Abstract
The Horned Screamer (Anhima cornuta) is one of the three species of the family Anhimidae occurring in South America. It is widely distributed in the Amazon, the Cerrado, and the Atlantic Forest biomes, but detailed studies on its biology are lacking. The objective of this study was to examine aspects of the breeding of A. cornuta in the Brazilian Cerrado. Evidences of breeding birds were obtained through a search for records (photographs) in the WikiAves database in November 2019. Documented records (n = 46) of breeding birds were obtained by citizens between 2009 and 2019 in 30 municipalities. Nests with eggs (n = 15) were found mainly in marshes and grassy areas, often adjacent to the water surface. They had 2–5 white eggs and were found in two periods of the year—one comprising mainly the dry season, and the other, the rainy season. Other records (n = 31) involved families with young birds. The periods of occurrence of yellow young were highly overlapped with those of eggs. More developed young with downy plumage were found throughout the year. Young with plumage similar to that of adults (juveniles) were eventually found. Records accompanied by comments provided by citizens indicated that nests and young are cared by both parents. It was observed that birds can breed successfully outside full protected conservation areas. This study suggests that marshes should be considered a major habitat for the conservation of A. cornuta in the Brazilian Cerrado.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anhimidae family (Anseriformes) is restricted to South America, where three species occur—the Horned Screamer Anhima cornuta (Linnaeus, 1766), the Northern Screamer Chauna chavaria (Linnaeus, 1766), and the Southern Screamer Chauna torquata (Oken, 1816) (Remsen et al. 2019). They are large birds weighting about 3.0 kg and having a gallinaceous aspect, robust and short legs, enormous toes, and a bill with rudimentary lamellae (Sick 1997; Mata et al. 2006).
The Horned Screamer is widely distributed through most of northern and central South America (Piland 2010; Carboneras et al. 2019). In Brazil, it occurs mainly through extensive areas in the Amazon, the Cerrado, and the Atlantic Forest biomes (Sick 1997; WikiAves 2019). They use a wide range of habitats, including swamps, marshes, grassy meadows near rivers, lakes, rivershores, moist forests, and wetlands (Matta et al. 2006; Piland 2010; Carboneras et al. 2019). Horned Screamers are herbivorous birds and feed mainly on succulent parts of aquatic plants, including leaves, stems, roots, and flowers, and arthropods (Piland 2010; Carboneras et al. 2019). They occur as single birds, in pairs, and eventually in groups with up to six individuals (Gill et al. 1974; Sick 1997; Piland 2010). Populations are declining through the whole distribution mainly due to habitat modification by agriculture and cattle ranching (Delany and Scott 2006; Alava et al. 2009; BirdLife International 2016).
Major reviews have reported that Horned Screamers breed mainly during the spring and summer, and that the breeding season can vary geographically; clutches are of 2–7 eggs, whose colors range from white to light brown (Piland 2010; Carboneras et al. 2019). The most detailed study, carried out in Colombia, has shown that breeding occurs between late November and early May, and that eggs are incubated by both parents throughout a 47-day period (Naranjo 1986). For Brazil, it was reported that their nests are floating platforms, and that clutches are of two olive-brown eggs (Sick 1997). Additionally, eggs and incubating adults were found in September, October, and February in a lake located at a forest fragment in Tocantins state, Brazil (Pascoal et al. 2016). Information on its breeding in Brazil remains scarce, as ornithological studies have briefly reported only on abundance or seasonal occurrence in specific regions (e.g., Pinheiro and Dornas 2009; Pacheco and Olmos 2010) or localities (e.g., Simon and Lima 2004; Motta-Junior et al. 2008; Gomes et al. 2012).
This study aimed to investigate the breeding of the Horned Screamer in the Brazilian Cerrado. More specifically, this study focused on the breeding season, clutch and brood sizes, and nest characteristics. Results were discussed in terms of its breeding in the Cerrado and other South American eco-regions, as most information on its breeding has been obtained in countries adjacent to Brazil.
Methods
Study area
The Cerrado is the vegetation province that dominates central Brazil, bordered by four major ecosystems—the Amazon, the Atlantic Forest, the Pantanal, and the Caatinga (Eiten 1972). This savanna eco-region occupies about 2 million km2 in Brazil and covers smaller areas in Bolivia and Paraguay (Oliveira and Marquis 2002; Scariot et al. 2005). Major matrix types of landscapes usually are savanna woodlands, grasslands, and semideciduous forests, while less dominant physiognomies are gallery forests, rocky grasslands, marshes, and veredas—wet grasslands with scattered palm trees (Eiten 1972; Oliveira-Filho and Ratter 2002). According to Cunha et al. (2015), marshes (brejos) are small humid areas with herbaceous plant communities that occur in low-slope terrain. They often occur on impermeable soils that reduce or prevent water infiltration. Marshes have a permanent humid core area that is surrounded by a zone that expands or retracts according to the precipitation regime (Cunha et al. 2015).
The climate is tropical and marked by two well-defined periods: the rainy season occurs between October and April, and the dry season occurs between May and September. Annual rainfall ranges between 1200 and 2000 mm, and is basically restricted to the rainy season. Temperatures usually range between 10 °C in the winter (June–July) and 40 °C in the summer (January–February) (Assad 1994).
Data collection
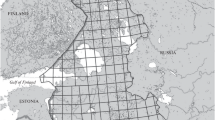
I included in the study records (photographs) of A. cornuta obtained through a search in the WikiAves database carried out between 19 and 23 November 2019. All of them were obtained by citizens in Brazilian Cerrado landscapes (Fig. 1) and involved evidences of breeding activities—nests, eggs, and young. When I could note that two or more photographs obtained in a given municipality within 1–2 years referred to the same breeding pair/family, only one was randomly selected to avoid pseudoreplication. As the database usually had no records of breeding pairs in a given locality within 1–2 years, it is unlikely that this compilation includes replications for a given family. I contacted authors of photographs in late November to confirm incubation and ask about the number of eggs and birds involved in each record, as some could be out of frame. Also, I contacted authors to ask about the location of the nest in the landscape and to verify if nests or birds were located within a conservation unit. I received permission from authors to include photographs in this paper.
Geographic distribution of the Cerrado eco-region (salmon color) in Brazil, with the location of 30 municipalities where citizens obtained records of breeding Horned Screamers (Anhima cornuta) between 2009 and 2019. The three types of localities are those with records of young (black circles), those with records of nests (white circles), and those with records of both nests and young (red circles). Photographs were gathered in the WikiAves database in November 2019
Data analysis
Based on size and plumage, young accompanied by adults were grouped into four categories/stages of development: (1) “yellow young,” i.e., small chicks with yellow downy plumage and short neck, being shorter than the adult’s belly and having the aspect of a small duck (Fig. 2a, b); (2) “intermediate young,” i.e., mid-size young with a mix of whitish-yellow and light gray downy plumage, being nearly as high as the adult’s breast (Fig. 2c, d); (3) “gray young,” i.e., mid-size young covered almost totally by a gray downy plumage, being as high as the adult’s breast or taller than this (Fig. 2e, f); (4) “juveniles,” i.e., large young with feathers similar to those of adults, including a white belly and black wings, head, neck, and breast; tail absent or short (Fig. 2g).
Young Horned Screamers (Anhima cornuta) recorded by citizens in the Brazilian Cerrado between 2009 and 2019: a three yellow young swimming at Dois Irmãos do Tocantins, TO (photo by André Grassi); b five yellow young in a nest at Três Lagoas, MS (photo by Júlio Silveira); c three intermediate young with adults at Inhumas, GO (photo by Amarildo Gonçalves); d two intermediate young with an adult at Uruana de Minas, MG (photo by Paulo Vale); e three gray young with an adult on floating vegetation at Piracanjuba, GO (photo by Wagner de Araújo Batista); f a more developed gray young with adult on a grassy area at Uruana de Minas, MG (photo by Paulo Vale); g a juvenile at Itauçu, GO (photo by Jorge Antônio do Couto). Photographs were gathered in the WikiAves database in November 2019
The mean clutch size and brood sizes of young at different stages of development were compared with a one-way ANOVA by using the program BioEstat 5.3 (Ayres et al. 2007). This analysis considered only records whose authors confirmed the number of young present in each family. A level of significance of 5% was considered in this study.
Results
Distribution of records
A total of 46 documented records of breeding Horned Screamers were included in this study. They represent 5.5% of the total number of records (n = 843) of this species obtained in the Cerrado, and available in the WikiAves database during the search period. These 46 records were obtained by citizens between 2009 and 2019 in 30 municipalities of six Brazilian states, of which Goiás and Minas Gerais had together most records (61%; Tables 1 and 2). Only a few localities were in western and eastern Cerrado (Fig. 1). Most of them were located in southern, central, and northern Cerrado, and were mainly concentrated in central Goiás and northwestern Minas Gerais. Most localities (n = 16; 53%) had only records of young, while nine (30%) localities had only records of nests; a small portion (n = 5; 17%) of the localities had records of both young and nests. These three types of localities appeared to be randomly distributed within the range in which records of breeding Horned Screamers were obtained (Fig. 1). No records were obtained in conservation units (protected areas) of integral (full) protection. Thus, breeding birds were found in rural properties or in unprotected government land.
Records of nests
Fifteen records of nests with eggs—those containing exposed eggs or associated with an incubating adult—were found in 13 municipalities of six states, mainly in Goiás and Maranhão (Table 1). They were found mainly in marshes or grassy areas, and only one was in a dry lake. About half of the nests were adjacent to the water surface, two were immediately surrounded by water, and, for six nests, it was not possible to know their distance from water. Six (40%) nests were platforms formed by the accumulation of substantial amounts of vegetal matter (Table 1). Five of them were small platforms (diameter ~ 1 m), while one was taller and wider—a large platform (diameter ~ 3 m). Other seven nests were formed by lesser amounts of matter, thus having only some long and dry leaves disposed in a nearly circular manner on short vegetation (Fig. 3). For two records, the incubating adult (confirmed by the author) could be sighted, but obstruction caused by surrounding vegetation did not allow visualization of the nest (Table 1). Eggs were white and some appeared to be dirty due to mud.
Nests of the Horned Screamer (Anhima cornuta) recorded by citizens in the Brazilian Cerrado between 2009 and 2019: a a nest with five eggs at Bataguassu, MS (photo by Valcir Muzi); b a large platform surrounded by water at Caxias, MA (photo by Firmino Freitas Soares Filho); c a nest formed by a few leaves at Itauçu, GO (photo by Jorge Antônio do Couto); d a small platform surrounded by water of a river at Itauçu, GO (photo by Jorge Antônio do Couto). Photographs were gathered in the WikiAves database in November 2019
Nests with eggs were found in two periods of the year (Fig. 4). A long period of their detection encompassed mainly the dry season, lasting from early June to mid-October, with a concentration of records in August and September. On the other hand, a short period involving the detection of nests with eggs was observed between mid-December and early February (Fig. 4), corresponding to the rainy season in the Cerrado.
Temporal record distribution of eggs and young of the Horned Screamer (Anhima cornuta) in the Brazilian Cerrado, based on photographs obtained by citizens between 2009 and 2019. Periods I, II, and III refer to days 1–10, 11–20, and 21–31 of each month, respectively. Records (n = 46) were compiled through a search in the WikiAves database in November 2019
Most (73%) nests containing eggs were closely associated with one adult on the moment of their encounters. Other nests were photographed when having 2–3 adults nearby (Table 1).
Records of young
Records involving young in variable stages of development (n = 31) were obtained in 21 municipalities of six states, of which Minas Gerais and Goiás had the highest numbers of records (Table 2). Yellow young comprised eight records, of which five (63%) involved birds in the nest, highlighting their intimate association with nests during this phase of development. Others were walking or swimming to pass through marshy areas accompanied by adults. These photographs of yellow young were grouped into two periods—a long period lasting from mid-June to late October, and a short one in the rainy season (between mid-January and early April). As a consequence, a high degree of overlap of long periods in which eggs and yellow young were detected was observed. This overlap lasted from the early dry season (June) to the early rainy season (October). Similarly, the short periods of detection of these two phases of development also were overlapped, at lesser degree. This overlap occurred in the rainy season (December to April) (Fig. 4).
Records (n = 12) of families with intermediate young have shown them walking through marshy areas. Nests never appeared in the photographs, indicating that young were no more associated to them at this phase of development. Intermediate young were recorded throughout the year, with a slight temporal division of records. The longest intervals without records lasted from mid-February to mid-April, and from mid-September to late October. With this, two periods of occurrence of intermediate young could be noted—one from late April to early September, and other lasting from early November to early February (Fig. 4).
Seven records involved gray young walking within marshes (Table 2). These records were spread throughout the year, with no remarkable temporal separation of records. However, three long intervals without records of birds at this phase of development occurred—from mid-November to late January, from mid-February to early April, and between late May and mid-July. Additionally, only four records of juveniles were obtained. They occurred in both seasons and were not concentrated in a given period of the year (Fig. 4).
The recorded families had one or two caring adults in the immediate proximities of their young. A close young care by two adults, documented by photographs obtained by citizens, was predominant in families having young at all stages of development: yellow young (75%), intermediate young (75%), gray young (57%), and juveniles (75%) (Table 2).
Clutch and brood sizes
Clutches had 2–5 eggs, being most frequent those with 2 and 3 eggs. It was not possible to know the clutch size of five nests, as citizens have not disturbed incubating adults. The number of yellow young found per family ranged from 2 to 5, with 2 being the most frequent brood size. Further, the range for intermediate young was 1–5, with 3 being the most frequent brood size. The most frequent numbers of gray young encountered with parents were 1 and 3, values that represented the range. On the other hand, 1–4 juveniles were found in families, which most often had a single young with parents (Table 2). The mean clutch and brood sizes were not significantly different (F = 0.6395, d.f. = 4, P = 0.64).
Discussion
This study presents the largest sample of breeding Horned Screamers in South American landscapes, as previous studies on its reproduction reported less than five nests (Gill et al. 1974; Hilty and Brown 1986; Naranjo 1986; Pascoal et al. 2016). Also, it is the most detailed study to investigate brood size, as the only investigation reporting on the number of young in the wild was based on a single record (Pascoal et al. 2016). Further, the approach of considering data acquisition by citizens allowed the examination of breeding through a large area, thus not being restricted to a particular locality, as occurred in previous studies (Gill et al. 1974; Hilty and Brown 1986; Alava et al. 2009; Pascoal et al. 2016).
Record distribution
Horned Screamers appear to breed throughout most of the Cerrado, as records were obtained in 30 municipalities of six states, mainly in its central, southern, and northern portions. Further observations might record breeding horned Screamers in Cerrado portions with no records obtained by citizens, such as its western and eastern regions. The fact that all records were obtained in non-protected landscapes might be a result of the photographers’ easy access to these areas, when compared with a more restricted access to parks and other conservation units. Tourism activities can bring information about birds inhabiting some Cerrado conservation units through citizen science (Pinheiro 2019). Thus, through tourism or leisure, citizens also could improve the acquisition of data regarding A. cornuta and other waterbirds in protected areas, not only in non-protected landscapes.
Most localities with records of breeding Horned Screamers were spottily distributed through most of the Cerrado’s extension. On the other hand, this compilation of records obtained by citizens detected two Cerrado regions with relatively high numbers of records of breeding birds. A region in central Goiás had 10 (21%) records and encompassed five municipalities—Itauçu, Palmeiras de Goiás, Trindade, Brazabrantes, and Inhumas. The other region with concentration of localities had six (13%) records and included five municipalities—Arinos, Uruana de Minas, Unaí, Paracatu, and João Pinheiro. These two regions do not harbor conservation units of integral protection, such as ecological stations and national parks, and their landscapes have been severely modified (INPE 2020; MMA 2020). Further studies, especially in these two regions, would be necessary to identify and protect wetlands where these birds are abundant. Also, studies at breeding sites through the Cerrado involving landscape ecology would provide information on environmental characteristics that allow them to breed successfully.
Breeding season
Information on the breeding season of A. cornuta has been brief and restricted to birds living in Colombia and Brazil. Gill et al. (1974) reported four nests with eggs in August and December (wet season) in the Colombian Llanos. Additionally, Barrow et al. (1986) observed a pair copulating in the Colombian Amazon in December, also in the rainy season. For a nest found in the Cerrado, Pascoal et al. (2016) mentioned an incubating adult in October and a young in December. Two years later, they found two nests with eggs, in September and February, in this same locality of Tocantins state.
With this, nests with eggs had been found in South America between September and February, thus involving both the rainy and dry seasons. This implies in a substantial overlap with the period of egg detection observed in my search in the WikiAves database—from early June to early February. Also, this 9-month period with eggs in the Cerrado (this study) encompasses the period of egg detection in Tocantins (Pascoal et al. 2016). Thus, eggs of A. cornuta can be found in the dry and the rainy seasons in the Cerrado. As incubation lasts for about 47 days (Naranjo 1986), egg laying also occurs in both seasons. This is because eggs found in July–September might have been laid in May–August (dry season), while those found in December–February might have been laid since October–November (rainy season). Further studies are necessary to know what environmental factors, such as food availability and climatic variables, determined the onset of breeding season of A. cornuta in the Cerrado.
Comparisons of current results involving young Horned Screamers with those of previous studies become unsatisfactory, as the only record (Pascoal et al. 2016) did not provide information on young development. The period of detection of yellow young was even longer than that of eggs—it lasted for 10 months (June to April). These two phases of breeding were substantially overlapped with each other and both were marked by an interval of about 60 days with no records. This co-occurrence of two periods with records separated by this interval makes me consider unlikely that this pattern is a result of poor sampling. I consider more likely that it results of factors relative to the environment and bird physiology, which could vary along the year. Thus, factors such as seasonal variation in rainfall, temperature, day length, and food acquisition might be leading to the observed separation in two periods.
In agreement, the seasonal occurrence of intermediate and gray young appears to be in line with the development of yellow young. First, the occurrence of intermediate and gray young between October and February would be in agreement with the occurrence of yellow young in previous months (July to October). Second, the presence of intermediate and gray young from April to September appears to be in agreement with the development of young detected between December and April. The few records of juveniles do not allow conclusions on their seasonal occurrence.
Characteristics and position of nests
Nests of A. cornuta have been described as floating platforms built within emergent vegetation in lakes, lagoons, or marshes (e.g., Gill et al. 1974; Naranjo 1986; Sick 1997; Pascoal et al. 2016). The dimensions of the small platforms found in the current database are similar to those nests recorded in previous studies (Gill et al. 1974; Naranjo 1986; Pascoal et al. 2016). However, the finding of a large platform and nests with a few leaves had not been reported previously. Further, it was observed that nest-building in the Cerrado is associated with aquatic environment (Pascoal et al. 2016). This is because numerous nests were photographed in marshes, while few nests were detected in dry lakes and grassy areas near water. Although it was noted that some of the nests were built over floating vegetation, it was not possible to know the proportion of nests representing this situation. However, observation of photographs and answers provided by authors allowed me to conclude that most nest were built over vegetation (floating or not) located very close to the water surface of marshy areas.
Role of adults in parental care and incubation
Most previous studies on breeding A. cornuta have not informed about the care of nests and young. However, Sick (1997) and Naranjo (1986) reported that incubation is performed by both sexes. The methodology adopted here does not allow broad conclusions on incubation, as it was based on photographs. Most records showing a single adult involved an incubating bird or families with one adult. In the majority of both cases, one adult of the breeding pair was out of frame, as informed by authors of photographs (pers. com.). This fact and the predominance of records with two adults indicate that both sexes participate actively in nest and young care.
Clutch and brood sizes
The range of 2–5 eggs per nest found in this study is similar to previous studies. This is because a compilation by Kear (2005) reported that females lay clutches of 2–7 eggs, while studies based on a few nests found clutches of one and four eggs in Brazil (Pascoal et al. 2016), and 3–5 eggs in Colombia (Gill et al. 1974; Naranjo 1986). Studies involving nest monitoring are necessary to know hatching success.
My study is the only to examine brood size of wild A. cornuta. It was observed that brood size was comparable for all the four categories of age/development. This occurred because the numbers of juveniles and gray, intermediate, and yellow young found per family tended to range between 2 and 3, similarly with what occurred with eggs. Although predation might occur since the egg phase, these comparable ranges and the frequent record of families with 3–4 well-developed young indicate that A. cornuta can successfully breed in non-protected landscapes in the Cerrado. The care by two adults, their large size, and structures such as strong legs and wings, and spurs, might help the protection of eggs and young against predators.
Concluding remarks
This study has shown that marshes are a major habitat for the breeding of A. cornuta in the Cerrado. This type of humid area was used for nesting and also by ranging families. Since adult birds are often encountered perched on trees (Wikiaves 2019), it becomes clear that appropriate conservation of A. cornuta outside protected areas in the Cerrado will require the conservation of vegetation patches bordering marshes. This concern was reinforced by some authors—while one adult accompanied young in marshy areas, another remained nearby (10–30 m), acting as a sentinel while perched on nearby trees (pers. com.). This behavior of the pair is in agreement with results of studies carried out in Colombia and Ecuador, and that reported territories including floating vegetation in lakes and the bordering vegetation (Gill et al. 1974; Barrow et al. 1986; Naranjo 1986; Alava et al. 2009).
Birds associated with marshes have not received much attention from scientists that investigate the Cerrado (see Macedo 2002). To help reduce the lack of information on the biology of waterbirds, future studies should investigate landscape complementation and supplementation (sensu Dunning et al. 1992) by A. cornuta and other aquatic birds to verify at which extension they use habitats adjacent to marshes for vigilance or other functions, such as feeding, resting, and shelter. Supporting this need of investigations and the conservation of native boundaries in modified landscapes are studies about landscape supplementation by Cerrado’s land birds (Tubelis 2004, 2009; Tubelis et al. 2004; Maruyama et al. 2019).
Although this study has not accompanied individually the development of the breeding phases of birds, the adopted approach of data collection provided novel information on some aspects of its reproduction. The number of breeding events of A. cornuta reported in scientific investigations carried out in the Cerrado until today (e.g., Pascoal et al. 2016) is several times lower than that compiled in this study. This fact highlights that citizens can substantially contribute to improve the knowledge regarding the breeding of aquatic birds inhabiting the Cerrado and other biomes elsewhere.
References
Alava JJ, Constantino M, Astudillo E, Arosemenas X, Penafiel M (2009) Population, seasonality and conservation threats of the Horned Screamer (Anhima cornuta) in southwestern Ecuador. Waterbirds 32:81–86. https://doi.org/10.1675/063.032.0109
Assad ED (1994) Chuva nos cerrados: análise e espacialização. EMBRAPA/SPI, Brasília
Ayres M, Ayres MM, Ayres DL, Santos AS (2007) BioEstat 5.0 – Aplicações estatísticas nas áreas das ciências biomédicas. Sociedade Civil Mamirauá/MCT-CNPq/Conservation International, Belém
Barrow JH, Black JM, Walter WB (1986) Behaviour patterns and their function in the Horned Screamer. Wildfowl 137:156–162
BirdLife International (2016) The IUCN Red List of Threatened Species 2016: Anhima cornuta. https://doi.org/10.2305/IUCN.UK.2016-3.RLTS.T22679723A92826187.en. Accessed 26 Nov 2019
Carboneras C, Boesman P, Kirwan GM (2019) Horned Screamer (Anhima cornuta). In: del Hoyo J, Elliott A, Sargatal J, Christie DA, de Juana E (eds) Handbook of the birds of the world alive. Lynx Editions, Barcelona https://www.hbw.com/node/52790. Accessed 25 Nov 2019
Cunha CN, Piedade MTF, Junk WJ (2015) Classificação e delineamento das áreas úmidas brasileiras e de seus macrohábitats. EdUFMT, Cuiabá
Delany S, Scott D (2006) Waterbird population estimates. Wetlands International, Wageningen
Dunning JB, Danielson BJ, Pulliam HR (1992) Ecological processes that affect populations in complex landscapes. Oikos 65:169–175
Eiten G (1972) The Cerrado vegetation of Brazil. Bot Rev 38:205–341
Gill FB, Stokes FJ, Stokes CC (1974) Observations on the Horned Screamer. Wilson Bull 86:43–50
Gomes FBR, Crozariol MA, Valério L (2012) Registro sobre a ocorrência de Anhima cornuta e notas sobre sua distribuição (Anseriformes: Anhimidae) em Taubaté, no Vale do Paraíba, São Paulo. Atual Ornitol 166:9–11
Hilty SL, Brown WL (1986) A guide to the birds of Colombia. Princeton University Press, Princeton
INPE (2020) Projeto Monitoramento Cerrado. http://cerrado.obt.inpe.br. Accessed 28 May 2020
Kear J (2005) Ducks, geese, and swans. Oxford University Press, New York
Macedo RHF (2002) The avifauna: ecology, biogeography and behavior. In: Oliveira PS, Marquis RJ (eds) The Cerrados of Brazil: ecology and natural history of a Neotropical savanna. Columbia University Press, New York, pp 242–265
Maruyama PK, Melo C, Pascoal C, Vicente E, Cardoso JCF, Brito VLG, Oliveira PE (2019) What is on the menu for frugivorous birds in the Cerrado? Fruiting phenology and nutritional traits highlight the importance of habitat complementarity. Acta Bot Bras 33:572–583. https://doi.org/10.1590/0102-33062019abb0221
Mata JRR, Erize F, Rumboll M (2006) A field guide to the birds of South America. Harper Collins Publishers Ltd, London
MMA (2020) Cadastro Nacional de Unidades de Conservação. https://www.mma.gov.br/areas-protegidas. Accessed 28 May 2020
Motta-Junior JC, Granzinolli MAM, Develey PF (2008) Birds of the Estação Ecológica de Itirapina, State of São Paulo, Brazil. Biota Neotrop 8:bn00308032008. https://doi.org/10.1590/S1676-06032008000300019
Naranjo LG (1986) Aspects of the biology of the Horned Screamer in southeastern Colombia. Wilson Bull 98:243–256
Oliveira PS, Marquis RJ (2002) The Cerrados of Brazil: ecology and natural history of a Neotropical savanna. Columbia University Press, New York
Oliveira-Filho AT, Ratter JA (2002) Vegetation physiognomies and woody flora of the Cerrado biome. In: Oliveira PS, Marquis RJ (eds) The Cerrados of Brazil: ecology and natural history of a neotropical savanna. Columbia University Press, New York, pp 91–120
Pacheco JF, Olmos F (2010) As aves do Tocantins, Brasil – 2: Jalapão. Rev Brasil Ornitol 18:1–18
Pascoal W, Dantas S, Weber L, Duks C (2016) Levantamento preliminar da avifauna do Campus da EMVZ da Universidade Federal do Tocantins, Araguaína – TO, com observações sobre a reprodução de algumas espécies. Atual Ornitol 189:45–56
Piland N (2010) Horned Screamer (Anhima cornuta), version 1.0. In: Schulenberg TS (ed) Neotropical birds online. Cornell Lab of Ornithology, Ithaca. https://doi.org/10.2173/nb.horscr1.01 Accessed 22 Nov 2019
Pinheiro RT (2019) Turismo de observação de aves nas unidades de conservação da região de Ilha do Bananal, Cantão (TO). Rev Brasil Ecotur 12:400–433. https://doi.org/10.34024/rbecotur.2019.v12.6740
Pinheiro RT, Dornas T (2009) Bird distribution and conservation on Cantão region, State of Tocatins: Amazon/Cerrado ecotone. Biota Neotrop 9:bn02609012009. https://doi.org/10.1590/S1676-06032009000100019
Remsen JV, Areta JI, Bonaccorso E, Claramunt S, Jaramillo A, Pacheco JF, Ribas C, Robbins MB, Stiles FG, Stotz DF, Zimmer KJ (2019) A classification of the bird species of South America. Version [13 November 2019]. American Ornithological Society. https://www.museum.lsu.edu/%20~Remsen/SACCBaseline.htm. Accessed 25 Nov 2019
Scariot A, Sousa-Silva JC, Felfili JM (2005) Cerrado: ecologia, biodiversidade e conservação. Ministério do Meio Ambiente, Brasília
Sick H (1997) Ornitologia brasileira. Editora Nova Fronteira, Rio de Janeiro
Simon JE, Lima SR (2004) Primeiro registro documentado de Anhima cornuta (Linnaeus, 1766) (Anseriformes: Anhimidae) para o Estado do Espírito Santo, Brasil. Ararajuba 12:151–152
Tubelis DP (2004) Species composition and seasonal occurrence of mixed-species flocks of forest birds in savannas in central Cerrado, Brazil. Ararajuba 12:105–111
Tubelis D (2009) Veredas and their use by birds in the Cerrado, South America: a review. Biota Neotrop 9:363–374. https://doi.org/10.1590/S1676-06032009000300031
Tubelis DP, Cowling A, Donnelly C (2004) Landscape supplementation in adjacent savannas and its implications for the design of corridors for forest birds in the central Cerrado, Brazil. Biol Conserv 118:353–364. https://doi.org/10.1016/j.biocon.2003.09.014
WikiAves (2019) Mapa de registros da espécie Anhuma (Anhima cornuta). https://www.wikiaves.com.br/mapaRegistros_anhuma. Accessed 25 Nov 2019
Acknowledgments
I thank Universidade Federal Rural do Semi-Árido for logistic support, and citizens that made their photographs available in the WikiAves database, and those that answered my questions regarding Horned Screamers. I am especially grateful to the authors of photographs included in this paper: Amarildo Gonçalves, André Grassi, Firmino Freitas Soares Filho, Jorge Antônio do Couto, Júlio Silveira, Kennedy Borges, Paulo Vale, Valcir Muzi, and Wagner de Araújo Batista. Ivinna Kariny da Costa Vieira elaborated the map. Leandro Bugoni, Luís Inoue, and two anonymous reviewers provided numerous comments on an earlier version of this manuscript.
Availability of data and material
Tables and figures contain all records involved in this study. Evidence of these records can be obtained in the WikiAves database.
Code availability
Not applicable
Author information
Authors and Affiliations
Contributions
Not applicable
Corresponding author
Ethics declarations
Conflict of interest
Not applicable
Ethics approval
Not applicable, as data was obtained in an online database
Consent to participate
Not applicable, as there is only one author
Consent for publication
Springer/Ornithology Research can publish this manuscript.
Additional information
Communicated by: Leandro Bugoni
Rights and permissions
About this article
Cite this article
Tubelis, D.P. Breeding biology of the Horned Screamer (Anhima cornuta) in non-protected areas in the Brazilian Cerrado. Ornithol. Res. 28, 115–124 (2020). https://doi.org/10.1007/s43388-020-00015-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43388-020-00015-0