Abstract
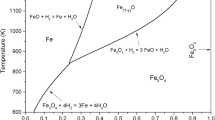
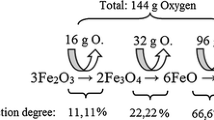
Reduction kinetics of fine iron ore powder in different gas mixtures were investigated in high-temperature fluidized bed at a scale of kilograms. Influence of processing parameters, such as particle size, gas flow velocity, height of charge, temperature, compositions of gas mixture, and percentage of inert components, on reduction kinetics was experimentally determined under the condition of fluidization. The equations for calculating instantaneous and average oxidation rates were deduced. It was found that an increasing H2O percentage in the gas mixture could obviously decrease the reduction rate because the equilibrium partial pressure of H2 decreased with increasing content of H2O in the gas mixture and then the driving force of reduction reaction was reduced. When the H2 content was high, the apparent reaction rate was so rapid when the average size of iron ore fines was less than 1 mm that the reaction temperature can be as low as 750 °C; when the average size of iron ore fines was more than 1 mm, a high reaction temperature of 800 °C was required. In addition, it was also found that the content of H2O should be less than 10% for efficient reduction.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
D. W. Zhang, Fundamental Research on the New Ironmaking Process Based on Fast Reduction at Low Temperature, Central Iron and Steel Research Institute, Beijing, 2007.
X. D. Jin, Iron and Steel 33 (1998) No. 4, 9–12.
R. J. Fruehan, Y. Li, L. Brabie, E. J. Kim, Scand. J. Metall. 34 (2005) 205–212.
A. A. El-geassy, M. I. Nasr, ISIJ Int. 30 (1990) 417–425.
S. K. Dutta, A. Ghosh, ISIJ Int. 33 (1993) 1168–1173.
P. Pourghahramani, E. Forssberg, Thermochim. Acta 454 (2007) 69–77.
A. Pineau, N Kanari, I. Gaballah, Thermochim. Acta 447 (2006) 89–100.
A. Unal, A. V. Bradshaw, Metall. Trans. B 14 (1983) 743–752.
M. Farren, S. P. Matthew, P. C. Hayes, Metall. Trans. B 21 (1990) 135–139.
R. A. D. Rodriguez, A. N. Conejo, E. B. Bedolla, I&SM 30 (2003) 25–33.
P. Zhao, P. M. Guo, D. W. Zhang, Iron and Steel 41 (2006) No. 8, 12–15.
P. Zhao, P. M. Guo, in: CSM 2005 Annual Meeting Proceedings (Vol.3), Metallurgical Industry Press, Beijing, 2005, pp. 677–681.
P. Zhao, P. M. Guo, Iron and Steel 40 (2005) No. 6, 6–9.
P. M. Guo, D. W. Zhang, P. Zhao, Spectroscopy and Spectral Analysis 27 (2007) 816–818.
Y. X. Hua, Metallurgical Process Dynamics Introduction, Metallurgical Industry Press, Beijing, 2004.
A. M. Squires, C. A. Johnson, J. Metals 1 (1957) 586–590.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation Item: Item Sponsored by National Environmental Protection Public Welfare Profession Scientific Special Plan of China (201209023)
Rights and permissions
About this article
Cite this article
Pang, Jm., Guo, Pm. & Zhao, P. Reduction kinetics of fine iron ore powder in mixtures of H2-N2 and H2-H2O-N2 of fluidized bed. J. Iron Steel Res. Int. 22, 391–395 (2015). https://doi.org/10.1016/S1006-706X(15)30017-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1016/S1006-706X(15)30017-0