Abstract
Background
GABAergic neurotransmission is involved in long-term potentiation, a neurophysiological basis for learning and memory. On the other hand, GABA-enhancing drugs may impair memory and learning in humans and animals. The present study aims at investigating the effect of GAT1 inhibitor tiagabine on memory and learning.
Methods
Albino Swiss (CD-1) and C57BL/6J mice were used in the passive avoidance (PA), Morris water maze (MWM) and radial arm water maze (RAWM) tasks. Scopolamine (1 mg/kg ip) was applied to induce cognitive deficits.
Results
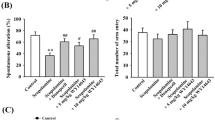
In the retention trial of PA scopolamine reduced step-through latency as compared to vehicle-treated mice, and pretreatment with tiagabine did not have any influence on this effect. In MWM the results obtained for vehicle-treated mice, scopolamine-treated group and combined scopolamine + tiagabine-treated mice revealed variable learning abilities in these groups. Tiagabine did not impair learning in the acquisition trial. In RAWM on day 1 scopolamine-treated group made nearly two-fold more errors than vehicle-treated mice and mice that received combined scopolamine and tiagabine. Learning abilities in the latter group were similar to those of vehicle-treated mice in the corresponding trial block on day 1, except for the last trial block, during which tiagabine + scopolamine-injected mice made more errors than control mice and the scopolamine-treated group. In all groups a complete reversal of memory deficits was observed in the last trial block of day 2.
Conclusions
The lack of negative influence of tiagabine on cognitive functions in animals with scopolamine-induced memory impairments may be relevant for patients treated with this drug.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CNS:
-
central nervous system
- GABA:
-
γ-aminobutyric acid
- GAT:
-
GABA transporter
- MWM:
-
Morris water maze
- PA:
-
passive avoidance
- RAWM:
-
radial arm water maze
References
Schousboe A, Madsen KK, Barker-Haliski ML, White HS. The GABA synapse as a target for antiepileptic drugs: a historical overview focused on GABA transporters. Neurochem Res 2014;39(10):1980–7.
Conti F, Minelli A, Melone M. GABA transporters in the mammalian cerebral cortex: localization, development and pathological implications. Brain Res Brain Res Rev 2004;45(3):136–212.
Sałat K, Kulig K. GABA transporters as targets for new drugs. Future Med Chem 2011;3(2):211–22.
Schousboe A, Sarup A, Larsson OM, White HS. GABA transporters as drug targets for modulation of GABAergic activity. Biochem Pharmacol 2004;68(8):1557–63.
Zhao X, Pabel J, Höfner GC, Wanner KT. Synthesis and biological evaluation of 4-hydroxy-4-(4-methoxyphenyl)-substituted proline and pyrrolidin-2-ylacetic acid derivativesas GABA uptake inhibitors. Bioorg Med Chem 2013;21(2):470– 84.
Dalby NO. Inhibition of gamma-aminobutyric acid uptake: anatomy, physiology and effects against epileptic seizures. Eur J Pharmacol 2003;479(1– 3):127–37.
Sitka I, Allmendinger L, Fülep G, Höfner G, Wanner KT. Synthesis of N-substituted acyclic β-amino acids and their investigation as GABA uptake inhibitors. Eur J Med Chem 2013;65:487–9.
Quandt G, Höfner G, Pabel J, Dine J, Eder M, Wanner KT. First photoswitchable neurotransmitter transporter inhibitor: light-induced control of γ-aminobutyric acid transporter 1 (GAT1) activity in mouse brain. J Med Chem 2014;57(15):6809–21.
Sałat K, Kulig K, Sałat R, Filipek B, Malawska B. Analgesic and anticonvulsant activity of new derivatives of 2-substituted 4-hydroxybutanamides in mice. Pharmacol Rep 2012;64(1):102–12.
Sałat K, Więckowska A, Więckowski K, Höfner GC, Kamiński J, Wanner KT, et al. Synthesis and pharmacological properties of new GABA uptake inhibitors. Pharmacol Rep 2012;64(4):817–33.
Kowalczyk P, Sałat K, Höfner GC, Guzior N, Filipek B, Wanner KT, et al. 2-Substituted 4-hydroxybutanamides as potential inhibitors of γ-aminobutyric acid transporters mGAT1-mGAT4: synthesis and biological evaluation. Bioorg Med Chem 2013;21(17):5154–67.
Kowalczyk P, Sałat K, Höfner GC, Mucha M, Rapacz A, Podkowa A, et al. Synthesis, biological evaluation and structure-activity relationship of new GABA uptake inhibitors, derivatives of 4-aminobutanamides. Eur J Med Chem 2014;83:256–73.
Kragholm B, Kvist T, Madsen KK, Jørgensen L, Vogensen SB, Schousboe A, et al. Discovery of a subtype selective inhibitor of the human betaine/GABA transporter 1 (BGT-1) with a non-competitive pharmacological profile. Biochem Pharmacol 2013;86(4):521–8.
Pabel J, Faust M, Prehn C, Wörlein B, Allmendinger L, Höfner G, et al. Development of an (S)-1-{2-[tris(4-methoxyphenyl)methoxy]ethyl}piperidine-3-carboxylic acid [(S)-SNAP-5114] carba analogue inhibitor for murine γ-aminobutyric acid transporter type 4. ChemMedChem 2012;7(7):1245–55.
Sałat K, Podkowa A, Kowalczyk P, Kulig K, Dziubina A, Filipek B, et al. Anticonvulsant active inhibitor of GABA transporter subtype 1, tiagabine, with activity in mouse models of anxiety, pain and depression. Pharmacol Rep 2015;67:465–72.
Thoeringer CK, Erhardt A, Sillaber I, Mueller MB, Ohl F, Holsboer F, et al. Long-term anxiolytic and antidepressant-like behavioural effects of tiagabine, a selective GABA transporter-1 (GAT-1) inhibitor, coincide with a decrease in HPA system activity in C57BL/6 mice. J Psychopharmacol 2010;24(5):733–43.
Carpenter LL, Schecter JM, Tyrka AR, Mello AF, Mello MF, Haggarty R, et al. Open-label tiagabine monotherapy for major depressive disorder with anxiety. J Clin Psychiatry 2006;67(1):66–71.
Novak V, Kanard R, Kissel JT, Mendell JR. Treatment of painful sensory neuropathy with tiagabine: a pilot study. Clin Auton Res 2001;11(6):357–66.
Pollack MH, Roy-Byrne PP, Van Ameringen M, Snyder H, Brown C, Ondrasik J, et al. The selective GABA reuptake inhibitor tiagabine for the treatment of generalized anxiety disorder: results of a placebo-controlled study. J Clin Psychiatry 2005;66(11):1401–8.
Schwartz TL, Nihalani N. Tiagabine in anxiety disorders. Expert Opin Pharmacother 2006;7(14):1977–87.
Todorov AA, Kolchev CB, Todorov AB. Tiagabine and gabapentin for the management of chronic pain. Clin J Pain 2005;21(4):358–61.
Fritz N, Glogau S, Hoffmann J, Rademacher M, Elger CE, Helmstaedter C. Efficacy and cognitive side effects of tiagabine and topiramate in patients with epilepsy. Epilepsy Behav 2005;6(3):373–81.
Ortinski P, Meador KJ. Cognitive side effects of antiepileptic drugs. Epilepsy Behav 2004;5(Suppl. 1):60–5.
Myhrer T. Neurotransmitter systems involved in learning and memory in the rat: a meta-analysis based on studies of four behavioral tasks. Brain Res Brain Res Rev 2003;41(2–3):268–87.
Castellano C, Zocchi A, Cabib S, Puglisi-Allegra S. Psychopharmacology of memory modulation: evidence for multiple interaction among neurotransmitters and hormones. Behav Brain Res 1996;77:1–21.
Collinson N, Kuenzi FM, Jarolimek W, Maubach KA, Cothliff R, Sur C, et al. Enhanced learning and memory and altered GABAergic synaptic transmission in mice lacking the alpha 5 subunit of the GABAA receptor. J Neurosci 2002;22(13):5572–80.
Shi J, Cai Y, Liu G, Gong N, Liu Z, Xu T, et al. Enhanced learning and memory in GAT1 heterozygous mice. Acta Biochim Biophys Sin 2012;44(4):359–66.
Tellez R, Gómez-Víquez L, Meneses A. GABA, glutamate, dopamine and serotonin transporters expression on memory formation and amnesia. Neurobiol Learn Mem 2012;97(2):189–201.
Kwan P, Brodie MJ. Neuropsychological effects of epilepsy and antiepileptic drugs. Lancet 2001;357(9251):216–22.
Schmitt U, Hiemke C. Tiagabine, a gamma-amino-butyric acid transporter inhibitor impairs spatial learning of rats in the Morris water-maze. Behav Brain Res 2002;133(2):391–4.
Klinkenberg I, Blokland A. The validity of scopolamine as a pharmacological model for cognitive impairment: a review of animal behavioral studies. Neurosci Biobehav Rev 2010;34(8):1307–50.
Patil SS, Sunyer B, Höger H, Lubec G. Evaluation of spatial memory of C57BL/6J and CD1 mice in the Barnes maze, the Multiple T-maze and in the Morris water maze. Behav Brain Res 2009;198(1):58–68.
Park SJ, Kim DH, Jung JM, Kim JM, Cai M, Liu X, et al. The ameliorating effects of stigmasterol on scopolamine-induced memory impairments in mice. Eur J Pharmacol 2012;676(1–3):64–70.
Puzzo D, Lee L, Palmeri A, Calabrese G, Arancio O. Behavioral assays with mouse models of Alzheimer’s disease: practical considerations and guidelines. Biochem Pharmacol 2014;88(4):450–67.
Bialuk I, Dobosz K, Potrzebowski B, Winnicka MM. CP55,940 attenuates spatial memory retrieval in mice. Pharmacol Rep 2014;66(6):931–6.
Alamed J, Wilcock DM, Diamond DM, Gordon MN, Morgan D. Two-day radialarm water maze learning and memory task; robust resolution of amyloid-related memory deficits in transgenic mice. Nat Protoc 2006;1(4):1671–9.
Wilcock DM, Rojiani A, Rosenthal A, Subbarao S, Freeman MJ, Gordon MN, et al. Passive immunotherapy against Abeta in aged APP-transgenic mice reverses cognitive deficits and depletes parenchymal amyloid deposits in spite of increased vascular amyloid and microhemorrhage. J Neuroinflamm 2004;1(1):24.
Sudduth TL, Weekman EM, Brothers HM, Braun K, Wilcock DM. β-Amyloid deposition is shifted to the vasculature and memory impairment is exacerbated when hyperhomocysteinemia is induced in APP/PS1 transgenic mice. Alzheimers Res Ther 2014;6(3):32.
Wang Y, Kan H, Yin Y, Wu W, Hu W, Wang M, et al. Protective effects of ginsenoside Rg1 on chronic restraint stress induced learning and memory impairments in male mice. Pharmacol Biochem Behav 2014;120:73–81.
Sankar R, Holmes GL. Mechanisms of action for the commonly used antiepileptic drugs: relevance to antiepileptic drug-associated neurobehavioral adverse effects. J Child Neurol 2004;19(Suppl. 1):S6–14.
Mula M, Trimble MR. Antiepileptic drug-induced cognitive adverse effects: potential mechanisms and contributing factors. CNS Drugs 2009;23(2):121– 37.
Ferreira DD, Stutz B, de Mello FG, Reis RA, Kubrusly RC. Caffeine potentiates the release of GABA mediated by NMDA receptor activation: Involvement of A1 adenosine receptors. Neuroscience 2014;281C:208–15.
O’Connell AW, Fox GB, Kjøller C, Gallagher HC, Murphy KJ, Kelly J, et al. Anti-ischemic and cognition-enhancing properties of NNC-711, a gamma-aminobutyric acid reuptake inhibitor. Eur J Pharmacol 2001;424(1):37–44.
Gong N, Li Y, Cai GQ, Niu RF, Fang Q, Wu K, et al. GABA transporter-1 activity modulates hippocampal theta oscillation and theta burst stimulation-induced long-term potentiation. J Neurosci 2009;29(50):15836–45.
Hu JH, Ma YH, Jiang J, Yang N, Duan SH, Jiang ZH, et al. Cognitive impairment in mice over-expressing gamma-aminobutyric acid transporter 1 (GAT1). Neuroreport 2004;15(1):9–12.
Gulcan HO, Unlu S, Esiringu I, Ercetin T, Sahin Y, Oz D, et al. Design, synthesis and biological evaluation of novel 6H-benzo[c]chromen-6-one, and 7,8,9,10-tetrahydro-benzo[c]chromen-6-one derivatives as potential cholinesterase inhibitors. Bioorg Med Chem 2014;22(19):5141–54.
Paul CM, Magda G, Abel S. Spatial memory: theoretical basis and comparative review on experimental methods in rodents. Behav Brain Res 2009;203(2):151–64.
D’Hooge R, De Deyn PP. Applications of the Morris water maze in the study of learning and memory. Brain Res Brain Res Rev 2001;36(1):60–90.
Sharma S, Rakoczy S, Brown-Borg H. Assessment of spatial memory in mice. Life Sci 2010;87(17–18):521–36.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sałat, K., Podkowa, A., Mogilski, S. et al. The effect of GABA transporter 1 (GAT1) inhibitor, tiagabine, on scopolamine-induced memory impairments in mice. Pharmacol. Rep 67, 1155–1162 (2015). https://doi.org/10.1016/j.pharep.2015.04.018
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2015.04.018