Abstract
Purpose
To establish whether common degenerative lumbar spine conditions have a predictable sagittal profile and associated range of lordosis.
Summary of background data
The spinopelvic balance of a normal population and normal ranges are well described in the literature. There is also evidence that certain degenerative conditions can lead to a preponderance of loss of lordosis at specific spinal levels. There is limited literature on the range and magnitude of loss of lordosis for known degenerative lumbar spine pathologies.
Methods
A retrospective analysis of prospectively obtained radiographs from a dual surgeon database was performed and imaging analysed for spinopelvic parameters. Degenerative conditions studied were; Lumbar degenerative spondylolisthesis (L3/4 and L4/5 analysed separately), L5/S1 degenerative disc disease, L5/S1 isthmic spondylolisthesis. Pelvic incidence, sacral slope, pelvic tilt, segmental and global lumbar lordosis, vertebral lordosis and lumbar vertical axis were measured.
Results
The range of change in segmental lordosis was normally distributed for all studied degenerative spinal conditions except L5/S1 isthmic spondylolisthesis. L5/S1 degenerative disc disease affected younger adults (mean age 37), whilst degenerative spondylolisthesis at L3/4 and L4/5 affected older adults (mean ages 69.5 and 68.9 respectively). Removing an outlying high-grade L5/S1 isthmic spondylolisthesis made the data distribution approach a normal distribution.
Conclusion
Most degenerative spinal pathologies cause a normally distributed spectrum of deformity which should be addressed and corrected with a tailored, individualised surgical plan for each patient. Universal treatment recommendations should be interpreted with caution.
Level of evidence
4.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With increased recognition that variability in pelvic shape as measured by pelvic incidence (PI) is associated with variation in sacral slope and the consequent lumbar lordosis (LL) required to maintain sagittal alignment [1, 2], there is increased recognition that spinal reconstruction with fusion needs to optimise sagittal realignment so as to optimise clinical outcomes and reduce adjacent segment degeneration [3], with its potential to cause late failure and the need for revision surgery.
Whilst LL is an acceptable global measure of lumbar sagittal alignment, it consists of five mobile articulations each of which contributes to the overall alignment. The individual contributions of each lumbar segment have been characterised [4,5,6,7,8]. Failure to recreate normal segmental alignment within a fusion requires compensation at adjacent segments with altered biomechanics that may lead to accelerated degeneration, but which may not be detected with an overall measure of multi-segmental lordosis—the LL [5].
Common pathologies that lead to low lumbar fusion procedures are associated with variable disc degeneration. This degeneration is associated with disc desiccation, loss of disc height, facet joint degeneration, annular bulging and loss of lordosis particularly in the lower lumbar spine where the motion segments are the major contributors to lordosis [9].
There is increasing evidence that different reconstruction options for lumbar fusion using a variety of interbody techniques result in variable lordosis gain [10,11,12,13,14,15]. It is becoming increasingly evident that if pathology occurring in the low lumbar spine requires treatment with fusion, treatment should attempt to optimise lordosis recreation to match the normal lordotic contribution of the treated segment within the overall lumbar lordosis [6].
If there is a goal to reconstruct the optimum lordosis at a pathological segment, there needs to be an understanding of what the normal lordosis at the operated segment should be, and the deformity created by the pathological process for which the surgery is indicated. The difference between the two will guide the magnitude of lordosis gain necessary in the reconstruction. To the best of our knowledge there is no definition of the deformity, the loss of normal sagittal alignment, associated with common pathologies of the low lumbar spine that may require intervention.
The purpose of this study is to document the sagittal deformity (loss of lordosis) associated with common lower lumbar disorders for which fusion surgery is indicated. We wished to specifically examine degenerative spondylolisthesis (DS) at both L3/4 and L4/5, degenerative disc disease (DDD) at L5/S1 (disc degeneration and loss of height without listhesis or due to degenerative anatomical change to the posterior elements), and isthmic spondylolisthesis (IS) at L5/S1, looking at the sagittal alignment of the relative motion segment in a population of patients coming forward for reconstructive surgery.
Methods
Patient cohort
A surgical database was interrogated and data of patients who had undergone single-level fusion for L3/4 DS, L4/5 DS, L5/S1 DDD and L5/S1 IS were retrieved. Data retrieved included: patient demographics, surgical procedure and age.
Radiology
Erect lateral radiograph of the lumbar spine including the hip joints had been performed on all patients prospectively prior to surgery for the purposes of surgical planning.
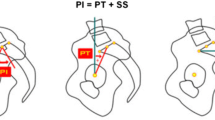
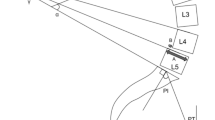
The radiological measurements were; pelvic incidence (PI), sacral slope (SS), pelvic tilt (PT), lumbar lordosis (LL), segmental lordosis at each lumbar intervertebral disc (SL) (measured from caudal endplate of superior vertebra to cranial endplate of inferior vertebra, i.e. bottom of L4 to top of L5 for the L4/5 disc), vertebral lordosis at each vertebra (VL), and lumbar vertical axis (LVA—the distance from the posterior edge of the S1 superior endplate to the plumb line from the centre of the L1 vertebral body). Kyphosis was recorded as a negative value.
Exclusion criteria included; inadequate radiographs, abnormal vertebral differentiation, e.g. sacralisation of lumbar vertebra or lumbarisation of sacral vertebra and previous fusion or dual pathologies (e.g. L5/S1 DDD and L4/5 DS).
Measurements were performed on Inteleviewer Picture Archiving and Communication System (PACS) (Intelerad, 800 Boulevard De Maisonneuve East, Montreal, Canada). Measurement accuracy was ensured by a single observer using the software on the same PACS display system. Blinded repeat measurements were made for ten random cases and the accuracy was assessed. The sum of the vertebral lordosis and segmental lordoses was equivalent to the lumbar lordosis, and the difference between these components and the total was also assessed for accuracy.
Statistical analysis was performed with t test using GraphPad (GraphPad Software, 2365 Northside Dr. Suite 560, San Diego, CA 92108). A p value of < 0.05 was taken to be statistically significant.
Results
165 surgical cases were identified, and after exclusion of cases without adequate radiographs 89 patients remained: L3/4 DS—9; L4/5 DS—49; L5/S1 DDD—19; and L5/S1 IS—12 (see Fig. 1).
The mean and range of ages of the groups were L3/4DS—69.555.0–78.6); L4/5DS—68.9 (56.3–86.9); L5/S1DDD—37 (25.5–45.8); and L5/S1IS—55 (26.3–96.5). There was a statistically significant difference between IS and DDD p < 0.05, IS and 3/4DS p < 0.05, IS and 4/5DS p < 0.05, L5/S1DDD and 3/4DS p < 0.05, 5/1DDD and 4/5DS p < 0.05, but not between 3/4DS and 4/5DS p = 0.43.
The mean PIs of the groups were L3/4DS—59.7 deg; L4/5DS—62.1 deg; L5/S1DDD—49 deg; and L5/S1IS—66.2 deg. The PI was significantly lower for L5/S1DDD compared with the other three diagnoses (vs L3/4DS p < 0.05, vs L4/5DS p < 0.05, vs L5/S1 IS p < 0.05), but there was no difference of the PI between L3/4DS, L4/5DS and L5/S1IS (L3/4DS vs L4/5 DS p = 0.27, L3/4DS vs L5/S1IS p = 0.15, L4/5DS vs L5/S1IS p = 0.14).
The mean and range of segmental lordosis at each operated level prior to surgery were L3/4DS 5.2 deg (0–11); L4/5DS 4.1 deg (− 12 to 16); L5/S1DDD 7.9 deg (0–17); and L5/S1IS − 1.8 deg (− 40 to 14).
The mean and range of LL-PI for each condition was L3/4DS—7.9 deg (− 34 to − 1); L4/5DS—11.1 deg (− 38 to 8); L5/S1DDD— − 4.8 deg (− 4 to 22); and L5/S1IS—6.8 deg (− 25 to 12).
Mean PT was > 20 deg in L3/4DS, L4/5 DS, and L5/S1IS, but not in L5/S1DDD. This information is demonstrated in Tables 1, 2, 3, and 4.
The range of SL for the operated level in each condition is shown in histogram form in Figs. 2, 3, 4, and 5. Included is a reference line for the normal segmental lordosis for that anatomical level as derived from literature norms.
L4/5 degenerative spondylolisthesis
The Shapiro–Wilk test for normality of L4/5 DS was p = 0.12, revealing the data to be normally distributed.
W = 0.96 (0.95–1.00).
L3/4 degenerative spondylolisthesis
The Shapiro–Wilk test for normality of L3/4 DS was p = 0.27, revealing the data to be normally distributed.
W = 0.90 (0.83–1.00).
L5/S1 degenerative disc disease
The Shapiro–Wilk test for normality of L5/S1 DDD was p = 0.13, revealing the data to be normally distributed.
W = 0.92 (0.90–1.00).
L5/S1 isthmic spondylolisthesis
The Shapiro–Wilk test for normality of L5/S1 IS was p = 0.005, revealing the data to NOT be normally distributed.
W = 0.78 (0.86–1.00).
Removing the single outlier for significant kyphosis (Grade III isthmic spondylolisthesis with a segmental lordosis at L5/S1 of − 40°) does not alter this outcome although the W value does approach a normal value. p = 0.03, W = 0.84 (0.85–1.00).
There was no statistically significant difference between the SL at L4/5 between the L4/5 DS and L3/4 DS patients p = 0.0546.
The difference in SL at L5/S1 was significant between DDD and L5/S1 IS, p = 0.0084.
Measurement accuracy was assessed. A single observer performed the measurements on a single PACS compliant Computer Display. Measurements were repeated for ten cases at a separate sitting to confirm reliability. All repeat measurements varied by less than 1%. Summation of all of the VL and SLs provided a combined total equal to LL. This allowed accuracy assessment within each case as LL was also measured separately. All measurements of sum VL and SL were within 1 degree of the LL for each case.
Discussion
This study has demonstrated that loss of normal sagittal alignment at the abnormal level can be very significant, but is highly variable in patients coming forward for single-level fusion surgery. The normative values for the lumbar disc contribution to lordosis are well established [4]. The maximum contribution to lordosis from the discs comes from L5/S1 and L4/5 levels where the mean lordotic angle of the disc is 15° and 12°, respectively [4]. Commonly treated pathologies at these levels, where disc height has been dramatically reduced as is often seen, will require operative procedures capable of dramatically improving lordosis of the segment if alignment is to be optimised and biomechanical forces at adjacent segments minimised.
The clinical outcomes of patients treated are not included within this paper as this was not the purpose of the paper. It is accepted that restoration of the spinopelvic balance decreases the risks of adjacent segment disease and clinical outcomes as demonstrated by Radovanovic amongst others [45].
From the study population three relatively common pathologies for which fusion is a common component of surgical treatment were selected, with degenerative spondylolisthesis being subdivided into cases at L3/4 and L4/5. This separation of the DS group was performed because of the recognised difference in lordosis in disc at the differing levels in the normal lumbar spine. The differences in the ages at the time of presentation for surgery represented the well-recognised differences in the pathologies. DS is symptomatic later in life, whereas IS is symptomatic across adult life [16, 17]. DDD included painful motion segments, and back dominant pain after disc prolapse treated with or without surgery, and this occurs in a younger demographic [18]. This undoubtedly influences the ability of the other motion segments of the spine to ‘compensate’ for deformity as demonstrated by the reduction in lordosis across uninstrumented motion segments [43] whilst the more aged spine has less flexibility and therefore less ability to compensate for the acquired deformity for example in DS patients.
The difference in the mean PI values is also consistent with the hypothesised influence of the PI and SS on lumbar spine disorders: IS and DS result in translation of the cephalad vertebra due to increased shear forces upon the posterior elements related to increased vertebral slope as seen in the lower lumbar spine where there is greater SS [19], whilst DDD is associated with more horizontal disc alignment due to a lower PI and SS, and this may result in greater loads through the anterior column of the spine, where the disc may be more susceptible to failure under axial loading [20].
The published literature suggests a wide range of lordotic gain associated with various surgical techniques used to perform fusion [12, 21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36]. Fusion in situ, with or without rigid instrumentation, is unlikely to significantly increase lordosis. At the other extreme experience suggests that the maximum gain of lordosis occurs where operative intervention involves wide posterior column osteotomy and facetectomy, and this can be combined with excision of the anterior longitudinal ligament with ALIF to maximise lordosis gain [37]. In general, minimal bone resection as seen in minimally invasive procedures reduces lordosis gain [38,39,40] with conflicting evidence as to which technique produces the most lordosis, including minimally invasive surgery and expandable cages [38, 41, 42]. It is clear that the wide range of operative options resulting in variable increases in SL [12, 21] could ideally be matched with the wide range of reductions in SL demonstrated in the study across each of the pathologies. It is highly likely that a single technique will not be suitable for the range of SL seen across any single pathology coming to fusion surgery. Schwab’s classification of osteotomies [37] provides a useful escalation of posterior-based osteotomies, whilst transforaminal, anterior only, posterior only or combined anterior–posterior techniques provide increasing lordotic gains whilst respecting the surgeon’s preferred, individual technique.
This study again questions the relevance of using the global measure of lordosis (LL) when looking at pathologies with potential for focused alignment abnormalities within the multi-articulated lumbar spine. The measure of mismatch between PI and LL, PI–LL < 10°, was not exceeded in L3/4DS, L5/S1IS or L5/S1DDD, and only marginally exceeded for L4/5 DS. This is not surprising. In younger spines, compensation occurs at levels adjacent to the pathology with the frequent appearance of compensatory hyperlordosis above a hypolordotic segment [43] and the global LL remains normal. The modest mismatch between PI and LL seen in the L4/5 DS group likely represents increasing stiffness with increasing spondylosis and lesser ability to compensate for regional abnormalities in this older population [44]. What is also evident from the data is the wide spectrum of all measurements of alignment, including PT and sagittal balance (as measured by LVA), which must be considered on an individual basis when addressing these segmental deformity corrections and the array of surgical treatment options and their respective lordosis gains should be considered when assessing individual patients and their segmental deformities.
Whilst it is tempting to recommend an optimum case by case increase in SL based on the SL associated with the pathology before surgery, it must be recognised that the normative anatomical data is based on population means. Given that the ‘shearing’ pathologies of IS and DS are associated with a higher PI and SS, it may well be that the disc contribution to the greater LL needed to match the PI in these disorders is greater than the published means. Whilst there is some evidence that more lordotic lumbar spines are associated with greater lordotic contributions from the upper lumbar spine [6], it seems likely that such spines also have a greater contribution to the LL by the SL in the lower discs at L4/5 and L5/S1. To the best of our knowledge there is no normative data for the disc contribution to the LL for the extremes of PI, yet it is the extremes of PI (low for DDD and high for DS and IS) which are commonly associated with these common pathologies requiring intervention.
This paper is the first that attempts to characterise the focal sagittal deformities associated with common lumbar pathologies requiring fusion as part of the treatment. It clearly demonstrates that the wide range of deformities means that a ‘typical’ loss of SL cannot be described for a specific pathology, and therefore attempts to both characterise the deformity and prescribe universal treatment recommendations are flawed. Individualised planning is required dependent upon the individual SL if reconstruction aims to optimise lordosis at the fused segment.
References
Vaz G, Roussouly P, Berthonnaud E et al (2002) Sagittal morphology and equilibrium of pelvis and spine. Eur Spine J 11(1):80–87. https://doi.org/10.1007/s005860000224
Labelle H, Roussouly P, Berthonnaud E et al (2005) The importance of spino-pelvic balance in L5–s1 developmental spondylolisthesis: a review of pertinent radiologic measurements. Spine (Phila Pa 1976) 30(6 Suppl):S27-34. https://doi.org/10.1097/01.brs.0000155560.92580.90
Rothenfluh DA, Mueller DA, Rothenfluh E et al (2015) Pelvic incidence-lumbar lordosis mismatch predisposes to adjacent segment disease after lumbar spinal fusion. Eur Spine J 24(6):1251–1258. https://doi.org/10.1007/s00586-014-3454-0(Epub 2014 Jul 14)
Damasceno LHF, Catarin SRG, Campos AD et al (2006) Lumbar lordosis: a study of angle values and of vertebral bodies and intervertebral discs role. Acta Ortopéd Brasileira 14(4):193–198
Pesenti S, Lafage R, Stein D et al (2018) The amount of proximal lumbar lordosis is related to pelvic incidence. Clin Orthop Relat Res 476(8):1603–1611. https://doi.org/10.1097/CORR.0000000000000380
Anwar HA, Butler JS, Yarashi T et al (2015) Segmental Pelvic Correlation (SPeC): a novel approach to understanding sagittal plane spinal alignment. Spine J 15(12):2518–2523
Roussouly P, Gollogly S, Berthonnaud E et al (2005) Classification of the normal variation in the sagittal alignment of the human lumbar spine and pelvis in the standing position. Spine (Phila Pa 1976) 30(3):346–353
Barrey C, Jund J, Noseda O et al (2007) Sagittal balance of the pelvis-spine complex and lumbar degenerative diseases. A comparative study about 85 cases. Eur Spine J 16:1459–1467
Murray KJ, Le Grande MR, de Mues AO et al (2017) Characterisation of the correlation between standing lordosis and degenerative joint disease in the lower lumbar spine in women and men: a radiographic study. BMC Musculoskelet Disord 18(1):330. https://doi.org/10.1186/s12891-017-1696-9
Moe JH, Denis F (1977) The iatrogenic loss of lumbar lordosis. Orthop Trans 1:131
Barrey C, Darnis A (2015) Current strategies for the restoration of adequate lordosis during lumbar fusion. World J Orthop 6(1):117–126. https://doi.org/10.5312/wjo.v6.i1.117
Robertson PA, Armstrong WA, Woods DL et al (2018) Lordosis re-creation in TLIF and PLIF: a cadaveric study of the influence of surgical bone resection and cage angle [published online ahead of print, 2018 Apr 24]. Spine. https://doi.org/10.1097/BRS.0000000000002705.
Landham PR, Don AS, Robertson PA (2017) Do position and size matter? An analysis of cage and placement variables for optimum lordosis in PLIF reconstruction. Eur Spine J26:2843–2850. https://doi.org/10.1007/s00586-017-5170-z
Saadeh YS, Joseph JR, Smith BW et al (2019) Comparison of segmental lordosis and global spinopelvic alignment after single-level lateral lumbar interbody fusion or transforaminal lumbar interbody fusion. World Neurosurg 126:e1374–e1378. https://doi.org/10.1016/j.wneu.2019.03.106
Elmekaty M, Kotani Y, Mehy EE et al (2018) Clinical and radiological comparison between three different minimally invasive surgical fusion techniques for single-level lumbar isthmic and degenerative spondylolisthesis: minimally invasive surgical posterolateral fusion versus minimally invasive surgical transforaminal lumbar interbody fusion versus midline lumbar fusion. Asian Spine J 12(5):870–879. https://doi.org/10.31616/asj.2018.12.5.870
Rosenberg NJ (1975) Degenerative spondylolisthesis. Predisposing factors. J Bone Joint Surg Am 57(4):467–474
Ganju A (2002) Isthmic spondylolisthesis. Neurosurg Focus 13(1):E1. https://doi.org/10.3171/foc.2002.13.1.2
Brinjikji W, Diehn FE, Jarvik JG et al (2015) MRI findings of disc degeneration are more prevalent in adults with low back pain than in asymptomatic controls: a systematic review and meta-analysis. AJNR Am J Neuroradiol 36(12):2394–2399. https://doi.org/10.3174/ajnr.A4498
Roussouly P, Pinheiro-Franco JL (2011) Biomechanical analysis of the spino-pelvic organization and adaptation in pathology. Eur Spine J 20(Suppl 5):609–618. https://doi.org/10.1007/s00586-011-1928-x
Strube P, Pumberger M, Sonnow L et al (2018) Association between lumbar spinal degeneration and anatomic pelvic parameters. Clin Spine Surg 31(6):263–267. https://doi.org/10.1097/BSD.0000000000000660
Phan K, Thayaparan GK, Mobbs RJ (2015) Anterior lumbar interbody fusion versus transforaminal lumbar interbody fusion–systematic review and meta-analysis. Br J Neurosurg 29(5):705–711. https://doi.org/10.3109/02688697.2015.1036838
Watkins RGT, Hanna R, Chang D et al (2014) Sagittal alignment after lumbar interbody fusion: comparing anterior, lateral, and transforaminal approaches. J Spinal Disord Tech 27(5):253–256
Rice JW, Sedney CL, Daffner SD et al (2016) Improvement of segmental lordosis in transforaminal lumbar interbody fusion: a comparison of two techniques. Glob Spine J 6(3):229–233
Sembrano JN, Yson SC, Horazdovsky RD et al (2015) Radiographic comparison of lateral lumbar interbody fusion versus traditional fusion approaches: analysis of sagittal contour change. Int J Spine Surg 9:16
Pawar AY, Hughes AP, Sama AA et al (2015) A Comparative study of lateral lumbar interbody fusion and posterior lumbar interbody fusion in degenerative lumbar spondylolisthesis. Asian Spine J 9(5):668–674
Sembrano JN, Horazdovsky RD, Sharma AK et al (2017) Do lordotic cages provide better segmental lordosis versus non-lordotic cages in lateral lumbar interbody fusion (LLIF)? Clin Spine Surg 30(4):E338–E343
Saville PA, Kadam AB, Smith HE et al (2016) Anterior hyperlordotic cages: early experience and radiographic results. J Neurosurg Spine:1–7
Baghdadi YM, Larson AN, Dekutoski MB et al (2014) Sagittal balance and spinopelvic parameters after lateral lumbar interbody fusion for degenerative scoliosis: a case-control study. Spine (Phila Pa 1976) 39(3):E166–E173
Kim CH, Chung CK, Park SB et al (2017) A change in lumbar sagittal alignment after single-level anterior lumbar interbody fusion for lumbar degenerative spondylolisthesis with normal sagittal balance. Clin Spine Surg 30(7):291–296
Fujimori T, Le H, Schairer WW, Berven SH et al (2015) Does transforaminal lumbar interbody fusion have advantages over posterolateral lumbar fusion for degenerative spondylolisthesis? Glob Spine J 5(2):102–109
Yson SC, Santos ER, Sembrano JN et al (2012) Segmental lumbar sagittal correction after bilateral transforaminal lumbar interbody fusion. J Neurosurg Spine 17(1):37–42
Dorward IG, Lenke LG, Bridwell KH et al (2013) Transforaminal versus anterior lumbar interbody fusion in long deformity constructs: a matched cohort analysis. Spine 38(12):E755–E762
Hsieh PC, Koski TR, O’shaughnessy BA et al (2007) Anterior lumbar interbody fusion in comparison with transforaminal lumbar interbody fusion: implications for the restoration of foraminal height, local disc angle, lumbar lordosis, and sagittal balance. J Neurosurg Spine 7:379–386
Jagannathan J, Sansur CA, Oskouian RJ et al (2009) Radiographic restoration of lumbar alignment after transforaminal lumbar interbody fusion. Neurosurgery 64(5):955–963
Tohmeh AG, Khorsand D, Watson B et al (2014) Radiographical and clinical evaluation of extreme lateral interbody fusion: effects of cage size and instrumentation type with a minimum of 1-year followup. Spine 39(26):E1582-1591
Acosta FL, Liu J, Slimack N, Moller D et al (2011) Changes in coronal and sagittal plane alignment following minimally invasive direct lateral interbody fusion for the treatment of degenerative lumbar disease in adults: a radiographic study. J Neurosurg Spine 15(1):92–96
Schwab F, Blondel B, Chay E et al (2014) The comprehensive anatomical spinal osteotomy classification. Neurosurgery 74(1):112–120. https://doi.org/10.1227/NEU.0000000000000182o
Carlson BB, Saville P, Dowdell J et al (2019) Restoration of lumbar lordosis after minimally invasive transforaminal lumbar interbody fusion: a systematic review. Spine J 19(5):951–958. https://doi.org/10.1016/j.spinee.2018.10.017
Sharma AK, Kepler CK, Girardi FP et al (2011) Lateral lumbar interbody fusion: clinical and radiographic outcomes at 1 year: a preliminary report. J Spinal Disord Tech 24(4):242–250
Anand N, Hamilton JF, Perri B et al (2016) Cantilever TLIF With structural allograft and RhBMP2 for correction and maintenance of segmental sagittal lordosis long-term clinical, radiographic, and functional outcome. Spine 31(20):E748–E753
Kim CW, Doerr TM, Luna IY et al (2016) Minially invasive transforaminal lumbar interbody fusion using expandable technology: a clinical and radiographic analysis of 50 patients. World Neurosurg 90(C):228–235
Alimi M, Shin B, Macielak M et al (2015) Expandable polyaryl-ether-ether-ketone spacers for interbody distraction in the lumbar spine. Glob Spine J 5:169–178
Kamlanathan V, Kieffer WKM, Zhu M et al (2020) Lordosis recreation with PLIF surgery - what is the influence on adjacent segment lordosis?—an observational study of radiological alignment change [published online ahead of print, 2020 Mar 19]. Spine (Phila Pa 1976). https://doi.org/10.1097/BRS.0000000000003492
Lee ES, Ko CW, Suh SW et al (2014) The effect of age on sagittal plane profile of the lumbar spine according to standing, supine, and various sitting positions. J Orthop Surg Res 9(1):11
Radovanovic I, Urquhart JC, Ganapathy V et al (2017) Influence of postoperative sagittal balance and spinopelvic parameters on the outcome of patients surgically treated for degenerative lumbar spondylolisthesis. J Neurosurg Spine 26(4):448–453
Funding
No funding was received for this work.
Author information
Authors and Affiliations
Contributions
WKMK, AD, AF, PAR: made substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data; or the creation of new software used in the work; drafted the work or revised it critically for important intellectual content; approved the version to be published; and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflict of interest disclosures relevant to this work.
Ethics committee
The work was reviewed by the Health and Disabilities Ethics Committees (HDEC) and granted exemption.
Copyright
There is no copyrighted material or patient identifiable data/consent forms within this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kieffer, W.K.M., Don, A., Field, A. et al. Lordosis loss in degenerative spinal conditions. Spine Deform 10, 1407–1414 (2022). https://doi.org/10.1007/s43390-022-00533-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43390-022-00533-5