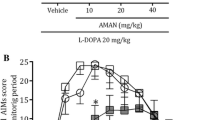
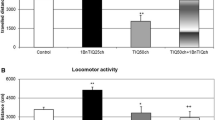
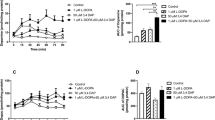
Abstract
Background
Interaction between dopaminergic and nitrergic neurotransmission in the brain plays a crucial role in the control of motor function and in the regulation of blood pressure (BP). In Parkinson’s disease (PD), dopaminergic denervation of the striatum leads to disturbances in the nitrergic system in the basal ganglia. Recently, it has been demonstrated that addition of a low dose of the nitric oxide donor molsidomine to l-DOPA therapy improves dopaminergic neurotransmission in the denervated nigrostriatal system and weakens dyskinesias in rodent models of the disease.
Methods
The aim of the present study was to examine the impact of chronic administration of molsidomine (2 mg/kg) and l-DOPA (25 mg/kg), alone and in combination, on systolic (SBP) and diastolic (DBP) blood pressure in the anesthetized, unilaterally 6-OHDA-lesioned rats. The measurement of SBP and DBP was performed 24 h after the penultimate and immediately after the last drug doses.
Results
In 6-OHDA-lesioned rats receiving saline, spontaneous, small decreases in SBP and DBP were observed during the measurements lasting 60 min. Administration of molsidomine alone or in combination with l-DOPA distinctly decreased the BP in 6-OHDA-lesioned rats already after 10 min compared to those treated with saline or l-DOPA alone, respectively. In both groups, the molsidomine-mediated declines in BP persisted till the end of measurement but they disappeared after 24 h.
Conclusions
Our results indicate that in this PD model molsidomine evokes a short-lasting decrease in BP in contrast to conventional antihypertensive drugs that maintain long-term effect and worsen orthostatic hypotension in parkinsonian patients.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BP:
-
blood pressure
- DA:
-
dopamine
- MFB:
-
medial forebrain bundle
- NO:
-
nitric oxide
- nNOS:
-
neuronal nitric oxide synthase
- 6-OHDA:
-
6-hydroxydopamine
- OH:
-
orthostatic hypotension
- PD:
-
Parkinson’s disease
- SNc:
-
substantia nigra pars compacta
- STR:
-
striatum
References
Togashi H, Sakuma I, Yoshioka M, Kobayashi T, Yasuda H, Kitabatake A, et al. A central nervous system action of nitric oxide in blood pressure regulation. J Pharmacol Exp Ther 1992;262(1):343–7.
Zanzinger J, Czachurski J, Seller H. Inhibition of basal and reflex-mediated sympathetic activity in the RVLM by nitric oxide. Am J Physiol 1995;268(4 Pt 2):R958–62.
Horn T, Smith PM, McLaughlin BE, Bauce L, Marks GS, Pittman QJ, et al. Nitric oxide actions in paraventricular nucleus: cardiovascular and neurochemical implications. Am J Physiol 1994;266(1 Pt 2):R306–13.
Resstel LB, Corrêa FM. Involvement of the medial prefrontal cortex in central cardiovascular modulation in the rat. Auton Neurosci 2006;12:6–127 130–8.
Resstel LB, Corrêa FM. Injection of l-glutamate into medial prefrontal cortex induces cardiovascular responses through NMDA receptor—nitric oxide in rat. Neuropharmacology 2006;51(1):160–7.
Busnardo C, Crestani CC, Tavares RF, Resstel LB, Correa FM. Cardiovascular responses to L-glutamate microinjection into the hypothalamic paraventricular nucleus are mediated by a local nitric oxide-guanylate cyclase mechanism. Brain Res 2010;1344:87–95.
van den Buuse M. Central effects of quinpirole on blood pressure of spontaneously hypertensive rats. J Pharmacol Exp Ther 1992;262(1):303–11.
Lin MT, Yang JJ. Stimulation of the nigrostriatal dopamine system produces hypertension and tachycardia in rats. Am J Physiol 1994;266(6 Pt 2):H2489–96.
Cornish JL, van den Buuse M. Pressor responses to electrical and chemical stimulation of the rat brain A10 dopaminergic system. Neurosci Lett 1994;176(2):142–6.
Böckelmann R, Wolf G, Ransmayr G, Riederer P. NADPH-diaphorase/nitric oxide synthase containing neurons in normal and Parkinson’s disease putamen. J Neural Transm Park Dis Dement Sect 1994;7(2):115–21.
Eve DJ, Nisbet AP, Kingsbury AE, Hewson EL, Daniel SE, Lees AJ, et al. Basal ganglia neuronal nitric oxide synthase mRNA expression in Parkinson’s disease. Brain Res Mol Brain Res 1998;63(1):62–71.
de Vente J, Markerink-van Ittersum M, van Abeelen J, Emson PC, Axer H, Steinbusch HW. NO-mediated cGMP synthesis in cholinergic neurons in the rat forebrain: effects of lesioning dopaminergic or serotonergic pathways on nNOS and cGMP synthesis. Eur J Neurosci 2000;12(2):507–19.
Sancesario G, Giorgi M, D’Angelo V, Modica A, Martorana A, Morello M, et al. Down-regulation of nitrergic transmission in the rat striatum after chronic nigrostriatal deafferentation. Eur J Neurosci 2004;20(4):989–1000.
Pierucci M, Galati S, Valentino M, Di Matteo V, Benigno A, Pitruzzella A, et al. Nitric oxide modulation of the basal ganglia circuitry: therapeutic implication for Parkinson’s disease and other motor disorders. CNS Neurol Disord Drug Targets 2011;10(7):777–91.
Kuiper MA, Visser JJ, Bergmans PL, Scheltens P, Wolters EC. Decreased cerebrospinal fluid nitrate levels in Parkinson’s disease, Alzheimer’s disease and multiple system atrophy patients. J Neurol Sci 1994;121(1):46–9.
Czarnecka A, Lenda T, Domin H, Konieczny J, Śmialowska M, Lorenc-Koci E. Alterations in the expression of nNOS in the substantia nigra and subthalamic nucleus of 6-OHDA-lesioned rats: the effects of chronic treatment with l-DOPA and the nitric oxide donor, molsidomine. Brain Res 2013;1541:92–105.
Sammut S, Dec A, Mitchell D, Linardakis J, Ortiguela M, West AR. Phasic dopaminergic transmission increases NO efflux in the rat dorsal striatum via a neuronal NOS and a dopamine D(1/5) receptor-dependent mechanism. Neuropsychopharmacology 2006;31(3):493–505.
Sammut S, Bray KE, West AR. Dopamine D2 receptor-dependent modulation of striatal NO synthase activity. Psychopharmacology (Berl) 2007;191(3):793–803.
Park DJ, West AR. Regulation of striatal nitric oxide synthesis by local dopamine and glutamate interactions. J Neurochem 2009;111(6):1457–65.
Hoque KE, Indorkar RP, Sammut S, West AR. Impact of dopamine-glutamate interactions on striatal neuronal nitric oxide synthase activity. Psychopharmacology 2010;207(4):571–81.
Huot P, Johnston TH, Koprich JB, Fox SH, Brotchie JM. The pharmacology of L-DOPA-induced dyskinesia in Parkinson’s disease. Pharmacol Rev 2013;65(1):171–222.
Lorenc-Koci E, Czarnecka A, Lenda T, Kamińska K, Konieczny J. Molsidomine, a nitric oxide donor, modulates rotational behavior and monoamine metabolism in 6-OHDA lesioned rats treated chronically with L-DOPA. Neurochem Int 2013;63(8):790–804.
Solís O, Espadas I, Del-Bel EA, Moratalla R. Nitric oxide synthase inhibition decreases l-DOPA-induced dyskinesia and the expression of striatal molecular markers in Pitx3(-/-) aphakia mice. Neurobiol Dis 2015;73:49–59.
Pathak A, Senard JM. Blood pressure disorders during Parkinson’s disease: epidemiology, pathophysiology and management. Expert Rev Neurother 2006;6(8):1173–80.
Jain S, Goldstein DS. Cardiovascular dysautonomia in Parkinson disease: from pathophysiology to pathogenesis. Neurobiol Dis 2012;46(3):572–80.
Goldstein DS, Eldadah BA, Holmes C, Pechnik S, Moak J, Saleem A, et al. Neurocirculatory abnormalities in Parkinson’s disease with orthostatic hypotension: independence from levodopa treatment. Hypertension 2005;46(6):1333–9.
Ziemssen T, Reichman H. Cardiovascular autonomic dysfunction in Parkinson’s disease. J Neurol Sci 2010;289:74–80.
Kelly PH, Iversen SD. Selective 6-OHDA-induced destruction of mesolimbic dopamine neurons: abolition of psychostimulant-induced locomotor activity in rats. Eur J Pharmacol 1976;40(1):45–56.
Paxinos G, Watson C. The rat brain in stereotaxic coordinates. San Diego: Academic Press; 1986.
Ungerstedt U. Postsynaptic supersensitivity after 6-hydroxy-dopamine induced degeneration of the nigro-striatal dopamine system. Acta Physiol Scand Suppl 1971;367:69–93.
Deumens R, Blokland A, Prickaerts J. Modeling Parkinson’s disease in rats: an evaluation of 6-OHDA lesions of the nigrostriatal pathway. Exp Neurol 2002;75(2):303–17.
Slack K, Billing R, Matthews Allbutt HN, Einstein R, Henderson JM. Subtle cardiovascular dysfunction in the unilateral 6-hdroxydopamine-lesioned rats. Parkinsons Dis 2010427810:, doi:https://doi.org/10.4061/2010/427810.
Lorenc-Koci E, Lenda T, Kamińska K, Czarnecka A, Konieczny J. Neurochemical and behavioral changes corresponding to the advanced Parkinson’s disease with coexistent depresion in rats treated unilaterally with 6-OHDA. Parkinsonism Relat Disord 2012;18(Supp 2):S197.
Braak H, Ghebremedhin E, Rüb U, Bratzke H, Del Tredici K. Stages in the development of Parkinson’s disease-related pathology. Cell Tissue Res 2004;318(1):121–34.
Coote JH. A role for the paraventricular nucleus of the hypothalamus in the autonomic control of heart and kidney. Exp Physiol 2005;90(2):169–73.
Krasowski MD, Harrison NL. General anaesthetic actions on ligand-gated ion channels. Cell Mol Life Sci 1999;55(10):1278–303.
Ariza D, Sisdeli L, Crestani CC, Fazan R, Martins-Pinge MC. Dysautonomias in Parkinson’s disease: cardiovascular changes and autonomic modulation in conscious rats after infusion of bilateral 6-OHDA in substantia nigra. Am J Physiol Heart Circ Physiol 2015;308(3):H250–7.
Banegas I, Prieto I, Segarra AB, Durán R, Vives F, Alba F, et al. Blood pressure increased dramatically in hypertensive rats after left hemisphere lesions with 6-hydroxydopamine. Neurosci Lett 2011;500(2):148–50.
Ossowska K. The role of excitatory amino acids in experimental models of Parkinson’s disease. J Neural Transm Park Dis Dement Sect 1994;8(1-2):39–71.
Mazza A, Ravenni R, Antonini A, Casiglia E, Rubello D, Pauletto P. Arterial hypertension, a tricky side of Parkinson’s disease: physiopathology and therapeutic features. Neurol Sci 2013;34(5):621–7.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lorenc-Koci, E., Czarnecka, A., Kamińska, K. et al. Contribution of the nitric oxide donor molsidomine and the antiparkinsonian drug l-DOPA to the modulation of the blood pressure in unilaterally 6-OHDA-lesioned rats. Pharmacol. Rep 69, 29–35 (2017). https://doi.org/10.1016/j.pharep.2016.09.022
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2016.09.022