the mechanism of erythrocyte Na+/K+-ATPase inhibition by nitric oxide and peroxynitrite anion. J Appl Toxicol 2003;23:275–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vezir, Ö., Çömelekoğlu, Ü., Sucu, N. et al. N-Acetylcysteine-induced vasodilatation is modulated by KATP channels, Na+/K+-ATPase activity and intracellular calcium concentration: An in vitro study. Pharmacol. Rep 69, 738–745 (2017). https://doi.org/10.1016/j.pharep.2017.03.019
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2017.03.019