Abstract
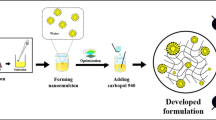
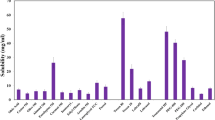
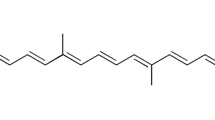
Solid lipid nanoparticles incorporating Curcuma longa L., Zingiberaceae, curcuminoids were produced by the hot melt emulsion method. A Box-Behnken factorial design was adopted to study the nanoparticles production at different levels of factors such as the percentage of curcuminoids, time of homogeniza-tion and surfactant ratio. The optimized nanoparticles were incorporated into hydrogels for stability, drug release and skin permeation tests. The average nanoparticle sizes were 210.4nm; the zeta poten-tial of -30.40 ±4.16; the polydispersivity was 0.222 ±0.125. The average encapsulation efficiency of curcumin and curcuminoids was 52.92 ± 5.41% and 48.39 ± 6.62%, respectively. Solid lipid nanocapsules were obtained with curcumin load varying from 14.2 to 33.6% and total curcuminoids load as high as 47.7%. The topical formulation containing SLN-Curcuminoids showed good spreadability and stability when subjected to mechanical stress test remained with characteristic color, showed no phase separa-tion and no significant change in pH. As a result of slow release, the nanoparticles were able to avoid permeation or penetration in the pig ear epidermis/dermis during 18h. The topical formulation is sta-ble and can be used in further in vivo studies for the treatment of inflammatory reactions, in special for radiodermitis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aggarwal, B., Harikumar, K.B., 2009. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int. J. Biochem. Cell Biol. 1, 40–59.
Anvisa, 2004. Guia de Estabilidade de Produtos Cosméticos. In: Ministério da Saúde. Agência Nacional de Vigilância Sanitária, 1st ed. Anvisa, Brasília (Série Qualidade em Cosméticos, 1).
Attama, A.A., Muller-Goymann, C.C., 2008. Effects of beeswax modifications on the lipid matrix of solid lipid nanoparticles crystallinity. Colloids Surf. A 315, 189–195.
Augustyniak, A., Bartosz, G., Cipak, A., Duburs, G., Kov L’Ubica, H., Luczaj, W., Majekova, M., Odysseos, A.D., Rackova, L., Skrzydlewska, E., Stefek, M., Strosov, M., Tirzitis, G., Venskutonis, P.R., Viskupicova, J., Vraka, P.S., Zarkovi, N., 2010. Natural and synthetic antioxidants: an updated overview. Free Radic. Res. 44, 1216–1262.
Bisht, S., Feldmann, G., Soni, S., Ravi, R., Karikar, C., Maitra, A., Maitral, A., 2007. Polymeric nanoparticle-encapsulated curcumin (“nanocurcumin”): a novel strategy for human cancer therapy. J. Nanobiotechnol., https://doi.org/10.1186/1477-3155-5-3.
Box, G.E.P., Hunter, W.G., Hunter, J.S., 1978. Statistics for Experimenters. John Wiley & Sons, New York.
Cevc, G., Vieri, U., 2010. Nanotechnology and the transdermal route. A state of the art review and critical appraisal. J. Control. Release 141, 277–299.
Contri, R.V., Fiel, L.A., Pohlmann, A.R., Guterres, S.S., Beck, R.C.R., 2011. Transport of substances and nanoparticles across the skin and in vitro models to evaluate skin permeation and/or penetration. In: Beck, R.C.R., Guterres, S.S., Pohlmann, A.R. (Eds.), Nanocosmetics and Nanomedicine., pp. 3–35.
Das, R.K., Kasoju, N., Bora, U., 2010. Encapsulation of curcumin in alginate-chitosan-pluronic composite nanoparticles for delivery to cancer cells. Nanomed. Nanotechnol. 6, 153–160.
Farmacopeia Brasileira, 2010. Ministério da Saúde. Agência Nacional de Vigilância Sanitária, Brasília http://www.anvisa.gov.br/hotsite/cd_farmacopeia/ pdf/volume1%2020110216.pdf (accessed September 2011).
Gao, Y., Li, Z., Sun, M., Guo, C., Yu, A., Xi, Y., Cui, J., Lou, H., Zhai, G., 2011. Preparation and characterization of intravenously injectable curcumin nanosuspension. Drug Deliv. 18, 131–142.
Goel, A., Kunnumakkara, A.B., Aggarwal, B.B., 2008. Curcumin as “Curecumin”: from kitchen to clinic. Biochem. Pharmacol. 75, 787–809.
Guterres, S.S., Alves, M.P., Pohlmann, A.R., 2007. Polymeric nanoparticles, nanospheres and nanocapsules forcutaneous applications. Drug Target Insights 2, 147–157.
Irving, G.R., Karmokar, A., Berry, D.P., Brown, K., Steward, W.P., 2011. Curcumin: the potential for efficacy in gastrointestinal diseases. Best Pract. Res. Clin. Gastroenterol. 25, 519–534.
Jayaprakasha, G.K., Rao, L.J., Sakariah, K.K., 2006. Antioxidant activities of curcumin, demethoxycurcumin and bisdemethoxycurcumin. Food Chem. 98, 720–724.
Jebors, S., Leydier, A., Wu, Q., Ghera, B.B., Malbouyre, M., Coleman, A.W., 2010. Solid lipid nanoparticles (SLNs) derived from para-acyl-calix[9]-arene: preparation and stability. J. Microencapsul. 27, 561–571.
Jenning, V., Gysler, A., Schafer-Korting, M., Gohla, S.H., 2000. Vitamin A loaded solid lipid nanoparticles for topical use: occlusive properties and drug targeting to the upper skin. Eur. J. Pharm. Biopharm. 49, 211–218.
Küchler, S., Abdel-Mottaleb, M., Lamprecht, A., Radowski, M.R., Haag, R., Schäfer-Korting, M., 2009. Influence of nanocarrier type and size on skin delivery of hydrophilic agents. Int. J. Pharm. 377, 169–172.
Luengo, J., Weiss, B., Schneider, M., Ehlers, A., Stracke, F., König, K., Kostka, K.H., Lehr, C.M., Schaefer, U.F., 2006. Influence of nanoencapsulation on human skin transport of flufenamic acid. Skin Pharmacol. Appl. Skin Physiol. 19, 190–197.
Lyra, M.A.M., Soares-Sobrinho, J.L., Brasileiro, M.T., Roca, M.F.L., Barraza, J.A., Viana, O.S., Rolim-Neto, P.J., 2007. Sistemas matriciais hidrofílicos e mucoadesivos para liberac¸ão controlada de fármacos Lat. Am. J. Pharm. 26, 784–793.
Martin, A.N., 1993. Physical Pharmacy: Physical Chemical Principles in the Pharma-ceutical Sciences, 2nd ed. Lippincott Williams & Wilkins, Philadelphia, PA, USA, pp. 622.
Martins, M.R.F.M., Veiga, F., 2002. Promotores de permeac¸ão para a liberac¸ão trans-dérmica de fármacos: uma nova aplicac¸ão para as ciclodextrinas. Rev. Bras. Farmacol. 38, 33–54.
Martins, R.M., 2014. Influência de micro e nanopartículas lipídicas sólidas na eficiência de formulac¸ões fotoprotetoras bioativas. Tese de Doutorado, Faculdade de Ciências Farmacêuticas de Ribeirão Preto da Universidade de São Paulo, Ribeirão Preto, pp. 181.
Mehnert, W., Mader, K., 2001. Solid lipid nanoparticles: production, characterization and applications. Adv. Drug Deliv. Rev. 47, 165–196.
Mourtas, S., Canovi, M., Zona, C., Aurilia, D., Niarakis, A., La Ferla, B., Salmona, M., Nicotra, F., Gobbi, M., Antimisiaris, S.G., 2011. Curcumin-decorated nanoliposomes with very high affinity for amyloid-FJ 1-42 peptide. Biomaterials 32, 1635–1645.
Nair, H.B., Sung, B., Yadav, V.R., Kannappan, R., Chaturvedi, M.M., Aggarwal, B.B., 2010. Delivery of antiinflammatory nutraceuticals by nanoparticles forthe pre-vention and treatment of cancer. Biochem. Pharmacol. 80, 1833–1843.
Nayak, A.P., Tiyaboonchai, W., Patankar, S., Madhusudhan, B., Souto, E.B., 2010. Curcuminoida-loaded lipid nanoparticles: novel approach towards malaria treatment. Colloid Surf. B 81, 263–273.
Oliveira, R.C.S., 2008. Desenvolvimento, formulac¸ão e avaliac¸ão de sistemas de libertac¸ão transdérmica incorporando sistemas ternários de complexac¸ão (Tese de Doutorado). Universidade do Porto, Portugal.
Paulucci, V.P., Couto, R.O., Teixeira, C.C.C., Freitas, L.A.P., 2013. Optimization of the extraction of curcumin from Curcuma longa rhizomes. Rev. Bras. Farmacogn. 23, 94–100.
Puglia, C., Frasca, G., Musumeci, T., Rizza, L., Puglisi, G., Bonina, F., Chiechio, S., 2012. Curcumin loaded NLC induces histone hypoacetylation in the CNS after intraperitoneal administration in mice. Eur.J. Pharm. Biopharm. 81, 288–293.
Rusig, O., Martins, M.C., 1992. Efeito da temperatura, do pH e da luz sobre extratos de oleorresina de cúrcuma (Curcuma longa L.) e curcumina. Rev. Bras. Cor. Nat. 1, 158–164.
Sartorelli, P., Anderson, H.R., Angerer, J., Corish, J., Drexler, H., Goen, T., 2000. Percu-taneous penetration studies for risk assessment. Environ. Toxicol. Pharmacol. 8, 133–152.
Serra, M.L.G., Vásquez, M.L.R., Villafuerte, L.R., Garcia, B.F., Hernandez, A.L., 2009. Efecto de los componentes de la formulación en las propiedades de las nanopartículas lipídicas sólidas. Rev. Mex. Cienc. Farm. 40, 26–40.
Suwantong, O., Waleetorncheepsawat, S., Sanchavanakit, N., Pavasant, P., Cheepsunthorn, P., Bunaprasert, T., Supaphol, P., 2007. In vitro biocompatibility of electrospun poly(3-hydroxybutyrate) and poly(3-hydroxybutyrate-co-3-hydroxyvalerate) fiber mats. Int. J. Biol. Macromol. 40, 217–223.
Tønnesen, H.H., Karlsen, J., 1985. Studies on curcumin and curcuminoids: VIkinet-ics of curcumin degradation in aqueous solution. Z. Lebensm. Unters. For. 180, 402–404.
Yallapu, M.M., Jaggi, M., Chauhan, S.C., 2012. Curcumin nanoformulations: a future nanomedicine for cancer. Drug Discov. Today 17, 71–80.
Yallapu, M.M., Jaggi, M., Chauhan, S.C., 2013. Curcumin nanomedicine: a road to cancer therapeutics. Curr. Pharm. Design 19, 1994–2010.
Wissing, S.A., Lippacher, A., Muller, R.H., 2001. Investigations onthe occlusive properties of solid lipid nanoparticles (SLN). J. Cosmet. Sci. 52, 313–324.
Zamarioli, C.M., 2014. Formulac¸ão tópica para prevenc¸ão e tratamento de radio-dermites: desenvolvimento de nanopartículas lipídicas sólidas (NLS) contendo curcuminoidese estudo invitro. Dissertac¸ão de Mestrado, Escola de Enfermagem de Ribeirão Preto da Universidade de São Paulo, Ribeirão Preto, pp. 139.
Zetasizer Nano-ZS, 2009. User Instructions. NBTC User Instructions.
Zhou, H., Beevers, C.S., Huang, S., 2011. The targets of curcumin. Curr. DrugTargets 12, 332–347.
Acknowledgments
Authors acknowledge the financial support from CAPES (MSc Scholarship), Fapesp (2011/20872-7) and CNPq (PQ-2).
Author information
Authors and Affiliations
Contributions
CMZ (MSc student) was responsible for most of experimental work (nanocapsules, gel and characterization); RMM developed the nanoencapsulation technique and contributed in all laboratory work and chromatographic analysis. ECC idealized the work and contributed to critical reading of the manuscript. LAPF designed the study, supervised the experiments, critically read and defined the final version of the manuscript. All the authors have read the final manuscript and approved the submission.
Corresponding author
Ethics declarations
The authors declare no conflicts of interest.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zamarioli, C.M., Martins, R.M., Carvalho, E.C. et al. Nanoparticles containing curcuminoids (Curcuma longa): development of topical delivery formulation. Rev. Bras. Farmacogn. 25, 53–60 (2015). https://doi.org/10.1016/j.bjp.2014.11.010
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2014.11.010