Abstract
Compared to Western populations, Chinese and Asians possess distinct genetics, lifestyles, and dietary habits. They tend to have shorter stature, lower Body Mass Index (BMI), and higher body fat percentages than Western populations. The aim of this study was to compare disparities in maternal–fetal outcomes by combining pre-pregnancy BMI and gestational weight gain (GWG) based on distinct US and Chinese guidelines. A total of 2,271 pregnant women who received perinatal care at Fooyin University Hospital from 2016 to 2021 were included. Logistic regression analysis categorized women into twelve groups based on the two criteria to explore the relationships between BMI and GWG, and maternal–fetal outcomes. Among the subjects, only 23.2% and 21.8% women had a normal weight BMI and adequate GWG, based on US and Chinese criteria, respectively. As BMI and GWG increase, the likelihood of developing complications such as gestational diabetes, gestational hypertension or preeclampsia, Cesarean section, and Large for Gestational Age also rises. Conversely, underweight women with excessive GWG exhibited lower risk of preterm birth either by US or Chinese guidelines. Two criteria exhibited similar odds for investigated outcomes, except for gestational hypertension or preeclampsia. Women had more than double the odds of developing gestational hypertension or preeclampsia when using US criteria compared to Chinese criteria. Therefore, it is essential for Asian, especially Chinese women, to be aware of the differences in adverse outcomes such as gestational hypertension or preeclampsia when using US criteria.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Compared to Western populations, Chinese and Asians possess distinct genetics, lifestyles, and dietary habits. They tend to have shorter stature, lower Body Mass Index (BMI), variations in body composition and fat distribution, with higher body fat percentages than Western populations. These differences lead to increased risk of metabolic diseases among Chinese and Asian populations even with a lower BMI [1,2,3,4,5].
The BMI classification by the World Health Organization (WHO) is the most commonly used internationally, which classifies BMI into four major categories: 1) underweight: under 18.5 kg/m2; 2) normal weight: 18.5–24.9 kg/m2; 3) overweight: 25.0–29.9 kg/m2 and 4) obese: greater than or equal to 30 kg/m2. However, Chinese people have different body shape and physical structure, the Working Group on Obesity in China (WGOC) classifies BMI into groups as follows: 1) underweight: under 18.5 kg/m2; 2) normal weight: 18.5–23.9 kg/m2; 3) overweight: 24.0–27.9 kg/m2 and 4) obese: greater than or equal to 28.0 kg/m [2, 6]. The US National Academy of Medicine (NAM, formerly the Institute of Medicine) recommended healthy gestational weight gain (GWG) of pregnant women according to prepregnancy BMI as follows: 1) underweight (BMI < 18.5): 12.5 to 18.0 kg; 2) normal weight BMI (18.5–24.9): 11.5 to 16.0 kg; 3) overweight BMI (25.0–29.9): 7 to 11.5 kg; and 4) obese BMI (over 30.0): 5 to 9 kg [7]. However, Chinese people have different recommendation for GWG according to prepregnancy BMI due to the aforementioned reasons, they classify GWG as follows: 1) underweight (BMI < 18.5): 11.0 to 16.0 kg; 2) normal weight BMI (18.5–23.9): 8.0 to 14.0 kg; 3) overweight BMI (24.0–27.9): 7.0 to 11.0 kg; and 4) obese BMI (over 28.0): 5.0 to 9.0 kg [8].
According to WHO obesity atlas 2022, the estimated global prevalence of adults living with obesity increased from 15.5% (696 million) in 2010 to 25.1% (1,469 million) in 2030 and it is expected that 1 in 5 women and 1 in 7 men will be living with obesity by 2030 [9]. Pregnant women face the same issue of obesity. Furthermore, in order to support the growth and development of the fetus, pregnant women often consume more calories, which lead to excess GWG. Consequently, both high pre-pregnancy BMI and excessive GWG lead to increase the incidence of maternal–fetal adverse outcomes [10].
Currently, in many Asian countries, there are no established guidelines or standards specifically for pre-pregnancy BMI and GWG for their women, and instead rely on US guidelines to assess the risk of perinatal complications. This reliance may result in the underestimation or misinterpretation of the risk odds [11]. Therefore, the purpose of the present study was to combine pre-pregnancy BMI and GWG into 12 groups using distinct US and Chinese criteria, to compare the disparities in maternal–fetal complications between two guidelines.
Materials and Methods
Subjects and Study Design
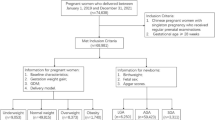
This study included 2,271 singleton pregnant women who received comprehensive prenatal and delivery care from October 2016 to June 2021 at Fooyin University Hospital. We combined four BMI categories—underweight (UW-BMI), normal weight (NM-BMI), overweight (OW-BMI), obesity (OB-BMI) and three GWG groups—inadequate (IA-GWG), adequate (AD-GWG), excessive (EX-GWG) and categorized these pregnant women into twelve cohorts according to US and Chinese guidelines [6,7,8]. The maternal-neonatal characteristics and adverse outcomes, including gestational diabetes (GDM), preeclampsia (PE), gestational hypertension (GHT), cesarean delivery (CS), preterm birth (PTB), small and large size for gestational age at birth (SGA and LGA), shoulder dystocia (SD), and neonatal intensive care unit (NICU) care, were obtained from the hospital information system (HIS). Among these women, 1999 (88%) were Taiwanese, 272 (12%) were immigrants from Asian countries who married Taiwanese husbands, with none originating from Western countries. Specifically, there were 189 from Vietnam, accounting for 8.3%, 30 from China (1.3%), 33 from Indonesia (1.5%), and 20 from other Southeast Asian countries (0.9%). Thus, the Asian immigrant wives exhibited similar physical stature and dietary habits to local Chinese women. The Institutional Review Board of Fooyin University Hospital approved this study (IRB No: FYH-IRB-112–06-02-A).
Definitions of Measurements
GDM was defined as any degree of glucose intolerance with onset or first recognition during pregnancy which was checked by two-step approach in a glucose threshold value > 140 mg/dL [12]. GHT was identified as a systolic blood pressure of higher than or equal to 140 mmHg and/or a diastolic blood pressure of greater than or equal to 90 mmHg which first happened after 20 weeks of gestation. The PE refers to the new onset of hypertension and proteinuria or the new onset of hypertension after 20 weeks of gestation [13]. PTB was defined as birth before 37 weeks of pregnancy. SGA and LGA were defined as a birthweight less or greater than 10th and 90th percentile for gestational age according to Taiwanese criteria [14]. Any complications meant at least one of the following adverse outcomes: GDM, GHT, PE, CS, LGA, SGA, PTB, SD, and NICU care.
Statistical Analysis
This study analyzed BMI, GWG, maternal–fetal characteristics and their perinatal adverse complications. The continuous variables are presented in means ± standard deviation (SD); while the categorical variables as percentages (%), except, crude and adjusted odds ratios (OR and aOR) with a 95% confidence interval (CI), which were measured by univariate and multivariate logistic regression models before and after adjusted possible factors to realize the odds of maternal–fetal adverse outcomes [15, 16]. All statistical tests were calculated using two-tailed tests, and p-values less than 0.05 were considered statistically significant. Data were analyzed using SPSS software (v. 25.0; IBM, Chicago, IL, USA).
Results
Table 1 compares the maternal characteristics and labor and delivery outcomes between women with or without complications. Significant differences were observed in age, hepatitis B infection, betel nut chewing, and first parity between the two groups (p < 0.05). According to US criteria, normal weight BMI and adequate GWG accounted for 24.8% in the group without complications and 21.9% in the group with complications, respectively. Similarly, under Chinese criteria, these figures were 24.1% and 20.2%, respectively. The incidences of GDM, PTB, and CS were 6.4%, 8.0%, and 45.4%, respectively, among all pregnant women. Additionally, the incidences of SGA, LGA, GHT or PE, SD, and NICU care were 3.3%, 6.3%, 3.7%, 0.4%, and 3.3%, respectively. Table 2 presents the number and percentage of complications in each BMI-GWG group based on US and Chinese criteria. Women who had at least one of the investigated complications accounted for 56.8% (1290/2271) of the total sample. Figure 1 illustrates specific adverse outcomes among the groups classified by BMI and GWG. Only 23.2% and 21.8% of women had a normal pre-pregnancy BMI and adequate weight gain during pregnancy; 48.3% and 62.5% of women were either obese, overweight, or had a normal BMI but experienced excessive weight gain according to US and Chinese criteria, respectively.
Tables 3 and 4 demonstrate the crude ORs and adjusted ORs (aORs) of maternal–fetal outcomes across different levels of BMI-GWG relative to the reference group. We found that the risk of GDM was significantly higher (ps < 0.05) in all groups categorized as OB-BMI and OW-BMI & EX-GWG, based on both US and Chinese standards. Compared to normal BMI & AD-GWG group, the OW-BMI & EX-GWG, OB-BMI & IA-GWG, OB-BMI & AD-GWG, OB-BMI & EX-GWG groups had higher aOR values ranging from 2.25 (95% CI: 2.25–3.92) to 4.02 (95% CI: 1.47–11.0) according to US criteria, and from 1.97 (95% CI: 1.09–3.55) to 4.08 (95% CI: 1.60–10.4) according to Chinese criteria. Furthermore, the OW-BMI & AD-GWG, OW-BMI & EX-GWG, OB-BMI & AD-GWG, OB-BMI & EX-GWG groups showed higher risks of GHT or PE, with aORs ranging from 4.95 (95% CI: 1.78–13.7) to 15.1 (95% CI: 5.94–38.1) based on US criteria. The OW-BMI & EX-GWG, and all OB-BMI groups also showed higher risks of GHT or PE, with aORs ranging from 2.42 (95% CI: 1.13–5.16) to 6.67 (95% CI: 3.22–13.8) based on Chinese criteria.
In terms of CS incidence, the OW-BMI & AD-GWG, OW-BMI & EX-GWG, and OB-BMI & EX-GWG groups had higher risks of CS rate, with aORs ranging from 1.61 (95% CI: 1.18–2.18) to 2.58 (95% CI: 1.63–4.08) according to US criteria; while the OB-BMI & AD-GWG, OW-BMI & EX-GWG, OB-BMI & AD-GWG, OB-BMI & EX-GWG groups also had higher risks of CS rates, the aORs ranged from 1.81 (95% CI: 1.09–3.01) to 2.14 (95% CI: 1.49–3.06) according to Chinese criteria. The odds of LGA were higher in the OW-BMI & EX-GWG and OB-BMI & EX-GWG groups compared to the normal BMI & AD-GWG groups. The aOR were 2.79 (95% CI: 1.58–4.92) and 5.51 (95% CI: 2.89–10.5), respectively, based on US criteria, and similar patterns were observed, with aOR of 2.90 (95% CI: 1.60–5.24), 3.03 (95% CI: 1.06–8.64), 5.12 (95% CI: 2.75–9.51), respectively, when the OW-BMI & EX-GWG, OB-BMI & IA-GWG and OB-BMI & EX-GWG groups compared to normal BMI & AD-GWG group. Additionally, compared to the reference group, the UW-BMI & IA-GWG and UW-BMI & AD-GWG groups had higher odds of SGA, with aOR of 3.39 (95% CI: 1.27–9.08) and 2.69 (95% CI: 1.04–6.98), respectively, based on the US standards. In the context of the Chinese standards, the UW-BMI & AD-GWG group exhibited even higher odds, with an aOR of 3.54 (95% CI: 1.55–8.08).
Compared to normal BMI & AD-GWG group, the UW-BMI & IA-GWG and NM-BMI & IA-GWG presented higher odds of PTB rates, the aORs were 2.75 (95% CI: 1.32–5.73) and 2.20 (95% CI: 1.41–3.44), respectively, by US criteria. The NW-BMI & IA-GWG and OW-BMI & IA-GWG also presented higher risks of PTB rates; the aORs were 2.31 (95% CI: 1.28–4.17) and 2.22 (95% CI: 1.04–4.76), respectively, by Chinese criteria. However, the UW-BMI & EX-GWG presented lower odds of PTB, with aORs of 0.21 (95% CI: 0.05–0.87) by Chinese criteria. There were no significant differences between groups in the incidences of SD and NICU care by either US or Chinese criteria (p > 0.05 s).
Finally, we compared the incidences of any investigated adverse complications between all groups. Women in all OW groups, OB-BMI & AD-GWG, and OB-BMI & EX-GWG groups had higher odds of experiencing at least one of the investigated complications compared to NM-BMI & AD-GWG women. The aORs ranged from 1.59 (95% CI: 1.16–2.18) to 2.37 (95% CI: 1.45–3.83) according to US criteria. Women in the OW-BMI & AD-GWG, OW-BMI & EX-GWG, OB-BMI & AD-GWG, OB-BMI & EX-GWG groups also showed similar patterns to those of US criteria, the aORs ranged from 1.82 (95% CI: 1.35–2.45) to 2.22 (95% CI: 1.27–3.89) according to Chinese criteria. However, the UW-BMI & EX-GWG groups showed a lower likelihood, with aOR of 0.43 (95% CI: 0.25–0.74) and 0.65 (95% CI: 0.43–0.99) by US and Chinese criteria, respectively.
Discussion
In the present study, we found that 27.3% women were overweight or obese according to WHO BMI criteria; while 33.9% women were overweight or obese according to Chinese criteria. The percentages were lower than those reported for women in the US and United Arab Emirates based on WHO criteria [17,18,19], but higher than those reported for Chinese, Brazilian and Slovakian women according to WHO criteria as well as rural Chinese according to Chinese criteria [20,21,22,23].
We found that 25.1% and 10.6% women had inadequate GWG and 39.1% and 54.4% had excessive GWG according to the NAM and Chinese guidelines, respectively. The results showed higher rates of excessive GWG compared to those reports for Tianjin and Beijing (China), Riyadh (Saudi Arabia) and Bratislava (Slovakia) according to NAM guidelines, as well as Tianjin women based on Chinese criteria [22, 24,25,26]. In the present study, compared to reference group, we observed an increased likelihood of GDM, LGA, CS, GHT or PE in women with different levels of overweight and obese combined GWG, as assessed by both US or Chinese criteria. Consistent with previous studies [11, 27,28,29,30,31,32,33,34,35], higher BMI and GWG were associated with increased odds of GDM, CS, LGA (macrosomia), possibly due to systemic inflammation and metabolic dysfunction caused by adipose tissue in women with high BMI. In addition, excessive GWG during pregnancy may lead to increased fat accumulation in the pelvic canal, resulting in reduced pelvic space, heightened resistance and decreased muscle contraction of pelvic canal, and consequently increased risks of maternal–fetal complications [27,28,29,30,31]. Furthermore, high pre-pregnancy BMI and excessive GWG superimposed the effect of increasing risk of HT and PE [32]. Notably, the US criteria, which use higher BMI and GWG threshold, resulted in nearly double the odds of HGT or PE relative to Chinese criteria in the present study.
Wang et al. revealed that overweight was a risk factor for LGA, whereas obesity was associated with higher GDM, CS, PTB, as compared with the reference normal group [33]. Sun et al. has shown overweight and inadequate GWG were risk factors for GDM, overweight and obesity were risk factors for GHT. Overweight, obesity and excessive GWG were risk factors for macrosomia as compared to the reference group [20]. Wang et al. also demonstrated being overweight and obese were linked to heighten odds of both maternal and fetal complications [33]. Similarly, Ke et al. found that Chinese women with normal weight and excessive GWG, as well as those who were overweight or obese with adequate or excessive GWG had increased odds of any complications compared to the reference group [34].
Choi et al. revealed overweight or obese Korean women with excess weight gain had the highest 3.5-fold odds, while women with underweight BMI and adequate GWG exhibited the lowers risk for any adverse outcomes as compared to reference group [35]. In a study encompassing nine million individuals in the US found that elevated maternal pre-pregnancy BMI was associated with increased the higher likelihood of macrosomia (OR 1.5 to 2.9) and LGA (OR 1.6 to 3.2) compared to mothers with normal BMI [36]. Another systematic review and meta-analysis involving over twenty million pregnant women also revealed that overweight and obese mothers had higher odds of CS (aOR: 1.4 in overweight, 2.0 in obese women), GDM (aOR: 2.1 in overweight, 4.1 in obese women), LGA (aOR: 1.7 in overweight, 2.4 in obese women), macrosomia (aOR: 1.7 in overweight, 2.3 in obese women), NICU care (aOR: 1.1 in overweight, 1.4 in obese women), GHT (aOR: 2.2 in overweight, 4.8 in obese women), PE (aOR: 1.9 in overweight, 3.6 in obese women) compared to mothers with normal BMI [11].
Our analysis of SGA and PTB across different groups reveled that women with underweight BMI were primarily at risk for SGA. Vats’s meta-analysis study showed1.5-folds increase in the risk of SGA among underweight women [11]. However, excessive weight gain during pregnancy appeared to mitigate the risk. Conversely, women with inadequate weight gain during pregnancy tended to have PTB. In the context of Chinese standards, women who were underweight and normal BMI, but experienced excessive weight gain during pregnancy, appeared to reduce the risk of PTB. Gong et al. similarly demonstrated that excessive weight gain could reduce the odds both SGA and PTB by nearly half, aligning with both Chinese and US guidelines [37].
It is recommended that overweight or obese women seek preconception and nutritional counseling prior to pregnancy [38]. The US Preventive Services Task Force Recommendation Statement (USPSTF) advocates for physicians to offer pregnant women effective behavioral counseling interventions for keeping healthy weight gain and adequate GWG during pregnancy which will help reduce the maternal-infant adverse outcomes [39, 40]. The Canadian study also suggested that counseling on pre-pregnancy weight loss actually can reduce the risk of poor perinatal outcomes, even a 10% reduction of prepregnancy BMI can lower the risk of PE, GDM diabetes, PTB, macrosomia, and stillbirth [41]. Physicians should provide guidance on managing and controlling pregnancy weight gain to reduce the risk of adverse pregnancy outcomes. Furthermore, women of childbearing age should be educated on the importance of maintaining optimal BMI when planning pregnancy. In Taiwan, medical institutions provide perinatal education and consulting service to pregnant women to maintain optimal pregnancy weight. Taiwan recommended that the ideal GWG of pregnant women is 10–14 kg, and the Taiwan Health Promotion Administration Bureau has also added the NAM guidelines into the maternal health book [42]. However, in this study, we found the GWG values were not quite satisfactory. More intensive strategies are needed to keep optimal GWG during pregnancy to mitigate the maternal–infant adverse outcomes.
The strength of this study lies in its comprehensive analysis, which combined BMI and GWG and divided subjects into 12 subgroups to compare the odds of adverse maternal and infant outcomes based on two major guidelines. To our knowledge, this is the first analysis comparing differences in the incidence of perinatal complications.
However, this study has some limitations. It was restricted to singleton women who received prenatal examination services and birth services at a regional hospital, potentially excluding women who had multiple births or did not received prenatal examination or birth services at our hospital. Furthermore, our data were limited to one hospital, and additional data from other hospitals may be needed to enhance the database and obtain more comprehensive results.
Conclusion
In conclusion, we demonstrated the relationship between BMI and GWG based on Chinese and US guidelines and their association with maternal–fetal outcomes. Both criteria showed similar odds of BMI-GWG on adverse complications, except for GHT or PE, where women had more than double the odds of developing GHT or PE when using US criteria compared to Chinese criteria. Therefore, it is essential for Asian, especially Chinese women, to be aware of the differences in adverse outcomes such as gestational hypertension or preeclampsia when using US criteria.
Data Availability
The data that support the findings of this study are available from the corresponding author, CC Lin, upon reasonable request.
Code Availability
Not applicable.
References
Sun C, Kovacs P, Guiu-Jurado E. Genetics of Body Fat Distribution: Comparative Analyses in Populations with European, Asian and African Ancestries. Genes (Basel). 2021;12(6):841.
Chen F, Wang P, Wang J, et al. Analysis and comparison of early childhood nutritional outcomes among offspring of Chinese women under the Chinese 2021 and US 2009 gestational weight gain guidelines. JAMA Netw Open. 2022;5(9):e2233250.
Rhee EJ. The Influence of Obesity and Metabolic Health on Vascular Health. Endocrinol Metab (Seoul). 2022;37(1):1–8.
Lear SA, Humphries KH, Kohli S, Chockalingam A, Frohlich JJ, Birmingham CL. Visceral adipose tissue accumulation differs according to ethnic background: results of the Multicultural Community Health Assessment Trial (M-CHAT). Am J Clin Nutr. 2007;86(2):353–9.
Wong RJ, Chou C, Sinha SR, Kamal A, Ahmed A. Ethnic disparities in the association of body mass index with the risk of hypertension and diabetes. J Community Health. 2014;39(3):437–45.
Liu L, Ma Y, Wang N, Lin W, Liu Y, Wen D. Maternal body mass index and risk of neonatal adverse outcomes in China: a systematic review and meta-analysis. BMC Pregnancy Childbirth. 2019;19(1):105.
Rasmussen KM, Yaktine AL. Institute of medicine (US) and national research council (US) committee to reexamine IOM pregnancy weight guidelines. In: Institute of Medicine and National Research Council (ed) Weight gain during pregnancy: reexamining the guidelines. Washington, DC: National Academies Press (US); 2009. https://doi.org/10.17226/12584.
Chinese Nutrition Society. Weight monitoring and evaluation during pregnancy period of Chinese women: group standard T/CNSS 009–2021 [in Chinese]. 2021. Available at: https://www.cnsoc.org/otherNotice/392100200.html. Accessed 12 July 2023.
Lobstein T, Brinsden H, Neveux M. World obesity atlas 2022. World Obesity Federation; 2022. Available at: https://www.worldobesityday.org/assets/downloads/World_Obesity_Atlas_2022_WEB.pdf. Accessed 12 July 2023.
Jin Y, Dai X, Wang N, Hu Y, Quan L, Zhu S. Clinical observation of effects of prepregnancy body mass index and weight gain during pregnancy on neonatal weight and delivery outcome. Evid Based Complement Alternat Med. 2021;2021:8323189.
Vats H, Saxena R, Sachdeva MP, Walia GK, Gupta V. Impact of maternal pre-pregnancy body mass index on maternal, fetal and neonatal adverse outcomes in the worldwide populations: A systematic review and meta-analysis. Obes Res Clin Pract. 2021;15(6):536–45.
Kung WJ, Kuo HY, Lee CH, Zen YH, Kong LC, Lin CC. Association between gestational abnormal glucose tolerance and maternal-fetal outcomes. J Obstet Gynaecol Res. 2022;48(10):2505–13.
Gestational Hypertension and Preeclampsia. ACOG Practice Bulletin, Number 222. Obstet Gynecol. 2020;135(6):e237–60.
Hsieh WS, Wu HC, Jeng SF, Liao HF, Su YN, Lin SJ, et al. Nationwide singleton birth weight percentiles by gestational age in Taiwan, 1998–2002. Acta Paediatr Taiwan. 2006;47(1):25–33.
Wu PW, Tsai S, Lee CY, et al. Contribution of insulin resistance to the relationship between sugar-sweetened beverage intake and a constellation of cardiometabolic abnormalities in adolescents. Int J Obes (Lond). 2021;45(4):828–39.
Lee CH, Lee KW, Fang FM, et al. The neoplastic impact of tobacco-free betel-quid on the histological type and the anatomical site of aerodigestive tract cancers. Int J Cancer. 2012;131(5):E733–43.
Akinyemi OA, Tanna R, Adetokunbo S, Omokhodion O, Fasokun M, Akingbule AS, et al. Increasing Pre-pregnancy Body Mass Index and Pregnancy Outcomes in the United States. Cureus. 2022;14(9):e28695.
Mischkot BF, Hersh AR, Greiner KS, Garg B, Caughey AB. Maternal and infant hospitalization costs associated with maternal pre-pregnancy body mass index in California, 2007–2011. J Matern Fetal Neonatal Med. 2022;35(23):4451–60.
Cheng TS, Ali N, Elbarazi I, et al. Sociodemographic determinants of prepregnancy body mass index and gestational weight gain: The Mutaba’ah study. Obes Sci Pract. 2021;8(3):308–19.
Sun Y, Shen Z, Zhan Y, et al. Effects of pre-pregnancy body mass index and gestational weight gain on maternal and infant complications. BMC Pregnancy Childbirth. 2020;20(1):390.
Dias MDS, Matijasevich A, Menezes AMB, et al. Association between maternal prepregnancy body mass index with offspring cardiometabolic risk factors: analysis of three Brazilian birth cohorts. J Dev Orig Health Dis. 2022;13(2):161–7.
Simko M, Totka A, Vondrova D, et al. Maternal body mass Index and gestational weight gain and their association with pregnancy complications and perinatal conditions. Int J Environ Res Public Health. 2019;16(10):1751.
Pan Y, Zhang S, Wang Q, et al. Investigating the association between prepregnancy body mass index and adverse pregnancy outcomes: a large cohort study of 536 098 Chinese pregnant women in rural China. BMJ Open. 2016;6(6):e011227.
Teede HJ, Goldstein R, Harrison C. Comparison of Chinese vs US gestational weight gain guidelines for Chinese women. JAMA Netw Open. 2022;5(9):e2233256.
Gou BH, Guan HM, Bi YX, Ding BJ. Gestational diabetes: weight gain during pregnancy and its relationship to pregnancy outcomes. Chin Med J (Engl). 2019;132(2):154–60.
Fayed A, Wahabi HA, Esmaeil S, Elkouny R, Elmorshedy H, Bakhsh H. Independent effect of gestational weight gain and prepregnancy obesity on pregnancy outcomes among Saudi women: A sub-cohort analysis from Riyadh mother and baby cohort study (RAHMA). PLoS ONE. 2022;17(1):e0262437.
Kawai T, Autieri MV, Scalia R. Adipose tissue inflammation and metabolic dysfunction in obesity. Am J Physiol Cell Physiol. 2021;320(3):C375–91.
Morales-Suárez-Varela M, Clemente-Bosch E, Peraita-Costa I, Llopis-Morales A, Martínez I, Llopis-González A. Maternal physical activity during pregnancy and the effect on the mother and newborn: A systematic review. J Phys Act Health. 2021;18(1):130–47.
Goldstein RF, Abell SK, Ranasinha S, et al. Gestational weight gain across continents and ethnicity: systematic review and meta-analysis of maternal and infant outcomes in more than one million women. BMC Med. 2018;16(1):153.
Tela FG, Bezabih AM, Adhanu AK. Effect of pregnancy weight gain on infant birth weight among mothers attending antenatal care from private clinics in Mekelle City, Northern Ethiopia: A facility based follow-up study. PLoS ONE. 2019;14(3):e0212424.
Nilses C, Persson M, Lindkvist M, Petersson K, Mogren I. High weight gain during pregnancy increases the risk for emergency caesarean section - Population-based data from the Swedish Maternal Health Care Register 2011–2012. Sex Reprod Healthc. 2017;11:47–52.
Gong X, Li J, Jiang Y, et al. Risk of preeclampsia by gestational weight gain in women with varied prepregnancy BMI: A retrospective cohort study. Front Endocrinol (Lausanne). 2022;13:967102.
Wang T, Li L, Wu C, et al. Body mass index and gestational weight gain are associated with maternal and neonatal outcomes based on Chinese women. J Diabetes Res. 2021;2021:4542367.
Ke JF, Liu S, Ge RL, Ma L, Li MF. Associations of maternal pre-pregnancy BMI and gestational weight gain with the risks of adverse pregnancy outcomes in Chinese women with gestational diabetes mellitus. BMC Pregnancy Childbirth. 2023;23(1):414.
Xu JC, Coelho Â. Association between body mass index and gestational weight gain with obstetric and neonatal complications in pregnant women with gestational diabetes. Acta Med Port. 2022;35(10):718–28.
Zong X, Wang H, Yang L, et al. Maternal pre-pregnancy body mass index categories and infant birth outcomes: A population-based study of 9 million mother-infant pairs. Front Nutr. 2022;9:789833.
Gong X, Wu T, Zhang L, et al. Comparison of the 2009 Institute of Medicine and 2021 Chinese guidelines for gestational weight gain: A retrospective population-based cohort study. Int J Gynaecol Obstet. 2023;162(3):1033–41.
Wei YM, Yang HX, Zhu WW, et al. Risk of adverse pregnancy outcomes stratified for pre-pregnancy body mass index. J Matern Fetal Neonatal Med. 2016;29(13):2205–9.
US Preventive Services Task Force, Davidson KW, Barry MJ, et al. Behavioral counseling interventions for healthy weight and weight gain in pregnancy: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(20):2087–2093.
Cantor AG, Jungbauer RM, McDonagh M, et al. Counseling and behavioral interventions for healthy weight and weight gain in pregnancy: Evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2021;325(20):2094–109.
Schummers L, Hutcheon JA, Bodnar LM, Lieberman E, Himes KP. Risk of adverse pregnancy outcomes by prepregnancy body mass index: a population-based study to inform prepregnancy weight loss counseling. Obstet Gynecol. 2015;125(1):133–43.
Taiwan Health Promotion Administration. Maternal health booklet. Ministry of Health and Welfare; 2022. Available at: https://health99.hpa.gov.tw/material/6852. Accessed 13 Oct 2023.
Acknowledgements
The authors gratefully acknowledge partial financial support from Fooyin University Hospital [grant number: FH-HR-112-10].
Funding
The study was supported by the Fooyin University Hospital under Grant No. FH-HR-112–10.
Author information
Authors and Affiliations
Contributions
WJ Kung and CC Lin were the principal investigators and drafted the article. HY Kuo and CF Chang formulated the study. CF Chang and YH Zen collected and interpreted the data, WJ Kung, HY Kuo, CC Lin wrote the manuscript and critically revised the article. All authors contributed to the intellectual content and study design and approved the final version of the article for publication.
Corresponding author
Ethics declarations
Ethics Approval
The Institutional Review Board of Fooyin University Hospital approved this study (IRB No: FYH-IRB-112–06-02-A).
Informed Consent
Not applicable.
Consent for Publication
All of the authors have read and agreed to the publication of the manuscript.
Competing Interests
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kung, WJ., Kuo, HY., Chang, CF. et al. Investigation and Comparison of Maternal Pre-Pregnancy Body Mass Index Coupled with Gestational Weight Gain on Maternal–Fetal Complications Based on US and Chinese Guidelines: A Retrospective Study. Reprod. Sci. 31, 2379–2391 (2024). https://doi.org/10.1007/s43032-024-01525-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-024-01525-8