Abstract
Objectives
To study factors causing postoperative change of PI after surgical correction of ASD and to assess the effect of this variability on postoperative PI-LL mismatch.
Background
PI is used as an individual constant to define lumbar lordosis (LL) correction goal (PI-LL < 10). Postoperative changes of PI were shown but with opposite vectors. The impact of the PI variability on the postoperative PI–LL has not been studied.
Methods
The medical and radiographic data analyzed for patients who underwent long posterior instrumented spinal fusion. Inclusion criteria are age, ≥ 20 years old; ASD due to degenerative disk disease (DDD) or scoliosis (DS); ≥ 3 levels fused; and 2-year follow-up or revision. Studied parameters are LL (L1–S1), PI, sacral slope (SS), pelvic tilt (PT), and PI-LL. Measurement error and postoperative changes were defined. Statistical analysis includes ANOVA, correlation, regression, and risk assessment by odds ratio; P ≤ 0.05 considered statistically significant.
Results
Eighty patients were included: mean age, 62.4 years-old (SD, 11.1); female, 63.7%; mean body mass index (BMI), 27.1 (SD, 5.6). Distribution of patients by follow-ups includes preoperative 100%; postoperative (1–3 weeks), 100%; 11–13 months. 90%; 22–26 months, 58%; and revision: 24%.
Pre- versus postoperative PI (∆PI) changed both positively and negatively and the absolute value of change|∆PI| exceeded measurement error (P ≤ 0.05) reaching as high as 31°, and progressed with time; R2 dropped from 0.73 to 0.45 (P < 0.001); ∆PI depended on disproportional changes of SS and PT, preoperative PI, and change of LL. Obesity, DS, and absence of sacroiliac fixation increased |∆PI|. The risk of LL insufficient correction (PI–LL > 10°) associated with a |∆PI|> 6°, P = 0.05. Sacroiliac fixation diminished PI variability only during the first postoperative year.
Conclusion
Preoperative variability and postoperative instability of PI diminish the applicability of the PI–LL < 10° goal to plan correction of LL. An alternative method is offered.
Level of evidence
IV.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Initially, pelvic incidence (PI) was considered an individual constant characteristic determined by sacral slope (SS) and pelvic tilt (PT): PI = SS + PT [1]. According to this concept, PI is currently regarded as an invariable input parameter as a criterion for spinal correction: PI–LL < 10°, where lumbar lordosis (LL) is a variable index [2, 3]. This is used for spinal correction planning. However, application of this criterion in surgery yielded the recommended range in only ≈ 50%, while deterioration was observed in ≈ 14% at 1-year follow-up after instrumented correction of adult spine deformity (ASD) [4]. This revealed that PI might change after long instrumented spinal correction [5,6,7]. One study showed an increase on average, 11.4° without sacroiliac fixation, and 5.9° with sacroiliac fixation at more than 1-year follow-ups [6]. Other studies revealed a decrease of PI immediately after correction with sacroiliac fixation [5, 7] and then increase at 1-year follow-up that was more prominent if the iliac screws loosened [7]. The immediate postoperative increase of PI ≥ 10° amplified the risk of sagittal imbalance at 1-year postoperative and risk of iliac screw loosening [7]. It was also shown that the magnitude of PI change increases with increasing age; is proportional to the body mass index (BMI) [8]; and changes from extension to flexion with differences reaching 12° [9]. Changes of PI after surgical correction of severe ASD had association with preoperative sagittal imbalance and level of it correction, preoperative PI, number of spinal levels fused, and caudal level of fusion [10]. This effect can be explained by the physiological and/or pathological spatial displacing of the pelvic and mobility of the sacrum relative to the pelvis through the sacroiliac joints. Radiographically, this mobility may be defined as disproportional changes of SS(∆SS) versus PT(∆PT), the algebraic sum of which equal the PI change: ∆PI = ∆SS + ∆PT [9]. Overall, previous studies showed that PI is not constant, and its postoperative changes may have clinical consequences. However, some clinically important details require elucidation: (a) factors influencing the PI instability at short- and long-term follow-ups besides sacroiliac fixation and (b) impact of the PI instability on postoperative PI–LL. Thus, the purpose of this study is to reveal factors causing postoperative variability of PI after surgical correction of ASD at short- and long-term follow-ups and to assess the influence of this variability on postoperative PI–LL mismatch.
Materials and methods
A retrospective, case series study performed after the Institutional Review Board approval (#14-1258). The medical records and radiographic images collected and analyzed for a consecutive group of patients who underwent long posterior instrumented spinal fusion (PIF) for correction of ASD between 2006 and 2016. Inclusion criteria are age, ≥ 20 years old; primary or revision surgery for ASD secondary to degenerative disk disease (DDD) or idiopathic scoliosis (DS); use or not use of vertebral interbody fusion (VIF) and/or osteotomy; ≥ 3 spinal levels PIF construct; postoperative observation during 2 years or revision/reoperation; and available adequate quality radiographic images. The exclusion criteria: < 20 years old; < 3 levels PIF; malignancy; infection; osteomyelitis; inherited and/or severe metabolic diseases; and trauma of spine, pelvis, hips or extremities, and absence of satisfactory radiographic images. The X-rays included the standard standing sagittal imaging of the full spine and pelvis with hip joints performed preoperatively and at postoperative follow-ups: immediate (1–3 weeks), 6 (5–7) months, 12 (11–13) months, and 24 (22–26) months.
Demographic and clinical data were extracted from the medical records by an experienced researcher under supervision of the principal investigator (PI). The radiographic images were obtained from the clinical electronic database. The studied parameters included LL (L1–S1), PI, SS, PT, and PI–LL. All measurements were taken in accordance with previously described and accepted methods using Surgimap (New York, NY, USA) software [1, 2]. An experienced researcher completed all the measurements (first independent evaluation); after that, another experienced researcher blind to the results of the first evaluation remeasured all the studied variables (second independent evaluation); then, 20% of images from the image pool were randomly selected and a third independent experienced evaluator blind to the results of these two evaluations taken measurements of the studied characteristics (third independent evaluation). Coincidence between these independent evaluations was assessed by the linear regression analysis. The inter-measurement reliability was defined by the coefficient of determination (R2) and statistical significance by the F Test. The root mean square error (RMSE) of the linear fit of variables obtained by the different independent evaluations was considered as the measurement error [11]. The collected demographic, clinical, and radiographic variables were entered into an electronic database designed for further statistical analysis. The data quality control was performed by the researchers, principal investigator, and the statistician. Disagreements were resolved by discussion. The principal investigator made the final decision concerning the inclusion or exclusion of each case based on the results of discussion. Statistical analysis included: (1) analysis of variances (ANOVA) to define preoperative status and the general trend of the postoperative changes at each follow-up, P value was defined by the paired two-tailed t test; (2) linear regression analysis to define the inter-measurement reliability, and linkage between the studied continuous characteristics at different follow-ups, P value was assessed by the F test; (3) logistic regression to assess an association between categorical and continuous characteristics, P value was defined by the χ2-test; and (4) odds ratio to define an association between the risk of the studied events with categorical characteristics, P value was defined by the Fisher’s exact test; P ≤ 0.05 considered as statistically significant [11]. The statistical program JMP® Pro 13.2.1 (2016, SAS Institute Inc.; http://www.jmp.com) was used for analysis. To better understand the preoperative status and trends of the postoperative changes, we used parameters typical for subjects without spinal deformity as optimal values: LL(L1–S1), 40°–70° [8]; PT, < 20° [3]; SS, 28°–48° [8]; PI, 39°–62° [8]; PI–LL < 10° [8].
Results
The medical records of 80 patients were included in the study: the mean age, 62.4 (SD, 11.1); female, 63.7%; the mean BMI, 27.1 (SD, 5.6); DDD, 60%; DS, 40%; smoking, 8.7%; osteoporosis, 32.5%; primary index operation, 26.3%; use of osteotomy, 58.7%; the mean number of spinal levels fused, 7.3(SD, 3.4); use of vertebral interbody fusion, 47.5% (anterior, 17.5%; transforaminal, 21.3%; axial, 6.2; and combination, 2.5%); and distal level of fusion: lumbar, 27.5%; lumbosacral, 51.2%; and sacroiliac with bolts and iliac screws, 21.7%, Table 1. All included patients had satisfactory quality X-ray images preoperatively, immediately after operation and at 6-month follow-up; 90% at 12-month follow-up, and 58% at 24-month follow-up; 24% were excluded from further analysis due to revision/reoperation after the index operation; 19% had unsatisfactory quality or absence of radiographic images at last follow-up. Postoperative complications during the whole period of observation occurred in 38% of patients including: proximal junctional kyphosis/failure, 32.5%; infection, 2.5%; vertebral fracture, 15%; instrumentation failure, 6.3%; pseudarthrosis, 6.3%; distal segment degeneration, 1.3%, Table 2.
The inter-measurement reliability was high for all studied parameters with R2 ranging from 0.76 to 0.86 (P < 0.001), and the root mean square error (RMSE) from 3.9° to 6.2° which was regarded as the measurement error, Table 3.
The mean preoperative LL was 38.0° (SD, 6.0°; min., 2°; max., 85°), 40% were optimal. It increased after correction on average 10.4°–11.4°. This effect was statistically significant (P < 0.001) at all follow-ups. The percentage of optimal values rose to 61–67%, Table 4.
The mean preoperative PI was 54.7° (SD, 13.5°; min., 25°; max., 86°), 56% were optimal. It tended to increase after correction on average 1.2°–2.4° but these changes were not statistically significant (0.1 > P < 0.2). The percentage of optimal values rose to 61–69%, Table 4. However, differences between postoperative and preoperative PI in individual patients varied from − 31° to + 27° reaching a maximum at 24-month follow-up, the RMSE between the preoperative and postoperative values ranged from 7.1° to 9.0° exceeding the measurement error, 4.7° (Table 3), Fig. 1. The rate of patients with postoperative change of PI exceeding the measurement error in absolute value was immediately postoperative, 36.3% (P = 0.005); at 12-month follow-up, 47.2% (P < 0.001); and at 24-month follow-up, 46.6% (P = 0.003).
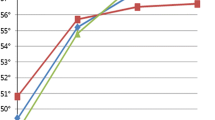
The linear regression (y = b + a × x) of the postoperative PI(y) by the preoperative PI(x) after surgical correction of adult spine deformity (ASD) at different follow-ups. (1) The expected regression line based on the hypothesis that PI is constant: PI (postop.) = PI(preop.); accordingly, the coefficient of determination, R2 = 1; a = 1; b = 0; and the root mean square error (RMSE) approximate the mean difference between two independent measurements in absolute value (measurement error) equal 4.7° in the current study, Table 3. (2) The regression line immediately after correction: a = 0.9; b = 8.4; R2 = 0.73; RMSE = 7.1; N = 80; P < 0.001. (3) The regression line at 12-month follow-up: a = 0.75; b = 15.3; R2 = 0.71; RMSE = 8.1; N = 72; P < 0.001. (4) The regression line at 24-month follow-up: a = 0.66; b = 20.7; R2 = 0.45; RMSE = 9.0; N = 46; P < 0.001
The mean preoperative PI–LL was 16.7° (SD, 17.0°; min., − 21°; max., 55°), 35% were optimal. It decreased after correction on average from − 8.4° to − 9.8° significantly at all follow-ups (P < 0.005); the rate of optimal values increased to 66% immediately after operation, but then dropped to 57% at 24-month postoperative, Table 4.
The mean preoperative PT was 24.3° (SD, 9.9°; min., 2°; max., 47°), 34% were optimal. It decreased on average − 4.3° immediately postoperatively (P < 0.0001), but then correction decreased steadily to only − 2.4° at 24-month follow-up which is not significant (P = 0.15); the rate of optimal values was 48% immediately after operation, but then decreased to 39% at 24-month postoperative, Table 4.
The mean SS was 29.7° (SD, 11.2°; min., 1°; max., 60°) before correction, 50% were optimal; SS increased on average 5.4° immediately postoperative (P < 0.0001), but then correction decreased to 4.5° at 24-month follow-up but was still significant (P = 0.015); the rate of optimal values ranged from 56% immediately after the operation to 67% at 24-month postoperative, Table 4.
The regression analysis showed that the deviation between the postoperative and preoperative PI increased steadily during two postoperative years: R2 dropped from 0.73 immediately postoperative to 0.45 at 24-month follow-up; the RMSE exceeded the measurement error at all follow-ups and reached 9° at 24-month follow-up. Of note, these changes had different vectors depending on the preoperative PI: < 39° increase; > 62° decrease; 39°–62° had minimal changes. This effect was more prominent at 12- and 24-month follow-ups, Fig. 1. The sacroiliac fixation somewhat diminished the PI instability immediately after the operation and at 1-year postoperative, but not at 2-year follow-up, Fig. 2.
The linear regression (y = b + a × x) of the postoperative PI(y) by the preoperative PI(x) after surgical correction of adult spine deformity (ASD) with sacroiliac fixation at different follow-ups. (1) The expected regression line based on the hypothesis that PI is constant: PI (postop.) = PI (preop.); accordingly, the coefficient of determination, R2 = 1; a = 1; b = 0; and the root mean square error (RMSE) approximate the mean difference between two independent measurements in absolute value (measurement error), which is 4.7° in the current study, Table 3. (2) The regression line immediately after correction: a = 0.85; b = − 6.1; R2 = 0.85; RMSE = 4.5; N = 17; P < 0.0001. (3) The regression line at 12-month follow-up: a = 0.79; b = 14.9; R2 = 0.63; RMSE = 6.2; N = 17; P < 0.001. (4) The regression line at 24-month follow-up: a = 0.28; b = 52.5; R2 = 0.28; RMSE = 10.3; N = 14; P = 0.36
The postoperative changes of PI(ΔPI) at 12-month follow-up had significant negative Spearman’s correlation with preoperative PI (ρ = − 0.34, P = 0.004) and SS (ρ = − 0.25, P = 0.04), and positive correlation with postoperative changes: ∆LL (ρ = 0.4, P < 0.001), ∆SS (ρ = 0.62, P < 0.001), and the algebraic sum of ∆SS and ∆PT (ρ = 1.0, P < 0.001), Table 5. Of note, the postoperative ∆SS strongly correlated with ∆LL (0.55 ≥ ρ ≤ 0.7, < 0.0001) and with ΔPI (0.48 ≥ ρ ≤ 0.68, < 0.001) at all follow-ups.
The regression analysis confirmed that postoperative ∆LL correlated with postoperative ∆PI, and this correlation was highest at 24-month follow-up, Fig. 3. The same trend was observed in patients with sacroiliac fixation, but it was less prominent and statistically insignificant due to the small number of cases.
The linear regression (y = b + a × x) of the postoperative change of pelvic incidence (∆PI = y) by the postoperative change of lumbar lordosis (∆LL = x) after surgical correction of adult spine deformity (ASD) at different follow-ups. (1) The expected regression line based on the hypothesis that PI is constant, and therefore, postoperative changes of LL do not have any correlation with postoperative changes of PI. Postoperative changes of PI should approximate to O and not exceed a measurement error: accordingly, the coefficient of determination, R2 = 0; a = 0; b = 0; and the root mean square error (RMSE) approximate the measurement error, 4.7° in the current study, Table 3. (2) The regression line immediately after correction: a = 0.18; b = − 0.8; R2 = 0.15; RMSE = 6.7; N = 80; P < 0.001. (3) The regression line at 12-month follow-up: a = 0.18; b = 0.09; R2 = 0.14; RMSE = 7.2; N = 72; P = 0.001. (4) The regression line at 24-month follow-up: a = 0.36; b = 0.8; R2 = 0.22; RMSE = 8.5; N = 46; P = 0.001
The postoperative ∆SS and ∆PT were not equivalent. In particular, the ∆PT were on average 40%–45% less than the corresponding ∆SS, and this disproportion was higher at 12- and 24-month follow-ups, Fig. 4. The algebraic sum of postoperative ∆SS and ∆PT reflects the difference between these changes and maximally ranged from -31° to 27° determining the postoperative ∆PI, Table 5.
The linear regression (y = b + a × x) of the postoperative change of pelvic tilt (∆PT = y) by the postoperative change of sacral slope (∆SS = x) after surgical correction of adult spine deformity (ASD) at different follow-ups. (1) The expected regression line based on the hypothesis that PI is constant; therefore, postoperative changes of PT and SS are equal in absolute values but have opposed vectors: ∆SS (preop. -postop.) = -– 1 × ∆PT (postop. -preop.); accordingly, the coefficient of determination, R2 = 1; a = − 1; b = 0; and the root mean square error (RMSE) approximate the mean absolute difference between two independent measurements (measurement error), 3.9° in the current study, Table 3. (2) The regression line immediately after correction: a = − 0.63; b = − 0.8; R2 = 0.47; RMSE = 6.4; N = 80; P < 0.001. (3) The regression line at 12-month follow-up: a = 0.49; b = − 0.87; R2 = 0.44; RMSE = 6.5; N = 72; P < 0.001. (4) The regression line at 24-month follow-up: a = 0.32; b = − 0.55; R2 = 0.44; RMSE = 8.5; N = 46; P < 0.001
Additional factors which may influence postoperative instability of PI were a diagnosis (DS rather than DDD), BMI (obesity), and caudal level of fusion (lumbar, lumbosacral, and sacroiliac) by the results of multiple regression analysis at 12-month follow-up, Fig. 5.
Impact of confounding factors on the postoperative deviation of pelvic incidence |∆PI) by results of the multiple regression analysis. Prediction formula: |∆PI|= 0.18 + \({X}_{1}\) + \({0.24X}_{2}\)+\({X}_{3}\), where: |∆PI|, the postoperative change of PI in absolute value at 12-month follow-up; \({X}_{1}\), match primary diagnosis degenerative disk disease (DDD), (− 1.4), and degenerative scoliosis (DS), 1.4; \({X}_{2}\), BMI; \({X}_{3}\), match caudal fixation: lumbar, 1.2; lumbosacral, 0.25; and sacroiliac, − 1.5; coefficient of determination, R2 = 0.13; root mean square error, RMSE = 5.4; P = 0.045
Factors associated with insufficient correction (postoperative PI–LL > 10°) at 12- and 24-month follow-up were preoperative: PI > 62° (P < 0.05), preoperative LL < 37° (P < 0.05), correction of LL < 10° (P < 0.02), and postoperative deviation of PI (|∆PI|> 6°, P < 0.01) at 24-month follow-up, Table 6.
Discussion
It has previously been shown that PI might change after surgical correction of ASD confirming inaccuracy of the previous concept concerning PI constancy [5,6,7, 10]. This effect was explained by the mobility of the sacrum relative to the pelvis through the sacroiliac joints due to physiologic properties and/or degenerative changes [6, 7, 9]. However, this assumption was not confirmed by computed tomography (CT). Therefore, we performed an additional research and revealed those patients from the studied group who had CT examination of lumbosacral spine before and after the index operation during two postoperative years, 28 such patients were revealed. The obtained results confirmed that position of sacrum relatively to pelvis may change after instrumented correction of ASD, and this change correlates with the postoperative change of PI. Besides, it was revealed that patients with ASD often have substantial preoperative spatial displacing of pelvic with the coronal (frontal) and/or axial (rotational) obliquity. These obliquities reached 8°–10° in supine position during CT scanning and had negative correlation with PI and SS defined at sagittal plane radiographic images obtained in standard standing position. It means that higher obliquity of pelvic in coronal or axial projection or their combination corresponded with smaller values of SS and PI. Postoperative elimination or decrease of these obliquities linked with increase of PI, while increase of these obliquities linked with postoperative decrease of PI (negative correlation). These additional results contribute to better understanding the mechanisms of the postoperative PI instability. It was assumed that sacroiliac/pelvic fixation should eliminate postoperative instability of spinopelvic characteristics. However, further research showed that PI may increase [6] or decrease [5, 7] even after sacroiliac fixation, and this effect is time-dependent [5,6,7]. The opposite directions of the PI change were not explained [7]. The current study revealed a few factors, which contribute to the PI postoperative instability and linked with a vector of the change. One of them is the preoperative value of PI: if PI < 39°, it tends to increase; if PI > 62°, it tends to decrease; 39° ≥ PI ≤ 62° is the most stable after correction. This finding may be linked with the preoperative spatial displacing of pelvis which may change after correction. The changes of PI continued during two postoperative years, Fig. 1. Likely, this is a result of postoperative mechanical stress in the lumbosacral and sacroiliac joints, which slowly results in a shifting of the pelvic spatial position and position of sacrum relative to the pelvis. Sacroiliac fusion mitigates this stress and diminishes the mobility but only immediately after the operation, and during the first postoperative year, after that, stabilization reduced, Fig. 2. This may be due to influence of following factors: (1) sacroiliac fixation does not eliminate postoperative changes of pelvic position including coronal and axial; and (2) time-dependent decrease of inter-surface connection between bone and implanted screws is caused by the permanent strain. This provokes bone resorption around the screws, decreases the stiffness of fixation, and increases displacement of sacrum relatively to pelvic through sacroiliac joints contributing to the postoperative PI change [11]. However, this requires further research. The next factor is disproportionality in changes of SS and PT after the correction, which determine value and vector of the PI changes: ∆PI = ∆SS + ∆PT. If the postoperative increase of SS is equivalent to the decrease of PT, PI is constant. If the postoperative increase of SS exceeds the decrease of PT, PI increases [6]. If the postoperative increase of SS is less than the decrease of PT, PI decreases [7]. Of note, SS defined as a projection of S1 cranial endplate at sagittal plane radiographic image highly depend on coronal and axial obliquity of sacrum/pelvic which may substantially decrease value of this parameter, and correspondingly PI. In some patients, SS and PT may increase simultaneously reflecting a substantial postoperative spatial displacement of the pelvic in different projections and the sacrum relatively to the pelvis. In such cases, PI may increase dramatically. It may be accompanied by the loss of sagittal balance, Fig. 6. If not, substantial retroversion of the pelvis may affect hip joints, Fig. 7. The preoperative spinopelvic alignment including PI, LL, SS, and PT, level of LL correction, and type of low-level fixation (lumbar, lumbosacral, or sacroiliac) correlates with postoperative changes of SS, PT, and PI. Of note, changes of LL correlate with the changes of SS and PI, and this correlation increases with time, Table 5, Fig. 3. Diagnosis (DS) and obesity also enhanced postoperative variability of PI, Fig. 5. The impact of obesity has been shown previously [8, 9]. It was recently shown that changes of PI after surgical instrumented correction of ASD associated with severity of preoperative sagittal imbalance, level of it correction, preoperative PI, number of spinal levels fused, and caudal level of fixation [10]. This well corresponds with the results of current study concerning postoperative instability of PI and compliments it.
Postoperative change of low preoperative pelvic incidence (PI) after correction of severe kyphoscoliosis. a Preoperative status: 67-year-old man with preoperative severe idiopathic kyphoscoliosis complicated by degenerative disk disease and lumbar stenosis. Relatively small PI (32°, bold black lines) due to the small sacral slope (SS = 11°) while the pelvic tilt (PT) close to the optimal range (21°), lumbar lordosis is also small (LL = 25°). Insufficient superposition of hip joints is due to spatial displacing of pelvic as an element of spine deformity. b Postoperative status immediate after correction by T3-L4 posterior instrumented fusion with L3/L4 decompression, transforaminal interbody fusion by cage placement (1), and multilevel (T5–T11) Smith–Petersen osteotomy. The result: LL increased to the level of preoperative PI (PI–LL = 1°); however, PI also increased immediately after the correction to 50° due to the increase of SS from 11° to 21° and PT from 21° to 29° suggesting the displacement of sacrum relatively to pelvis; correspondingly, PI–LL increased from 7° to 19° instead of expected decrease. Insufficient superposition of hip joints is due to spatial displacing of pelvic as an element of spine deformity which was not corrected. c Postoperative status at 35-month follow-up: distal segment degeneration with deformation and/or fracture of L3 (2), subsidence of the interbody cage (3), collapse of L4/L5 and L5/S1 intervertebral discs(4) with L5/S1 spondylolisthesis, loss of sagittal balance and the corrected LL, and progression of proximal junctional kyphosis; PI is still higher than before correction, 51°; tendency to progression of the pelvis retroversion indicated by the further increase of PT to 32°; PI–LL increased to 39°. a It seems that initial correction of LL to the optimal range in healthy subjects (40°–70°) rather than to the preoperative PI (32°) would have contributed to preventing the loss of correction and improve the postoperative spinopelvic alignment in this case. Well superposition of hip joins suggests optimization of the pelvic spatial position
Progressive increase of PI after repetitive surgical treatment of severe degenerative disk disease (DDD) by instrumented spinal fusion. a Preoperative status: Female, 60 years old, primary diagnosis: DDD with radiculopathy, back pain, and lumbar stenosis, status after previous decompression with posterior instrumented lumbar fusion (L3/L4). Pelvic incidence (PI) is relatively high (73.2°) due to high PT (28.9°) suggesting retroversion of the pelvis, SS is optimal (44°); lumbar lordosis (LL) looks optimal (59.1°) but PI–LL = 14° exceeding the recommended threshold (10°) due to high PI, no sagittal imbalance (SVA = 6.3 mm). Insufficient superposition of hip joints is due to spatial displacing of pelvic as an element of spine deformity. b Postoperative status immediate after correction. The previous posterior fusion extended (T10-S1) with lumbosacral fixation and cross-link hardware between L2 and L3. PI increased to 89.2° due to increase of PT to 39.6° and SS to 49.6° suggesting displacement of the sacrum relatively to the pelvis with additional retroversion of the pelvis; LL did not change substantially but PI–LL increased to 26.4° due to increasing PI, no sagittal imbalance (SVA = 16.4 mm). Insufficient superposition of hip joints is due to spatial displacing of pelvic as an element of spine deformity which was not corrected. c Postoperative status at 10-month follow-up: a further increase of PT to 43.7°, SS to 53°, and PI to 96.7°. It suggests continuing retroversion of the pelvis. Additionally, kyphosis increases proximally the upper fused level, LL increased to 71.4°, PI–LL still high, 25.3°, while sagittal balance is normal (SVA = 16.6 mm). This case shows instability of sacroiliac joint after long spinal fusion with lumbosacral fixation. This indicated by the progressive increase of PI caused by increasing PT while the sagittal balance (SVA), LL, and SS are within or close to the optimal range. It means that progressive retroversion of pelvis may be without loss of sagittal balance, but it influences hip joints due to change their physiologic position
The significant changes of the mean PI–LL toward improvement were at all follow-ups, Table 4. However, the recommended range (PI–LL < 10°) was reached in only 66% immediately after the correction and then dropped to 57% at 24-month follow-up, Table 4. This well corresponds with the previously published data [4]. However, we revealed that the insufficient correction (PI–LL ≥ 10°) is associated with high preoperative PI (> 62°), low preoperative LL (< 37°), correction of LL < 10°, and postoperative deviation of PI, ∆PI >|6°|, Table 6. It means that in patients with high preoperative PI and low LL, it is difficult to reach the recommended level of correction. This raises the question concerning the adequacy of the criterion PI–LL < 10 in some cases. For example, if a patient has preoperative PI = 80° and LL = 25°, the corrected LL should be > 70°, and LL should be increased by ≥ 45°. Of note, this may change PI, Figs. 3, 7, which decreases the likelihood to reach the planned result. Besides, an increase of LL > 30° and PI > 10° raises the risk of mechanical complications such as rod fracture and screw loosening [7, 11, 12]. Therefore, we propose an alternative approach based on two key parameters: (1) the minimum optimal value of LL in healthy subjects (40°) and (2) the maximum LL that does not increase the risk of mechanical complications, equal to the preoperative LL + 30°, 55° in this example. Thus, the corrected LL should be between 40° and 55°. Theoretically, this is enough to keep sagittal balance, minimize the risk of instrumentation failure, and is much easier to reach. If preoperative PI is low (< 39°) applying of the PI–LL < 10° criterion provides under-correction of LL which then may cause loss of sagittal balance and other complications as an example at Fig. 6. The presented alternative approach is applicable in this case too. Theoretically, this method could decrease the risk of complications and contribute to better outcomes after surgical correction of ASD.
The main limitations of the current study are retrospective design and dropout at last follow-up, which diminishes the level of evidence. Therefore, the results and conclusions are preliminary requiring confirmation.
Conclusions
-
1.
The obtained results confirm previous findings concerning inconstancy of PI after surgical correction of ASD. The postoperative changes of PI depend on time and the impact of several factors that determine value and vector of these changes.
-
2.
The main factors, which determine postoperative changes of PI, are: preoperative PI, the level of LL and SS correction, disproportionality in the postoperative changes of SS and PT, preoperative spatial position of pelvic including obliquity in the coronal and axial projections, it postoperative changes, and postoperative displacement of sacrum relatively to pelvic through sacroiliac joints. The following confounders contribute to the postoperative instability of PI: diagnosis of degenerative scoliosis, BMI (obesity), and absence of sacroiliac fixation.
-
3.
Criterion of LL correction as PI–LL < 10° has limited applicability. An alternative more universal and safe approach is proposed based on the population data of healthy subjects and the maximum acceptable level of LL correction.
Material and/or Code availability
Only routine medical records that have limited availability were used.
References
Legaye J, Duval-Beaupere G, Hecquet J et al (1998) Pelvic incidence: a fundamental pelvic parameter for three-dimensional regulation of spinal sagittal curves. Eur Spine J 7:99–103
Schwab F, Patel A, Ungar B, Farcy JP et al (2010) Adult spinal deformity-postoperative standing imbalance: how much can you tolerate? An overview of key parameters in assessing alignment and planning corrective surgery. Spine 35:2224–2231
Schwab F, Ungar B, Blondel B et al (2012) Scoliosis Research Society-Schwab adult spinal deformity classification: a validation study. Spine 37:1077–1082
Moal B, Schwab F, Ames CP et al (2014) Radiographic outcomes of adult spinal deformity correction: a critical analysis of variability and failures across deformity patterns. Spine Deform 2:219–225
Cecchinato R, Redaelli A, Martini C et al (2017) Long fusions to S1 with or without pelvic fixation can induce relevant acute variations in pelvic incidence: a retrospective cohort study of adult spine deformity surgery. Eur Spine J 26:436–441
Lee JH, Na KH, Kim JH et al (2016) Is pelvic incidence a constant, as everyone knows? Changes of pelvic incidence in surgically corrected adult sagittal deformity. Eur Spine J 25:3707–3714
Oba H, Ebata S, Takahashi J et al (2019) Loss of pelvic incidence correction after long fusion using iliac screws for adult spinal deformity: cause and effect on clinical outcome. Spine 44:195–202
Noshchenko A, Hoffecker L, Cain CM et al (2017) Spinopelvic parameters in asymptomatic subjects without spine disease and deformity: a systematic review with meta-analysis. Clin Spine Surg. 30:392–403
Schroeder N, Noschenko A, Burger E et al (2018) Pelvic incidence changes between flexion and extension. Spine Deform 6(6):753–761. https://doi.org/10.1016/j.jspd.2018.03.008
Kleck CJ, Calabrese D, Reeves BJ et al (2020) Long-term treatment effect and predictability of spinopelvic alignment after surgical correction of adult spine deformity with patient-specific spine rods. Spine 7:E387–E396
Glantz SA (2005) Primer of biostatistics, 6th edn. McGraw-Hill Medical Pub. Division, New York
Barton C, Noshchenko A, Patel VV et al (2017) Different types of mechanical complications after surgical correction of adult spine deformity with osteotomy. World J Meta-Anal 5:132–149
Acknowledgments
The authors acknowledge David Calabrese, Francisco Rodriguez Fontan and Bradley J Reeves for their contribution into the collection of primary data for the current study in particular, demographic, clinical and radiographic data, and their preparation for statistical analysis.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by CJK, AN, CMJC, EB, and VVp. The first draft of the manuscript was written by AN and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest (financial or non-financial) to declare that are relevant to the content of this article.
Ethics approval
The presented study was performed in accordance with the ethical standards of the 1964 Helsinki Declaration and its later amendments. Ethical approval was waived by the local Ethics. Committee, the Colorado Multiple Institutional Review Board (COMIRB), approval #14–1258).
Informed consent
Informed consent for participation and publication of the results were nor required due to retrospective design of the study which used just results of routine medical care investigations.
Data transparency
All authors confirm that all data and materials as well as software application support their published claims and comply with field standards.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kleck, C.J., Noshchenko, A., Burger, E.L. et al. Postoperative pelvic incidence (PI) change may impact sagittal spinopelvic alignment (SSA) after instrumented surgical correction of adult spine deformity (ASD). Spine Deform 9, 1093–1104 (2021). https://doi.org/10.1007/s43390-020-00283-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43390-020-00283-2