Abstract
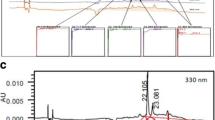
The hepatoprotective activities of two traditionally used plants, Cleome droserifolia (Forssk.) Delile, Cleo-maceae, and Artemisia annua L., Asteraceae, were recently reported. However, the biologically active metabolites responsible for this activity were not identified. The aqueous extract of C. droserifolia aerial parts, and the polar fraction of A. annua leaves were screened for their antioxidant activities using the 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid) assay. The in vitro viability of HepG-2 cells treated with CCl4 and the extracts were assessed by MTT assay. The effects of the extracts on the liver enzymes and the total soluble protein in CCl4-intoxicated HepG-2 cells were investigated. An HPLC/PDA/ESI/MS-MS based analysis was carried out for extract of C. droserifolia and polar fraction of A. annua. Both exhibited pronounced free radical scavenging activities (86 and 83%, respectively). Both showed a significant increase in cell viability: 86.43% for the extract of C. droserifolia and 79.32% for polar fraction of A. annua. Only the extract of C. droserifolia (39.6 ± 5.41 and 20.4 ± 6.91 IU/dl, respectively) and polar fraction of A. annua (40.8 ±2.14 and 24.5 ±3.11 IU/dl, respectively) restored the levels of liver enzymes (aspartate transaminase and alanine transaminase, respectively) compared to the CCl4 intoxicated group (87.5 ± 4.34 and 34.1 ± 8.12 IU/dl, respectively) and other herbal extracts. More than fifty phenolic secondary metabolites were identified in the extracts under investigation. The significant hepatoprotective activities of both extracts seemed to be strongly connected to their content of hydroxycinnamoyl quinic acids and flavonoids.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abdel-Hameed, E.S., Bazaid, SA, Salman, M.S., 2013. Characterization of the phyto-chemical constituents of Taif rose and its antioxidant and anticancer activities. BioMed. Res. Int., https://doi.org/10.1155/2013/345465.
Abdel-Kader, M.S., Al-Qasoumi, S.I., AL-Taweel, A.M., 2009. Hepatoprotective constituents from Cleome droserifolia. Chem. Pharm. Bull. 57, 620–624.
Abdel-Kawy, M.A., El-Deib, S., El-Khyat, Z., Mikhail, Y.A., 2000. Chemical and biological studies of Cleome droserifolia (Forssk.) Del. PartI. Egypt J. Biomed. Sci. 6, 204–218.
Abdel Motaal, A., Ezzat, S.M., El-Askary, H., 2014. Antihyperglycemic activity and standardization of the bioactive extract of Cleome droserifolia growing in Egypt. Phcog. J. 6, 15–21.
Abdullah, W., Elsayed, W.E., Abdelshafeek, K.A., Nazif, N.M., Singab, A.N., 2016. Chemical constituents and biological activities of Cleome genus: a brief review. Int. J. Pharmaco. Phytochem. Res. 8, 777–787.
Abou El Azm, A.R., Yousef, M., Mansour, N., Awad, A., El Dardiry, S., Abdel Aziz, I., 2014. New insights on non-B non-C hepatocellular carcinoma in mid Delta region. Egypt J. Gastrointest. Cancer 45, 276–283.
Aboul Ela, MA, El-Lakany, A.M., Abdel-Kader, M.S., Alqasoumi, S.I., Shams-El-Din, S.M., Hammoda, H.M., 2012. New quinic acid derivatives from hepatoprotective Inula crithmoides root extract. Helv. Chim. Acta 95, 61–66.
Aboushoer, M.I., Fathy, H.M., Abdel-Kader, M.S., Goetz, G., Omar, A.A., 2010. Terpenes and flavonoids from an Egyptian collection of Cleome droserifolia. Nat. Prod. Lett. 24, 687–696.
Abu-Reidah, I.M., Ali-Shtayeh, M.S., Jamous, R., Arráez-Román, D., Segura-Carretero, A., 2015. HPLC-DAD-ESI-MS/MS screening of bioactive components from Rhus coriaria L. (Sumac) fruits. Food Chem. 166, 179–191.
Barros, L., Duen˜as, M., Carvalho, A., Ferreira, I., Santos-Buelga, C., 2012. Characterization of phenolic compounds in flowers of wild medicinal plants from Northeastern Portugal. Food Chem. Toxicol. 50, 1576–1582.
Beekman, A.C., Wierenga, P.K., Woerdenbag, HJ., Van Uden, W., Pras, N., Konings, A.W., El-Feraly, F.S., Galal, A.M., Wikström, H.V., 1998. Artemisinin- derived sesquiterpene lactones as potential antitumour compounds: cytotoxic action against bone marrow and tumour cells. Planta Med. 64, 615–619.
Bilia, A.R., Melillo de Malgalhaes, P., Bergonzi, M.C., Vincieri, F.F., 2006. Simultaneous analysis of artemisinin and flavonoids of several extracts of Artemisia annua L. obtained from a commercial sample and a selected cultivar. Phytomedicine 13, 487–493.
Catarino, M.D., Silva, A.M., Saraiva, S.C., Sobral, A.J., Cardoso, S.M., 2015. Characterization of phenolic constituents and evaluation of antioxidant properties of leaves and stems of Eriocephalus africanus. Arab. J. Chem., https://doi.org/10.1016/j.arabjc.2015.04.018.
Chen, H., Zhang, Q., Wang, X., Yang, J., Wang, Q., 2010. Qualitative analysis and simultaneous quantification of phenolic compounds in the aerial Parts of Salvia miltiorrhiza by HPLCDAD and ESI/MSn. Phytochem. Anal. 22, 247–257.
Clifford, M.N., Johnston, K.L., Knight, S., Kuhnert, N., 2003. Hierarchical scheme for LC-MSn identification of chlorogenicacids. J. Agric. Food Chem. 51, 2900–2911.
Clifford, M., Knight, S., Kuhnert, N., 2005. Discriminating between the six isomers of dicaffeoylquinic acid by LC-MSn. J. Agric. Food Chem. 53, 3821–3832.
Dou, I., Lee, V.S., Tzen, J., Lee, M., 2007. Identification and comparison of phenolic compounds in the preparation of Oolong tea manufactured by semifermentation and dryingprocesses. J. Agric. Food Chem. 55, 7462–7468.
El-Askary, H., Gala, A., Abou-Hussein, D.R., El-Ghawwas, E., 2004. Cultivation of Artemisia annua in Egypt and production of its anti-malarial drug (Artemisinin). Bull. Fac. Pharm. Cairo Univ. 42, 99–105.
El Sayed, A., Ezzat, S., El Naggar, M., El Hawary, S., 2016. In vivo diabetic wound healing effect and HPLC-DAD-ESI-MS/MS profiling of the methanol extracts of eight Aloe species. Rev. Bras. Farmacogn. 26, 352–362.
Ezzat, S.M., Abdel Motaal, A., 2012. Isolation of new cytotoxic metabolites from Cleome droserifolia growing in Egypt. Z. Naturforsch. C 67, 266–274.
Farag, M.A., Handoussa, H., Fekry, M.I., Wessjohann, L., 2016. Metabolites profiling in 18 Saudi date palm fruit cultivars and their antioxidant potential via UPLC-qTOF- MS and multivariate data analyses. Food Funct. 7, 1077–1086.
Ferreira, J.F., Simon, J.E., Janick, J., 1995. Developmental studies of Artemisia annua: flowering and artemisinin production under greenhouse and field conditions. Planta Med. 61, 167–170.
Ferreira, J.F., Luthria, D.L., Sasaki, T., Heyerick, A., 2010. Flavonoids from Artemisia annua L. as antioxidants and their potential synergism with artemisinin against malaria and cancer. Molecules 15, 3135–3170.
Gite, S., Yadav, S., Nilegaonkar, S., Agte, V., 2014. Evaluation of hepatoprotective potential of functional food formulations using in vitro and in vivo models of CCl4 radical induced toxicity. IJIMS 1, 6–13.
Gouveia, S., Castilho, P.C., 2011. Characterisation of phenolic acid derivatives and flavonoids from different morphological parts of Helichrysum obconicum by a RP-HPLC-DAD-ESI-MSn method. Food Chem. 129, 333–344.
Handoussa, H., Hanafi, R., Eddiasty, I., El-Gendy, M., El Khatib, A., Linscheid, M., Mahran, L., Ayoub, N., 2013. Anti-inflammatory and cytotoxic activities of dietary phenolics isolated from Corchorus olitorius and Vitis vinifera. J. Funct. Food 5, 1204–1216.
Ibrahim, A.S., Sobh, M.A., Eid, H.M., Salem, A., Elbelasi, H.H., El-Naggar, M.H., Abdel-Bar, F.M., Sheashaa, H., Sobh, M.A., Badria, F.A., 2014. Gingerol-derivatives: emerging new therapy against human drug-resistant MCF-7. Tumour Biol. 35, 9941–9948.
Johnson, CE., Lin, L.Z., Harnly, J.M., Oladeinde, F.O., Kinyua, A.M., Michelin, R., Bron-ner, Y., 2014. Identification of the phenolic components of Vernonia amygdalina and Russelia equisetiformi. J. Nat. Prod. 4, 57–64.
Kargbo, M., Gbago, O., Song, Y., 2015. In vitro anti-diabetic activity and phenolic compound profile of ethanol extracts of Anisophyllea laurina R. Br. ex Sabine leaves and stem bark. Euro Acad. Res. 2, 16089–16106.
Lin, L., Harnly, J.M., 2010. Identification of the phenolic components of Chrysanthemum flower (Chrysanthemum morifolium Ramat). Food Chem. 120, 319–326.
Maianski, N.A., Geissler, J., Srinivasula, S.M., Alnemri, E.S., Roos, D., Kuijpers, T.W., 2004. Functional characterization of mitochondria in neutrophils: a role restricted to apoptosis. Cell Death Differ. 11, 143–153.
Mittal, A., Kadyan, P., Gahlaut, A., Dabur, R., 2013. Nontargeted identification of the phenolic and other compounds of Saraca asoca by high performance liquid chromatography-positive electrospray ionization and quadrupole time-offlight mass spectrometry. ISRN Pharmaceutics, https://doi.org/10.1155/2013/293935.
Motaal, A.A., Ezzat, S.M., Haddad, P.S., 2011. Determination of bioactive markers in Cleome droserifolia using cell-based bioassays for antidiabetic activity and isolation of two novel active compounds. Phytomedicine 19, 38–41.
Nassar, M.I., Gamal-Eldeen, A.M., 2003. Potential antioxidant activity of flavonoids from Hypericum triquetrifolium Turra and Cleome droserifolia (Forssk.) Del. Bull. Fac. Pharm. Cairo Univ. 41, 107–115.
Pereira, O.R., Peres, A.M., Silva, A.M., Domingues, M.R., Cardoso, S.M., 2013. Simultaneous characterization and quantification of phenolic compounds in Thymus citriodorus using a validated HPLC-UV and ESI-MS combined method. Food Res. Int. 54, 1773–1780.
Shalaby, E.A., Shanab, S.M.M., 2013. Comparison of DPPH and ABTS assays for determining antioxidant potential of water and methanol extracts of Spirulina platensis. Indian J. Mar. Sci. 42, 556–564.
Simirgiotis, M.J., 2013. Antioxidant capacity and HPLC-DAD-MS profiling of Chilean Peumo (Cryptocaryaalba) fruits and comparison with German Peumo (Crataegus monogyna) from Southern Chile. Molecules 18, 2061–2080.
Torres-Gonzalez, L., Munoz-Espinosa, L.E., Rivas-Estilla, A.M., TrujilloMurillo, K., Salazar-Aranda, R., De Torres, N.W., Cordero-Perez, P., 2011. Protective effect of four Mexican plants against CCl4 induced damage onthe Huh 7 human hepatoma cell line. Ann. Hepatol. 10, 73–79.
Zheng, W., Wang, S.Y., 2001. Antioxidant activity and phenolic compounds in selectedherbs. J. Agric. Food Chem. 49, 5165–5170.
Zou, Y., Chang, S.K., Gu, Y., Qian, S., 2011. Antioxidant activity and phenolic compositions of lentil (Lens culinaris var. morton) extract and itsfractions. J. Agric. Food Chem. 59, 2268–2276.
Żuchowski, J., Pecio, L., Stochmal, A., 2014. Novel flavonol glycosides from the aerial parts of lentil (Lens culinaris). Molecules 19, 18152–18178.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
HE and AAM contributed in collecting plant samples and extract preparation. AHE and MWL performed qualitative chemical profiling of the phytoconstituents. HH and AA carried out the data analysis. FB carried out the in vitro assays. HE and AAM designed the study and contributed to the critical reading of the manuscript. All authors read and approved the final manuscript.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
El-Askary, H., Handoussa, H., Badria, F. et al. Characterization of hepatoprotective metabolites from Artemisia annua and Cleome droserifolia using HPLC/PDA/ESI/MS-MS. Rev. Bras. Farmacogn. 29, 213–220 (2019). https://doi.org/10.1016/j.bjp.2018.10.001
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2018.10.001