Abstract
Objective
The primary objective was to quantify the prognostic association between various D-dimer thresholds and 30-day PE diagnosis among emergency department (ED) patients with suspected SARS-CoV-2 infection.
Methods
This was a retrospective study of patients enrolled in the Canadian COVID-19 ED Rapid Response Network (CCEDRRN) registry from March 1, 2020 to July 2, 2021. We included consecutive adults (≥ 18 years) presenting to 49 EDs with chest pain, shortness of breath, hypoxia, syncope, presyncope, or hemoptysis who were tested for both SARS-CoV-2 and D-dimer at index ED visit. The primary outcome measure was the sensitivity, specificity, and negative predictive value of D-dimer test thresholds for the outcome of 30-day PE diagnosis.
Results
Among 10,837 patients included in our study, 404 (3.7%) were diagnosed with PE at 30-days. A standard D-Dimer threshold of 500 ng/mL had a sensitivity of 97.8% (95% confidence interval [CI] 95.8–99.0%), specificity of 40.9% (95% CI 39.9–41.8%), and negative predictive value of 99.8% (95% CI 99.6–99.9%). An age-adjusted D-dimer threshold had a sensitivity of 96.0% (95% CI 93.6–97.7%), specificity of 48.5% (95% CI 47.5–49.4%), and negative predictive value of 99.7% (95% CI 99.5–99.8%). D-dimer testing had slightly lower prognostic performance among SARS-CoV-2 positive compared to SARS-CoV-2 negative patients in predicting 30-day PE diagnosis.
Conclusions
Among ED patients with suspected SARS-CoV-2, the standard 500 ng/mL and age-adjusted D-dimer thresholds were comparable for the prediction of PE at 30-days. The prognostic performance of D-dimer was lower among SARS-CoV-2 positive patients.
Trial registration
Clinicaltrials.gov, NCT04702945.
Résumé
Objectif
L'objectif principal était de quantifier l'association pronostique entre différents seuils de D-dimères et le diagnostic d'EP à 30 jours chez les patients des services d'urgence suspectés d'être infectés par le SRAS-CoV-2.
Méthodes
Il s'agissait d'une étude rétrospective des patients inscrits au registre du réseau canadien de réponse rapide aux urgences COVID-19 (CCEDRRN) du 1er mars 2020 au 2 juillet 2021. Nous avons inclus des adultes consécutifs (>18 ans) se présentant dans 49 services d'urgence pour une douleur thoracique, un essoufflement, une hypoxie, une syncope, une présyncope ou une hémoptysie et qui ont été testés à la fois pour le SRAS-CoV-2 et les D-dimères lors de la visite de référence aux urgences. Le principal critère d'évaluation était la sensibilité, la spécificité et la valeur prédictive négative des seuils du test des D-dimères pour le diagnostic de l'EP à 30 jours.
Résultats
Parmi les 10 837 patients inclus dans notre étude, 404 (3,7 %) ont reçu un diagnostic d'EP à 30 jours. Un seuil standard de D-Dimer de 500 ng/mL avait une sensibilité de 97,8 % (intervalle de confiance [IC] à 95 % 95,8-99,0 %), une spécificité de 40,9 % (IC à 95 % 39,9-41,8 %) et une valeur prédictive négative de 99,8 % (IC à 95 % 99,6-99,9 %). Un seuil de D-dimères ajusté à l'âge avait une sensibilité de 96,0% (IC à 95 % 93,6-97,7 %), une spécificité de 48,5% (IC à 95 % 47,5-49,4 %) et une valeur prédictive négative de 99,7 % (IC à 95 % 99,5-99,8 %). Le test des D-dimères avait une performance pronostique légèrement inférieure chez les patients positifs pour le SRAS-CoV-2 par rapport aux patients négatifs pour le SRAS-CoV-2 en ce qui concerne la prédiction du diagnostic d'EP à 30 jours.
Conclusions
Chez les patients des urgences suspectés d'être atteints du SRAS-CoV-2, les seuils standard de 500 ng/ml et les seuils de D-dimères ajustés à l'âge étaient comparables pour la prédiction de l'EP à 30 jours. La performance pronostique des D-dimères était plus faible chez les patients positifs pour le SRAS-CoV-2.
Enregistrement de l'essai
Clinicaltrials.gov, NCT04702945.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
What is known about the topic? |
Pulmonary embolism (PE) risk stratification is particularly challenging in the setting of concurrent suspected SARS-CoV-2 infection. |
What did this study ask? |
What was the prognostic association between various D-dimer thresholds and 30-day PE diagnosis among ED patients with suspected SARS-CoV-2 infection? |
What did this study find? |
In this multi-centre study of ED patients tested for SARS-CoV-2, standard and age-adjusted D-dimer testing strategies ruled out PE diagnosis at 30-days with similarly high sensitivity and negative predictive value, irrespective of patient SARS-CoV-2 status. |
Why does this study matter to clinicians? |
In ED patients with suspected SARS-CoV-2, a negative standard or age-adjusted D-dimer test at index visit is associated with very low incidence of PE within the following 30-days. |
Introduction
Background
Patients with SARS-CoV-2 infection appear to be at elevated risk of venous thromboembolic disease (VTE) [1]. Most studies estimating VTE risk in patients with SARS-CoV-2 infection have focused on hospitalized patients, in whom pulmonary embolism (PE) has been reported in 13–32% of patients depending on SARS-CoV-2 infection severity [2,3,4].
While prior research has explored the diagnostic performance of standard (500 ng/mL) and age-adjusted D-dimer for PE diagnosis in the setting of SARS-CoV-2 [5, 6], as well as the prognostic association between D-dimer and mortality among hospitalized SARS-CoV-2 patients [7, 8], an evaluation of the prognostic association between D-dimer and subsequent PE risk in a cohort of ED patients with suspected SARS-CoV-2 infection is lacking.
Importance
Patients with suspected SARS-CoV-2 often undergo investigation for PE in the ED due to symptom overlap and a paucity of evidence on optimal utilization of D-dimer testing in this patient population [9, 10]. Approximately 1 in 10 patients with SARS-CoV-2 infection have two or more ED visit events over the first 30-days following index diagnosis, often resulting in repeat testing for persistent or worsening symptoms [9].
Evaluating the prognostic association between initial D-dimer values and subsequent PE risk in the acute to a subacute phase of SARS-CoV-2 infection may inform decision-making for ED evaluation of nonspecific cardiorespiratory symptoms, particularly in the setting of recurrent ED visits for persistent symptoms or in resource-limited settings where transport to sites with confirmatory imaging capability is costly and resource-intensive.
Goals of this investigation
Our primary aim was to evaluate the prognostic utility of standard and age-adjusted D-dimer threshold concentrations for predicting 30-day PE diagnosis in ED patients with suspected SARS-CoV-2 infection. Our secondary aim was to compare the prognostic performance of D-dimer thresholds with 30-day PE diagnosis according to SARS-CoV-2 status.
Methods
Study design and setting
This registry-based prognostic association study used data from the multicenter Canadian COVID-19 Emergency Department Rapid Response Network (CCEDRRN) [11]. Details about CCEDRRN, including data collection methods, data quality and validation, standard operating procedures, governance and patient engagement have previously been published [11], along with completed risk score development studies [12,13,14]. The research ethics boards of all participating sites reviewed and approved the study with a waiver for informed consent for registry enrolment. The funding organizations had no role in the study conduct, data analysis, manuscript preparation. CCEDRRN’s patient engagement committee reviewed and provided feedback on the analytic protocol and the manuscript. This study follows the Standards for Reporting Diagnostic accuracy studies (STARD) reporting recommendations [15].
Participants
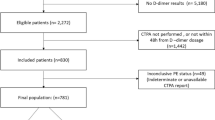
The CCEDRRN registry enrolls consecutive patients tested for SARS-CoV-2 at participating EDs (with a small number of sites enrolling only consecutive patients who tested positive). Our study includes patients enrolled in the registry between March 1, 2020 and July 2, 2021 (Supplementary Table 1) who were 18 years of age or older, tested for SARS-CoV-2 in the ED or within 24 h of arrival at index visit, tested for D-dimer at index ED visit, and presented with one or more of the following presenting triage complaints: chest pain, shortness of breath, hypoxia, syncope, presyncope, or hemoptysis. This symptom list was adapted from prior work evaluating presenting complaint codes that identify ED patients most likely to undergo PE investigation [16, 17].
We defined suspected SARS-CoV-2 infection as administration of a SARS-CoV-2 nucleic acid amplification test at or within 24 h of the index ED visit. At the time of the study, CCEDRRN sites exclusively used nucleic acid amplification testing to diagnose SARS-CoV-2 infection. For patients with multiple ED visits within the enrollment period, we considered the first documented visit within CCEDRRN as the index visit and utilized D-dimer concentrations measured during the first ED index visit. There were no other exclusion criteria.
Data collection
The CCEDRRN registry collected the following data from 49 participating sites: demographic and social variables, vital signs, symptoms, comorbid conditions (derived from the International Severe Acute Respiratory and Emerging Infection Consortium (ISARIC) reporting form [18]) infection risk factors, the results of laboratory tests and diagnostic imaging, health resource utilization and patient outcomes. Trained research assistants abstracted data at each site using electronic medical record extraction and manual review of health records, depending on site-specific documentation practices. Research assistants were blinded to the objectives of this study. The reliability of health record data abstraction was verified by comparing key clinical variables abstracted retrospectively from the health record with prospective data collection in a sample of patients [11]. The CCEDRRN central coordinating office conducted regular data quality checks and verified extreme and outlying values at each participating site to ensure data quality, and a national coordinator reviewed site study logs to ensure capture of ≥ 99% of consecutive eligible patients.
D-dimer measurement
D-dimer assay types at each enrolling site are summarized in Supplementary Table 2. We converted all D-dimer concentrations to ng/mL fibrinogen equivalent units (FEU) by using unit conversions where necessary and using the formula FEU = 2 × D-dimer Units (DDU) [19]. For the standard D-dimer threshold, we considered a concentration less than 500 ng/ml FEU negative for all patients. For the age-adjusted threshold, we considered a concentration less than 500 ng/ml FEU negative in patients younger than age 50 and in patients 50 years of age or older, we considered the D-dimer negative if the concentration was lower than a value of the patient’s age multiplied by 10 [20]. For patients with multiple ED visits, we only evaluated the D-dimer value drawn at index ED visit, although PE outcomes observed on subsequent ED visits within the 30-day follow-up period were included as outcomes.
Outcomes
The primary outcome was the diagnosis of PE at 30-days based on either a positive CTPA test or an ED or hospital diagnosis of PE abstracted by trained research assistants (RAs) reviewing records at each site for any CCEDRRN documented encounter within 30 days of index ED visit. RAs manually reviewed patient records (either electronically or physically depending on site-specific chart storage protocols) and coded outcomes as PE positive if any of the following specific terms were identified on the ED or hospital diagnosis fields: “PE”, “pulmonary embolism”, or “acute pulmonary embolism”. RAs were instructed to code any diagnoses with the word “suspected” as “other” rather than PE positive. ICD-10 codes were not used in the data abstraction protocol. Ventilation Perfusion (VQ) scan results were not captured in CCEDRRN. Outcomes were ascertained using health record reviews for a minimum of 30 days, including any subsequent visits or hospitalizations for patients who were not admitted at the index visit or whose index admission was less than the full 30-day follow-up period [11]. For patients with hospitalization events lasting > 30-days, data were censored at 30-day follow-up.
Statistical analysis
The primary analysis was the prognostic association of standard and age-adjusted D-dimer thresholds and 30-day PE diagnosis. Secondary analyses included evaluation of prespecified D-dimer thresholds (age-adjusted and 500–5000 ng/mL thresholds in 500 ng/mL increments) for 30-day PE diagnosis and prognostic performance stratified by SARS-CoV-2 status.
We calculated the sensitivity, specificity, negative likelihood ratio, and negative predictive value for all D-dimer concentrations with 95% confidence intervals (CIs). Chi-square analysis was used to calculate p-values. We considered a p-value less than 0.05 statistically significant. We conducted all analyses using R version 4.05 and RStudio version 1.3.1093. To ensure patient privacy, a cell size restriction policy prevented us from publishing cell counts under five.
Results
Study population
The CCEDRRN registry enrolled 125,630 patients between March 1, 2020 and July 2, 2021, of which 10,837 (8.6%) met our inclusion criteria (Fig. 1). Among those, 4311 (39.8%) tested positive for SARS-CoV-2. By 30-days follow-up from the index visit, 404 (3.7%) had been diagnosed with a PE based on either CTPA results or ED or hospital diagnosis, with the majority of PE diagnoses made at the time of the index ED visit (Table 1).
Compared with SARS-CoV-2 negative cases, SARS-CoV-2 positive patients were more likely to arrive by ambulance, more likely to present with shortness of breath or hypoxia rather than chest pain, had less smoking or vaping history, had shorter ED length of stay, and were more likely to be hospitalized (60.1% vs 38.8%). The proportion of patients with PE diagnosis at 30 days was lower among SARS-CoV-2 positive patients as compared to SARS-CoV-2 negative patients (3.1% vs 4.1%, respectively; p = 0.004).
Prognostic association of d-dimer testing and 30-day PE
The standard D-dimer threshold of 500 ng/mL had a sensitivity of 97.8% (95% CI 95.8–99.0%), specificity of 40.9% (95% CI 39.9–41.8%), and negative predictive value of 99.8% (95% CI 99.6–99.9%), while the age-adjusted D-dimer had a sensitivity of 96.0% (95% CI 93.6–97.7%), specificity of 48.5% (95% CI 47.5–49.4%), and negative predictive value of 99.7% (95% CI 99.5–99.8%) for the diagnosis of 30-days PE among all enrolled patients (Table 2). All other D-dimer thresholds had sensitivities < 90% but preserved high negative predictive values (Table 2). The prognostic performance of D-dimer was lower among SARS-CoV-2 positive as compared to negative patients. This difference in prognostic performance between SARS-CoV-2 positive as compared to negative patients held true for both the standard 500 ng/mL threshold (sensitivity 96.2% vs 98.5% and specificity 31.4% vs 47.2%, respectively) and the age-adjusted threshold (sensitivity 93.9% vs 97.1% and specificity 38.9% vs 54.9%) (Table 3).
Discussion
Interpretation of findings
Among patients with suspected SARS-CoV-2 infection, both standard (500 ng/mL) and age-adjusted D-dimer thresholds demonstrated strong prognostic associations for identifying patients at low risk of 30-days PE diagnosis, irrespective of final SARS-CoV-2 status. We found prognostic performance to be slightly lower among SARS-CoV-2 positive patients compared to SARS-CoV-2 negative patients.
The observed association between SARS-CoV-2 infection [1] and elevated VTE risk introduces a significant challenge for ED physicians managing patients with persistent non-specific cardiorespiratory symptoms where PE remains on the differential diagnosis. Beyond its utility as a prognostic marker of SARS-CoV-2 infection severity and mortality risk [7, 8], low D-dimer values below a standard (500 ng/mL) or age-adjusted threshold appear to predict a very low risk of 30-days PE occurrence, even among patients with confirmed SARS-CoV-2 infection.
Despite a higher proportion of hospitalizations among SARS-CoV-2 positive patients, we observed a lower 30-day PE prevalence in this cohort. It is possible that this reflects the broader application of D-dimer for indications beyond VTE diagnosis in patients with confirmed SARS-CoV-2 infection (ie. trending infection severity). Alternatively, it may be due to baseline imbalances in pre-test probability for PE between the two groups, which we were unable to ascertain utilizing data from the CCEDRRN registry.
Comparison to previous studies
Our findings expand upon prior literature demonstrating the prognostic role of D-dimer among SARS-CoV-2 patients [7, 8] and suggests that symptomatic ED patients with negative index visit D-dimer (according to either standard or age-adjusted thresholds) are at low risk of subsequent 30-days PE complications, irrespective of SARS-CoV-2 status. These findings should not be directly compared to existing diagnostic accuracy studies given methodological differences, however, results may help inform clinician considerations for potentially low-value repeat PE testing in patients with recurrent ED visits for nonspecific cardiorespiratory symptoms in the setting of suspected SARS-CoV-2.
We found a 30-day PE prevalence (3.7%) lower than prior literature focused on VTE prevalence in hospitalized SARS-CoV-2 patients [2,3,4]. This likely reflects the broad range of both symptom severity and pre-test probability for PE captured within our cohort.
Strengths and limitations
Our analysis captured a broad and representative spectrum of symptomatic ED patients including 53% who were well enough to be discharged home. The inclusion of all patients with suspected SARS-CoV-2, irrespective of final SARS-CoV-2 test result, reflects the clinical context in which most ED physicians currently practice, where SARS-CoV-2 status is often not confirmed at the first point of contact, and where both SARS-CoV-2 infection and PE remain on the differential diagnosis.
Our study was unable to capture clinician gestalt for pre-test probability of PE, and other variables required for clinical risk stratification tools (such as the Wells PE and YEARS criteria) [21, 22]. We were therefore unable to evaluate the combined prognostic performance of D-dimer with pre-test probability tools in this study population. By including all eligible patients who received D-dimer testing in our cohort, we potentially captured an overinclusive sample where D-dimer testing was performed for SARS-CoV-2 prognostication rather than PE investigation. This could have skewed the population either higher or lower for overall pre-test probability compared to a general ED population undergoing PE testing (ie. higher if it was selected for more symptomatic patients, lower if it excluded patients who were felt to be high enough risk that they proceeded directly to diagnostic imaging without D-dimer risk stratification). Despite this, our results demonstrate high negative predictive value across all tested patients at both standard and age-adjusted D-dimer test thresholds. Readers should keep in mind that negative predictive value is affected by disease prevalence and should be interpreted in the context of our low (3.7%) observed event rate. Ventilation perfusion (VQ) scan results were unavailable in the CCEDRRN database and therefore were not included in the study outcome. This would likely have only missed a small number of 30-days PE cases as CTPA testing was generally the preferred modality for PE imaging during the pandemic [23]. We were unable to capture mortality data due to provincial restrictions on data access. Finally, the multicenter nature of this study relied on D-dimer testing across 49 Canadian ED sites with varying D-dimer analytic platforms and assays (Supplementary Table 2). Study findings reflect aggregate performance across all test assays.
Clinical implications
ED patients with suspected SARS-CoV-2 infection and negative index-visit D-dimer results by either standard (500 ng/mL) or age-adjusted thresholds are at low risk of 30-day PE complications. This may inform patient-centered discussions around the utility of PE imaging at the index encounter or subsequent encounters for persistent symptoms.
Research implications
Future studies that integrate pre-test probability estimates and quantitative D-dimer results will be important to enable precise risk stratification of patients undergoing PE workup in the setting of suspected SARS-CoV-2 symptoms.
Conclusion
Among ED patients with suspected SARS-CoV-2 infection, a negative D-dimer test result at either standard (500 ng/mL) or age-adjusted test thresholds during the index ED visit predicts a low risk of 30-day PE complications, irrespective of final SARS-CoV-2 status.
Data availability
The data that support the findings of this study are available on request from the corresponding author, KL.
References
Kollias A, Kyriakoulis KG, Lagou S, et al. Venous thromboembolism in COVID-19: a systematic review and meta-analysis. Vasc Med. 2021;26:415–25. https://doi.org/10.1177/1358863X21995566.
Gervaise A, Bouzad C, Peroux E, et al. Acute pulmonary embolism in non-hospitalized COVID-19 patients referred to CTPA by emergency department. Eur Radiol. 2020;30:6170–7. https://doi.org/10.1007/s00330-020-06977-5.
Zhang C, Shen L, Le K-J, et al. Incidence of venous thromboembolism in hospitalized coronavirus disease 2019 patients: a systematic review and meta-analysis. Front Cardiovasc Med. 2020;7:151. https://doi.org/10.3389/fcvm.2020.00151.
Malas MB, Naazie IN, Elsayed N, et al. Thromboembolism risk of COVID-19 is high and associated with a higher risk of mortality: a systematic review and meta-analysis. EClinicalMedicine. 2020;29–30: 100639. https://doi.org/10.1016/j.eclinm.2020.100639.
Elberts S, Bateman R, Koutsoubis A, et al. The impact of COVID-19 on the sensitivity of D-dimer for pulmonary embolism. Acad Emerg Med. 2021;28(10):1142–9.
Machowski M, Poganska A, Galecka-Nowak M, et al. Age-adjusted D-dimer levels may improve diagnostic assessment for pulmonary EMbolism in COVID-19 patients. J Clinic Med. 2022;11(12):3298.
Poudel A, Poudel Y, Adhikari A, et al. D-dimer as a biomarker for assessment of COVID-19 prognosis: D-dimer levels on admission and its role in predicting disease outcome in hospitalized patients with COVID-1. PLoS One. 2021;16(8): e0256744.
Zhang L, Yan X, Fan Q, et al. D-dimer levels on admission to predict in-hospital mortality in patients with COVID-19. J Thromb Haemostat. 2020;18(6):1324–9.
Hohl CM, Rosychuk RJ, Hau JP, et al. Treatments, resource utilization, and outcomes of COVID-19 patients presenting to the emergency department across pandemic waves. Emerg Med. 2021. https://doi.org/10.1101/2021.07.30.21261288.
Rostami M, Mansouritorghabeh H. D-dimer level in COVID-19 infection: a systematic review. Expert Rev Hematol. 2020;13:1265–75. https://doi.org/10.1080/17474086.2020.1831383.
Hohl CM, Rosychuk RJ, McRae AD, et al. Development of the Canadian COVID-19 Emergency Department Rapid Response Network population-based registry: a methodology study. CMAJ Open. 2021;9:261–70. https://doi.org/10.9778/cmajo.20200290.
Hohl CM, Rosychuk RJ, Archambault PM, et al. The CCEDRRN COVID-19 Mortality Score to predict death among nonpalliative patients with COVID-19 presenting to emergency departments: a derivation and validation study. CMAJ Open. 2022;10:E90–9. https://doi.org/10.9778/cmajo.20210243.
Davis P, Rosychuk RJ, Hau JP, et al. Diagnostic yield of screening for SARS-CoV-2 among patients admitted for alternate diagnoses. medRxiv. 2021. https://doi.org/10.1101/2021.09.23.21264036. (Published Online First: 27 September 2021).
McRae AD, Hohl CM, Rosychuk R, et al. CCEDRRN COVID-19 Infection Score (CCIS): development and validation in a Canadian cohort of a clinical risk score to predict SARS-CoV-2 infection in patients presenting to the emergency department with suspected COVID-19. BMJ Open. 2021;11: e055832. https://doi.org/10.1136/bmjopen-2021-055832.
Bossuyt PM, Reitsma JB, Bruns DE, et al. STARD 2015: an updated list of essential items for reporting diagnostic accuracy studies. BMJ. 2015. https://doi.org/10.1136/bmj.h5527.
Burles K, Innes G, Senior K, et al. Limitations of pulmonary embolism ICD-10 codes in emergency department administrative data: let the buyer beware. BMC Med Res Methodol. 2017;17:89. https://doi.org/10.1186/s12874-017-0361-1.
Andruchow JE, Grigat D, McRae AD, et al. Decision support for computed tomography in the emergency department: a multicenter cluster-randomized controlled trial. CJEM. 2021;23:631–40. https://doi.org/10.1007/s43678-021-00170-3.
ISARIC. Clinical Data Collection – The COVID-19 Case Report Forms (CRFs). 2020. https://isaric.org/research/covid-19-clinical-research-resources/covid-19-crf/
Soo T. The D-dimer Debate. Maimonides Emerg. Med. https://www.maimonidesem.org/blog/the-d-dimer-debate (accessed 28 Apr 2022).
Righini M, Van Es J, Den Exter PL, et al. Age-adjusted D-dimer cutoff levels to rule out pulmonary embolism: the ADJUST-PE study. JAMA. 2014;311:1117. https://doi.org/10.1001/jama.2014.2135.
Wells PS, Anderson DR, Rodger M, et al. Excluding pulmonary embolism at the bedside without diagnostic imaging: management of patients with suspected pulmonary embolism presenting to the emergency department by using a simple clinical model and d-dimer. Ann Intern Med. 2001;135:98–107. https://doi.org/10.7326/0003-4819-135-2-200107170-00010.
van der Hulle T, Cheung WY, Kooij S, et al. Simplified diagnostic management of suspected pulmonary embolism (the YEARS study): a prospective, multicentre, cohort study. Lancet Lond Engl. 2017;390:289–97. https://doi.org/10.1016/S0140-6736(17)30885-1.
Schaefer W, Knollman D, Meyer P. V/Q SPECT/CT in the time of COVID-19: changing the order to improve safety without sacrificing accuracy. J of Nut Med. 2021;62(7):1022–4.
Acknowledgements
We thank the CCEDRRN trial investigators, the clinical research coordinators, and the research support teams at all participating centers for their partnership in the data collection required to complete this study.
Funding
The network is funded by the Canadian Institutes of Health Research (447679), Ontario Ministry of Colleges and Universities (C-655-2129), Saskatchewan Health Research Foundation (5357), Genome BC (COV024 and VAC007) Fondation du CHU de Québec (Octroi No. 4007) Sero-Surveillance and Research (COVID-19 Immunity Task Force Initiative) provided peer-reviewed funding. The BC Academic Health Science Network and BioTalent Canada provided non-peer-reviewed funding. These organizations are not-for-profit and had no role in study conduct, analysis, or manuscript preparation.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The study authors have no conflicts of interest to declare. All authors vouch for the accuracy and completeness of the data, and for adherence to the protocol.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, K., Xu, K., Daoust, R. et al. Prognostic association between d-dimer thresholds and 30-day pulmonary embolism diagnosis among emergency department patients with suspected SARS-CoV-2 infection: a Canadian COVID-19 Emergency Department Rapid Response Network study. Can J Emerg Med 25, 134–142 (2023). https://doi.org/10.1007/s43678-022-00440-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43678-022-00440-8
Keywords
- COVID-19
- Coronavirus disease 2019
- SARS-COV-2
- D-dimer
- Pulmonary embolism
- Venous thromboembolism
- Computed tomography pulmonary angiogram