Abstract
Background
Toyocamycin, an antibiotic agent isolated from Streptomyces species, has been shown to have anticancer and chemopreventive effects on various cancer cells. Until now, Toyocamycin-induced apoptosis has not been reported to be involved in the regulation between mitogen-activated protein kinases (MAPKs) and reactive oxygen species (ROS) production.
Methods
Cell viability assay, western blot, cell-cycle arrest, annexin V/propidium iodide assay, reactive oxygen species (ROS) production, mitochondrial membrane potential and intracellular Ca2+ flux were assayed.
Results
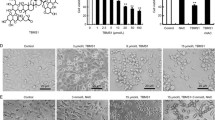
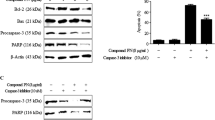
We investigated the apoptotic effect of Toyocamycin and the underlying molecular mechanism in prostate cancer PC-3 cells. Toyocamycin treatment resulted in reduced cell viability of PC-3 cells, but not of non-malignant RWPE-1 cells. Toyocamycin enhanced apoptosis, mitochondrial dysfunction, and ROS production in PC-3 cells. In addition, MAPK proteins were activated upon Toyocamycin treatment. The p38 and extracellular signal-regulated kinases (ERK) activities were regulated by ROS-mediated signaling pathway underlying the Toyocamycin-induced apoptosis. Pretreatment with N-acetyl-l-cysteine (NAC) recovered the Toyocamycin-induced mitochondrial dysfunction, ROS, and apoptosis. Additionally, p38 stimulated ROS production and inhibitory effects on ERK activation, while ERK inhibited the ROS production and had no effect on p38 activation.
Conclusion
ROS-mediated activation of p38/ERK partially contributes to Toyocamycin-induced apoptosis, and p38/ERK MAPKs regulate the ROS production in PC-3 cells.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dunn MW, Kazer MW. Prostate cancer overview. Semin Oncol Nurs 2011;27:241–50.
Ito K. Prostate cancer in Asian men. Nat Rev Urol 2014;11:197–212.
Feldman BJ, Feldman D. The development of androgen-independent prostate cancer. Nat Rev Cancer 2001;1:34–45.
Taplin ME, Balk SP. Androgen receptor: a key molecule in the progression of prostate cancer to hormone independence. J Cell Biochem 2004;91:483–90.
Kim KY, Yu SN, Lee SY, Chun SS, Choi YL, Park YM, et al. Salinomycin-induced apoptosis of human prostate cancer cells due to accumulated reactive oxygen species and mitochondrial membrane depolarization. Biochem Biophys Res Commun 2011;413:80–6.
Zhang DX, Gutterman DD. Mitochondrial reactive oxygen species-mediated signaling in endothelial cells. Am J Physiol Heart Circ Physiol 2007;292: H2023–31.
D’Autreaux B, Toledano MB. ROS as signalling molecules: mechanisms that generate specificity in ROS homeostasis. Nat Rev Mol Cell Biol 2007;8:813–24.
Zhang W, Liu HT. MAPK signal pathways in the regulation of cell proliferation in mammalian cells. Cell Res 2002;12:9–18.
Li JX, Shen YQ, Cai BZ, Zhao J, Bai X, Lu YJ, et al. Arsenic trioxide induces the apoptosis in vascular smooth muscle cells via increasing intracellular calcium and ROS formation. Mol Biol Rep 2010;37:1569–76.
Lepage C, Leger DY, Bertrand J, Martin F, Beneytout JL, Liagre B. Diosgenin induces death receptor-5 through activation of p38 pathway and promotes TRAIL-induced apoptosis in colon cancer cells. Cancer Lett 2011;301:193–202.
Sakon S, Xue X, Takekawa M, Sasazuki T, Okazaki T, Kojima Y, et al. NF-kappaB inhibits TNF-induced accumulation of ROS that mediate prolonged MAPK activation and necrotic cell death. EMBO J 2003;22:3898–909.
Cagnol S, Chambard JC. ERK and cell death: mechanisms of ERK-induced cell death-apoptosis, autophagy and senescence. FEBS J 2010;277:2–21.
Yen L, Magnier M, Weissleder R, Stockwell BR, Mulligan RC. Identification of inhibitors of ribozyme self-cleavage in mammalian cells via high-throughput screening of chemical libraries. RNA 2006;12:797–806.
Osada H, Sonoda T, Tsunoda K, Isono K. A new biological role of sangivamycin; inhibition of protein kinases. J Antibiot (Tokyo) 1989;42:102–6.
Osada H, Cui CB, Onose R, Hanaoka F. Screening of cell cycle inhibitors from microbial metabolites by a bioassay using a mouse cdc2 mutant cell line, tsFT210. Bioorg Med Chem 1997;5:193–203.
Nishioka H, Sawa T, Hamada M, Shimura N, Imoto M, Umezawa K. Inhibition of phosphatidylinositol kinase by toyocamycin. J Antibiot (Tokyo) 1990;43:1586–9.
Ri M, Tashiro E, Oikawa D, Shinjo S, Tokuda M, Yokouchi Y, et al. Identification of Toyocamycin, an agent cytotoxic for multiple myeloma cells, as a potent inhibitor of ER stress-induced XBP1 mRNA splicing. Blood Cancer J 2012;2:e79.
Lee SA, Jung M. The nucleoside analog sangivamycin induces apoptotic cell death in breast carcinoma MCF7/adriamycin-resistant cells via protein kinase Cdelta and JNK activation. J Biol Chem 2007;282:15271–83.
Cohen MB, Glazer RI. Comparison of the cellular and RNA-dependent effects of sangivamycin and toyocamycin in human colon carcinoma cells. Mol Pharmacol 1985;27:349–55.
Droge W. Free radicals in the physiological control of cell function. Physiol Rev 2002;82:47–95.
McCubrey JA, Lahair MM, Franklin RA. Reactive oxygen species-induced activation of the MAP kinase signaling pathways. Antioxid Redox Signal 2006;8:1775–89.
Fey D, Croucher DR, Kolch W, Kholodenko BN. Crosstalk and signaling switches in mitogen-activated protein kinase cascades. Front Physiol 2012;3:355.
Park HY, Kim MK, Moon SI, Cho YH, Lee CH. Cell cycle arrest and apoptotic induction in LNCaP cells by MCS-C2, novel cyclin-dependent kinase inhibitor, through p53/p21WAF1/CIP1 pathway. Cancer Sci 2006;97:430–6.
Hughes BG, Robins RK. 2′,5′-Oligoadenylates and related 2′,5′-oligonucleotide analogues 2. Effect on cellular proliferation, protein synthesis, and endoribonuclease activity. Biochemistry 1983;22:2127–35.
Gross A, McDonnell JM, Korsmeyer SJ. BCL-2 family members and the mitochondria in apoptosis. Genes Dev 1999;13:1899–911.
Xia Z, Dickens M, Raingeaud J, Davis RJ, Greenberg ME. Opposing effects of ERK and JNK-p38 MAP kinases on apoptosis. Science 1995;270:1326–31.
Kang YH, Lee SJ. The role of p38 MAPK and JNK in Arsenic trioxide-induced mitochondrial cell death in human cervical cancer cells. J Cell Physiol 2008;217:23–33.
Brozovic A, Osmak M. Activation of mitogen-activated protein kinases by cisplatin and their role in cisplatin-resistance. Cancer Lett 2007;251:1–16.
Porras A, Zuluaga S, Black E, Valladares A, Alvarez AM, Ambrosino C, et al. P38 alpha mitogen-activated protein kinase sensitizes cells to apoptosis induced by different stimuli. Mol Biol Cell 2004;15:922–33.
Zuluaga S, Gutierrez-Uzquiza A, Bragado P, Alvarez-Barrientos A, Benito M, Nebreda AR, et al. p38alpha MAPK can positively or negatively regulate Rac-1 activity depending on the presence of serum. FEBS Lett 2007;581:3819–25.
Wagner EF, Nebreda AR. Signal integration by JNK and p38 MAPK pathways in cancer development. Nat Rev Cancer 2009;9:537–49.
Bragado P, Armesilla A, Silva A, Porras A. Apoptosis by cisplatin requires p53 mediated p38alpha MAPK activation through ROS generation. Apoptosis 2007;12:1733–42.
Wakao K, Watanabe T, Takadama T, Ui S, Shigemi Z, Kagawa H, et al. Sangivamycin induces apoptosis by suppressing Erk signaling in primary effusion lymphoma cells. Biochem Biophys Res Commun 2014;444:135–40.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, SG., Kim, SH., Kim, KY. et al. Toyocamycin induces apoptosis via the crosstalk between reactive oxygen species and p38/ERK MAPKs signaling pathway in human prostate cancer PC-3 cells. Pharmacol. Rep 69, 90–96 (2017). https://doi.org/10.1016/j.pharep.2016.10.014
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2016.10.014