Abstract
Purpose of Review
This review highlights the importance of exploring microorganisms with probiotic potential—mainly lactic acid bacteria and yeasts—which are isolated from traditional fermented products that come from Africa, Asia, and Latin America. We greatly underline the potential these microorganisms have when it comes to conceiving non-dairy foods. After all, these microorganisms resulted in products with greater nutritional value and improved sensory characteristics, such as aroma, texture, and flavor.
Recent Findings
Studies indicate that demand for products containing probiotics is increasing in markets as consumers seek healthier options for their diets. In response to this trend, the food industry has strived to produce more fermented products based on cereals, legumes, roots, and tubers so as to serve specific target groups. It is well known that consuming probiotics has several health benefits, including reduced acute inflammation and decreased risk of cardiovascular disease. These benefits occur due to the ability these microorganisms have of reducing cholesterol through the deconjugation of bile salts.
Summary
Microorganisms play a fundamental role in our existence, being essential assets in the production of a variety of fermented foods and drinks that are part of our diets. Much research has focused on elucidating the benefits these microorganisms can bestow our health. This study emphasizes the relevance of microorganisms with probiotic potential, which are isolated from various food sources, including cereals, roots, tubers, and vegetables. These sources are of great economic and social importance in African, Asian, and South American countries, as they represent a source of income for thousands of families and small farmers, in addition to comprising the nutritional foundation of these populations. The utilization of these probiotic strains presents a significant economic impact, as it improves the quality and safety of food as well as contributing towards the improvement of human health.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A wide variety of fermented foods are produced and consumed in different regions of the world [1••]. They are defined as food produced through controlled microbial growth and the enzymatic transformation of major and minor components [2]. Fermented products are important food sources in Asia, Africa, and Latin America due to their sensory properties, high nutritional value, and ease of digestion [3]. Furthermore, these foods can act on the human body, as it is an important vehicle for probiotics [4••].
Fermentation is an old food processing method employed throughout the world that emerged with the goal of food conservation. Moreover, it is a part of the cultures of people whose knowledge spans generations. These products serve nutritional sources for many families in which several individuals live on account of the production of these foods [5•]. These fermented foods are produced from local raw materials and develop important products in the overall composition of diets [6], as well as foods with high nutritional value and with more enticing aromas, flavors, and textures [5•]. Fermented foods are produced from a variety of raw materials such as milk, cereals, legumes, roots, fruits, vegetables, meats, and fish [5•]. Africa is one of the continents that rely heavily on fermented foods in order to attain balanced and ideal diets. In Latin America, fermented food has been produced and associated with its tradition since pre-Hispanic times [4••]. Asians consume a variety of vegetables—both domesticated and wild—across different recipes, using indigenous knowledge of bio preservation [7].
A variety of traditional fermented products containing probiotic bacteria are sold in popular establishments in Asia [8]. Among these products, one can highlight fermented soy and vegetables such as cabbage, beet, turnips, cauliflower, green beans, and cucumber [9]. The food found in African culture plays a key role: It is prepared from cereals such as corn, rice, millet, or sorghum. The native population of Latin America produces fermented foods and beverages mainly from maize, rice, and cassava that are important nutritional sources for these communities [10, 11]. Despite the abundance of fermented foods and their microbiological variety, there are still several challenges when it comes to deciphering the real health benefits obtained from the consumption of these products [12].
Preparation, fermentation time, and raw materials differ across the globe [13•]. The fermentation process used to obtain traditional products happens spontaneously and in rustic environments, with a diverse microbiota found in its raw materials and environment (Table 1) [13•, 14]. These fermented foods are also produced via traditional methods which employ pure and mixed cultures of various microorganisms such as lactic acid bacteria, yeasts, and non-lactic acid bacteria and fungi [4••, 8]. Some species of LAB and yeast possess characteristic antibiotic compounds with biotechnological applications. They also have probiotic effects, which result in intestinal modulation, prevention and the treatment of gastrointestinal diseases, hypocholesterolemia effects, and antihypertensive, hypoglycemic, and immunomodulator properties [15••, 16]. To be used in food, these probiotic microorganisms must be able to survive the passage through the digestive tract in order to grow within the intestine. This means that they must either resist gastric fluid and be able to grow in the presence of bile under intestinal conditions, or be consumed in a food vehicle that allows them to survive during the passage through the stomach and exposure to bile (Fig. 1) [17].
Demand for probiotic foods is increasing due to their health benefits and growing consumer preferences. Although most probiotic products are derived from milk, lactose intolerance, milk protein allergy, high cholesterol content, high contents of saturated fatty acids, and vegetarianism limit their consumer population and inspired the idea of developing dairy-free probiotic products [47]. Therefore, non-dairy fermented foods with unique organoleptic properties derived from raw materials (cereals, tubers, legumes, and roots) need to be further explored. The food industry uses probiotic microorganisms isolated predominantly from substrates of animal origin such as dairy fermented food; however, they are also found in non-dairy fermented foods [5•]. Therefore, this review aims to present different microbial strains that have probiotic potential found in non-dairy fermented foods.
Probiotic Potential of Traditional Fermented Food
Microorganisms with probiotic potential are found in traditional fermented foods [4, 7]. Probiotics are defined as live microorganisms that provide health benefits to the host when administered according to specific needs [17, 23, 52, 53]. Strains considered probiotics have various characteristics, including safety and physiological features and technological properties such as antibiotic susceptibility, acid tolerance, bile salt deconjugation, cell surface hydrophobicity, self-aggregation, co-aggregation with pathogens, activity against pathogens, and survival in gastrointestinal conditions which must all be taken into account. In the food matrix, NaCl tolerance, exopolysaccharides, and lipolytic and proteolytic enzyme production should also be considered (Fig. 1) [15••, 54].
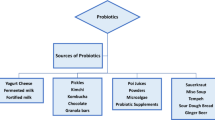
In this review, we will introduce some of the main traditional fermented products from Asia, Africa, and Latin America, while highlighting the presence of microbial strains that possess probiotic potential. Among the products described in this study are gari, agbelima, lafun, fufu, jalebi, tapuy, puba, tucupi, sour cassava starch, calugi, yakupa, tarubá, cauim, masato of yuca, chicha, dégué, tempe, among others (Table 1).
Lactic-Acid Bacteria
Products fermented by lactic-acid bacteria (LAB) represent a wide variety of functional foods and can provide probiotic strains. These microorganisms could be attractive for the food industry, mainly for producing gluten-free, lactose-free, and plant-based products [13•]. LABs can produce an active probiotic substance such as BLISs (bacteriocin-like substances)—a cellular complex—that allows the interaction with the host’s mucous membranes, modulating the immune system beneficially [5•, 55]. Since they stimulate the interaction between microorganisms and hosts, promoting their survival within the intestine and ensuring intestinal barrier protection through various mechanisms, the adhesion of lactic acid bacteria to epithelial cells and mucosal surfaces has been employed as a potential probiotic marker [26].
Lactobacillus, Lactiplantibacillus, Limosilactobacillus, and Lacticaseibacillus
Lactiplantibacillus plantarum stands out among other LABs as one of the microorganisms most frequently found in the vegetable fermentation process. This species is predominant in several products from Africa, such as gari, ogi, uji, agbelima, ting, lafun, and fufu [4, 5•, 12, 18, 27]; from Asia, such as jalebi, tapuy, and chinese steamed bun (comme) [9, 19, 24]; and from Latin America, such as puba, tucupi, sour cassava starch, calugi, yakupa, tarubá, cauim, tocosh, pozol, masato of yuca, chicha, and atol agrio [14, 20, 29].
Strains of L. plantarum and Lactobacillus acidophilus could tolerate the high-temperature thermal treatment (50–56°C) and were selected to verify their probiotic ability. These strains also presented high inhibitory activity against Salmonella enterica serovar Enteritidis, Salmonella enterica serovar Typhimurium, Escherichia coli, Staphylococcus aureus, and Listeria monocytogenes with a medium inhibition diameter higher than 15 mm. These species also showed tolerances to pH 3.0 and 0.3% (v/v) oxgall bile. Hence, the authors concluded that due to their properties, both strains are potentially probiotic and can be employed in the industrial processing of probiotic products in the future [21].
The CCMA 0743 strain of L. plantarum isolated from cassava cauim, a beverage produced by indigenous people in the North of Brazil, was able to tolerate pH 2.0 and grow in the presence of 0.3% biliary salts. This resistance is required in order to reach the human intestine and showcases their probiotic potential. In this paper, it was also observed that the L. plantarum isolates reached the desirable levels for a product to be considered probiotic (8.0 log UFC/mL) [10]. A probiotic food must contain at least 108 UFC/g of viable microorganisms, depending on the microorganism and the consumer’s physiological conditions [28, 52]. Rebaza‑Cardenas et al. [51] examined different strains of L. plantarum isolated from the poli-cereal mixture chicha. One of the L. plantarum strains showed better resistance to in vitro gastrointestinal traffic and a major ability of adhesion to the intestinal epithelium in comparison to commercial probiotic strains [51].
Limosilactobacillus fermentum strains of these microorganisms were isolated from a wide variety of fermented foods, such as gari, ogi, uji, hussuwa, ting, agbelima, kunu-zaki, dégué, lafun, fufu [4••, 5•, 18, 27, 46, 48•, 50], jalebi and tempe [7, 19], puba, sour cassava starch, tucupi, yakupa, caxiri, pozol, and cassava’s chicha [14, 30, 32, 36, 38, 40,41,42, 45, 56].
Nascimento et al. [49] reported that the ingestion of L. fermentum strains from different sources, including fermented food, is capable of improving the composition of intestinal microbiota, reducing acute systemic inflammation, enhancing the short-chain fat acid compound in the colon, lowering blood pressure and dyslipidemia, and enhancing antioxidant defenses in the colon, liver, heart, and kidneys [49]. Lacerda et al. [31] evaluated various studies that reported the use of L. fermentum strains isolated from different sources, such as Chinese rice noodle wastewater—among others—with probiotic potential concerning the management of diabetes mellitus and observed that the consumption of different strains provided varying effects. Furthermore, some strains showed advantages when handling type 2 diabetes and their daily ingestion could be responsible for mitigating the complications of type 1 diabetes [31].
Strains of Lactobacillus gasseri are predominant in the small intestine and are also found in different fermented foods such as dégué, sour cassava starch, and puba [12, 38, 41, 50]. Studies reported that these strains contribute to immunological regulation, preventing bacterial and viral infections, reducing allergic symptoms, inhibiting lipid absorption, and exerting anti-tumor effects [43, 57].
In Africa, Levilactobacillus brevis strains are part of the microbial diversity of dégué, mahewu, gari, and agbelima [5•, 27, 50, 58, 59]. In Latin products, L. brevis strains are found in agrio, rice cauim, corn chicha, tarubá, sour cassava starch, and puba [14, 33, 37, 38, 41, 42, 56, 60, 61]. In Asia, they are present in sourdough [24]. Several strains of this species are classified as probiotic because different studies showed that L. brevis improves immunity and mind health, relieving gastrointestinal tract irritation and enhancing the natural resistance to pathogens [62, 63].
Lacticaseibacillus casei can be isolated from food, but also from human reproductive and gastrointestinal tracts, which explains its wide implementation as a probiotic [64]. L. casei strains were identified in some fermented foods like dégué, fufu, ogi, and kunu-zaki (Africa) [50, 59]; Chinese steamed bun (comme) (Asia) [24]; and pozol, tocosh, rice chicha, and puba (Latin America) [33, 41, 65, 66]. Several strains of this species represent not only potential but actual probiotic bacteria. Studies showed that L. casei strains are effective in immunological response modulations, in the protection against diabetes mellitus, in exerting anti-inflammatory effects over intestinal epithelial cells, in reducing time and preventative measures required for respiratory infections, as well as anti-arthritic properties, the modulation of intestinal microbiota [64].
Lacticaseibacillus paracasei compounds the microbiota of Uji and Mahewu [50, 67]. Recently, new strains were described as potential probiotics due to their ability to inhibit pathogens and functional properties. Hence, this species is a good candidate for a stater culture with probiotic claims [67]. In addition, results pointed out the ability to inhibit the growth of common pathogenic bacteria, highlighting their probiotic potential. Thus, these results can be useful for a deeper comprehension of L. paracasei as well as for the development of new starter cultures and probiotic preparations based on this LAB in the future [67].
Lactobacillus delbrueckii are the predominant bacteria during the mahewu fermentation process and can be found in ogi and fufu fermentation [48•, 50, 59, 68]. In Asia, L. delbrueckii subsp. lactis was isolated in tempe and the L. delbrueckii species in kha nhom Jeen (rice noodle) and tauco [7, 24]. Between the Latin American products, L. delbrueckii was found in cassava chicha, pozol, and puba [36, 41, 65].
A study performed by Guglielmotti et al. [34] showed that different strains of L. delbrueckii presented a limited resistance to bile, while most strains tolerated lysozymes, growing in bile salts and fermenting in prebiotic presence. Moreover, they showed strong antibacterial activity because of the lactic acid production. The studied strains were also capable of joining Caco-2/TC-7 monolayers and significantly inhibited the invasion of Salmonella enterica serovar Enteritidis in Caco-2/TC-7cell. Hence, all tested strains presented probiotic characteristics, suggesting an important probiotic potential role for L. delbrueckii [34].
Pediococcus spp
Pediococcus spp. are frequently found in foods obtained from vegetable fermentation processes. Among the African and Asian products obtained from vegetable fermentation, Pediococcus were isolated from uji (Ped. acidilactici and Ped. pentosaceus); hussuwa, sufu, maesura, and rabadi (Ped. acidilactici); and ogi, kunu-zaki, fufu, selroti, sourdough, and tape ketan (Ped. pentosaceus) [5•, 7, 24, 48•, 50]. In Latin American products, they were found in atole agrio (Ped. pentosaceus), caxiri, corn chicha (Ped. acidilactici), and rice cauim (Ped. guilliermondii) [14, 25, 60, 61].
Ped. acidilactici, isolated from milk products, exhibits a probiotic potential due to its ability to reduce high blood sugar levels and low-density lipoprotein and inhibit pathogens. The capacity to reduce blood sugar levels is more effective than metformin. Consequently, this potential probiotic prevents hyperglycemia, hypercholesterolemia, and gastrointestinal infections [69]. Likewise, Ped. pentosaceus produces BLISs with antibacterial properties and anti-inflammatory, anticancer, antioxidant, detox, and lipid reducer abilities [39].
Leuconostoc
The probiotic claim for Leuconostoc resides in antimicrobial activities due to the production of BLISs [70]. Leuconostoc species were already found in Asian products such as doenjang, idli, selroti, puto, and saké (Leuc. mesenteroides) and sourdough (Leuc. citreum) [7, 9, 24]. Leuconostoc strains were also isolated from African foods, such as mahewu (Leuconostoc spp.), gari (Leuc. fallax and Leuc. pseudomesenteroides), fufu (Leuc. mesenteroides), agbelima (Leuc. mesenteroides), and lafun (Leuconostoc sp.) [4, 5•, 18, 27, 46, 48•, 50]. Across Latin American products, Leuconostoc strains were identified in atole agrio (Leuc. pseudomesenteroides), corn chicha (Leuc. mesenteroides and Leuc. lactis), rice chicha (Leuc. lactis), tarubá (Leuc. mesenteroides and Leuc. lactis), sour cassava starch (Leuc. mesenteroides, Leuc. pseudomesenteroides, and Leuc. citreum), and puba (Leuc. mesenteroides) [33, 37, 41, 56, 60, 66, 71].
Lactococcus lactis
Lactococcus lactis was identified in Latin-American products, such as atole agrio, sour cassava starch, and puba [33, 38, 41, 56, 61, 71], as well as in African products such as mahewu and gari [46, 50]. In Asia, this species was isolated from kha nhom Jeen (rice noodles) [24].
Lactococcus lactis is an LAB that is widely used for the fermentation of dairy and non-dairy foods, such as cheese, yogurt, and sauerkraut. This LAB species is generally recognized as safe (GRAS) by the Food and Drug Administration (FDA). Beyond the acid production, these bacteria are capable of producing bacteriocin, presenting antagonistic effects against pathogens [70]. Lc. lactis has gastrointestinal tract resistance, immunomodulatory activity, anti-allergy, anti-cancer, and healing capabilities [72, 73]. A study with two strains of Lc. lactis reported the resistance to bile salts, low pH, osmotic stress, and adhesion to intestinal cells, in addition to presenting antiproliferative effects in colorectal cancer cells [44, 73]. A study with an Lc. lactis strain isolated from a fermented product showed healing activities for induced colitis in animal models and inhibited intestinal inflammation through the suppression of inflammatory factors, improvement of the epithelial intestinal barriers and short-chain fat acid concentration regulation. A verified inflammation inhibition mechanism was the inhibition of the NF-κB and MAP, signaling routes that are key regulators during inflammatory processes [44].
Yeasts
Yeasts, due to their natural resistance to antibacterials [74], have advantageous characteristics when used as probiotics. They can help treat gastrointestinal conditions, whether acute, such as retroviral and bacterial diarrhea, or chronic, such as inflammatory diseases. Additionally, yeasts can reinforce humoral, innate immunity, and improve the composition of intestinal microflora in healthy individuals [75].
Saccharomyces
Saccharomyces strains are directly associated to the fermentation of several African, Asian, and Latin American products. Saccharomyces cerevisiae ferments foods such as ogi, lafun, gari, fufu [4, 5•, 18, 27, 46, 48•, 68], selroti, saké, idli, jalebi, takju [7, 9], calugi, yakupa, cauim, chicha, and champús [11, 14, 25, 30, 36, 45, 76, 77].
S. cerevisiae 905, isolated from cachaça, was able to reach potentially functional population levels in the gastrointestinal regions of mice, against the enteropathogenic bacteria Salmonella Typhimurium and Clostridium difficile. However, antagonism against any pathogenic bacteria was not shown by this yeast in gnotobiotic mices’ the digestive tracts. The mortality was lower, and the liver tissue was better preserved in conventional animals treated with yeast against S. Typhimurium comparison to the control group. Intestinal histopathological results showed a good protective effect against oral problems with C. difficile in gnotobiotic mice [35].
Menezes et al. [78] evaluated the probiotic potential of S. cerevisiae strains isolated from cassava caxiri with a chewed sweet potato inoculum. The isolated majority showed the capability of co-aggregating with the Escherichia coli pathogens, while some also showed this ability with Salmonella Enteriditis and Listeria monocytogenes. Yeasts reduced pathogen adhesion and bacterial infection in Caco-2 cells [78]. In a previous study performed with S. cerevisiae strains isolated from caxiri, Menezes et al. [16] reported the capability the isolates showcased of tolerating the gastrointestinal tract conditions (low pH, bile salts, and 37°C temperature). Furthermore, strains were able to produce phytase, increasing nutrient availability. Yeasts showed high hydrophobicity, auto-aggregation, and co-aggregation with E. coli and Caco-2 cell adhesion. These characteristics are relevant for new probiotic strain research [16].
Debaryomyces hansenii
Debaryomyces hansenii, involved in the fermentation of ogi [46] and Asian products such as idli, selroti, doenjang, and tapuy [7, 9], has probiotic properties. Oral administration of these species’ strain result in immunostimulatory effects, intestinal microbiota modulation, increases in cell proliferation and differentiation as well as improvements in digestive function [79].
Pichia spp
Pichia strains are part of the microbial diversity of some African and Latin American products such as P. kudriavzevii (ogi and yakupa), P. scutulata, P. kudriavzevii, P. rhodanensis (lafun), P. scutulata (gari and sour cassava starch), P. guilliermondii (rice cauim), P. fermentans (champús), P. kluyveri, and P. kudriavzevii (rice chicha) [11, 18, 22, 27, 30, 38, 48•, 50, 68, 77, 80].
Two strains of P. kudriavzevii isolated from ogi showed the optimal ability of auto-aggregation, capabilities to remove cholesterol and eliminate free radicals on the methanol reaction system, and enhanced co-aggregation for a strain of P. kudriavzevii, and Escherichia coli [80].
Kluyveromyces marxianus
Kluyveromyces marxianus can be found during the fermentation process of African products such as gari and lafun [18, 27, 48•, 50].
Five strains of K. marxianus, tested in a simulated gastrointestinal environment, showcased the best survival capabilities compared to the probiotic yeast strain S. boulardii MYA-796. Additionally, the five strains showed different lactose, xylitol, d-sorbitol and lactate assimilation, great cell surface hydrophobicity, biofilm production, and auto-aggregation capabilities. These characteristics indicate a major survival potential in the host’s gastrointestinal tract, suggesting its use as a probiotic [81].
Conclusion
This review focuses on the microbial diversity in fermented foods in Africa, Asia, and Latin America, highlighting the importance of the probiotic potential of the microorganisms involved in these fermentative processes. In this way, fermented foods become vehicles for probiotics and can be considered natural reservoirs of new strains with this potential, providing different health benefits to the consumer. Probiotic abilities include functional properties, nutritional enrichment, immunological effects, and protective properties.
In recent years, interest in and consumption of fermented products has increased due to the natural presence or direct incorporation of probiotics. However, choosing appropriate food systems to deliver probiotics is crucial. Always consider when developing probiotic foods. Although most probiotic products are sold as dairy products, there is growing interest in creating new probiotic foods, mainly plant-based products. Furthermore, despite the strains probiotic potential, they must meet strict safety, functionality, and quality criteria. Although probiotics are GRAS (recognized as safe) organisms, variation in regulatory standards between countries adds another layer of complexity to incorporating probiotics into food products.
It is essential to raise awareness among those producing probiotic foods and teach them about advanced production technologies. Furthermore, adopting policies and guidelines that provide food security and promote sustainability is necessary. During the development of probiotic food, several technological difficulties arise, such as the selection of the food matrix for the probiotic, the appropriate choice of strain for industrial production, the microbial stability of the starter culture, sensory acceptance, the nutritional value of the new product, stability during storage, and the type of packaging, which can also affect the survival of probiotics. Economic aspects, health benefits, and the practicality of developed foods also play a crucial role. Studies point to the feasibility of using probiotic starter cultures derived from plant products, legumes, and cereals.
The future of probiotic foods will lie in product innovation, such as plant-based foods fermented with probiotic starter cultures. Recognizing the potential of these foods is just the beginning. It is equally crucial to dedicate significant efforts to uncovering different probiotic capabilities.
Data availability
Not applicable.
References
Papers of particular interest, publish recently, have been highlighted as: • Of importance •• Of major importance
Adebo OA, Chinma CE, Obadina AO, Soares AG, Panda SK, Gan R-Y. An insight into indigenous fermented foods for the tropics. Adebo OA, Chinma CE, Obadina AO, Soares AG, Panda SK, Gan R-Y, organizadores. Indi Ferm Foods for the Trop. 2023;1–12. https://doi.org/10.1016/B978-0-323-98341-9.00003-7. This book chapter discusses the importance of fermented products in Africa, Asia, and South America and provides a brief overview of the fermented food products available. Many of the products mentioned here have their entire production process described in this book.
Marco ML, Heeney D, Binda S, Cifelli CJ, Cotter PD, Foligné B, et al. Health benefits of fermented foods: microbiota and beyond. Curr Opin Biotechnol. 2016;44:94–102. https://doi.org/10.1016/j.copbio.2016.11.010.
Wood BJB. Microbiology of Fermented Foods. 1998;2. https://doi.org/10.1007/978-1-4613-0309-1.
Cuamatzin‐García L, RodríguezRrugarcía P, El‐Kassis EG, Galicia G, Meza‐jiménez M de L, Baños‐Lara MDR, et al. Traditional fermented foods and beverages from around the world and their health benefits. Microorganisms. 2022:10. https://doi.org/10.3390/microorganisms10061151. This work focuses on studies that use traditional fermented foods and beverages as important in promoting human health. Many microorganisms mentioned here were described in this paper.
Ilango S, Antony U. Probiotic microorganisms from non-dairy traditional fermented foods. Trends Food Sci Technol. 2021;118:617–38. https://doi.org/10.1016/j.tifs.2021.05.034. This paper is an attempt to compare scientific progress regarding probiotics in fermented foods from Africa and Asia, where these foods can serve as affordable probiotic supplements. Some foods and microorganisms mentioned here are described in this article.
Gomes FCO, Lacerda ICA, Libkind D, Lopes CA, Carvajal J, Rosa CA. Traditional foods and beverages from South America: microbial communities and production strategies. Em: Krause J, Fleischer O, Organizadores. Industrial Fermentation: food processes, nutrient sources and producting strategies. Nova Science Pub; 2010. pp. 386.
Tamang B, Tamang JP. Role of lactic acid bacteria and their functional properties in goyang, a fermented leafy vegetable product of the sherpas. J Hill Res. 2007;20(2):53–61.
Murooka Y, Yamshita M. Traditional healthful fermented products of Japan. Ind Microbiol Biotechnol. 2008;35:791–8. https://doi.org/10.1007/s10295-008-0362-5.
Rhee SJ, Lee J-E, Lee C-H. Importance of lactic acid bacteria in Asian fermented foods. Microbial Cell Factories. 2011;10. https://doi.org/10.1186/1475-2859-10-S1-S5.
Freire AL, Ramos CL, Schwan RF. Effect of symbiotic interaction between a fructooligosaccharide and probiotic on the kinetic fermentation and chemical profile of maize blended rice beverages. Food Res Int. 2017;100:698–707. https://doi.org/10.1016/j.foodres.2017.07.070.
Schwan RF, Almeida EG, Souza-Dias MAG, Jespersen L. Yeast diversity in rice-cassava fermentations produced by the indigenous Tapirapé people of Brazil. FEMS Yeast Res. 2007;7:966–72. https://doi.org/10.1111/j.1567-1364.2007.00241.x.
Anukam KC, Reid G. African traditional fermented foods and probiotics. J Med Food. 2009;12:1177–84. https://doi.org/10.1089/jmf.2008.0163.
Carboni AD, Martins GN, Gómez-Zavaglia A, Castilho PC. Lactic acid bacteria in the production of traditional fermented foods and beverages of Latin America. Fermentation. 2023;9:315. https://doi.org/10.3390/fermentation9040315. This review summarizes the main characteristics of fermented foods and beverages made with LAB traditionally elaborated by the native population of Latin America. Some products mentioned here are described in this article.
Ramos CL, de Almeida EG, de Melo Pereira GV, Cardoso PG, Dias ES, Schwan RF. Determination of dynamic characteristics of microbiota in a fermented beverage produced by Brazilian Amerindians using culture-dependent and culture-independent methods. Int J Food Microbiol. 2010;140:225–31. https://doi.org/10.1016/j.ijfoodmicro.2010.03.029.
Pimentel TC, Gomes de Oliveira LI, de Lourdes Chaves Macedo E, Costa GN, Dias DR, Schwan RF, et al. Understanding the potential of fruits, flowers, and ethnic beverages as valuable sources of techno-functional and probiotics strains: current scenario and main challenges. Trends in Food Science & Technology. 2021;114:25–59. https://doi.org/10.1016/j.tifs.2021.05.024. This review explored the added-value microorganisms isolated from unconventional sources and their techno-functional and probiotic properties. Some microorganisms with probiotic potential cited here are discussed in this article.
Menezes AGT, Ramos CL, Cenzi G, Melo DS, Dias DR, Schwan RF. Probiotic potential, antioxidant activity, and phytase production of indigenous yeasts isolated from indigenous fermented foods. Probiotics Antimicro Prot. 2020;12:280–8. https://doi.org/10.1007/s12602-019-9518-z.
FAO/WHO. Probiotics in food: Health and nutritional properties and guidelines for evaluation. Rome: FAO and WHO; 2006.
Padonou SW, Nielsen DS, Hounhouigan JD, Thorsen L, Nago MC, Jakobsen M. The microbiota of Lafun, an African traditional cassava food product. Int J Food Microbiol. 2009;133:22–30. https://doi.org/10.1016/j.ijfoodmicro.2009.04.019.
Shangpliang HNJ, Tamang JP. Metagenomics and metagenome-assembled genomes mining of health benefits in jalebi batter, a naturally fermented cereal-based food of India. Food Res Int. 2023;172:113130. https://doi.org/10.1016/j.foodres.2023.113130.
Jiménez E, Yépez A, Pérez-Cataluña A, Ramos Vásquez E, Zúñiga Dávila D, Vignolo G, et al. Exploring diversity and biotechnological potential of lactic acid bacteria from tocosh - traditional Peruvian fermented potatoes - by high throughput sequencing (HTS) and culturing. LWT. 2018;87:567–74. https://doi.org/10.1016/j.lwt.2017.09.033.
Fossi BT, Ndjouenkeu R. Probiotic potential of thermotolerant lactic acid bacteria isolated from “Gari “a cassava-based African fermented food. J App Biol Biotech. 2017;5:001–5. https://doi.org/10.7324/JABB.2017.50401.
Osorio-Cadavid E, Chaves-López C, Tofalo R, Pararella A, Suzzi G. Detection and identification of wild yeasts in Champús, a fermented Colombian maize beverage. Food Microbiol. 2008;25:771–7. https://doi.org/10.1016/j.fm.2008.04.014.
Wojtyniak K, Szajewska H. Systematic review: probiotics for functional constipation in children. Eur J Pediatr. 2017;176:1155–62. https://doi.org/10.1007/s00431-017-2972-2.
Ashaolu TJ, Reale A. A holistic review on Euro-Asian lactic acid bacteria fermented cereals and vegetables. Microorganisms. 2020;8:1176. https://doi.org/10.3390/microorganisms8081176.
do Santos CCA, de Almeida EG, de Melo GVP, Schwan RF. Microbiological and physicochemical characterisation of caxiri, an alcoholic beverage produced by the indigenous Juruna people of Brazil. Int J Food Microbiol. 2012;156:112–21. https://doi.org/10.1016/j.ijfoodmicro.2012.03.010.
Sharma S, Kanwar SS. Adherence potential of indigenous lactic acid bacterial isolates obtained from fermented foods of Western Himalayas to intestinal epithelial Caco-2 and HT-29 cell lines. J Food Sci Technol. 2017;54:3504–11. https://doi.org/10.1007/s13197-017-2807-1.
Flibert G, Abel T, Aly S. African cassava traditional fermented food: the microorganism’s contribution to their nutritional and safety values-a review. Int J Curr Microbiol App Sci. 2016;5:664–87. https://doi.org/10.20546/ijcmas.2016.510.074.
Vinderola CG, Reinheimer JA. Lactic acid starter and probiotic bacteria: a comparative “in vitro” study of probiotic characteristics and biological barrier resistance. Food Res Int. 2003;36:895–904. https://doi.org/10.1016/S0963-9969(03)00098-X.
Almeida EG, Rachid CCTC, Schwan RF. Microbial population present in fermented beverage ‘cauim’ produced by Brazilian Amerindians. Int J Food Microbiol. 2007;120:146–51. https://doi.org/10.1016/j.ijfoodmicro.2007.06.020.
Freire AL, Ramos CL, de Almeida EG, Duarte WF, Schwan RF. Study of the physicochemical parameters and spontaneous fermentation during the traditional production of yakupa, an indigenous beverage produced by Brazilian Amerindians. World J Microbiol Biotechnol. 2014;30:567–77. https://doi.org/10.1007/s11274-013-1476-0.
Lacerda DC, da Costa PCT, Pontes PB, dos Santos LAC, Neto JPRC, Luis CCS, et al. Potential role of Limosilactobacillus fermentum as a probiotic with anti-diabetic properties: a reviw. World J Diabetes. 2022;13:717–28. https://doi.org/10.4239/wjd.v13.i9.717.
Brito B de N do C, Chisté RC, Lopes AS, Glória MBA, Chagas Junior GCA, Pena R da S. Lactic acid bacteria and bioactive amines identified during Manipueira fermentation for Tucupi production. Microorganisms. 2022;10. https://doi.org/10.3390/microorganisms10050840.
Pérez-Cataluña A, Elizaquível P, Carrasco P, Espinosa J, Reyes D, Wacher C, et al. Diversity and dynamics of lactic acid bacteria in Atole agrio, a traditional maize-based fermented beverage from South-Eastern Mexico, analysed by high throughput sequencing and culturing. Antonie Van Leeuwenhoek. 2018;111:385–99. https://doi.org/10.1007/s10482-017-0960-1.
Guglielmotti DM, Marcó MB, Golowczyc M, Reinheimer JA, del Quiberoni AL. Probiotic potential of Lactobacillus delbrueckii strains and their phage resistant mutants. Int Dairy J. 2007;17:916–25. https://doi.org/10.1016/j.idairyj.2006.11.004.
Martins FS, Nardi RMD, Arantes RME, Rosa CA, Neves MJ, Nicoli JR. Screening of yeasts as probiotic based on capacities to colonize the gastrointestinal tract and to protect against enteropathogen challenge in mice. J Gen Appl Microbiol. 2005;51:83–92. https://doi.org/10.2323/jgam.51.83.
Freire AL, Zapata S, Mosquera J, Mejia ML, Trueba G. Bacteria associated with human saliva are major microbial components of Ecuadorian indigenous beers (chicha). PeerJ. 2016;2016. https://doi.org/10.7717/peerj.1962.
Ramos CL, de Sousa ESO, Ribeiro J, Almeida TMM, do Santos CCA, Abegg MA, et al. Microbiological and chemical characteristics of tarubá, an indigenous beverage produced from solid cassava fermentation. Food Microbiol. 2015;49:182–8. https://doi.org/10.1016/J.FM.2015.02.005.
Penido FCL, Piló FB, de Cicco Sandes SH, Nunes ÁC, Colen G, de Souza OE, et al. Selection of starter cultures for the production of sour cassava starch in a pilot-scale fermentation process. Braz J Microbiol. 2018;49:823–31. https://doi.org/10.1016/j.bjm.2018.02.001.
Jiang S, Cai L, Lv L, Li L. Pediococcus pentosaceus, a future additive or probiotic candidate. Microb Cell Fact. 2021;20:1–14. https://doi.org/10.1186/s12934-021-01537-y.
López-Sánchez R, Hernández-Oaxaca D, Escobar-Zepeda A, Cerrillo BR, López-Munguía A, Segovia L. Analysing the dynamics of the bacterial community in pozol, a Mexican fermented corn dough. Microbiology 2023;169. https://doi.org/10.1099/mic.0.001355.46.
Crispim SM, Nascimento AMA, Costa PS, Moreira JLS, Nunes AC, Nicoli JR, et al. Molecular identification of Lactobacillus spp. associated with puba, a Brazilian fermented cassava food. Braz J Microbiol. 2013;44:15–21. https://doi.org/10.1590/S1517-83822013005000007.
Lacerda ICA, Miranda RL, Borelli BM, Nunes ÁC, Nardi RMD, Lachance MA, et al. Lactic acid bacteria and yeasts associated with spontaneous fermentations during the production of sour cassava starch in Brazil. Int J Food Microbiol. 2005;105:213–9. https://doi.org/10.1016/j.ijfoodmicro.2005.04.010.
Kasuga G, Tanaka M, Harada Y, Nagashima H, Yamato T, Wakimoto A, et al. Homologous expression and characterization of Gassericin T and Gassericin S, a novel class IIb bacteriocin produced by Lactobacillus gasseri LA327. Am Soc Microbiol 2019;85. https://doi.org/10.1128/AEM.02815-18.
Liu M, Zhang X, Hao Y, Ding J, Shen J, Xue Z, et al. Protective effects of a novel probiotic strain, Lactococcus lactis ML2018, in colitis: in vivo and in vitro evidence. Food Funct. 2019;10:1132–45. https://doi.org/10.1039/C8FO02301H.
Miguel MGCP, Collela CF, de Almeida EG, Dias DR, Schwan RF. Physicochemical and microbiological description of Caxiri – a cassava and corn alcoholic beverage. Int J Food Sci Technol. 2015;50:2537–44. https://doi.org/10.1111/ijfs.12921.
Tamang JP, Kailasapathy K. Fermented foods and beverages of the world. Boca Raton: CRC Press; 2010. https://doi.org/10.1201/EBK1420094954.
Yang Z, Zhu X, Wen A, Qin L. Development of probiotics beverage using cereal enzymatic hydrolysate fermented with Limosilactobacillus reuteri. Food Sci Nutr. 2022;10:3143–53. https://doi.org/10.1002/fsn3.2913.
Obafemi YD, Oranusi SU, Ajanaku KO, Akinduti PA, Leech J, Cotter PD. African fermented foods: overview, emerging benefits, and novel approaches to microbiome profiling. Nature Parther J. 2022;6. https://doi.org/10.1038/s41538-022-00130-w. This review provides an overview of African fermented foods, their microbiota, and the health-promoting potential of these foods and microorganisms. Some products mentioned here are very well described in this article.
do Nascimento LCP, Lacerda DC, Ferreira DJS, de Souza EL, de Brito Alves JL. Limosilactobacillus fermentum, current evidence on the antioxidant properties and opportunities to be exploited as a probiotic microorganism. Probiotics Antimicrob Proteins. 2022;14:960–79. https://doi.org/10.1007/s12602-022-09943-3.
Franz CMAP, Huch M, Mathara JM, Abriouel H, Benomar N, Reid G, et al. African fermented foods and probiotics. Int J Food Microbiol. 2014;190:84–96. https://doi.org/10.1016/j.ijfoodmicro.2014.08.033.
Rebaza-Cardenas TD, Silva-Cajaleón K, Sabater C, Delgado S, Montes-Villanueva ND, Ruas-Madiedo P. “Masato de Yuca” and “Chicha de Siete Semillas” two traditional vegetable fermented beverages from peru as source for the isolation of potential probiotic bacteria. Probiotics Antimicrob Proteins. 2023;15:300–11. https://doi.org/10.1007/s12602-021-09836-x.
Chugh B, Kamal-Eldin A. Bioactive compounds produced by probiotics in food products. Curr Opin Food Sci. 2020;32:76–82. https://doi.org/10.1016/j.cofs.2020.02.003.
Sánchez MT, Ruiz MA, Morales ME. Probiotic microorganisms and health. Ars Pharmaceutica. 2015;56:45–59. https://doi.org/10.4321/S2340-98942015000100007.
Albuquerque TMR, Garcia EF, de Oliveira AA, Magnani M, Saarela M, de Souza EL. In vitro characterization of Lactobacillus strains isolated from fruit processing by-products as potential probiotics. Probiotics Antimicro Prot. 2018;10:704–16. https://doi.org/10.1007/s12602-017-9318-2.
Naidu AS, Bidlack WR, Clemens RA. Probiotic spectra of lactic acid bacteria (LAB). Crit Rev Food Sci Nutr. 1999;39:13–126. https://doi.org/10.1080/10408699991279187.
Lacerda ICA, Gomes FCO, Borelli BM, Faria CLL, Franco GR, Mourão MM, et al. Identification of the bacterial community responsible for traditional fermentation during sour cassava starch, cachaça and minas cheese production using cultureindependent 16s rrna gene sequence analysis. Braz J Microbiol. 2011;42:650–7. https://doi.org/10.1590/S1517-83822011000200029.
Motevaseli E, Shirzad M, Akrami SM, Mousavi A-S, Mirsalehian A, Modarressi MH. Normal and tumour cervical cells respond differently to vaginal lactobacilli, independent of pH and lactate. J Med Microbiol. 2013;62:1065–72. https://doi.org/10.1099/jmm.0.057521-0.
Mokoena MP, Mutanda T, Olaniran AO. Perspectives on the probiotic potential of lactic acid bacteria from African traditional fermented foods and beverages. Food Nutr Res. 2016;60:1–12. https://doi.org/10.3402/fnr.v60.29630.
Olasupo NA, Olukoya DK, Odunfa SA. Identification of Lactobacillus species associated with selected African fermented foods. Zeitschrift fur Naturforschung Section C - Journal of Biosciences. 1997;52:105–8. https://doi.org/10.1515/znc-1997-1-218.
Elizaquível P, Pérez-Cataluña A, Yépez A, Aristimuño C, Jiménez E, Cocconcelli PS, et al. Pyrosequencing vs. culture-dependent approaches to analyze lactic acid bacteria associated to chicha, a traditional maize-based fermented beverage from Northwestern Argentina. Int J Food Microbiol. 2015;198:9–18. https://doi.org/10.1016/j.ijfoodmicro.2014.12.027.
Väkeväinen K, Valderrama A, Espinosa J, Centurión D, Rizo J, Reyes-Duarte D, et al. Characterization of lactic acid bacteria recovered from atole agrio, a traditional Mexican fermented beverage. LWT. 2018;88:109–18. https://doi.org/10.1016/j.lwt.2017.10.004.
Azam M, Saeed M, Ahmad T, Yamin I, Khan WA, Iqbal MW, et al. Characterization of biopolymeric encapsulation system for improved survival of Lactobacillus brevis. Food Measure. 2022;16:2292–9. https://doi.org/10.1007/s11694-022-01334-0.
Teixeira P. LACTOBACILLUS | Lactobacillus brevis. Em: Batt CA, Tortorello ML, organizadores. Encyclopedia of Food Microbiology. 2° ed Oxford: Academic Press; 2014. pp. 418–24. https://doi.org/10.1016/B978-0-12-384730-0.00178-6.
Gobbetti M. Lactobacillus casei. Em: Minirvini F, organizador. Encyclopedia of Food Microbiology. Oxford: Elsevier; 2014. pp. 432–8. https://doi.org/10.1016/B978-0-12-384730-0.00180-4.
Omar NB, Ampe F, Raimbault M, Guyot JP, Tailliez P. Molecular diversity of lactic acid bacteria from cassava sour starch (Colombia). Syst Appl Microbiol. 2000;23:285–91. https://doi.org/10.1016/S0723-2020(00)80016-8.
Puerari C, Magalhães-Guedes KT, Schwan RF. Bacaba beverage produced by Umutina Brazilian Amerindians: microbiological and chemical characterization. Braz J Microbiol. 2015;46:1207–16. https://doi.org/10.1590/S1517-838246420140964.
Moiseenko KV, Begunova AV, Savinova OS, Glazunova OA, Rozhkova IV, Fedorova TV. Biochemical and genomic characterization of two new strains of Lacticaseibacillus paracasei isolated from the traditional corn-based beverage of South Africa, Mahewu, and their comparison with strains isolated from Kefir grains. Foods. 2023;12:1–19. https://doi.org/10.3390/foods12010223.
Mgbodile FC, Nwagu TNT. Probiotic therapy, African fermented foods and food-derived bioactive peptides in the management of SARS-CoV-2 cases and other viral infections. Biotechnol Rep. 2023;38:1–15. https://doi.org/10.1016/j.btre.2023.e00795.
Al-Emran HM, Moon JF, Miah ML, Meghla NS, Reuben RC, Uddin MJ, et al. Genomic analysis and in vivo efficacy of Pediococcus acidilactici as a potential probiotic to prevent hyperglycemia, hypercholesterolemia and gastrointestinal infections. Sci Rep. 2022;12:20429. https://doi.org/10.1038/s41598-022-24791-5.
Shao X, Fang K, Medina D, Wan J, Lee J-L, Hong SH. The probiotic, Leuconostoc mesenteroides, inhibits Listeria monocytogenes biofilm formation. J Food Saf. 2020;40. https://doi.org/10.1111/jfs.12750.
Ampe F, Sirvent A, Zakhia N. Dynamics of the microbial community responsible for traditional sour cassava starch fermentation studied by denaturing gradient gel electrophoresis and quantitative rRNA hybridization. Int J Food Microbiol. 2001;65:45–54. https://doi.org/10.1016/S0168-1605(00)00502-X.
Song AA-L, In LLA, Lim SHE, Rahim RA. A review on Lactococcus lactis: from food to factory. Microb Cell Fact. 2017;16:55. https://doi.org/10.1186/s12934-017-0669-x.
Sałański P, Kowalczyk M, Bardowski JK, Szczepankowska AK. Health-promoting nature of Lactococcus lactis IBB109 and Lactococcus lactis IBB417 strains exhibiting proliferation inhibition and stimulation of interleukin-18 expression in colorectal cancer cells. Front Microbiol. 2022;13:822912. https://doi.org/10.3389/fmicb.2022.822912.
Shruthi B, Deepa N, Somashekaraiah R, Adithi G, Divyashree S, Sreenivasa MY. Exploring biotechnological and functional characteristics of probiotic yeasts: a review. Biotechnol Rep. 2022;28. https://doi.org/10.1016/j.btre.2022.e00716.
Sen S, Mansell TJ. Yeasts as probiotics: mechanisms, outcomes, and future potential. Fungal Genet Biol. 2020;137:103333. https://doi.org/10.1016/j.fgb.2020.103333.
da Cruz Pedrozo Miguel MG, Santos MRRM, Duarte WF, de Almeida EG, Schwan RF. Physico-chemical and microbiological characterization of corn and rice “calugi” produced by Brazilian Amerindian people. Food Res Int. 2012;49:524–32. https://doi.org/10.1016/j.foodres.2012.08.012.
Grijalva-Vallejos N, Aranda A, Matallana E. Evaluation of yeasts from Ecuadorian chicha by their performance as starters for alcoholic fermentations in the food industry. Int J Food Microbiol. 2020;317. https://doi.org/10.1016/j.ijfoodmicro.2019.108462.
Menezes AGT, Melo D de S, Ramos CL, Moreira SI, Alves E, Schwan RF. Yeasts isolated from Brazilian fermented foods in the protection against infection by pathogenic food bacteria. Microbial Pathogenesis. 2020;140. https://doi.org/10.1016/j.micpath.2020.103969.
Angulo M, Reyes-Becerril M, Medina-Córdova N, Tovar-Ramírez D, Angulo C. Probiotic and nutritional effects of Debaryomyces hansenii on animals. Appl Microbiol Biotechnol. 2020;104:7689–99. https://doi.org/10.1007/s00253-020-10780-z.
Ogunremi OR, Sanni AI, Agrawal R. Probiotic potentials of yeasts isolated from some cereal-based Nigerian traditional fermented food products. J Appl Microbiol. 2015;119:797–808. https://doi.org/10.1111/jam.12875.
Youn H-Y, Kim D-H, Kim H-J, Bae D, Song K-Y, Kim H, et al. Survivability of Kluyveromyces marxianus isolated from Korean Kefir in a simulated gastrointestinal environment. Front Microbiol. 2022;13. https://doi.org/10.3389/fmicb.2022.842097.
Funding
This study received financial support from Fundação do Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG, process number APQ-02451–21).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethical Approval
This article does not contain any studies with human participants or animals carried out by any authors.
Competing Interests
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Caetano, R.G., Xavier, I.B., Feldmann, V. et al. Traditional Fermented Products: Potential Origin for Probiotic Strains. Curr Food Sci Tech Rep 2, 201–211 (2024). https://doi.org/10.1007/s43555-024-00030-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43555-024-00030-y